Matters Arising |
Featured
-
-
Article
| Open Access Phase separation of FSP1 promotes ferroptosis
Phase separation of FSP1 promotes ferroptosis
An inhibitor of the ferroptosis-suppressing FSP1 induces phase separation of FSP1, thereby impairing its function and reducing tumour growth.
- Toshitaka Nakamura
- , Clara Hipp
- & Marcus Conrad
-
Article |
 Multifunctional biocatalyst for conjugate reduction and reductive amination
Multifunctional biocatalyst for conjugate reduction and reductive amination
A biocatalytic enzyme originating from bacteria, EneIRED, facilitates amine-activated conjugate alkene reduction followed by reductive amination, efficiently preparing chiral amine diastereomers, which are commonly used in pharmaceuticals and agrochemicals.
- Thomas W. Thorpe
- , James R. Marshall
- & Nicholas J. Turner
-
Article |
 The polar oxy-metabolome reveals the 4-hydroxymandelate CoQ10 synthesis pathway
The polar oxy-metabolome reveals the 4-hydroxymandelate CoQ10 synthesis pathway
18O2 labelling is used to identify metabolites in human cells that incorporate gaseous oxygen, including 4-hydroxymandelate, an intermediate in the synthesis of the coenzyme Q10 head group.
- Robert S. Banh
- , Esther S. Kim
- & Michael E. Pacold
-
Letter |
 Singlet molecular oxygen regulates vascular tone and blood pressure in inflammation
Singlet molecular oxygen regulates vascular tone and blood pressure in inflammation
Singlet molecular oxygen, produced by indoleamine 2,3-dioxygenase 1 activity, gives rise to a signalling molecule that regulates arterial relaxation under inflammatory conditions.
- Christopher P. Stanley
- , Ghassan J. Maghzal
- & Roland Stocker
-
Letter |
 Metal-free ribonucleotide reduction powered by a DOPA radical in Mycoplasma pathogens
Metal-free ribonucleotide reduction powered by a DOPA radical in Mycoplasma pathogens
A subclass of ribonucleotide reductase in Mycoplasma pathogens contains a stable radical formed from a modified tyrosine residue, overturning the presumed requirement for a dinuclear metal site in aerobic ribonucleotide reductase.
- Vivek Srinivas
- , Hugo Lebrette
- & Martin Högbom
-
Letter |
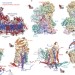 Structure of mammalian respiratory complex I
Structure of mammalian respiratory complex I
Electron cryomicroscopy structures are provided for all core and supernumerary protein subunits of mammalian complex I, a 45-subunit enzyme that powers eukaryotic respiration.
- Jiapeng Zhu
- , Kutti R. Vinothkumar
- & Judy Hirst
-
Letter |
 Repairing oxidized proteins in the bacterial envelope using respiratory chain electrons
Repairing oxidized proteins in the bacterial envelope using respiratory chain electrons
The identification of an enzymatic system repairing proteins containing oxidized methionine in the bacterial cell envelope, a compartment particularly susceptible to oxidative damage by host defence mechanisms.
- Alexandra Gennaris
- , Benjamin Ezraty
- & Frédéric Barras
-
Letter |
 X-ray structure of a mammalian stearoyl-CoA desaturase
X-ray structure of a mammalian stearoyl-CoA desaturase
The crystal structure of mouse SCD1 bound to fatty acid stearoyl-CoA is solved at 2.6 Å resolution; the structure reveals a novel geometry for the dimetal centre, and the acyl chain of the bound fatty acid is shown to be shielded and shaped to a particular conformation by the enzyme, providing a structural basis for the selectivity of fatty acid metabolism.
- Yonghong Bai
- , Jason G. McCoy
- & Ming Zhou
-
Letter |
 Trace-gas metabolic versatility of the facultative methanotroph Methylocella silvestris
Trace-gas metabolic versatility of the facultative methanotroph Methylocella silvestris
Distinct groups of microorganisms have been thought to grow on methane and on short-chain alkanes; now, the methanotroph Methylocella silvestris is shown to express two distinct soluble di-iron centre monooxygenases that allow it to use either methane or propane as a carbon and energy source.
- Andrew T. Crombie
- & J. Colin Murrell
-
Research Highlights |
Enzyme design inspired by cancer
-
Letter |
 Structure of a methyl-coenzyme M reductase from Black Sea mats that oxidize methane anaerobically
Structure of a methyl-coenzyme M reductase from Black Sea mats that oxidize methane anaerobically
The crystal structure of the enzyme MCR from methanogenic archaea shows that it is very similar to that of methanotrophic archaea; the differences observed may tune the enzymes for their respective biological context within the sea mats.
- Seigo Shima
- , Martin Krueger
- & Ulrich Ermler
-
Letter |
 Reductive glutamine metabolism by IDH1 mediates lipogenesis under hypoxia
Reductive glutamine metabolism by IDH1 mediates lipogenesis under hypoxia
- Christian M. Metallo
- , Paulo A. Gameiro
- & Gregory Stephanopoulos
-
Letter |
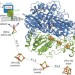 The crystal structure of an oxygen-tolerant hydrogenase uncovers a novel iron-sulphur centre
The crystal structure of an oxygen-tolerant hydrogenase uncovers a novel iron-sulphur centre
- Johannes Fritsch
- , Patrick Scheerer
- & Christian M. T. Spahn
-
Letter |
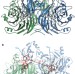 N2O binding at a [4Cu:2S] copper–sulphur cluster in nitrous oxide reductase
N2O binding at a [4Cu:2S] copper–sulphur cluster in nitrous oxide reductase
- Anja Pomowski
- , Walter G. Zumft
- & Oliver Einsle
-
Letter |
 X-ray crystal structure of the light-independent protochlorophyllide reductase
X-ray crystal structure of the light-independent protochlorophyllide reductase
The ability of plants to 'green' in the dark is attributed to the activity of the dark-operative protochlorophyllide oxidoreductase (DPOR). This enzyme catalyses the stereospecific reduction of the C17≡C18 double bond of protochlorophyllide to form chlorophyllide a, the direct precursor of chlorophyll a. The X-ray crystal structure of the catalytic component of DPOR has now been solved. A chemical mechanism is proposed by which the reduction of the double bond may occur.
- Norifumi Muraki
- , Jiro Nomata
- & Yuichi Fujita

 DHODH inhibitors sensitize to ferroptosis by FSP1 inhibition
DHODH inhibitors sensitize to ferroptosis by FSP1 inhibition