Featured
-
-
Article
| Open Access Crystal structures of ryanodine receptor SPRY1 and tandem-repeat domains reveal a critical FKBP12 binding determinant
Crystal structures of ryanodine receptor SPRY1 and tandem-repeat domains reveal a critical FKBP12 binding determinant
The ryanodine receptor (RyR) is a large multi-domain ion channel that functions to release calcium from the endoplasmic or sarcoplasmic reticulum. Here the authors present crystal structures of the SPRY1 and tandem repeat domains of RyR, allowing precise positioning of the domains and linking disease mutations to RyR function.
- Zhiguang Yuchi
- , Siobhan M. Wong King Yuen
- & Filip Van Petegem
-
Article
| Open Access Structural basis for the blockade of MATE multidrug efflux pumps
Structural basis for the blockade of MATE multidrug efflux pumps
MATE transporters confer bacterial multidrug resistance by transporting drugs across cell membranes. Here, the authors present new structures of the DinF and NorM MATE transporters and reveal insights into the mechanisms of substrate transport and MATE inhibition by small-molecule pharmaceuticals.
- Martha Radchenko
- , Jindrich Symersky
- & Min Lu
-
Article
| Open Access Harmonic force spectroscopy measures load-dependent kinetics of individual human β-cardiac myosin molecules
Harmonic force spectroscopy measures load-dependent kinetics of individual human β-cardiac myosin molecules
Single molecule methods for measuring load dependence are fundamental for molecular motor research. Here, Sung et al. introduce harmonic force spectroscopy, a method that randomly applies varying loads at high frequency, allowing the determination of load dependent parameters of human β-cardiac myosin at physiological ATP concentration.
- Jongmin Sung
- , Suman Nag
- & James A. Spudich
-
Article
| Open Access Conformational states of the full-length glucagon receptor
Conformational states of the full-length glucagon receptor
G protein-coupled receptors (GPCRs) are the largest family of transmembrane signaling proteins in humans and exhibit diverse activation mechanisms. Here, the authors combine electron microscopy, hydrogen deuterium exchange and Molecular dynamics simulations to gain insight into the activation mechanism of the glucagon receptor.
- Linlin Yang
- , Dehua Yang
- & Hualiang Jiang
-
Article
| Open Access Chemically related 4,5-linked aminoglycoside antibiotics drive subunit rotation in opposite directions
Chemically related 4,5-linked aminoglycoside antibiotics drive subunit rotation in opposite directions
Ratchet-like rotation of the small ribosomal subunit relative to the large is essential to the translation mechanism. Here, the authors show that chemically related aminoglycoside antibiotics have distinct impacts on the nature and rate of the subunit rotation process within the intact ribosome.
- Michael R. Wasserman
- , Arto Pulk
- & Scott C. Blanchard
-
Article
| Open Access Flow-driven assembly of VWF fibres and webs in in vitro microvessels
Flow-driven assembly of VWF fibres and webs in in vitro microvessels
3D microvessels with complex geometries and intact endothelium can be built in vitro. Using these engineered microvessels, here the authors show that the generation of the pathologic meshwork of the blood protein von Willebrand factor is affected by vessel architecture, flow and the proteolytic activity of ADAMTS13.
- Ying Zheng
- , Junmei Chen
- & José A. López
-
Article
| Open Access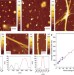 Infrared nanospectroscopy characterization of oligomeric and fibrillar aggregates during amyloid formation
Infrared nanospectroscopy characterization of oligomeric and fibrillar aggregates during amyloid formation
The onset of neurodegenerative disorders is associated at the molecular level with insoluble protein aggregates, named amyloids. Here, the authors characterize by infrared nanospectroscopy and nanomechanical studies, the amyloid aggregation at the individual species scale.
- F. S. Ruggeri
- , G. Longo
- & G. Dietler
-
Article
| Open Access Structures of FolT in substrate-bound and substrate-released conformations reveal a gating mechanism for ECF transporters
Structures of FolT in substrate-bound and substrate-released conformations reveal a gating mechanism for ECF transporters
Substrate specificity of ECF transporters is determined by the transmembrane EcfS protein subunit. Here Zhao et al.present substrate-bound and substrate-released structures of a bacterial folate transporter EcfS and suggest a gating mechanism for ECF transporters.
- Qin Zhao
- , Chengcheng Wang
- & Peng Zhang
-
Article
| Open Access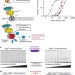 Assembly-driven activation of the AIM2 foreign-dsDNA sensor provides a polymerization template for downstream ASC
Assembly-driven activation of the AIM2 foreign-dsDNA sensor provides a polymerization template for downstream ASC
The AIM2 inflammasome complex is essential for defence against a number of human pathogens but how it assembles upon recognition of foreign DNA remains incompletely understood. Here Morrone et al.suggest the AIM2 pyrin domain acts in both DNA binding and filament assembly to generate a structural template for complex assembly.
- Seamus R. Morrone
- , Mariusz Matyszewski
- & Jungsan Sohn
-
Article
| Open Access Atomic view of the histidine environment stabilizing higher-pH conformations of pH-dependent proteins
Atomic view of the histidine environment stabilizing higher-pH conformations of pH-dependent proteins
In biological systems, large pH-induced conformational changes can be observed in certain proteins, a phenomenon poorly understood at the molecular level. Here the authors describe a peptide with the ability to self-organize into either small or large nanotubes in a pH-dependent manner and detail the mechanism driving the transition.
- Céline Valéry
- , Stéphanie Deville-Foillard
- & Franck Artzner
-
Article
| Open Access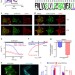 Inside-out Ca2+ signalling prompted by STIM1 conformational switch
Inside-out Ca2+ signalling prompted by STIM1 conformational switch
Depletion of calcium from intracellular stores induces interaction between the endoplasmic reticulum STIM1 protein and the plasma membrane ORAI1 channel that facilitates cellular calcium entry. Here Ma et al. characterize a STIM1 gain-of-function mutant and propose a conformational switch that controls ORAI1 gating.
- Guolin Ma
- , Ming Wei
- & Yubin Zhou
-
Article
| Open Access Locking GTPases covalently in their functional states
Locking GTPases covalently in their functional states
The cellular function of small GTPases is regulated by switching between active (GTP-bound) and inactive (GDP-bound) states. Here the authors develop nucleotide analogues that can be covalently linked to GTPases via a strategically placed cysteine residue to lock the target GTPase in defined activation states.
- David Wiegandt
- , Sophie Vieweg
- & Roger S. Goody
-
Article
| Open Access Cryo-EM structure of Hepatitis C virus IRES bound to the human ribosome at 3.9-Å resolution
Cryo-EM structure of Hepatitis C virus IRES bound to the human ribosome at 3.9-Å resolution
The Hepatitis C virus (HCV) relies on an internal ribosome entry site (IRES) for translation of all the proteins encoded by its single-stranded RNA genome. Here the authors present a near-atomic cryo-EM structure of the HCV IRES bound to the human ribosome, shedding light on the initiation mechanism of HCV's and related IRESs.
- Nick Quade
- , Daniel Boehringer
- & Nenad Ban
-
Article
| Open Access Discovery of intramolecular trans-sialidases in human gut microbiota suggests novel mechanisms of mucosal adaptation
Discovery of intramolecular trans-sialidases in human gut microbiota suggests novel mechanisms of mucosal adaptation
Mucosal sialoglycans contribute to host–microbe interactions at mucosal surfaces and impact bacterial colonization of the digestive system. Here the authors identify and characterize an intramolecular trans-sialidase produced by the gut bacterium R. gnavusATCC 29149 that may contribute to adaptation to the mucosal environment.
- Louise E. Tailford
- , C. David Owen
- & Nathalie Juge
-
Article
| Open Access Structural basis for cellobiose dehydrogenase action during oxidative cellulose degradation
Structural basis for cellobiose dehydrogenase action during oxidative cellulose degradation
Cellobiose dehydrogenases (CDHs) cooperate with lytic polysaccharide monooxygenases (LPMOs) to catalyse cellulose degradation. Here Tan et al. define the electron transfer pathway in CDH, providing a structural analysis of CDH conformers and of the interaction between CDH and LPMO during cellulose depolymerisation.
- Tien-Chye Tan
- , Daniel Kracher
- & Christina Divne
-
Article
| Open Access Microsecond protein dynamics observed at the single-molecule level
Microsecond protein dynamics observed at the single-molecule level
Single molecule spectroscopy can visualise dynamic changes in protein conformation on the submillisecond timescale. Here, Otosu et al. apply two-dimensional fluorescence lifetime correlation spectroscopy to visualise dynamics between seven conformers of cytochrome con the microsecond timescale.
- Takuhiro Otosu
- , Kunihiko Ishii
- & Tahei Tahara
-
Article
| Open Access Probing the target search of DNA-binding proteins in mammalian cells using TetR as model searcher
Probing the target search of DNA-binding proteins in mammalian cells using TetR as model searcher
During transcription, replication and repair, DNA-binding proteins must find specific interaction sites hidden within a vast excess of genomic DNA. Here the authors use single-molecule tracking to quantitatively determine the contributions of the different processes that underlie target search in human cells.
- Davide Normanno
- , Lydia Boudarène
- & Maxime Dahan
-
Article
| Open Access Cryo-EM structure of the bacteriophage T4 portal protein assembly at near-atomic resolution
Cryo-EM structure of the bacteriophage T4 portal protein assembly at near-atomic resolution
Tailed bacteriophages translocate the genome into and out of the capsid through a portal protein assembly located between the phage s head and tail. Here Sun et al. provide a cryo-EM structure of the bacteriophage T4 portal protein assembly, suggesting the functions and evolution of the portal structure.
- Lei Sun
- , Xinzheng Zhang
- & Michael G. Rossmann
-
Article |
 Allostery through the computational microscope: cAMP activation of a canonical signalling domain
Allostery through the computational microscope: cAMP activation of a canonical signalling domain
Allostery, communication between distant parts of a protein, is a key element of enzyme catalysis. Here the authors combine existing experimental data with molecular dynamics simulations and Markov state models to provide insight into the mechanism of ligand-induced allostery within the cyclicnucleotide-binding domain of protein kinase A.
- Robert D. Malmstrom
- , Alexandr P. Kornev
- & Rommie E. Amaro
-
Article
| Open Access Structural basis for catalytically restrictive dynamics of a high-energy enzyme state
Structural basis for catalytically restrictive dynamics of a high-energy enzyme state
Adenylate kinase (AdK) plays a key role in cellular energy homeostasis by catalysing the reversible magnesium-dependent formation of ADP from AMP and ATP. Here the authors present a detailed analysis of adenylate kinase’s conformational dynamics and characterize a high-energy state of AdK indispensable for catalysis.
- Michael Kovermann
- , Jörgen Ådén
- & Magnus Wolf-Watz
-
Article
| Open Access Cryo-EM reveals the conformation of a substrate analogue in the human 20S proteasome core
Cryo-EM reveals the conformation of a substrate analogue in the human 20S proteasome core
The proteasome is a highly regulated complex fundamental for cell homeostasis and a target for cancer therapy. Here the authors use cryo-EM and single-particle analysis to obtain a detailed map of the interactions between each active sites of the core 20S proteasome and the irreversible inhibitor AdaAhx3L3VS.
- Paula C.A. da Fonseca
- & Edward P. Morris
-
Article
| Open Access Crystallographic structure of a small molecule SIRT1 activator-enzyme complex
Crystallographic structure of a small molecule SIRT1 activator-enzyme complex
Sirtuins are NAD+-dependent deacylases implicated in the regulation of stress responses, bioenergetics and epigenetic control. Here the authors describe the crystal structure of a sirtuin-activating compounds (STAC)-sirtuin complex and begin to elucidate the mechanism of sirtuins activation by STACs.
- Han Dai
- , April W. Case
- & James L. Ellis
-
Article
| Open Access Protein conformational plasticity and complex ligand-binding kinetics explored by atomistic simulations and Markov models
Protein conformational plasticity and complex ligand-binding kinetics explored by atomistic simulations and Markov models
Conformational plasticity influences several aspects of protein function. Here the authors combine extensive MD simulations with Markov state models—using trypsin as model—to reveal new mechanistic details of how conformational plasticity influence ligand-receptors interactions.
- Nuria Plattner
- & Frank Noé
-
Article
| Open Access Mechanistic insights into metal ion activation and operator recognition by the ferric uptake regulator
Mechanistic insights into metal ion activation and operator recognition by the ferric uptake regulator
The regulation of iron levels is an important physiological process as excess cellular iron is highly toxic. Here the authors present several structures of a bacterial ferric uptake regulator (Fur) in complex with the Fe2+transport protein operator and Fur box, shedding light on how iron promotes DNA recognition by Fur.
- Zengqin Deng
- , Qing Wang
- & Zhongzhou Chen
-
Article
| Open Access Specificity and catalysis hardwired at the RNA–protein interface in a translational proofreading enzyme
Specificity and catalysis hardwired at the RNA–protein interface in a translational proofreading enzyme
The editing domain of aminoacyl-tRNA synthetases is responsible for removing non-cognate amino acids from mischarged tRNAs. Here the authors show that the D-aminoacyl-tRNA deacylase fold of archaeal ThrRS does not rely on protein side chains for substrate specificity and catalysis.
- Sadeem Ahmad
- , Sowndarya Muthukumar
- & Rajan Sankaranarayanan
-
Article
| Open Access Structure and gating of the nuclear pore complex
Structure and gating of the nuclear pore complex
Nuclear pore complexes (NPCs) are large macromolecular assemblies that mediate the exchange of molecules between the nucleus and cytoplasm. Here the authors present a ∼20 Å cryo-EM structure of the X. laevisNPC in different states of transport to propose a model for the architecture of the NPC’s molecular gate within its central channel.
- Matthias Eibauer
- , Mauro Pellanda
- & Ohad Medalia
-
Article
| Open Access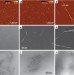 Understanding nanocellulose chirality and structure–properties relationship at the single fibril level
Understanding nanocellulose chirality and structure–properties relationship at the single fibril level
Cellulose is a material found in many different biological systems, but the fine structure at the single-molecule level is still being assessed. Here, the authors present high-resolution imaging of cellulose structures at the single particle level, finding evidence of chirality in bundles and fibrils.
- Ivan Usov
- , Gustav Nyström
- & Raffaele Mezzenga
-
Article
| Open Access Force-induced chemical reactions on the metal centre in a single metalloprotein molecule
Force-induced chemical reactions on the metal centre in a single metalloprotein molecule
The investigation of the chemical reactivity of metal centres in metalloproteins in aqueous solution is challenging. Here, the authors demonstrate the use of single molecule force spectroscopy to study the chemical reactivity of the iron-sulfur centre in rubredoxin in aqueous solution.
- Peng Zheng
- , Guilherme M. Arantes
- & Hongbin Li
-
Article
| Open Access Direct interaction between centralspindlin and PRC1 reinforces mechanical resilience of the central spindle
Direct interaction between centralspindlin and PRC1 reinforces mechanical resilience of the central spindle
The central spindle is an anti parallel bundle of microtubules that forms between segregating chromosomes and links the two halves of the mitotic spindle. Lee et al.reveal that interaction between two microtubule bundling proteins at the central spindle confers robustness to cortical pulling forces.
- Kian-Yong Lee
- , Behrooz Esmaeili
- & Masanori Mishima
-
Article
| Open Access Physical principles of membrane remodelling during cell mechanoadaptation
Physical principles of membrane remodelling during cell mechanoadaptation
Variations in cell shape must be accommodated by the cell membrane, but how the membrane adjusts to changes in area and volume is not known. Here the authors show that the membrane responds in a nearly instantaneous, purely physical manner involving the flattening or generation of membrane invaginations.
- Anita Joanna Kosmalska
- , Laura Casares
- & Pere Roca-Cusachs
-
Article
| Open Access Fluctuations between multiple EF-G-induced chimeric tRNA states during translocation on the ribosome
Fluctuations between multiple EF-G-induced chimeric tRNA states during translocation on the ribosome
EF-G enhances the rate of tRNA–mRNA translocation on the ribosome. Here the authors use single-molecule FRET to follow tRNA translocation in real time, identifying new chimeric intermediates and suggesting how EF-G binding and GTP hydrolysis change the energetic landscape of translocation to accelerate forward tRNA movement.
- Sarah Adio
- , Tamara Senyushkina
- & Marina V. Rodnina
-
Article |
 A network of molecular switches controls the activation of the two-component response regulator NtrC
A network of molecular switches controls the activation of the two-component response regulator NtrC
Nitrogen regulatory protein C (NtrC) is the effector of a two-component system activated by the sensor kinase NtrB. Vanatta et al.use molecular dynamics simulations to construct a Markov state model of NtrC activation pathways, revealing metastable conformations that could be targeted for inhibitor design.
- Dan K. Vanatta
- , Diwakar Shukla
- & Vijay S. Pande
-
Article |
 Free energy landscape of activation in a signalling protein at atomic resolution
Free energy landscape of activation in a signalling protein at atomic resolution
While active and inactive conformations of proteins have been characterised, pathways connecting these states remain largely obscure. Pontiggia et al.find that the inactive state of NtrC represents an ensemble of different conformers that interconvert to the active state via multiple pathways.
- F. Pontiggia
- , D.V. Pachov
- & D. Kern
-
Article
| Open Access Structural basis of asymmetric DNA methylation and ATP-triggered long-range diffusion by EcoP15I
Structural basis of asymmetric DNA methylation and ATP-triggered long-range diffusion by EcoP15I
Type III restriction–modification enzymes consists of two methylation and one or two restriction subunits. Here the authors report the structure of the full EcoP15I complex bound to DNA, which suggests mechanisms for ATP hydrolysis dependent diffusion along DNA and how a dimeric methyltransferase modifies only one DNA strand.
- Yogesh K. Gupta
- , Siu-Hong Chan
- & Aneel K. Aggarwal
-
Article |
 The molecular origin of high DNA-repair efficiency by photolyase
The molecular origin of high DNA-repair efficiency by photolyase
Photolyase is an enzyme responsible for repairing DNA which is damaged after exposure to UV light. Here, the authors use site directed mutagenesis and femtosecond spectroscopy to study how photolyase achieves its maximal repair efficiency.
- Chuang Tan
- , Zheyun Liu
- & Dongping Zhong
-
Article
| Open Access Conserved Omp85 lid-lock structure and substrate recognition in FhaC
Conserved Omp85 lid-lock structure and substrate recognition in FhaC
The fundamental processes of protein insertion and translocation at the outer membrane are mediated by Omp85 proteins. Here the authors report structures of the translocase FhaC, showing that the critical L6 loop adopts a conformation similar to that of related insertases; establishing a common structural basis for Omp85 function.
- Timm Maier
- , Bernard Clantin
- & Vincent Villeret
-
Article
| Open Access Architecture of TFIIIC and its role in RNA polymerase III pre-initiation complex assembly
Architecture of TFIIIC and its role in RNA polymerase III pre-initiation complex assembly
TFIIIC is a RNA polymerase III-specific general transcription factor complex essential for tRNA synthesis. Here the authors combine chemical crosslinking/mass spectrometry and X-ray crystallography to define the architecture of TFIIIC and suggest a model for the assembly of pre-initiation complexes at tRNA genes.
- Gary Male
- , Alexander von Appen
- & Christoph W. Müller
-
Article
| Open Access Ligand regulation of a constitutively dimeric EGF receptor
Ligand regulation of a constitutively dimeric EGF receptor
Whereas epidermal growth factor-induced dimerization is considered essential for EGFR signalling, the structurally related insulin receptor is a disulfide-linked dimer. Here the authors show that C. elegansEGFR is constitutively dimeric and undergoes subtle structural changes upon ligand binding that likely underlie allosteric activation.
- Daniel M. Freed
- , Diego Alvarado
- & Mark A. Lemmon
-
Article
| Open Access Signal-sequence induced conformational changes in the signal recognition particle
Signal-sequence induced conformational changes in the signal recognition particle
The signal recognition particle plays a key role in the co-translational protein targeting of membrane and secretory proteins. Here the authors report a crystal structure of the ternary SRP complex in signal sequence bound and unbound forms, providing insight into how signal sequence binding is coupled to SRP receptor interaction.
- Tobias Hainzl
- & A. Elisabeth Sauer-Eriksson
-
Article
| Open Access Molecular architecture of native fibronectin fibrils
Molecular architecture of native fibronectin fibrils
Fibronectin fibres are an important component of the extracellular matrix, supporting cell adhesion, growth and migration. Here the authors combine site-specific protein labelling with single-molecule localization microscopy to provide detailed insights into the molecular organization of native fibronectin fibrils.
- Susanna Maria Früh
- , Ingmar Schoen
- & Viola Vogel
-
Article
| Open Access Structural insights into the translational infidelity mechanism
Structural insights into the translational infidelity mechanism
Translation of mRNA into proteins is the least accurate process during genetic information transfer. Here the authors suggest—based on 11 high-resolution ribosome crystal structures—that the origin of protein missense errors involves molecular mimicry via tautomerism or ionization.
- Alexey Rozov
- , Natalia Demeshkina
- & Gulnara Yusupova
-
Article
| Open Access Vimentin filament organization and stress sensing depend on its single cysteine residue and zinc binding
Vimentin filament organization and stress sensing depend on its single cysteine residue and zinc binding
Vimentin is an intermediate filament protein that is targeted by oxidative modification at its single cysteine residue, C328. Here Pérez-Sala et al. reveal a role for this cysteine in vimentin organization and function, and uncover a protective role for zinc binding in filament stability.
- Dolores Pérez-Sala
- , Clara L. Oeste
- & F. Javier Cañada
-
Article
| Open Access Cooperative folding of intrinsically disordered domains drives assembly of a strong elongated protein
Cooperative folding of intrinsically disordered domains drives assembly of a strong elongated protein
Staphylococcal biofilm formation is promoted by the surface protein SasG. Here, the authors characterize the structure and remarkable mechanical strength of the repeat region of SasG, and show how elongation is achieved by obligate folding of the disordered regions within the repeating units.
- Dominika T. Gruszka
- , Fiona Whelan
- & Jane Clarke
-
Article
| Open Access Direct observation of TALE protein dynamics reveals a two-state search mechanism
Direct observation of TALE protein dynamics reveals a two-state search mechanism
TALEs are programmable DNA-binding proteins with practical use in genome engineering and synthetic biology. Here the authors use single-molecule fluorescence microscopy to establish that TALE proteins function using two distinct DNA-interaction modes during sequence-specific target search.
- Luke Cuculis
- , Zhanar Abil
- & Charles M. Schroeder
-
Article |
 eIF1A augments Ago2-mediated Dicer-independent miRNA biogenesis and RNA interference
eIF1A augments Ago2-mediated Dicer-independent miRNA biogenesis and RNA interference
miRNAs are incorporated into ribonucleoprotein complexes called RNA-induced silencing complexes (RISCs) to exert RNA interference. Here the authors show that translation initiation factor eIF1A interacts with the RISCs component Ago2 to promote miR-451 biogenesis and RNA interference.
- Tingfang Yi
- , Haribabu Arthanari
- & Gerhard Wagner
-
Article |
 Evolutionary-guided de novo structure prediction of self-associated transmembrane helical proteins with near-atomic accuracy
Evolutionary-guided de novo structure prediction of self-associated transmembrane helical proteins with near-atomic accuracy
While the transmembrane regions of single-pass transmembrane proteins play critical roles in receptor signalling, they remain difficult to characterize structurally. Here the authors present a computational approach for accurate structure prediction of associated single-pass transmembrane helical proteins.
- Y. Wang
- & P. Barth
-
Article
| Open Access Atomistic design of microbial opsin-based blue-shifted optogenetics tools
Atomistic design of microbial opsin-based blue-shifted optogenetics tools
Retinal-bound opsins are widely used tools for optical control of neuronal activity in vivo, so called optogenetics. Here, using molecular simulations, biochemistry, electrophysiology and X-ray crystallography, the authors present new molecular design principles for the generation of blue-shifted variants of microbial rhodopsins.
- Hideaki E. Kato
- , Motoshi Kamiya
- & Osamu Nureki
-
Article
| Open Access Green fluorescent protein nanopolygons as monodisperse supramolecular assemblies of functional proteins with defined valency
Green fluorescent protein nanopolygons as monodisperse supramolecular assemblies of functional proteins with defined valency
Supramolecular protein assemblies can provide novel nano-architectures with diverse structures and functions. Here, the authors report a fabrication strategy for a series of monodisperse protein oligomers, which allows valency-controlled display of various functional proteins.
- Young Eun Kim
- , Yu-na Kim
- & Yongwon Jung
-
Article
| Open Access RNA structure determination by solid-state NMR spectroscopy
RNA structure determination by solid-state NMR spectroscopy
The determination of RNA structures within high-molecular weight protein-RNA complexes in non-crystalline state is technically challenging. Here, the authors describe a solid-state NMR protocol for the determination of RNA structures at high resolution.
- Alexander Marchanka
- , Bernd Simon
- & Teresa Carlomagno

 Structural basis for drug-induced allosteric changes to human β-cardiac myosin motor activity
Structural basis for drug-induced allosteric changes to human β-cardiac myosin motor activity