In Your Element |
Featured
-
-
News & Views |
 Upcycling chlorinated trash into synthetic organic treasure
Upcycling chlorinated trash into synthetic organic treasure
Chlorine-containing waste streams pose potential risks to human health and the environment, so their remediation represents a significant challenge. Now, chlorinated wastes have been successfully repurposed as chlorinating reagents for use in the preparation of organic chemicals and pharmaceutical ingredients.
- Andrew Jordan
-
Article
| Open Access Mechanically triggered on-demand degradation of polymers synthesized by radical polymerizations
Mechanically triggered on-demand degradation of polymers synthesized by radical polymerizations
Radical polymerizations yield polymers that cannot easily be degraded. The co-polymerization of cyclobutene-based monomers with conventional vinyl monomers has now been shown to result in co-polymers with cyclobutane mechanophores in their backbone, which facilitate on-demand degradation through a combination of mechanical activation and hydrolysis. This approach offers a promising avenue for the degradation of all-carbon-bond-backbone polymers.
- Peng Liu
- , Sètuhn Jimaja
- & Nico Bruns
-
Article
| Open Access Upcycling of polyethylene to gasoline through a self-supplied hydrogen strategy in a layered self-pillared zeolite
Upcycling of polyethylene to gasoline through a self-supplied hydrogen strategy in a layered self-pillared zeolite
The development of new methodologies to convert plastics into fuels without relying on noble metal-based catalysts is desirable. Now it is shown that a layered self-pillared zeolite enables the conversion of polyethylene to gasoline with a selectivity of 99% and yields of >80% without the need to use external hydrogen.
- Ziyu Cen
- , Xue Han
- & Buxing Han
-
Article
| Open Access Tetrameric self-assembling of water-lean solvents enables carbamate anhydride-based CO2 capture chemistry
Tetrameric self-assembling of water-lean solvents enables carbamate anhydride-based CO2 capture chemistry
Carbon capture, utilization and storage is key for climate change mitigation and developing more environmentally friendly technologies. Now it has been shown that CO2 capture in single-component water-lean solvents is accompanied by the self-assembly of reverse-micelle-like tetrameric clusters in solution that enable the formation of various CO2-containing compounds.
- Julien Leclaire
- , David J. Heldebrant
- & Jaelynne King
-
Perspective |
 Bridging the information gap in organic chemical reactions
Bridging the information gap in organic chemical reactions
Lack of standardization, transparency and interaction creates information gaps in scientific publications. Through strategies such as voluntary information management, standardization of reaction set-ups, and smart screening approaches, this Perspective gives guidelines on how to improve data management in publications reporting chemical reactions, focusing on reproducibility, standardization and evaluation of synthetic transformations.
- Malte L. Schrader
- , Felix R. Schäfer
- & Frank Glorius
-
Article
| Open Access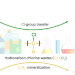 Tandem catalysis enables chlorine-containing waste as chlorination reagents
Tandem catalysis enables chlorine-containing waste as chlorination reagents
While chlorinated compounds are ubiquitous in chemical synthesis, they have a negative impact on human health and the environment. Now, a sustainable tandem catalytic process has been developed that uses chlorine-containing waste as chlorination reagents. This approach represents a promising way for the viable management of chlorinated compounds.
- Mingyang Liu
- , Xinbang Wu
- & Paul J. Dyson
-
Article |
 Chemical upcycling of commodity thermoset polyurethane foams towards high-performance 3D photo-printing resins
Chemical upcycling of commodity thermoset polyurethane foams towards high-performance 3D photo-printing resins
Current strategies for recycling cross-linked polyurethane foam waste are economically unattractive and/or lead to recycled products with inferior properties. Now it has been shown that a cost-effective chemical strategy can be used to turn the foam into high-performance value-added three-dimensional photo-printing resins.
- Zenghe Liu
- , Zizheng Fang
- & Tao Xie
-
Article
| Open Access Electrochemical synthesis of propylene from carbon dioxide on copper nanocrystals
Electrochemical synthesis of propylene from carbon dioxide on copper nanocrystals
The electrochemical reduction of carbon dioxide to propylene requires multiple C–C coupling steps and the transfer of 18 electrons, posing kinetic and thermodynamic barriers. Now the electrosynthesis of propylene from carbon dioxide using copper nanocrystals has been demonstrated, with analysis revealing that the key step for its formation is probably the coupling between carbon dioxide or carboxyl with intermediates involved in the ethylene pathway.
- Jing Gao
- , Alimohammad Bahmanpour
- & Michael Grätzel
-
Article |
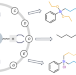 Stepwise on-demand functionalization of multihydrosilanes enabled by a hydrogen-atom-transfer photocatalyst based on eosin Y
Stepwise on-demand functionalization of multihydrosilanes enabled by a hydrogen-atom-transfer photocatalyst based on eosin Y
The controlled functionalization of multihydrosilanes is challenging. Now, using a hydrogen-atom-transfer photocatalyst based on neutral eosin Y, a method for the diverse functionalization of hydrosilanes has been developed, enabling the stepwise on-demand decoration of silicon atoms. This approach is distinguished by its atom-, step-, redox- and catalyst-economy, metal-free nature, its versatility (>150 examples), modularity, selectivity and scalability.
- Xuanzi Fan
- , Muliang Zhang
- & Jie Wu
-
Article
| Open Access Janus-type emission from a cyclometalated iron(iii) complex
Janus-type emission from a cyclometalated iron(iii) complex
Noble metals dominate the field of photosensitizers and luminophores. Now, an approach incorporating cyclometalating and carbene functions into FeIII complexes has been shown to enable dual emission from the opposing ligand-to-metal and metal-to-ligand charge-transfer states. The latter shows an exceptionally long lifetime of 4.6 ns and is quenched by oxygen and other quenchers.
- Jakob Steube
- , Ayla Kruse
- & Matthias Bauer
-
Article |
 Using waste poly(vinyl chloride) to synthesize chloroarenes by plasticizer-mediated electro(de)chlorination
Using waste poly(vinyl chloride) to synthesize chloroarenes by plasticizer-mediated electro(de)chlorination
The facile release of corrosive HCl gas and plasticizers from poly(vinyl chloride) (PVC) makes it a challenging material to recycle. Now, it has been shown that PVC waste can be directly used as a halogen source to synthesize chloroarenes. This paired electro(de)chlorination is mediated by a phthalate plasticizer already contained in PVC waste.
- Danielle E. Fagnani
- , Dukhan Kim
- & Anne J. McNeil
-
Thesis |
 Common elements, uncommon chemistry
Common elements, uncommon chemistry
Bruce Gibb suggests that chemists can play a central role in democratizing access to energy by focusing on common elements.
- Bruce C. Gibb
-
In Your Element |
 Ionic liquids charge ahead
Ionic liquids charge ahead
Choline 2-hexenoate is an ionic compound that is a liquid at room temperature, and is just one of a class of compounds that have huge potential in biomedical research and clinical applications, explains Eden E. L. Tanner.
- Eden E. L. Tanner
-
Article |
 Sustainable polyesters via direct functionalization of lignocellulosic sugars
Sustainable polyesters via direct functionalization of lignocellulosic sugars
Functionalizing an intact carbohydrate core with acetals allows for the dramatically simplified production of a plastic precursor directly during the initial fractionation of non-edible biomass. When polymerized, the rigid and polar carbohydrate core also leads to bioplastics with competitive material and end-of life properties.
- Lorenz P. Manker
- , Graham R. Dick
- & Jeremy S. Luterbacher
-
Thesis |
 Sustaining chemistry
Sustaining chemistry
Bruce C. Gibb offers some suggestions to chemists for how they can help save the world, one nudge at a time.
- Bruce C. Gibb
-
Review Article |
 Multistep enzyme cascades as a route towards green and sustainable pharmaceutical syntheses
Multistep enzyme cascades as a route towards green and sustainable pharmaceutical syntheses
Enzymes, either purified or as whole-cell biocatalysts, can be concatenated into catalytic cascades and used to produce pharmaceutically relevant molecules. This Review discusses the advantages and requirements of multistep enzyme cascades and also highlights how they can be harnessed to achieve highly sustainable and cost-efficient syntheses.
- Ana I. Benítez-Mateos
- , David Roura Padrosa
- & Francesca Paradisi
-
Article |
 Tunable acetylene sorption by flexible catenated metal–organic frameworks
Tunable acetylene sorption by flexible catenated metal–organic frameworks
Flexible metal–organic frameworks (MOFs) in which guest uptake and release occur above certain threshold pressures are attractive adsorbents. Now, the gated sorption behaviour of such a zinc-based mixed-ligand MOF has been tuned to match the narrow temperature and pressure range required for safe, efficient acetylene storage by adjusting the ratio of two different functional groups on its benzenedicarboxylate ligands.
- Mickaele Bonneau
- , Christophe Lavenn
- & Susumu Kitagawa
-
News & Views |
 White phosphorus first
White phosphorus first
Many of the methods used to make phosphines proceed via phosphorus trichloride-based intermediates, leading to chloride waste that is difficult to recycle. It has now been shown that this disadvantage can be overcome by using a method that directly converts white phosphorus into value-added phosphorus compounds.
- Hansjörg Grützmacher
-
Q&A |
 The steps to sustainability
The steps to sustainability
Miao Hong, based at the Shanghai Institute of Organic Chemistry, tells Nature Chemistry about her work in sustainable polymer design and her thoughts about the future of this field.
- Kathryn Ashe
-
Article |
 Fast CO2 hydration kinetics impair heterogeneous but improve enzymatic CO2 reduction catalysis
Fast CO2 hydration kinetics impair heterogeneous but improve enzymatic CO2 reduction catalysis
Carbonic anhydrase enzymatically catalyses CO2 hydration, and its effect on enzymatic and heterogeneous CO2 reduction has now been studied. Through the co-immobilization of carbonic anhydrase, it has been shown that faster CO2 hydration kinetics are beneficial for enzymatic catalysis (using formate dehydrogenase) but detrimental for heterogeneous catalysts, such as gold.
- Samuel J. Cobb
- , Vivek M. Badiani
- & Erwin Reisner
-
Article |
 A photosensitizer–polyoxometalate dyad that enables the decoupling of light and dark reactions for delayed on-demand solar hydrogen production
A photosensitizer–polyoxometalate dyad that enables the decoupling of light and dark reactions for delayed on-demand solar hydrogen production
Decoupling the processes of light harvesting and catalytic hydrogen evolution could be a potentially important step in storing solar energy. This has now been achieved with a single molecular unit: a light-harvesting ruthenium complex–polyoxometalate dyad that absorbs light, separates and stores charge and then generates hydrogen on demand following the addition of a proton donor.
- Sebastian Amthor
- , Sebastian Knoll
- & Carsten Streb
-
Article |
 Towards high-performance sustainable polymers via isomerization-driven irreversible ring-opening polymerization of five-membered thionolactones
Towards high-performance sustainable polymers via isomerization-driven irreversible ring-opening polymerization of five-membered thionolactones
Five-membered lactones are common in nature and are produced in large quantities from biomass, but a lack of ring strain means that ring-opening polymerization is usually thermodynamically unfavourable at ambient conditions. Now, an irreversible ring-opening polymerization of biomass-derived five-membered thionolactones—driven by S/O isomerization—has been developed, enabling their conversion into sustainable polymers at industrially relevant temperatures.
- Pengjun Yuan
- , Yangyang Sun
- & Miao Hong
-
Article |
 A dual cellular–heterogeneous catalyst strategy for the production of olefins from glucose
A dual cellular–heterogeneous catalyst strategy for the production of olefins from glucose
A dual cellular-then-heterogeneous catalysis strategy has been used to produce olefins from glucose. 3-Hydroxy acids are made using an engineered microbial host. A hydrolytic step then provides the driving force for fatty acid deoxygenation by simple heterogeneous Lewis acid catalysis. This decarboxylation–dehydration route to olefinic products avoids the need for an additional redox input typically required for deoxygenation of unmodified fatty acids.
- Zhen Q. Wang
- , Heng Song
- & Michelle C. Y. Chang
-
News & Views |
 From disposable diapers to adhesives
From disposable diapers to adhesives
Finding alternative fates for plastics that would otherwise end up in landfills requires innovative chemistry. Now, poly(acrylic acid) from diaper waste has been converted into valuable pressure-sensitive adhesives through an open-loop recycling method that is cost-effective and environmentally competitive.
- Meredith A. Borden
- & Frank A. Leibfarth
-
Article |
 Oxidative cleavage of C–C bonds in lignin
Oxidative cleavage of C–C bonds in lignin
The valorization of lignin is generally implemented through the cleavage of labile C–O bonds to produce aromatic monomers in up to 40 wt% yield. The remaining material consists of lignin dimers and oligomers connected by C–C bonds, but now a method has been developed for the oxidative cleavage of these C–C bonds using oxoammonium salts, to produce benzoquinones.
- Elena Subbotina
- , Thanya Rukkijakan
- & Joseph S. M. Samec
-
News & Views |
 Low strain, more gain
Low strain, more gain
Recycling polymers to their monomers would enable a circular polymer economy, but this can be challenging, especially for materials with all-carbon backbones. Now, by lowering the strain of cyclooctene through ring fusion, recyclable polymers with useful physical properties can be made by an olefin-metathesis-based route.
- Yutan Getzler
-
Article
| Open Access Selectivity control in hydrogenation through adaptive catalysis using ruthenium nanoparticles on a CO2-responsive support
Selectivity control in hydrogenation through adaptive catalysis using ruthenium nanoparticles on a CO2-responsive support
A multifunctional catalytic system composed of ruthenium nanoparticles immobilized on a silica surface decorated with an amine-functionalized polymer is used for the hydrogenation of biomass-derived furfural acetone and related substrates. The presence or absence of CO2 in the gas feed alters the selectivity of the hydrogenation—producing either a ketone or a saturated alcohol, respectively—in a fully reversible manner.
- Alexis Bordet
- , Sami El Sayed
- & Walter Leitner
-
Article |
 Iron-catalysed synthesis and chemical recycling of telechelic 1,3-enchained oligocyclobutanes
Iron-catalysed synthesis and chemical recycling of telechelic 1,3-enchained oligocyclobutanes
Iron-catalysed [2+2] cycloaddition/oligomerization of neat butadiene affords
(1,n′-divinyl)oligocyclobutane —a telechelic, crystalline material consisting of 1,3-enchained cyclobutyl units. This oligocyclobutane can be chemically recycled to pure butadiene using the same iron catalyst employed in its synthesis, demonstrating design principles for next-generation plastic materials that can be returned to pristine monomer.- Megan Mohadjer Beromi
- , C. Rose Kennedy
- & Paul J. Chirik
-
Article |
 Screening and characterization of a diverse panel of metagenomic imine reductases for biocatalytic reductive amination
Screening and characterization of a diverse panel of metagenomic imine reductases for biocatalytic reductive amination
High-throughput biocatalytic screening and metagenomics have been used to discover over 300 imine reductases (IREDs) and subsequently produce a sequence-diverse panel of IREDs suitable for optimizing the synthesis of chiral amines. Additional characterization identified biocatalysts that accommodate structurally demanding amines and ketones for enzymatic reductive aminations.
- James R. Marshall
- , Peiyuan Yao
- & Nicholas J. Turner
-
Article |
 Enzyme-free synthesis of natural phospholipids in water
Enzyme-free synthesis of natural phospholipids in water
It is unclear how phospholipid membranes formed on the early Earth, as modern cells synthesize the phospholipid constituents of their membranes enzymatically. Now, a combination of ion pairing and self-assembly has enabled transacylation of lysophospholipids with acyl donors in water, affording a variety of membrane-forming natural diacylphospholipids in high yields.
- Luping Liu
- , Yike Zou
- & Neal K. Devaraj
-
Article |
 Coupling N2 and CO2 in H2O to synthesize urea under ambient conditions
Coupling N2 and CO2 in H2O to synthesize urea under ambient conditions
Conventionally, urea is synthesized via two consecutive processes, N2 + H2 → NH3 followed by NH3 + CO2. Now, an electrocatalyst consisting of PdCu alloy nanoparticles on TiO2 nanosheets has been shown to directly couple N2 and CO2 in H2O to produce urea under ambient conditions.
- Chen Chen
- , Xiaorong Zhu
- & Shuangyin Wang
-
Article |
 Facile synthesis of precious-metal single-site catalysts using organic solvents
Facile synthesis of precious-metal single-site catalysts using organic solvents
Au/C single-site catalysts have been validated commercially for acetylene hydrochlorination, but they have previously been prepared using highly oxidizing acidic solvents or additional ligands. It has now been shown that they can be made by impregnation of a metal salt from an acetone solution—generating catalysts with comparable activity to those synthesized by the other methods.
- Xi Sun
- , Simon R. Dawson
- & Graham J. Hutchings
-
Article |
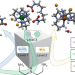 Simultaneous CO2 capture and metal purification from waste streams using triple-level dynamic combinatorial chemistry
Simultaneous CO2 capture and metal purification from waste streams using triple-level dynamic combinatorial chemistry
Carbon capture technologies are poised to play an important role in reducing CO2 emissions to mitigate global warming, but their cost prevents their extensive use. Now, CO2 capture and utilization have been combined using a triple-level dynamic combinatorial approach. CO2 was spontaneously captured by industrial polyamines to create libraries of ligands that can be used for the separation and recovery of metals.
- Jean Septavaux
- , Clara Tosi
- & Julien Leclaire
-
Article |
 A marine viral halogenase that iodinates diverse substrates
A marine viral halogenase that iodinates diverse substrates
A flavin-dependent halogenase with a remarkable preference for iodination has now been discovered. The halogenase (VirX1) was discovered using a bioinformatics-based approach and comes from a cyanophage. Structural characterization and kinetic studies show that VirX1 possesses broad substrate tolerance, making it an attractive tool for synthesis.
- Danai S. Gkotsi
- , Hannes Ludewig
- & Rebecca J. M. Goss
-
Article |
 A closed cycle for esterifying aromatic hydrocarbons with CO2 and alcohol
A closed cycle for esterifying aromatic hydrocarbons with CO2 and alcohol
Previous methods for functionalizing hydrocarbons with CO2 to form carboxylic acid derivatives have required stoichiometric energy-intensive reagents such as strong bases or electrophilic activators. Now, a simple solid base has been developed that transforms aromatic hydrocarbons, CO2 and methanol into aromatic esters via a two-step cycle requiring no additional reagents.
- Dianne J. Xiao
- , Emma D. Chant
- & Matthew W. Kanan
-
Article |
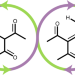 Closed-loop recycling of plastics enabled by dynamic covalent diketoenamine bonds
Closed-loop recycling of plastics enabled by dynamic covalent diketoenamine bonds
It is difficult to recover materials for re-manufacturing and re-use from plastics that are compounded with colourants, fillers and flame retardants. Now, it has been shown that alternative plastics based on dynamic covalent poly(diketoenamine)s depolymerize in strong aqueous acids and enable triketone and amine monomers to be isolated and upcycled into new plastics.
- Peter R. Christensen
- , Angelique M. Scheuermann
- & Brett A. Helms
-
Comment |
 Circular chemistry to enable a circular economy
Circular chemistry to enable a circular economy
By expanding the scope of sustainability to the entire lifecycle of chemical products, the concept of circular chemistry aims to replace today’s linear ‘take–make–dispose’ approach with circular processes. This will optimize resource efficiency across chemical value chains and enable a closed-loop, waste-free chemical industry.
- Tom Keijer
- , Vincent Bakker
- & J. Chris Slootweg
-
Article |
 From alkylarenes to anilines via site-directed carbon–carbon amination
From alkylarenes to anilines via site-directed carbon–carbon amination
Limitations associated with the primary amination of aryl C–H bonds include the poor control of regioselectivity with electron-rich substrates and the challenging nature of the reaction in the case of electron-deficient arenes. Now, site-directed C–C bond primary amination of simple alkylarenes and benzyl alcohols provides a route for the direct and efficient preparation of anilines.
- Jianzhong Liu
- , Xu Qiu
- & Ning Jiao
-
Article |
 Carbohydrate stabilization extends the kinetic limits of chemical polysaccharide depolymerization
Carbohydrate stabilization extends the kinetic limits of chemical polysaccharide depolymerization
Chemically depolymerizing biomass polysaccharides to simple sugars is often controlled by the balance between depolymerization and degradation kinetics, which has limited the concentration of solutions that can be obtained and overall yields. The reversible stabilization of carbohydrates by acetal formation pushes back these limits and creates stabilized sugars that have advantageous properties for further upgrading.
- Ydna M. Questell-Santiago
- , Raquel Zambrano-Varela
- & Jeremy S. Luterbacher
-
Article |
 Non-aqueous homogenous biocatalytic conversion of polysaccharides in ionic liquids using chemically modified glucosidase
Non-aqueous homogenous biocatalytic conversion of polysaccharides in ionic liquids using chemically modified glucosidase
As a consequence of high chemical resistance and low solubility in conventional solvents, deconstructing biomass into fuels and other useful chemical building blocks remains a challenge. Now, through enzyme modification and ionic liquid solvents, it is possible to homogeneously biocatalytically convert cellulose to sugars at a rate 30 times greater than is achievable in water.
- Alex P. S. Brogan
- , Liem Bui-Le
- & Jason P. Hallett
-
Article |
 Oxidase catalysis via aerobically generated hypervalent iodine intermediates
Oxidase catalysis via aerobically generated hypervalent iodine intermediates
Oxidation chemistry is critical to introducing molecular complexity during chemical synthesis. Development of sustainable oxidation chemistry demands strategies to harness O2 as a terminal oxidant. Access to hypervalent iodine compounds — a class of broadly useful chemical oxidants — from O2 increases the scope of aerobic oxidation chemistry that can be achieved.
- Asim Maity
- , Sung-Min Hyun
- & David C. Powers
-
Article |
 Enantioselective, intermolecular benzylic C–H amination catalysed by an engineered iron-haem enzyme
Enantioselective, intermolecular benzylic C–H amination catalysed by an engineered iron-haem enzyme
The intermolecular amination of C–H bonds is an enabling transformation for the synthesis of nitrogen-containing molecules; however, developing catalysts for this class of reactions is very challenging. Now, an iron-based enzyme for this reaction has been engineered, demonstrating that a protein can confer a difficult new function upon an otherwise unreactive base metal.
- Christopher K. Prier
- , Ruijie K. Zhang
- & Frances H. Arnold
-
-
Article |
 Aldehydes as alkyl carbanion equivalents for additions to carbonyl compounds
Aldehydes as alkyl carbanion equivalents for additions to carbonyl compounds
Methods utilizing renewable feedstocks are critical to accessing molecules of industrial importance in light of the present ecological and economic climate. Here, it is shown that umpolung reactivity of carbonyl compounds can be used for nucleophilic additions to yield a diverse array of valuable alcohols as an alternative to using stoichiometric organometallic reagents.
- Haining Wang
- , Xi-Jie Dai
- & Chao-Jun Li
-
Article |
 Rapid heteroatom transfer to arylmetals utilizing multifunctional reagent scaffolds
Rapid heteroatom transfer to arylmetals utilizing multifunctional reagent scaffolds
The direct transfer of primary amino and hydroxyl groups to arylmetals in a scalable and environmentally friendly fashion remains a formidable synthetic challenge. Here, it is demonstrated that bench-stable N–H and N–alkyl oxaziridines can be used as efficient multifunctional reagents, without deprotonation, for the direct primary amination and hydroxylation of (hetero)arylmetals.
- Hongyin Gao
- , Zhe Zhou
- & László Kürti
-
Commentary |
 One-world chemistry and systems thinking
One-world chemistry and systems thinking
The practice and overarching mission of chemistry need a major overhaul in order to be fit for purpose in the twenty-first century and beyond. The concept of 'one-world' chemistry takes a systems approach that brings together many factors, including ethics and sustainability, that are critical to the future role of chemistry.
- Stephen A. Matlin
- , Goverdhan Mehta
- & Alain Krief
-
News & Views |
 Redox flow batteries go organic
Redox flow batteries go organic
The use of renewable resources as providers to the electrical grid is hampered by the intermittent and irregular nature in which they generate energy. Electrical energy storage technology could provide a solution and now, by using an iterative design process, a promising anolyte for use in redox flow batteries has been developed.
- Wei Wang
- & Vince Sprenkle
-
Commentary |
 The role of chemistry in inventing a sustainable future
The role of chemistry in inventing a sustainable future
The Sustainable Development Goals adopted at a UN summit in September 2015 address many of the great challenges that our planet faces this century. Chemistry can make pivotal contributions to help realize these ambitious goals, but first it must undergo major changes in its priorities, approaches and practices.
- Stephen A. Matlin
- , Goverdhan Mehta
- & Alain Krief
-
Article |
 Chemoselective conversion of biologically sourced polyols into chiral synthons
Chemoselective conversion of biologically sourced polyols into chiral synthons
Biorenewable carbohydrate feedstocks can be efficiently converted into a diverse set of oxygen-functionalized chiral synthons using a combination of a tertiary silane and the catalyst B(C6F5)3. The deoxygenation mechanism involves cyclic intermediates, which provide a means of controlling chemo- and diastereoselectivity.
- Laura L. Adduci
- , Trandon A. Bender
- & Michel R. Gagné

 Deciding the future of adipic acid
Deciding the future of adipic acid Nitrate NO more
Nitrate NO more