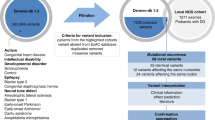
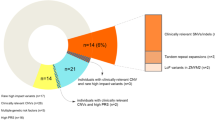
Abstract
Advances in genome sequencing technologies have begun to revolutionize neurogenetics, allowing the full spectrum of genetic variation to be better understood in relation to disease. Exome sequencing of hundreds to thousands of samples from patients with autism spectrum disorder, intellectual disability, epilepsy and schizophrenia provides strong evidence of the importance of de novo and gene-disruptive events. There are now several hundred new candidate genes and targeted resequencing technologies that allow screening of dozens of genes in tens of thousands of individuals with high specificity and sensitivity. The decision of which genes to pursue depends on many factors, including recurrence, previous evidence of overlap with pathogenic copy number variants, the position of the mutation in the protein, the mutational burden among healthy individuals and membership of the candidate gene in disease-implicated protein networks. We discuss these emerging criteria for gene prioritization and the potential impact on the field of neuroscience.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
Accessions
NCBI Reference Sequence
References
de Ligt, J. et al. Diagnostic exome sequencing in persons with severe intellectual disability. N. Engl. J. Med. 367, 1921–1929 (2012).
Rauch, A. et al. Range of genetic mutations associated with severe non-syndromic sporadic intellectual disability: an exome sequencing study. Lancet 380, 1674–1682 (2012).
O'Roak, B.J. et al. Sporadic autism exomes reveal a highly interconnected protein network of de novo mutations. Nature 485, 246–250 (2012).
Sanders, S.J. et al. De novo mutations revealed by whole-exome sequencing are strongly associated with autism. Nature 485, 237–241 (2012).
Iossifov, I. et al. De novo gene disruptions in children on the autistic spectrum. Neuron 74, 285–299 (2012).
Jiang, Y.-H. et al. Detection of clinically relevant genetic variants in autism spectrum disorder by whole-genome sequencing. Am. J. Hum. Genet. 93, 249–263 (2013).
Neale, B.M. et al. Patterns and rates of exonic de novo mutations in autism spectrum disorders. Nature 485, 242–245 (2012).
Gulsuner, S. et al. Spatial and temporal mapping of de novo mutations in schizophrenia to a fetal prefrontal cortical network. Cell 154, 518–529 (2013).
Xu, B. et al. De novo gene mutations highlight patterns of genetic and neural complexity in schizophrenia. Nat. Genet. 44, 1365–1369 (2012).
Fromer, M. et al. De novo mutations in schizophrenia implicate synaptic networks. Nature doi:10.1038/nature12929 (2014).
Allen, A.S. et al. De novo mutations in epileptic encephalopathies. Nature 501, 217–221 (2013).
Veltman, J.A. & Brunner, H.G. De novo mutations in human genetic disease. Nat. Rev. Genet. 13, 565–575 (2012).
Stefansson, H. et al. CNVs conferring risk of autism or schizophrenia affect cognition in controls. Nature 505, 361–366 (2014).
Kong, A. et al. Rate of de novo mutations and the importance of father's age to disease risk. Nature 488, 471–475 (2012).
Malaspina, D. et al. Advancing paternal age and the risk of schizophrenia. Arch. Gen. Psychiatry 58, 361–367 (2001).
Hultman, C.M., Sandin, S., Levine, S.Z., Lichtenstein, P. & Reichenberg, A. Advancing paternal age and risk of autism: new evidence from a population-based study and a meta-analysis of epidemiological studies. Mol. Psychiatry 16, 1203–1212 (2011).
McGrath, J.J. et al. A comprehensive assessment of parental age and psychiatric disorders. JAMA Psychiatry 71, 301–309 (2014).
Carvill, G.L. et al. Targeted resequencing in epileptic encephalopathies identifies de novo mutations in CHD2 and SYNGAP1. Nat. Genet. 45, 825–830 (2013).
Yang, Y. et al. Clinical whole-exome sequencing for the diagnosis of Mendelian disorders. N. Engl. J. Med. 369, 1502–1511 (2013).
O'Roak, B.J. et al. Multiplex targeted sequencing identifies recurrently mutated genes in autism spectrum disorders. Science 338, 1619–1622 (2012).
O'Roak, B.J. et al. Multiplex targeted sequencing identifies recurrently mutated genes in autism spectrum disorders. Science 338, 1619–1622 (2012).
O'Roak, B.J. et al. Exome sequencing in sporadic autism spectrum disorders identifies severe de novo mutations. Nat. Genet. 43, 585–589 (2011).
Cooper, G.M. et al. A copy number variation morbidity map of developmental delay. Nat. Genet. 43, 838–846 (2011).
Kaminsky, E.B. et al. An evidence-based approach to establish the functional and clinical significance of copy number variants in intellectual and developmental disabilities. Genet. Med. 13, 777–784 (2011).
Stefansson, H. et al. Large recurrent microdeletions associated with schizophrenia. Nature 455, 232–236 (2008).
Vulto-van Silfhout, A.T. et al. Clinical significance of de novo and inherited copy number variation. Hum. Mutat. 34, 1679–1687 (2013).
Møller, R.S. et al. Truncation of the Down syndrome candidate gene DYRK1A in two unrelated patients with microcephaly. Am. J. Hum. Genet. 1165–1170 (2008).
Huang, N., Lee, I., Marcotte, E.M. & Hurles, M.E. Characterising and predicting haploinsufficiency in the human genome. PLoS Genet. 6, e1001154 (2010).
van Bokhoven, H. & Brunner, H.G. Splitting p63. Am. J. Hum. Genet. 71, 1–13 (2002).
Bowen, M.E. et al. Loss-of-function mutations in PTPN11 cause metachondromatosis, but not Ollier disease or Maffucci syndrome. PLoS Genet. 7, e1002050 (2011).
Tartaglia, M. & Gelb, B. Noonan syndrome and related disorders. Annu. Rev. Genomics Hum. Genet. 6, 45–68 (2005).
Hoischen, A. et al. De novo mutations of SETBP1 cause Schinzel-Giedion syndrome. Nat. Genet. 42, 483–485 (2010).
Filges, I. et al. Reduced expression by SETBP1 haploinsufficiency causes developmental and expressive language delay indicating a phenotype distinct from Schinzel-Giedion syndrome. J. Med. Genet. 48, 117–122 (2011).
Marseglia, G. et al. 372 kb microdeletion in 18q12.3 causing SETBP1 haploinsufficiency associated with mild mental retardation and expressive speech impairment. Eur. J. Med. Genet. 55, 216–221 (2012).
Kamath, B.M. et al. NOTCH2 mutations in Alagille syndrome. J. Med. Genet. 49, 138–144 (2012).
Isidor, B. et al. Truncating mutations in the last exon of NOTCH2 cause a rare skeletal disorder with osteoporosis. Nat. Genet. 43, 306–308 (2011).
Simpson, M.A. et al. Mutations in NOTCH2 cause Hajdu-Cheney syndrome, a disorder of severe and progressive bone loss. Nat. Genet. 43, 303–305 (2011).
Kryukov, G.V., Pennacchio, L.A. & Sunyaev, S.R. Most rare missense alleles are deleterious in humans: implications for complex disease and association studies. Am. J. Hum. Genet. 80, 727–739 (2007).
Khurana, E. et al. Integrative annotation of variants from 1092 humans: application to cancer genomics. Science 342, 1235587 (2013).
Kircher, M. et al. A general framework for estimating the relative pathogenicity of human genetic variants. Nat. Genet. 46, 310–315 (2014).
Carter, H., Douville, C., Stenson, P.D., Cooper, D.N. & Karchin, R. Identifying Mendelian disease genes with the variant effect scoring tool. BMC Genomics 14 (suppl. 3), S3 (2013).
Gilman, S.R. et al. Rare de novo variants associated with autism implicate a large functional network of genes involved in formation and function of synapses. Neuron 70, 898–907 (2011).
Parikshak, N.N. et al. Integrative functional genomic analyses implicate specific molecular pathways and circuits in autism. Cell (in the press).
Helsmoortel, C. et al. A SWI/SNF-related autism syndrome caused by de novo mutations in ADNP. Nat. Genet. 46, 380–384 (2014).
Purcell, S.M. et al. A polygenic burden of rare disruptive mutations in schizophrenia. Nature 506, 185–190 (2014).
Willsey, A.J. et al. Co-expression networks implicate human mid-fetal deep cortical projection neurons in the pathogenesis of autism. Cell 155, 997–1007 (2013).
Ronan, J.L., Wu, W. & Crabtree, G.R. From neural development to cognition: unexpected roles for chromatin. Nat. Rev. Genet. 14, 347–359 (2013).
Santen, G.W.E. et al. Coffin-Siris syndrome and the BAF complex: genotype-phenotype study in 63 patients. Hum. Mutat. doi:10.1002/humu.22394 (2013).
Hoischen, A. et al. De novo nonsense mutations in ASXL1 cause Bohring-Opitz syndrome. Nat. Genet. 43, 729–731 (2011).
Gibson, W.T. et al. Mutations in EZH2 cause Weaver syndrome. Am. J. Hum. Genet. 90, 110–118 (2012).
Goriely, A. & Wilkie, A.O.M. Paternal age effect mutations and selfish spermatogonial selection: causes and consequences for human disease. Am. J. Hum. Genet. 90, 175–200 (2012).
Hudson, T.J. et al. International network of cancer genome projects. Nature 464, 993–998 (2010).
Schuurs-Hoeijmakers, J.H.M. et al. Recurrent de novo mutations in PACS1 cause defective cranial-neural-crest migration and define a recognizable intellectual-disability syndrome. Am. J. Hum. Genet. 91, 1122–1127 (2012).
Hoyer, J. et al. Haploinsufficiency of ARID1B, a member of the SWI/SNF-a chromatin-remodeling complex, is a frequent cause of intellectual disability. Am. J. Hum. Genet. 90, 565–572 (2012).
Santen, G.W.E. et al. Mutations in SWI/SNF chromatin remodeling complex gene ARID1B cause Coffin-Siris syndrome. Nat. Genet. 44, 379–380 (2012).
Girirajan, S. et al. Refinement and discovery of new hotspots of copy-number variation associated with autism spectrum disorder. Am. J. Hum. Genet. 92, 221–237 (2013).
Girirajan, S. et al. Phenotypic heterogeneity of genomic disorders and rare copy-number variants. N. Engl. J. Med. 367, 1321–1331 (2012).
Classen, C.F. et al. Dissecting the genotype in syndromic intellectual disability using whole exome sequencing in addition to genome-wide copy number analysis. Hum. Genet. 132, 825–841 (2013).
Zaidi, S. et al. De novo mutations in histone-modifying genes in congenital heart disease. Nature 498, 220–223 (2013).
Skarnes, W.C. et al. A conditional knockout resource for the genome-wide study of mouse gene function. Nature 474, 337–342 (2011).
Tweedie, S. et al. FlyBase: enhancing Drosophila Gene Ontology annotations. Nucleic Acids Res. 37, D555–D559 (2009).
Vulih-Shultzman, I. et al. Activity-dependent neuroprotective protein snippet NAP reduces tau hyperphosphorylation and enhances learning in a novel transgenic mouse model. J. Pharmacol. Exp. Ther. 323, 438–449 (2007).
van Bon, B.W.M. et al. Intragenic deletion in DYRK1A leads to mental retardation and primary microcephaly. Clin. Genet. 79, 296–299 (2011).
Fotaki, V. et al. Dyrk1A haploinsufficiency affects viability and causes developmental delay and abnormal brain morphology in mice. Mol. Cell. Biol. 22, 6636–6647 (2002).
Tejedor, F. et al. Minibrain: a new protein kinase family involved in postembryonic neurogenesis in Drosophila. Neuron 14, 287–301 (1995).
Kettleborough, R.N.W. et al. A systematic genome-wide analysis of zebrafish protein-coding gene function. Nature 496, 494–497 (2013).
Genovese, G. et al. Using population admixture to help complete maps of the human genome. Nat. Genet. 45, 406–414 (2013).
Sudmant, P.H. et al. Diversity of human copy number variation and multicopy genes. Science 330, 641–646 (2010).
Beyer, K. et al. New brain-specific beta-synuclein isoforms show expression ratio changes in Lewy body diseases. Neurogenetics 13, 61–72 (2012).
Karakoc, E. et al. Detection of structural variants and indels within exome data. Nat. Methods 9, 176–178 (2012).
Fromer, M. et al. Discovery and statistical genotyping of copy-number variation from whole-exome sequencing depth. Am. J. Hum. Genet. 91, 597–607 (2012).
Krumm, N. et al. Transmission disequilibrium of small CNVs in simplex autism. Am. J. Hum. Genet. 93, 595–606 (2013).
Lupski, J.R. Genetics. Genome mosaicism–one human, multiple genomes. Science 341, 358–359 (2013).
Greenman, C. et al. Patterns of somatic mutation in human cancer genomes. Nature 446, 153–158 (2007).
Alexandrov, L.B. et al. Signatures of mutational processes in human cancer. Nature 500, 415–421 (2013).
Hanahan, D. & Weinberg, R.A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Banka, S. et al. MLL2 mosaic mutations and intragenic deletion-duplications in patients with Kabuki syndrome. Clin. Genet. 83, 467–471 (2013).
Huisman, S.A., Redeker, E.J.W., Maas, S.M., Mannens, M.M. & Hennekam, R.C.M. High rate of mosaicism in individuals with Cornelia de Lange syndrome. J. Med. Genet. 50, 339–344 (2013).
Rodríguez-Santiago, B. et al. Mosaic uniparental disomies and aneuploidies as large structural variants of the human genome. Am. J. Hum. Genet. 87, 129–138 (2010).
Hiatt, J.B., Pritchard, C.C., Salipante, S.J., O'Roak, B.J. & Shendure, J. Single molecule molecular inversion probes for targeted, high-accuracy detection of low-frequency variation. Genome Res. 23, 843–854 (2013).
Shiroguchi, K., Jia, T.Z., Sims, P.A . & Xie, X.S. Digital RNA sequencing minimizes sequence-dependent bias and amplification noise with optimized single-molecule barcodes. Proc. Natl. Acad. Sci. USA 109, 1347–1352 (2012).
Klei, L. et al. Common genetic variants, acting additively, are a major source of risk for autism. Mol. Autism 3, 9 (2012).
Lee, S.H. et al. Genetic relationship between five psychiatric disorders estimated from genome-wide SNPs. Nat. Genet. 45, 984–994 (2013).
Yu, T.W. et al. Using whole-exome sequencing to identify inherited causes of autism. Neuron 77, 259–273 (2013).
Morrow, E.M. et al. Identifying autism loci and genes by tracing recent shared ancestry. Science 321, 218–223 (2008).
Najmabadi, H. et al. Deep sequencing reveals 50 novel genes for recessive cognitive disorders. Nature 478, 57–63 (2011).
He, X. et al. Integrated model of de novo and inherited genetic variants yields greater power to identify risk genes. PLoS Genet. 9, e1003671 (2013).
Levy, D. et al. Rare de novo and transmitted copy-number variation in autistic spectrum disorders. Neuron 70, 886–897 (2011).
Jacquemont, S. et al. A higher mutational burden in females supports a “female protective model” in neurodevelopmental disorders. Am. J. Hum. Genet. 94, 415–425 (2014).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
van Bon, B.W.M. et al. The 2q23.1 microdeletion syndrome: clinical and behavioural phenotype. Eur. J. Hum. Genet. 18, 163–170 (2010).
Talkowski, M.E. et al. Assessment of 2q23.1 microdeletion syndrome implicates MBD5 as a single causal locus of intellectual disability, epilepsy, and autism spectrum disorder. Am. J. Hum. Genet. 89, 551–563 (2011).
Makishima, H. et al. Somatic SETBP1 mutations in myeloid malignancies. Nat. Genet. 45, 942–946 (2013).
Piazza, R. et al. Recurrent SETBP1 mutations in atypical chronic myeloid leukemia. Nat. Genet. 45, 18–24 (2013).
Acknowledgements
We are grateful to T. Brown and C. Gilissen for assistance during manuscript preparation, F. Kooy for early preprint access and H.G. Brunner for sharing patient photographs used in Figure 3. A.H. is supported by a ZonMW grant (916–12–095); E.E.E. is supported by a US National Institute of Mental Health grant (1R01MH101221–01) and is an investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
E.E.E. is on the scientific advisory board of DNAnexus, Inc. and was a scientific advisory board member of Pacific Biosciences, Inc. (2009–2013) and SynapDx Corp. (2011–2013).
Supplementary information
Supplementary Text and Figures
Supplementary Table 1 (PDF 84 kb)
Glossary
- Exome
-
The exome is the part of a genome that encodes proteins, approximately 1% of the human genome.
- Trio family study
-
A trio family study is an analysis of probands and both of their parents. Sequencing-based trio studies often focus on de novo mutations that are present in the proband's genome, but are not detected in the genomes of his or her parents.
- Proband
-
A proband is an individual being studied or reported on. The term is often used to refer to an individual affected with a disease or disorder, as distinct from their unaffected relatives.
- de novo mutation (DNM)
-
A de novo mutation (DNM) is a mutation that is part of an individual's genome that is not detected in the genome of either parent (although it may have arisen from a mutation in the parental germline). With the exception of de novo mutations in monozygotic twins, or those shared by siblings as a result of germline mosaicism, most new mutations are not shared by relatives and do not contribute to heritability estimates.
- Rare variant
-
Rare variant describes variants that are private to individuals and families. In some usage, the term rare variant is used more expansively to include all variants that are not common.
- Copy number variation (CNV)
-
A copy number variation (CNV) is a type of submicroscopic genetic variation involving the deletion or duplication of a genomic region. Although CNVs can involve genomic segments as small as a kilobase or as large as several megabases, most CNVs detected are relatively large (100 kilobases or larger) because of the resolution of genotyping arrays; in the future, sequencing-based studies may analyze many smaller CNVs.
- Mendelian disease
-
A Mendelian disease describes a single gene disorder that is caused by the presence of one (dominant) or two (recessive) alleles.
- Next generation sequencing (NGS)
-
Next generation sequencing (NGS) refers to a set of technologies that sequence DNA in massively parallel ways; for example, by optically detecting the incorporation of specific bases into millions of different DNA molecules, spatially segregated on an imageable glass surface, at the same time.
- Candidate gene
-
A candidate gene is a pre-specified gene of potential interest. Candidate gene studies are often distinguished from unbiased genome-wide studies that analyze variation in all or most genes simultaneously.
- Locus
-
A locus is a place on a chromosome. A locus may contain one gene, multiple genes or no genes at all.
- Resequencing
-
Resequencing is the activity of sequencing a gene or genomic segment that has already been sequenced in other members of that species; for example, to identify genetic variations within the species.
- Case-control study
-
A case-control study is a study design that compares the distribution of a genetic or other variable between individuals affected with a disease (cases) and unaffected individuals (controls).
- Single-nucleotide variant (SNV)
-
A single-nucleotide variant (SNV) is a DNA sequence variation occurring when a single nucleotide differs between members of a species. The term SNV is often used to describe sequence variants that are not common, or whose allele frequency is not known.
- Genetic background
-
Genetic background refers to the genotype of all genes that may modify the expression or presentation of a phenotype related to a gene of interest.
- Complex disease
-
A complex disease describes a disorder caused by many contributing factors, both genetic and non-genetic, and does not display a simple pattern of inheritance.
Rights and permissions
About this article
Cite this article
Hoischen, A., Krumm, N. & Eichler, E. Prioritization of neurodevelopmental disease genes by discovery of new mutations. Nat Neurosci 17, 764–772 (2014). https://doi.org/10.1038/nn.3703
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3703
This article is cited by
-
Inversed Effects of Nav1.2 Deficiency at Medial Prefrontal Cortex and Ventral Tegmental Area for Prepulse Inhibition in Acoustic Startle Response
Molecular Neurobiology (2024)
-
Targeted sequencing and integrative analysis to prioritize candidate genes in neurodevelopmental disorders
Molecular Neurobiology (2021)
-
mTADA is a framework for identifying risk genes from de novo mutations in multiple traits
Nature Communications (2020)
-
Emerging themes in cohesin cancer biology
Nature Reviews Cancer (2020)
-
Pogz deficiency leads to transcription dysregulation and impaired cerebellar activity underlying autism-like behavior in mice
Nature Communications (2020)