Abstract
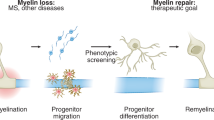
Multiple sclerosis involves an aberrant autoimmune response and progressive failure of remyelination in the central nervous system. Prevention of neural degeneration and subsequent disability requires remyelination through the generation of new oligodendrocytes, but current treatments exclusively target the immune system. Oligodendrocyte progenitor cells are stem cells in the central nervous system and the principal source of myelinating oligodendrocytes1. These cells are abundant in demyelinated regions of patients with multiple sclerosis, yet fail to differentiate, thereby representing a cellular target for pharmacological intervention2. To discover therapeutic compounds for enhancing myelination from endogenous oligodendrocyte progenitor cells, we screened a library of bioactive small molecules on mouse pluripotent epiblast stem-cell-derived oligodendrocyte progenitor cells3,4,5. Here we show seven drugs function at nanomolar doses selectively to enhance the generation of mature oligodendrocytes from progenitor cells in vitro. Two drugs, miconazole and clobetasol, are effective in promoting precocious myelination in organotypic cerebellar slice cultures, and in vivo in early postnatal mouse pups. Systemic delivery of each of the two drugs significantly increases the number of new oligodendrocytes and enhances remyelination in a lysolecithin-induced mouse model of focal demyelination. Administering each of the two drugs at the peak of disease in an experimental autoimmune encephalomyelitis mouse model of chronic progressive multiple sclerosis results in striking reversal of disease severity. Immune response assays show that miconazole functions directly as a remyelinating drug with no effect on the immune system, whereas clobetasol is a potent immunosuppressant as well as a remyelinating agent. Mechanistic studies show that miconazole and clobetasol function in oligodendrocyte progenitor cells through mitogen-activated protein kinase and glucocorticoid receptor signalling, respectively. Furthermore, both drugs enhance the generation of human oligodendrocytes from human oligodendrocyte progenitor cells in vitro. Collectively, our results provide a rationale for testing miconazole and clobetasol, or structurally modified derivatives, to enhance remyelination in patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
Primary accessions
Gene Expression Omnibus
Data deposits
RNAseq datasets have been deposited in GEO under accession number GSE63804. Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests. Readers are welcome to comment on the online version of the paper. Correspondence and requests for materials should be addressed to P.J.T. (paul.tesar@case.edu) or R.H.M. (rhm3@gwu.edu).
References
Goldman, S. A., Nedergaard, M. & Windrem, M. S. Glial progenitor cell-based treatment and modeling of neurological disease. Science 338, 491–495 (2012)
Chang, A., Tourtellotte, W. W., Rudick, R. & Trapp, B. D. Premyelinating oligodendrocytes in chronic lesions of multiple sclerosis. N. Engl. J. Med. 346, 165–173 (2002)
Najm, F. J. et al. Rapid and robust generation of functional oligodendrocyte progenitor cells from epiblast stem cells. Nature Methods 8, 957–962 (2011)
Tesar, P. J. et al. New cell lines from mouse epiblast share defining features with human embryonic stem cells. Nature 448, 196–199 (2007)
Brons, I. G. et al. Derivation of pluripotent epiblast stem cells from mammalian embryos. Nature 448, 191–195 (2007)
Mi, S. et al. Promotion of central nervous system remyelination by induced differentiation of oligodendrocyte precursor cells. Ann. Neurol. 65, 304–315 (2009)
Bai, L. et al. Hepatocyte growth factor mediates mesenchymal stem cell-induced recovery in multiple sclerosis models. Nature Neurosci. 15, 862–870 (2012)
Deshmukh, V. A. et al. A regenerative approach to the treatment of multiple sclerosis. Nature 502, 327–332 (2013)
Mei, F. et al. Micropillar arrays as a high-throughput screening platform for therapeutics in multiple sclerosis. Nature Med. 20, 954–960 (2014)
Fancy, S. P. et al. Overcoming remyelination failure in multiple sclerosis and other myelin disorders. Exp. Neurol. 225, 18–23 (2010)
Franklin, R. J. & Ffrench-Constant, C. Remyelination in the CNS: from biology to therapy. Nature Rev. Neurosci. 9, 839–855 (2008)
Dubois-Dalcq, M., Ffrench-Constant, C. & Franklin, R. J. Enhancing central nervous system remyelination in multiple sclerosis. Neuron 48, 9–12 (2005)
Barres, B. A., Lazar, M. A. & Raff, M. C. A novel role for thyroid hormone, glucocorticoids and retinoic acid in timing oligodendrocyte development. Development 120, 1097–1108 (1994)
Woodruff, R. H. & Franklin, R. J. The expression of myelin basic protein exon 1 and exon 2 containing transcripts during myelination of the neonatal rat spinal cord–an in situ hybridization study. J. Neurocytol. 27, 683–693 (1998)
Woodruff, R. H. & Franklin, R. J. The expression of myelin protein mRNAs during remyelination of lysolecithin-induced demyelination. Neuropathol. Appl. Neurobiol. 25, 226–235 (1999)
Jeffery, N. D. & Blakemore, W. F. Remyelination of mouse spinal cord axons demyelinated by local injection of lysolecithin. J. Neurocytol. 24, 775–781 (1995)
Kumar, S., Cole, R., Chiappelli, F. & de Vellis, J. Differential regulation of oligodendrocyte markers by glucocorticoids: post-transcriptional regulation of both proteolipid protein and myelin basic protein and transcriptional regulation of glycerol phosphate dehydrogenase. Proc. Natl Acad. Sci. USA 86, 6807–6811 (1989)
Morisaki, S. et al. Endogenous glucocorticoids improve myelination via Schwann cells after peripheral nerve injury: an in vivo study using a crush injury model. Glia 58, 954–963 (2010)
Ishii, A., Fyffe-Maricich, S. L., Furusho, M., Miller, R. H. & Bansal, R. ERK1/ERK2 MAPK signaling is required to increase myelin thickness independent of oligodendrocyte differentiation and initiation of myelination. J. Neurosci. 32, 8855–8864 (2012)
Fyffe-Maricich, S. L., Schott, A., Karl, M., Krasno, J. & Miller, R. H. Signaling through ERK1/2 controls myelin thickness during myelin repair in the adult central nervous system. J. Neurosci. 33, 18402–18408 (2013)
Hu, B. Y., Du, Z. W. & Zhang, S. C. Differentiation of human oligodendrocytes from pluripotent stem cells. Nature Protocols 4, 1614–1622 (2009)
Wang, S. et al. Human iPSC-derived oligodendrocyte progenitor cells can myelinate and rescue a mouse model of congenital hypomyelination. Cell Stem Cell 12, 252–264 (2013)
Najm, F. J. et al. Isolation of epiblast stem cells from preimplantation mouse embryos. Cell Stem Cell 8, 318–325 (2011)
Najm, F. J. et al. Transcription factor-mediated reprogramming of fibroblasts to expandable, myelinogenic oligodendrocyte progenitor cells. Nature Biotechnol. 31, 426–433 (2013)
Wisniewski, J. R., Nagaraj, N., Zougman, A., Gnad, F. & Mann, M. Brain phosphoproteome obtained by a FASP-based method reveals plasma membrane protein topology. J. Proteome Res. 9, 3280–3289 (2010)
Factor, D. C. et al. Epigenomic comparison reveals activation of “seed” enhancers during transition from naive to primed pluripotency. Cell Stem Cell 14, 854–863 (2014)
Cahoy, J. D. et al. A transcriptome database for astrocytes, neurons, and oligodendrocytes: a new resource for understanding brain development and function. J. Neurosci. 28, 264–278 (2008)
Trapnell, C., Pachter, L. & Salzberg, S. L. TopHat: discovering splice junctions with RNA-Seq. Bioinformatics 25, 1105–1111 (2009)
Trapnell, C. et al. Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nature Biotechnol. 28, 511–515 (2010)
Trapnell, C. et al. Differential analysis of gene regulation at transcript resolution with RNA-seq. Nature Biotechnol. 31, 46–53 (2013)
Acknowledgements
This research was supported by grants from the US NIH NS085246 (P.J.T. and R.H.M.), NS030800 (R.H.M.), and NS026543 (S.D.M.); New York Stem Cell Foundation (P.J.T.); Myelin Repair Foundation (P.J.T., R.H.M., and S.D.M.); Mt. Sinai Health Care Foundation (P.J.T.); and NIH predoctoral training grants T32GM008056 (R.T.K.) and F30CA183510 (T.E.M). Additional support was provided by the Cytometry & Imaging Microscopy, Proteomics, and Genomics core facilities of the Case Comprehensive Cancer Center (P30CA043703), the CWRU Council to Advance Human Health, and philanthropic support from the Goodman, Long, and Geller families. P.J.T. is a New York Stem Cell Foundation–Robertson Investigator. We are grateful to M. Hitomi, W. Harte, D. Adams, W. Seibel, M. Haag, P. Scacheri, J. Wanta, C. Fang, H. Olsen, T. LaFramboise, J. Song, F. Van den Akker, and M. Shoham for technical assistance and discussion, A. Lager and M. Elitt for comments on the manuscript, B. Trapp for the PLP1 antibody, and B. Barres and B. Zuchero for RNA from in vivo isolated OPCs.
Author information
Authors and Affiliations
Contributions
F.J.N., R.H.M., and P.J.T. designed the overall screening strategy; F.J.N. and P.J.T. generated mouse OPCs and performed the primary screen; F.J.N., K.Q., H.T., and R.P. designed analysis scripts for the primary screen; K.R.B., M.S., M.B.B., and A.J. performed 1,536-well external validation of primary screen; M.M., F.J.N., A.Z., E.S., C.K., and A.S. performed organotypic slice culture and in vivo assays; F.J.N. and K.Q. designed analysis scripts for the slice cultures; A.P.R., J.R.P., and S.D.M. designed and performed PLP EAE and immune cell experiments; R.T.K., Z.S.N., and F.J.N. generated and tested human OPCs; D.C.F., F.J.N., T.E.M., and P.J.T. generated and analysed RNAseq data; D.M.S., F.J.N., and P.J.T. generated and analysed the proteomics data; F.J.N., M.M., R.H.M., and P.J.T. analysed all of the data and wrote the paper. All authors edited and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Performance of the primary screen.
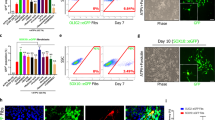
a, Representative flow cytometry plots showing co-expression of NG2 and CD140a in both batches of EpiSC-derived OPCs used for this study. The batches of EpiSC-derived OPCs were sorted to purity (circled areas of plots) before use in this study. b, RNaseq expression heat map showing downregulation of pluripotent stem cell transcripts and upregulation of OPC transcripts when EpiSCs were differentiated into OPCs. Fragments per kilobase exon per million reads (FPKM) for each transcript are shown compared with in vivo isolated mouse OPCs. c, Quantification of DMSO (v/v) tolerance of EpiSC-derived OPCs in 96-well plates shown as mean ± s.e.m. For reference, 0.05% (v/v) DMSO was used as vehicle for all in vitro experiments in this study; n = 16 wells per group with >690 cells scored per well. d, Quantification of cell viability of thyroid hormone (positive control) and DMSO vehicle treatments per well across all ten assay plates shown as mean ± s.e.m.; n = 80 wells per group with >6,800 cells scored per well. e, Signal to background (S/B) mean values with standard deviation (s.d.) of controls from the entire screen; n = 80 wells per group. f, Raw data of MBP process length from the primary screen for thyroid hormone treatment and DMSO vehicle across all plates shown as mean ± s.d.; n = 8 wells per group with >6,800 cells scored per well. g, Raw data of MBP process intensity from the primary screen for thyroid hormone treatment and DMSO vehicle shown as mean ± s.d.; n = 8 wells per group with >6,800 cells scored per well.
Extended Data Figure 2 Drug hit ranking and validation.
a, Chart ranking the 22 primary drug hits (single dose rank) into four tiers on the basis of calculation of EC50 to induce PLP1+ oligodendrocytes from OPCs and the concentration at which 50% of the cells were lost (50% Tox) calculated from a seven-point dose treatment; n = 4 wells per dose per drug using independently sourced drug and separate OPC batch from the primary screen. Tiers ranged from the most potent and least toxic effectors to the least potent and most toxic: tier a (green), tier b (grey), tier c (orange), and tier d (red). The 1,536-well format external validation of 14 out of 16 tested hits is also shown. Drugs were further ranked into groups of high (green), medium (grey), and low (orange) on the basis of their ability to increase MBP+ axonal ensheathment in mouse cerebellar brain slices relative to vehicle (DMSO)-treated controls as measured by HCA. NT, not tested. b, External validation whole 1,536-well images of MBP+ (green) oligodendrocytes generated from OPCs after 72 h of treatment. GE InCell HCA is shown with processes traced in yellow and nuclei in blue.
Extended Data Figure 3 Primary screen structure–activity analysis.
Chemoinformatic identification of two substructures consistently enriched in high-performing drugs in the OPC assay. Numerical activity rank in the primary screen is indicated with the top 22 shown in green, 23–50 shown in grey, and >51 in red. a, 1,3-Diazoles, mono-substituted at the 1-position showed consistent activity on OPCs. b, c, 1,3-Diazoles, poly-substituted at two or more of the R groups (b), or 1,2,4-triazoles, mono-substituted at the 1-position (c) showed no activity on OPCs. d, The sterane base structure showed enrichment in the top performing hits.
Extended Data Figure 4 Histological assessment of remyelination in the LPC-induced model of demyelination.
a, Representative electron micrographs showing remyelinated axons within lesions of miconazole treated mice at 8 d.p.l. Scale bar, 2 μm. b, Histological sections stained with toluidine blue showing the extent of remyelination in the lesions of treated animals at 12 d.p.l. Normal uninjured myelin appears to the left of the black dashed line demarcating the definitive lesion edge. Scale bar, 20 μm.
Extended Data Figure 5 Miconazole and clobetasol enhance myelination in vivo.
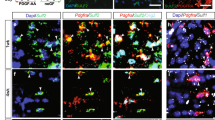
a, b, Representative immunohistochemical images of the lateral corpus callosum (CC) of postnatal day 6 mouse pups that had been injected intraperitoneally daily for 4 days previously starting on postnatal day 2 with vehicle, clobetasol (2 mg/kg), or miconazole (10 mg/kg). CC1 (red) marks newly generated oligodendrocytes (a) and MBP (green) shows the extent of developmental myelination (b). Clobetasol and miconazole treatment each induce a marked increase in the number of CC1-positive oligodendrocytes in the lateral corpus callosum (a) and a significant increase in the length of the corpus callosum covered with aligned MBP+ fibres (b). Scale bar, 200 μm. Two-tailed t-test, *P ≤ 0.05 and **P ≤ 0.01. Str, striatum. All graphs are presented as mean ± s.e.m.
Extended Data Figure 6 RNaseq time course of drug-treated OPCs.
a, Volcano plots of all genes from OPCs treated with clobetasol or miconazole relative to vehicle control, with differentially expressed genes highlighted (red). Significance (measured as −log10[q value]) is plotted in relation to expression change (log2[treatment/vehicle]). Time course was after 2, 6, and 12 h of drug treatment. b, Venn diagrams depicting the overlap of genes differentially expressed at any time point and increased in treatments versus vehicle (left), as well as those decreased in treatments versus vehicle (right). c, Significant canonical pathways perturbed by each drug treatment according to Ingenuity Pathway Analysis.
Extended Data Figure 7 Global phosphoproteomic analysis of miconazole-treated OPCs.
a, b, OPCs treated with miconazole for (a) 1 h or (b) 5 h followed by global phosphoproteomic analysis. Proteins highlighted in green were observed to have a twofold or greater increase in phosphorylation whereas those highlighted in red were observed to have a twofold or greater decrease in phosphorylation compared with time-point-matched vehicle-treated controls. Proteins highlighted in grey were detected in the analysis but were not changed compared with vehicle control. See Supplementary Table 3 for the full phosphoproteomic data set. c, Quantification of the percentage of MBP+ oligodendrocytes differentiated from mouse OPCs after 72 h of treatment with DMSO, miconazole (1 μM), or voriconazole (seven doses, 0.01–6.7 μM); n = 6 wells per condition with >6,000 cells scored per well. Graph presented as mean ± s.e.m. The chemical structure of voriconazole is shown.
Extended Data Figure 8 Miconazole and clobetasol enhance human OPC differentiation.
a, Representative phase contrast image of a hESC colony cultured on matrigel. b, Representative phase contrast image of hESC-derived OPCs. c, hESC-derived OPCs stain positive for Sox10. d, e, Representative images of hESC-derived OPCs (d) and hiPSC-derived OPCs (e) treated with vehicle (DMSO), miconazole (1 μM), or clobetasol (5 μM) for 21 days stained for MBP (red). f, g, HCA of hESC-derived (f) and hiPSC-derived (g) OPCs differentiated in the presence of drugs or vehicle over 21 days; n = 3–5 wells per condition with >120 cells scored per well. Graphs presented as mean ± s.e.m. Scale bars, 100 μm.
Extended Data Figure 9 Effects of miconazole and clobetasol on immune cell survival and function.
a–d, Quantification of cell proliferation (a, c) and differentiation (b, d) of naive CD4+ T cells from unprimed SJL/J mice after activation with plate-bound anti-CD3 under Th1 (a, b) or Th17 (c, d) cell driving conditions. e–j, Ex vivo recall assays quantifying cell proliferation (ΔCPM) (e, h), with IFN-γ (f, i) and IL-17 (g, j) cytokine production from lymphocytes of mice primed with PLP139–151 (e–g) or MOG35–55 (h–j). Cultures were treated with vehicle (DMSO), benztropine, clobetasol, or miconazole (10−9–10−5 M) and analysed after 4 days. Four independent replicates are shown for each assay.
Extended Data Figure 10 Histological improvements in MOG35–55EAE spinal cords after treatment with miconazole or clobetasol.
a, b, Representative images of luxol fast blue (LFB) staining (a) demonstrated a clear decrease in areas of white matter disruption in the spinal cords of drug-treated animals which coincides with increased MBP staining (b). c, IBA1 staining showed a small reduction of immune cell infiltration into the lesion areas, especially in clobetasol-treated animals, but not an abrogation. d, e, Representative images stained with toluidine blue (d) and electron micrographs (e) revealed a reduction in the areas of demyelination in drug-treated animals. Lesioned areas are outlined with black dotted lines. Insets in toluidine blue staining show higher magnification of myelination in the corresponding spinal cords. Scale bars, 100 μm (a–c, d) and 2 μm (e).
Supplementary information
Supplementary Information
This file contains Supplementary Table 1 and Supplementary Figures 1-2 (full scans of western blots presented in the Letter). (PDF 695 kb)
Supplementary Table 2
This table shows global kinase profiling. Results of the 414 kinase assays tested are listed in the first three tabs for each of the Z’LYTE, Adapta, and Lantha assays, respectively. (XLSX 83 kb)
Supplementary Table 3
This table shows global phosphoproteomics. Tab 1 is the raw data of all detected phosphorylated peptides after one and five hour treatments of DMSO, clobetasol, and miconazole of OPCs. Tab 2 contains peptides that met the two-fold change cutoff for each drug relative to the DMSO value at the same time point. Tab 3 shows IPA analysis of miconazole treatment after one and five hour treatments. (XLSX 563 kb)
Rights and permissions
About this article
Cite this article
Najm, F., Madhavan, M., Zaremba, A. et al. Drug-based modulation of endogenous stem cells promotes functional remyelination in vivo. Nature 522, 216–220 (2015). https://doi.org/10.1038/nature14335
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature14335
This article is cited by
-
Artificial axons as a biomimetic 3D myelination platform for the discovery and validation of promyelinating compounds
Scientific Reports (2023)
-
Inflammation in multiple sclerosis: consequences for remyelination and disease progression
Nature Reviews Neurology (2023)
-
An in vivo accelerated developmental myelination model for testing promyelinating therapeutics
BMC Neuroscience (2022)
-
Higher levels of myelin are associated with higher resistance against tau pathology in Alzheimer’s disease
Alzheimer's Research & Therapy (2022)
-
Thinking outside the box: non-canonical targets in multiple sclerosis
Nature Reviews Drug Discovery (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.