Abstract
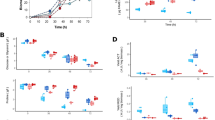
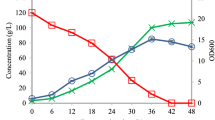
Secondary metabolite production by Streptomyces is often highly sensitive to oxygen supply, which can be limiting in large-scale fermentations. In an attempt to improve oxygen utilization by the cells, we expressed a heterologous bacterial hemoglobin gene in Streptomyces coelicolor and Streptomyces lividans. Hemoglobin expression was demonstrated by immunoblot analysis and carbon monoxide binding activity. In batch fermentations run under reduced aeration, the expression of hemoglobin in S. coelicolor resulted in a ten-fold increase in specific yields of the aromatic polyketide, actinorhodin. Actinorhodin yields were also much less sensitive to aeration conditions in the hemoglobin-expressing strain. In addition, hemoglobin-expressing S. lividans cells grown under reduced aeration had higher final cell densities and exhibited greater oxygen consumption rates than non-expressing cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Atkinson, B. and Mavituna, B. 1983. Biochemical Engineering and Biotechnology Handbook. Stockton Press, New York, NY.
Buckland, B. 1985. Fermentation exhaust gas analysis using mass spectrometry. Bio/Technology 3: 982–988.
Yegneswaran, P.K. and Gray, M.R. 1988. Effects of reduced oxygen on growth and antibiotic production in Streptomyces clavuligerus. Biotechnol. Lett. 10: 479–484.
Adlercreutz, P. and Mattiasson, B. 1982. Oxygen supply to immobilized cells. Eur. J. Appl. Microbiol. Biotechnol. 16: 165–170.
Khosla, C. and Bailey, J.E. 1988. Heterologous expression of a bacterial haemoglobin improves the growth properties of recombinant Escherichia coli. Nature 331: 633–635.
Orii, Y. and Webster, D.A. 1986. Photodissociation of oxygenated cytochrome o(s) (Vitreoscilla) and kinetic studies of reassociation. J. Biol. Chem. 261: 3544–3547.
Boreman, S. and Webster, D.A. 1982. Control of heme content in Vitreoscilla by oxygen. J. Gen. Appl. Microbiol. 28 35–43.
Wakabayashi, S., Matsubara, H. and Webster, D.A. 1986. Primary sequence of a dimeric bacterial hemoglobin from Vitreoscilla. Nature 322: 481–483.
Khosla, C. and Bailey, J.E. 1988. The Vitreoscilla hemoglobin gene: Molecular cloning, nucleotide sequence and genetic expression in Escherichia coli. Mol. Gen. Genet. 214: 158–161.
Khosla, C., Curtis, J.E., DeModena, J., Rinas, U. and Bailey, J.E. 1990. Expression of intracellular hemoglobin improves protein synthesis in oxygen-limited Escherichia coli. Bio/Technology 8: 849–853.
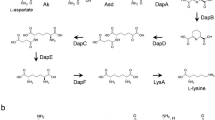
Cole, S.P., Rudd, B.A.M., Hopwood, D.A., Chang, C. and Floss, H.G. 1987. Biosynthesis of the antibiotic actinorhodin: Analysis of blocked mutants of Streptomyces coelicolor. J. Antibiotics. 40: 340–347.
Khosla, C. and Bailey, J.E. 1989. Evidence for partial export of Vitreoscilla hemoglobin into the periplasmic space in Escherichia coli. J. Mol. Biol. 210 79–89.
Kieser, T. and Melton, R.E. 1988. Plasmid pIJ699, a multi-copy positive-selection vector for Streptomyces. Gene 65 83–91.
Maniatis, T., Fritsch, E.F. and Sambrook, J. 1989. Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY.
Hobbs, G., Frazer, C.M., Gardner, C.J., Cullum, J.A. and Oliver, S.G. 1989. Dispersed growth of Streptomyces in liquid culture. Appl. Microbiol. Biotechnol. 31: 272–277.
Hopwood, D.A., Bibb, M.J., Chater, K.F., Kieser, T., Bruton, C.J., Kieser, H.M., Lydiate, D.J., Smith, C.P., Ward, J.M. and Schrempf, H. 1985. Genetic Manipulation of Streptomyces: A Laboratory Manual. John Innes Foundation, Norwich, UK.
Ausubel, F.M., Brent, R., Kingston, R.E., Moore, D.D., Seidman, J.G., Smith, J.A. and Struhl, K. 1987. Current Protocols in Molecular Biology. Current Protocols, U.S.A.
Webster, D.A. and Lu, C.Y. 1974. Reduced nicotinamide adenine dinucleotide cytochrome o reductase associated with cytochrome o purified from Vitreoscilla. J. Biol. Chem. 249: 4257–4260.
Bradford, M.M. 1976. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principal of protein dye binding. Anal. Biochem. 72: 248–254.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Magnolo, S., Leenutaphong, D., DeModena, J. et al. Actinorhodin Production By Streptomyces coelicolor and Growth of Streptomyces lividans Are Improved by the Expression of a Bacterial Hemoglobin. Nat Biotechnol 9, 473–476 (1991). https://doi.org/10.1038/nbt0591-473
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0591-473
This article is cited by
-
Vitreoscilla hemoglobin enhances the catalytic performance of industrial oxidases in vitro
Applied Microbiology and Biotechnology (2022)
-
Antimicrobial activities of actinomycetes isolated from unexplored regions of Sundarbans mangrove ecosystem
BMC Microbiology (2015)
-
Development of Intergeneric Conjugal Gene Transfer System in Streptomyces diastatochromogenes 1628 and Its Application for Improvement of Toyocamycin Production
Current Microbiology (2014)