Abstract
Milk-derived exosomes were identified as a novel mechanism of mother-to-child transmission of regulatory molecules, but their functions in intestinal tissues of neonates are not well-studied. Here, we characterized potential roles of porcine milk-derived exosomes in the intestinal tract. In vitro, treatment with milk-derived exosomes (27 ± 3 ng and 55 ± 5 ng total RNA) significantly promoted IPEC-J2 cell proliferation by MTT, CCK8, EdU fluorescence and EdU flow cytometry assays. The qRT-PCR and Western blot analyses indicated milk-derived exosomes (0.27 ± 0.03 μg total RNA) significantly promoted expression of CDX2, IGF-1R and PCNA and inhibited p53 gene expression involved in intestinal proliferation. Additionally, six detected miRNAs were significantly increased in IPEC-J2 cell, while FAS and SERPINE were significantly down-regulated relative to that in control. In vivo, treated groups (0.125 μg and 0.25 μg total RNA) significantly raised mice’ villus height, crypt depth and ratio of villus length to crypt depth of intestinal tissues, significantly increased CDX2, PCNA and IGF-1R’ expression and significantly inhibited p53′ expression. Our study demonstrated that milk-derived exosomes can facilitate intestinal cell proliferation and intestinal tract development, thus giving a new insight for milk nutrition and newborn development and health.
Similar content being viewed by others
Introduction
The development of the gastrointestinal tract (GIT) progresses through the prenatal and postnatal periods. During postnatal development, the GIT undergoes profound growth, morphological changes and functional maturation1. A close relationship exists between the degree of maturation and absorptive functions of the intestine. In the neonatal intestine, nutrient transport occurs along the whole crypt–villus axis, whereas in the adult intestine absorption of nutrients is shifted to the upper part of the villi2. Therefore, in mammalian nutrition, the GIT is responsible for the first physiological step of bringing nutrients to the body’s cells and plays a crucial role in the regulation of the development and health of infants3. The unique composition of breast milk may provide factors that stimulate developmental changes of the neonatal intestine that occur following birth4.
After birth, the neonatal GIT is stimulated chiefly by enteral nutritive and non-nutritive (biologically active) substances from colostrum and milk. Nutrition is a critical determinant in the functional growth and maturation of the GIT5. Thus, malnutrition in the postnatal period may restrict the morphologic and biochemical development of the GIT6. In milk, regulation of proteins, lipids and nucleotide synthesis to orchestrate anabolism, cell growth and proliferation occurs by activating the mTORC1 signaling pathway7. Hormones and growth-promoting peptides in milk, such as insulin, cortisol, epidermal growth factor (EGF) and insulin-like growth factor I (IGF-I), have been found to play a role in postnatal GIT development in newborns1. Differences in colostrum and milk composition between species and responses to milk-borne bioactive components may be expected due to ontogenic development of tissues and organs8.
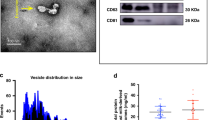
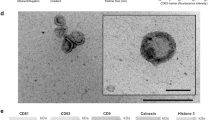
Exosomes are nanosized endosome-derived membrane vesicles (40–100 nm in diameter), which form from inward budding of early endosomes to become multivesicular endosomes (MVB) containing intralumenal vesicles9. Exosomes contain mRNA, microRNA (miRNA), DNA, proteins and lipids that can be transferred to cells to confer new functions or cell signaling events10,11,12, which are involved in cell-cell communication via the functionally-active cargo13. Exosomes are present in breast milk and are packed with abundant immune-related proteins (such as MHC class II, CD86 and the tetraspanin proteins, CD63 and CD81)10, as well as miRNAs. They were proposed to have the potential to influence the immune system of infants14,15. Human milk exosomes are capable of surviving digestion and being taken up by enterocytes into the nucleus and may affect gene expression through exosomal miRNAs16 or enter the infant’ systemic circulation to exert the functions of tissue-specific immunoprotective and development17. Bovine milk-derived extracellular vesicles (BMEVs) have been shown to be taken up by murine macrophage RAW264.7 cells, splenocytes and intestinal cells in vitro and to reduce the serum levels of MCP-1 and IL-6 in splenic cells18.
Even though milk-derived exosomes are known to be resistant to a harsh environment and are taken up by multiple cells, including intestinal cells, whether they regulate intestinal cell proliferation and intestinal tract development remains unclear. Therefore, based on our previous exploration of miRNAs, mRNAs and proteins in porcine milk19, the KEGG pathway analysis of potential targets of top 10 miRNAs in porcine milk were enriched in Notch signaling pathway which could play an early critical role in cell fate determination and later roles in the regulation of cell proliferation and differentiation20. Otherwise, the Notch signaling pathway could participate in regulation of human cervical cancer and mammary gland progression21,22. Thus, we hypothesized that porcine milk-derived exosomes are capable of regulating intestinal cell proliferation and intestinal tract development. We hope this study could provide a new evidence for the regulation of intestine tract development from breastmilk exosomes to offspring.
Results
Milk-derived exosomes promoted proliferation of IPEC-J2 cell
IPEC-J2 cells were seeded at a density between 4,000 to 12,000 cells per/well (96-well plate) and their growth peaked between 24 h to 72 h after seeding (see Supplementary Figure 1). Porcine milk-derived exosomes containing 27 ± 3 ng and 55 ± 5 ng total RNA were then added to the IPEC-J2 cell culture, followed by analysis using the MTT assay at 0, 24, 48 and 72 h post treatment. The MTT results showed that treatment with porcine milk exosomes could significantly raise the OD value at 24, 48 and 72 h and the 27 ± 3 ng total RNA group had the most significant effect (P < 0.01) (Fig. 1a).
IPEC-J2 cell proliferation detected by different methods.
(a) Both 27 ± 3 ng and 55 ± 5 ng total RNA groups showed significantly improved OD values in the MTT assay, but the effect of the 27 ± 3 ng treatment group was more obvious (n = 10). (b,c) Effects of 27 ± 3 ng and 55 ± 5 ng total RNA treatment groups. The 27 ± 3 ng treatment group showed a significantly improved OD value (P < 0.01) in the CCK8 compared with the 55 ± 5 ng treatment group (n = 10). (d,e) Fluorescence intensity of IPEC-J2 cells after treatment with 27 ± 3 ng and 55 ± 5 ng total RNA for 24 h and 48 h. The 27 ± 3 ng treatment group showed greater fluorescence intensity by EdU fluorescence microscopy (n = 6). (f,g) Positive ratio of the 0.27 ± 0.03 μg total RNA (6-well plate) treatment group at 24 h and 48 h, respectively. The positive ratio significantly increased up to 80% after treatment for 24 h and 67% at 48 h by Edu flow cytometry (n = 6, 24 h).
In the CCK8 assay, porcine milk exosomes containing 27 ± 3 ng and 55 ± 5 ng total RNA significantly improved the OD value of CCK8 compared with the control group and the 27 ± 3 ng total RNA group showed a more efficient effect than the 55 ± 5 ng total RNA group (Fig. 1b,c). These results were consistent with those of the MTT assay. Furthermore, in the EdU fluorescence assay, the number of fluorescent cells was obviously greater in treatment groups than that in the control group (Fig. 1d,e). As revealed by EdU flow cytometry, milk exosomes of the 0.27 ± 0.03 μg total RNA group (6-well plate) significantly increased the positive fluorescence rates up to 80% at 24 h (Fig. 1f) and 67% at 48 h (Fig. 1g) post treatment, which were greater than that of the control group (Supplementary Figure 2).
Effect on proliferation-related gene’s expression by milk-derived exosome
Analysis by qRT-PCR revealed that porcine milk-derived exosomes significantly improved the expression of CDX2, proliferating cell nuclear antigen (PCNA) and IGF-1R genes (Fig. 2a,b,d) and inhibited the expression of p53 (Fig. 2c) in IPEC-J2 cells. Similarly, Western blotting showed significantly increased protein expression of CDX2, PCNA and IGF-1R (Fig. 2e,f,h) and significantly inhibited expression of p53 (Fig. 2g, P < 0.01). Those changes in protein markers of cell proliferation are fully consistent with the increased cell proliferation observed above.
Expression of proliferation-related genes and proteins in IPEC-J2 cells.
(a–d) Expression of CDX2, PCNA, p53 and IGF-1R mRNAs, respectively. CDX2, PCNA and IGF-1R levels were significantly improved, while p53 was inhibited in the 0.27 ± 0.03 μg total RNA treatment group (P < 0.01, n = 6). (e–h) Expression of CDX2, IGF-1R, p53 and PCNA proteins, respectively. CDX2, IGF-1R and PCNA were increased significantly, while p53 was inhibited significantly (P < 0.01) (n = 6).
Milk-derived exosomes changed miRNA concentration and resulted in changing of mRNA level in IPEC-J2 cells
To testify if miRNAs in milk-derived exosomes could enter into IPEC-J2 cells, we determined the level of miR-7134, miR-1343, miR-2320, miR-181a, miR-769-3p and miR-128 in IPEC-J2 cells after incubation with exosomes. q-PCR confirmed those miRNAs showed significantly higher level in IPEC-J2 cells after milk-derived exosomes incubation (Fig. 3b,c) relative to that in control, respectively. Bioinformatics analyzed showed the FAS was targeted of miR-2320 and miR-181a and SERPINE was target of miR-769-3p and miR-128 in p53 signaling pathways19. More interestingly, we analyzed the FAS and SERPINE which are potential targets of milk exosomal miRNAs and involved in p53 pathways and found out mRNA levels of FAS (Fig. 4a) and SERPINE (Fig. 4b) were down-regulated after being treated with milk exosome. All those results hinted that the milk-derived exosomes could not only be uptake by IPEC-J2 cells, but also regulate gene expression in recipient cells.
Milk-derived exosomes affected intestinal tract growth in mice
To further explore effects of porcine milk exosomes on intestinal tract development, we designed an in vivo experiment in mice. Hematoxylin and eosin (H&E) staining of paraffin sections showed that exosome treatments in the 0.125 μg, 0.25 μg, 0.5 μg and 1 μg total RNA groups significantly increased villus height and crypt depth of the duodenum and jejunum relative to the control group, both at 3 weeks (Fig. 5a,c) and 2 weeks (Supplementary Figure 5) post treatment. More interestingly, daily administration of milk exosomes (0.125 μg and 0.25 μg total RNA) significantly increased (P < 0.01) the villus height and crypt depth and thus improved the V/C ratio (Fig. 5b,d) relative to the control. However, higher dosages of 0.5 μg and 1 μg total RNA resulted in a lower increase of the V/C ratio (Fig. 5b,d) than those of the 0.125 μg and 0.25 μg total RNA groups. Additionally, results at 2 weeks post treatment were similar with those at 3 weeks (Supplementary Figure 5). All of these findings suggested that the milk exosomes are capable of improving the development of the intestinal tract in mice.
Observation and analysis of intestinal histomorphology (3 weeks).
(a) The 0.125 μg, 0.25 μg, 0.5 μg and 0.1 μg treatment groups showed improvement in the villus height and crypt depth compared with the control group by microscopy observation of duodenum morphology (n = 6). (b) Statistical analysis of the 0.125 μg, 0.25 μg and 0.5 μg treatment groups showed significantly increased villus height, crypt depth and V/C ratio in the duodenum (n = 30). (c) The 0.125 μg, 0.25 μg, 0.5 μg and 0.1 μg treatment groups showed improvement in the villus height and crypt depth compared with the control group by microscopy observation of jejunum morphology (n = 6). (d) Statistical analysis of the 0.125 μg and 0.25 μg treatment groups showed significantly increased villus height, crypt depth and V/C ratio of the jejunum (n = 30).
Expression of proliferation-related genes in mouse intestinal tract
After morphological observation of the mouse intestinal tract, we selected the jejunum tissue (3 weeks) for gene expression analysis. Results showed that 0.125 μg and 0.25 μg of milk-derived exosome RNA significantly improved the expression of CDX2, PCNA and IGF-1R in the jejunum at 3 weeks post treatment (Fig. 6a–c) and significantly decreased the p53 expression (Fig. 6d) as revealed by qRT-PCR. More importantly, Western blot analysis also showed increased protein levels of PCNA, IGF-1R and CDX2 (Fig. 6e,g,h) and a decreased p53 protein level (Fig. 6f) in the jejunum. Intriguingly, results above were fully consistent with those of the in vitro analysis in IPEC-J2 cells. These findings strongly suggest that milk exosomes promote intestinal cell proliferation by regulating expression of CDX2, PCNA, IGF-1R and p53.
Expression of proliferation-related genes and proteins in intestinal tissue.
(a–d) Expression of CDX2, PCNA, IGF-1R and p53 mRNAs, respectively. Expression of CDX2, PCNA and IGF-1R genes significantly increased at different treatment concentrations (0.125 μg and 0.25 μg treatment groups), except for the expression of IGF-1R in the 0.125 μg treatment group (P = 0.055). Meanwhile, p53 significantly decreased (n = 6). (e–h) Expression of CDX2, p53, IGF-1R and PCNA proteins, respectively. Except for the IGF-1R protein expression in the 0.25 μg treatment group which did not change significantly, expression levels of other proteins were consistent with those of their corresponding mRNAs (n = 6).
Discussion
Exosomes are small (30–150 nm) membrane-derived extracellular vesicles that carry miRNAs, proteins and lipids and mediate intercellular communication. In this study, exposure to porcine milk exosomes resulted in significantly increased proliferation of IPEC-J2 cells, as revealed by analyses using the MTT assay, CCK8 assay and EdU fluorescence and flow cytometry. As a thymidine analogue, EdU can be integrated in cellular DNA during the S-phase of the cell cycle23. Thus, the cell fluorescence intensity and the proportion of cells in the S-phase may represent the rate of proliferation. As described in the results, the ratio of positive fluorescent IPEC-J2 cells increased up to 80% by addition of milk-derived exosomes (27 ± 3 ng total RNA group). In the mouse experiment, morphometric analysis showed that the villus height, crypt depth and V/C ratio were significantly increased after treatment with porcine milk exosomes. Intestinal cell proliferation is a primary driver of intestinal growth and development24 and the continuous renewal of the intestinal epithelium is closely related to the crypt/villus functional unit. A dynamic system directs this process involving cell generation and migration from the stem cell population, located near the bottom of the crypts, to the extrusion of terminally differentiated cells at the tips of the villi25. Indeed, epithelial cells of the different compartments of the crypt–villus axis are characterized by differential properties in regard to cellular proliferation as well as differentiation26. Both in vitro and in vivo experiments in this current study strongly suggest that milk exosomes are able to promote intestinal cell proliferation and digestive tract development.
To further explore how milk exosomes can affect intestinal cell proliferation, expression levels of genes and proteins related to intestinal cell proliferation were resolved by qRT-PCR and Western blotting, respectively. CDX2 is an intestine-specific transcription factor expressed in the nuclei of epithelial cells throughout the intestine, from duodenum to rectum27, which is directly involved in intestinal development, differentiation and maintenance of the intestinal phenotype28. CDX2 is mostly present in the villi or differentiated cell compartment of the small intestine29. PCNA is associated with the cell cycle and functions in both DNA replication and repair30. PCNA immunolocalization has been reported to be useful as an index of cell proliferation in normal tissues and lymphoid neoplasms31. The IGR-1R downstream Akt/Wnt signaling pathways play a critical role in cell proliferation. A previous report on colon cancer showed that attenuated IGF-1R protein levels could suppress cell proliferation and elevate apoptosis even in the presence of IGF-1 via suppression of IGF-1R/Akt/Wnt signaling pathways and activation of p5332. All three of these genes tested in this study were significantly up-regulated by milk exosomes, consistent with their functions.
The p53 protein is the main negative regulator of the cell cycle. Alteration or inactivation of p53, or interactions with oncogene products of DNA tumor viruses that depend on p53, would lead to cancer33. p53 controls the expression of the gene encoding the p21 cyclin-dependent protein kinase (CDK) regulator. In normal human cells, p21 is dependent on cyclin, CDK and PCNA and it controls CDK activity, thereby affecting cell-cycle control30. A rapid rise in p53 levels can inhibit cell growth and tight regulation of p53 function is critical for normal cell growth and development34. Meanwhile, Mdm2 is a potent inhibitor of p53, which can block its regulation of target genes and exert anti-proliferative effects35. In our results, p53 expression in IPEC-J2 cells and the mouse intestinal tract was significantly inhibited.
How milk exosomes affect gene expression is another topic of interest, which awaits further exploration. As we previously reported, porcine milk exosomes contain hundreds of miRNAs19. By bioinformatics analysis, miR-34a, miR-34c, miR-885-3p and miR-885-5p were predicted to play roles in cell fate control and development as related to the Notch signaling pathway36. A report in HeLa cells demonstrated that miR-223 targets IGF-1R and suppresses cell proliferation by activation of the downstream PI3K/Akt/mTOR/p70S6K pathway37. In ovarian cancer and other cancers, loss of miR-31 can lead to a defect in the p53 pathway38. Genes encoding miRNAs in the miR-34 family are direct transcriptional targets of p53, which suppresses tumor formation through integration of multiple transcriptional targets and miR-34 may act in concert with other effectors to inhibit inappropriate cell proliferation39. miR-885-5p leads to the accumulation of p53 protein and activates the p53 pathway, subsequently inhibiting proliferation and interfering with cell cycle progression and cell survival40. Although we do not yet have direct experimental evidence to show that milk exosomal miRNAs regulate CDX2, PCNA, IGF1-R or p53, confirming these findings would likely be possible in future studies.
To explore if the milk-derived exosome could be uptake by IPEC-J2 cell and resultantly influence gene expression, we quantitatively detected expression levels of miRNAs and mRNA in IPEC-J2 cells after incubation with milk exsosomes. Six miRNAs detected were significantly elevated after incubation with milk exosome, which indicates miRNA in milk exosome may be uptake by IPEC-J2 cells. Our results are similarly to the previous reports. It was reported that the DCs-derived (dendritic cells) exosomal miR-155 and miR-146a could be transferred to recipient DCs in immune responses41. Invasive and non-invasive UBC cell lines derived exosomes specific miRNAs were transferred into cancer cells and tumor-associated fibroblasts (TAFs)42. In our study, mRNA of FAS and SERPINE, who are potential targets of milk exosomal miRNAs19 and involved in p53 signaling pathway, were found to be down-regulated due to milk exosome treatment. Similarly, it was reported that pancreatic cancer (PC) derived exosomal miRNAs inhibited mRNA expression of dendritic cells and induced immune tolerance43. Many types of cells have been shown to absorb exosomal microRNAs, where they induced post-translational repression of target mRNAs. A recently study reported that the milk exosomal miRNA (miR)-29b and miR-200c could be absorbed by human, meanwhile, mimicking postprandial concentrations of miR-29b and miR-200c in human embryonic kidney 293 cells and reporter gene activities significantly decreased by 44% and 17%, respectively. These results demonstrated the miRNAs in milk are bioactive food compounds that regulate human genes44. Another study revealed the bovine milk exosomes were incorporated into differentiated THP-1 cells then functioned in human cells by containing RNA45. Taken together, milk exosome may be uptake by IPEC-J2 cells and resultantly regulated gene expression in cells.
Milk exosomes are biologically active vesicles. Human milk exosomes are reported to be capable of surviving digestion by treatment with pepsin and pancreatin and being taken up by enterocytes where they localize to the nucleus and may affect gene expression16. The commercial milk-derived extracellular vesicles are extremely stable under degrading conditions, including low pH, boiling and freezing temperatures and easily taken up by murine macrophages in vitro, subsequently facilitating T cell differentiation through their packaged bioactive TGF-β46. Transport of bovine exosomes also depends on cell and exosome surface glycoproteins in human and rat intestinal cells by endocytosis47. Otherwise, bovine milk exosomes are incorporated into differentiated THP-1 cells and affect human cells through their packaged RNAs45. Extracellular vesicle-encapsulated miRNAs also cross the intestinal mucosa by processes involving endocytosis and exocytosis48. Another study performed on splenocyte cells, RAW264.7 cells and intestinal cells showed that treatment with BMEVs could reduce the serum levels of MCP-1 and IL-6 produced by splenic cells. Via oral gavage, administration of BMEVs to IL-1Ra−/− mice and collagen-induced arthritis mice was shown to result in delayed onset of arthritis and diminished cartilage pathology and bone marrow inflammation; similarly, it diminished the anti-collagen IgG2a levels and was accompanied by reduced splenic Th1 (Tbet) and Th17 (RORγT) mRNA18. All these findings showed that milk exosomes could transfer their encapsulated products and exert their functions on target cells.
Furthermore, some studies showed that extracellular vesicles from commercial milk contained several immunomodulating miRNAs and membrane protein CD63. However, the concentrations of miRNAs in milk were significantly reduced during the milk processing49, possibly because of that the milk cells and lipid fractions were usually discarded from formula during the industrial milk preparation procedures17 and the human milk (HM) cell and HM fat contain more number of miRNAs than peripheral blood mononuclear cells (PMBCs) and plasma, whilst they were with a strong association in human milk50. Nonetheless, infant formulas are manufactured from bovine milk and their RNA concentrations were found to be significantly lower than that in raw milk. Moreover, the quality-of-milk product indicators miR-148a and miR-200c were significantly lower in the extensively hydrolyzed formula than in the standard and follow-on formulas51. The decreased miRNAs may be attributed to the disruption of exosome membranes in milk and exposure of miRNAs released from exosomes to milk RNases49, suggesting that milk exosomes play an important role in developmental regulation in neonates and their mechanism needs further investigation.
Conclusion
Our study is the first report on the regulation of intestinal cell proliferation and digestive tract development by milk exosomes. As described in this study, milk exosomes are a new type of regulator in milk which has promising practical applications. As digestive tract development is crucial for the growth and health of newborns, our results will facilitate the goal of providing better nutrition for newborns.
Materials and Methods
Sample collection
Fresh porcine milk samples were collected from 10 healthy Landrace female pigs that had been lactating for 1 to 5 days (after parturition) at the pig farm of the South China Agriculture University (Guangzhou, China). Milk samples were frozen immediately and kept at −80 °C until used.
Preparation of exosomes
Porcine milk exosomes were separated as previously described19. Briefly, about 80–100 mL fresh raw procine milk samples were centrifuged at 2000 × g for 30 min at 4 °C to remove milk fat globules (MFGs) and mammary gland-derived cells. Defatted samples were then subjected to centrifugation at 12,000 × g for 30 min at 4 °C to remove residual MFGs, casein and other debris. From the supernatant, the membrane fraction was prepared by ultracentrifugation at 110,000 × g for 2 h using an SW41T rotor (Beckman Coulter Instruments, Fullerton, CA, USA) for three times and the supernatant was collected as a control for in vitro and in vivo experiments. The total RNA was then extracted and the porcine milk-derived exosome concentration was quantified and expressed as μg total RNA/mL in PBS.
IPEC-J2 cell culture and treatment with porcine milk exosomes
The IPEC-J2 cell line was cultured in Dulbecco’s modified eagle medium (DMEM/Ham’s F-12 [1:1]) (Invitrogen, Life Technologies, Carlsbad, CA, USA) supplemented with 5% fetal calf serum (FCS) (Invitrogen), 5 ug/ mL insulin (Sigma, St. Louis, MO, USA), 5 ng/mL EGF; Peprotech, Rocky Hill, NJ, USA) and incubated at 37 °C with 5% CO2. The IPEC-J2 cells were routinely seeded at a density of 0.5 × 105 mL−1 with 10 mL medium in plastic tissue culture flasks (75 cm2 Corning, Corning, NY, USA). Cells formed a confluent monolayer within 4 days and then used in experiments23.
Cell lines were seeded in 6-well tissue culture plates (9.6 cm2/well) at between 2.5 × 105 to 3.0 × 105 cells per well in a 2 mL volume and the 96-well culture plates (0.32 cm2/well) were seeded with 5 × 103 to 8 × 103 cells per well in a 200 μL volume. All of the seeded cells were allowed to adhere for 24 h before being re-fed every other day to allow for confluency. The cells were maintained in at 37 °C with an atmosphere of 5% CO252. For treatment, after the seeded cells reached 70–80% confluency (about 12 h after seeding), exosomes were added. Each 30 mL whey pellet ultracentrifuged from 40–45 mL (27 ± 3 μg total RNA) of raw milk was mixed with 10 mL PBS and then filtered with 0.45 μm and 0.22 μm membranes for the treatment. Cells in 6-well plates were treated with 0.27 ± 0.03 μg total RNA/well and 96-well plates were treated with 27 ± 3 ng and 55 ± 5 ng total RNA/well.
MTT assay
The MTT assay has been confirmed to be feasible, rapid and reproducible. Moreover, its results have shown good correlation with those of other in vitro proliferation assays, such as the 3H-thymidine uptake assay53. For this study, the MTT kit was purchased from Beyotime Biotechnology (Shanghai, China) and used according to the manufacturer’s protocol. Briefly, IPEC-J2 cells were seeded in 96-well plates at the density of 5,000 cells per well with 200 μL of complete culture medium. After being allowed to adhere and spread for 12 h, the cells were treated with different concentrations of porcine milk exosomes for 24 h to 48 h. MTT assays were performed by incubating the exosome-treated IPEC-J2 cells with 20 μL (5 mg/mL) MTT labeling solution. After 4 h of incubation, IPEC-J2 cells were lyzed with 150 μL DMSO and the purple formazan crystals were solubilized for detection at 570 nm54.
CCK8 proliferation assay
The CCK8 kit was purchased from Beyotime Biotechnology and used according to the manufacturer’s protocol. IPEC-J2 cells were seeded and treated as described for the MTT method. After 24 h and 48 h, the supernatant was removed and 100 μL of DMEM/F12 medium containing 10 μL of CCK8 was added to each well for incubation for another 3 h at 37 °C. The culture plates were then shaken for 10 min and the OD values were read at 450 nm55.
EdU assay
EdU (5-ethynyl-2′-deoxyuridine) is a labeled nucleoside analog of thymidine. In the S-phase of the cell cycle, its incorporation during DNA synthesis can reveal late replication regions56 and offers a wide range of opportunities to analyze cellular proliferation, population homeostasis and cell marking procedures57.
The following steps were carried out according to the manual of the Cell-Light™ EdU Apollo®488 In Vitro Imaging Kit (C10310-3) (Ribobio, Guangzhou, China). After treatment for 24 h or 48 h, 50 μM of the EdU labeling medium was added to the cell culture for incubation for 2 h at 37 °C with 5% CO2. Thereafter, cultured IPEC-J2 cells were fixed with 4% paraformaldehyde (pH 7.4, 50 μL/well of a 96-well plate) for 30 min and incubated with glycine (2 mg/mL) for 5 min. The cells were then washed with PBS (100 μL/well) and staining with anti-EdU working solution was performed at room temperature for 30 min. Following washing with 0.5% TritonX-100 (100 μL/well) in PBS for 5 min, the cells were incubated with 5 μg/mL Hoechst 33342 dye at room temperature for 30 min, followed by observation under a fluorescence microscope (OLYMPUS, Tokyo, Japan)58.
The percentage of EdU-positive cells was calculated from the Cell-Light™ EdU Apollo®488 In Vitro Flow Cytometry Kit (C10338-3) (Ribobio, Guangzhou, China) with slight modification of the EdU labeling procedure as follows. First, IPEC-J2 cells were seeded in a 6-well plate and collected after 24 h or 48 h of treatment by centrifugation at 1500 rpm/min for 5 min. After the supernatant was removed, the cells were resuspended with PBS and then centrifuged again at 1500 rpm/min for 5 min, followed by removal of the supernatant. Subsequently, the protocol of the Imaging Kit was followed and at the last step, the percentage of EdU-positive cells was detected by flow cytometry with the Cytomics FC 500 MCL (Beckman Coulter, Brea, CA, USA) as previously reported59,60.
Detection of expression of miRNAs and proliferation-related genes by qRT-PCR
IPEC-J2 cells were harvested after treatment for 48 h with porcine milk-derived exosomes and then used for RNA and protein extractions as follows. Total RNA was first digested with DNase I (Promega, Madison, WI, USA) and 2 μg of total RNA was reverse transcribed with oligo (dT). The cDNA was diluted 2-fold with ddH2O and PCR was performed on a Bio-Rad system (Hercules, CA, USA) in a final 20 μL volume reaction, containing 2 μL PCR cDNA, 10 μL of 2× PCR Mix (Roche, Basel, Switzerland) and 1 mM of each primer. The real-time PCR thermal profile was as follows: 5 min at 95 °C, 40 cycles of 30 s at 94 °C, 30 s at the corresponding annealing temperature (Tm) and 72 °C for 30 s, followed by 72 °C at 10 min and β-actin was used as an internal control for the PCR19,61. The milk-derived exosomes and IPEC-J2 miRNAs quantitative detected according to the protocol of Mir-X miRNA First Strand Synthesis Kit (Takara Bio Company, Dalian, China). The mRNA and miRNAs primers were designed with Primer 5.0 (Table 1).
Western blotting
RIPA lysis buffer was used to extract IPEC-J2 cell proteins according to the assay kit protocol (Bioteke, Beijing, China). Briefly, 1 mM PMSF was added to the RIPA lysis buffer and 100–200 μL was added to porcine milk exosomes. Following complete exosome lysis, the sample was centrifuged at 10,000–14,000 × g for 3–5 min and the supernatant was subjected to further analysis. Proteins were stored at −80 °C until used.
Protein samples (20–30 μg) were measured by the BCA assay62 and separated using 10–15% SDS-PAGE, transferred to a 0.22 mm or 0.45 mm polyvinylidene difluoride membrane (Millipore, Bedford, MA, USA), incubated with specific and HRP-conjugated secondary antibodies and detected with an enhanced chemiluminescence kit (Roche) using FluorChem M (ProteinSimple)63. Anti-p53 (D120082), anti-CDX2 (D162691) and anti-PCNA (D120014) antibodies were purchased from BBI Antibody (Sangon Biotech, Shanghai, China). IGF-1R and β-actin were purchased from Cell Signaling Technology (Danvers, MA, USA). Protein concentrations were determined using the Pierce BCA Protein Assay Kit (Thermo Fisher, Waltham, MA, USA) using a bovine serum albumin standard and Image J software was used for gray scan analysis.
Animal treatment
Male Kunming mice aged 18 days old were obtained from the Laboratory Animal Services Centre of Guangdong Province, China and were kept in specific pathogen-free conditions. Sixty mice were evenly divided into five groups, including the control (supernatant), 0.125 μg, 0.25 μg, 0.5 μg and 1 μg total RNA dose groups (dissolved in 300 μL PBS). The time points for monitoring the mice were 2 and 3 weeks. Every day, each mouse was given the appropriate treatment by intragastric administration until to the end of experiment, at which time it was killed according to the guidelines of the Animal Experimentation Ethics Committee of South China of Agricultural University64. Three sections of duodenum and jejunum were collected. One section (3–5 cm) was fixed with 10% formalin for tissue sectioning and the others were frozen in −80 °C until used for RNA and protein analysis.
Intestinal histomorphology
Fixed intestinal tissues were taken for tissue sectioning and H&E staining. Sections were stained with H&E using standard pathologic procedures as previously reported65, including fixation in neutral buffered 10% formalin, embedding in paraffin and cutting into horizontal sections of 6 μm thickness. The villi height and crypt depth were determined under a light microscope (Nikon, Tokyo, Japan) at a magnification of 40× or 100×.
qRT-PCR
Total RNA of intestinal tissue was extracted by TRIzol reagent (Invitrogen) as previously reported66 and the qRT-PCR analysis was carried out according to the above method for IPEC-J2 cells.
Western blotting
Each tissue sample (0.5–1 μg) was ground in Fastprep-24 (MP Biomedicals, Santa Ana, CA, USA) with 300 μl RIPA lysis buffer according to the assay kit protocol (Bioteke) and then analyzed by Western blot according to the protocol used for IPEC-J2 cells.
Statistical analysis
All data are expressed as the mean ± standard error of the mean (SEM). The significance of differences was determined using t-test for comparison of 2 groups and one-way analysis of variance (ANOVA) with post hoc test of least significant difference (LSD) or Duncan test for multiple comparisons with SPSS 17.0. Differences were considered statistically significant at P < 0.05.
Declarations
Ethics Statement
The samples collected were according to the guidelines of Guangdong Province on the Review of Welfare and Ethics of Laboratory Animals approved by the Guangdong Province Administration Office of Laboratory Animals (GPAOLA). And the procedures were as the protocol of SCAU-AEC-2010-0416 approved by the Animal Ethics Committee of South China Agricultural University.
Additional Information
How to cite this article: Chen, T. et al. Porcine milk-derived exosomes promote proliferation of intestinal epithelial cells. Sci. Rep. 6, 33862; doi: 10.1038/srep33862 (2016).
References
Xu, R.-J. Development of the newborn GI tract and its relation to colostrum/milk intake: a review. Reproduction, Fertility and Development 8, 35–48 (1996).
Pácha, J. Development of intestinal transport function in mammals. Physiological reviews 80, 1633–1667 (2000).
Guilloteau, P., Zabielski, R., Hammon, H. M. & Metges, C. C. Nutritional programming of gastrointestinal tract development. Is the pig a good model for man? Nutrition Research Reviews 23, 4–22 (2010).
Sheard, N. F. & Walker, W. A. The role of breast milk in the development of the gastrointestinal tract. Nutrition Reviews 46, 1–8 (1988).
Alpers, D. H. Enteral feeding and gut atrophy. Current Opinion in Clinical Nutrition & Metabolic Care 5, 679–683 (2002).
Lebenthal, E., Lee, P. & Heitlinger, L. A. Impact of development of the gastrointestinal tract on infant feeding. The Journal of pediatrics 102, 1–9 (1983).
Melnik, B. C. Milk—A Nutrient System of Mammalian Evolution Promoting mTORC1-Dependent Translation. International journal of molecular sciences 16, 17048–17087 (2015).
Blum, J. W. & Baumrucker, C. R. In Bioactive components of milk 397–422 (Springer, 2008).
Reinhardt, T. A., Lippolis, J. D., Nonnecke, B. J. & Sacco, R. E. Bovine milk exosome proteome. Journal of Proteomics 75, 1486–1492 (2012).
Admyre, C. et al. Exosomes with immune modulatory features are present in human breast milk. The Journal of immunology 179, 1969–1978 (2007).
Hata, T. et al. Isolation of bovine milk-derived microvesicles carrying mRNAs and microRNAs. Biochemical and biophysical research communications 396, 528–533 (2010).
Lässer, C. et al. Human saliva, plasma and breast milk exosomes contain RNA: uptake by macrophages. Journal of translational medicine 9, 9 (2011).
Munagala, R., Aqil, F., Jeyabalan, J. & Gupta, R. C. Bovine milk-derived exosomes for drug delivery. Cancer letters 371, 48–61 (2016).
Zhou, Q. et al. Immune-related microRNAs are abundant in breast milk exosomes. International journal of biological sciences 8, 118 (2012).
Kosaka, N., Izumi, H., Sekine, K. & Ochiya, T. microRNA as a new immune-regulatory agent in breast milk. Silence 1, 7 (2010).
Lonnerdal, B., Du, X., Liao, Y. & Li, J. Human milk exosomes resist digestion in vitro and are internalized by human intestinal cells. The FASEB Journal 29, 121.123 (2015).
Alsaweed, M., Hartmann, P. E., Geddes, D. T. & Kakulas, F. MicroRNAs in breastmilk and the lactating breast: potential immunoprotectors and developmental regulators for the infant and the mother. International journal of environmental research and public health 12, 13981–14020 (2015).
Arntz, O. J. et al. Oral administration of bovine milk derived extracellular vesicles attenuates arthritis in two mouse models. Molecular nutrition & food research 59, 1701–1712 (2015).
Chen, T. et al. Exploration of microRNAs in porcine milk exosomes. BMC genomics 15, 100 (2014).
Conboy, I. M. & Rando, T. A. The regulation of Notch signaling controls satellite cell activation and cell fate determination in postnatal myogenesis. Developmental cell 3, 397–409 (2002).
Ramdass, B. et al. Coexpression of Notch1 and NF-κB signaling pathway components in human cervical cancer progression. Gynecologic oncology 104, 352–361 (2007).
Hynes, N. E. & Stoelzle, T. Key signalling nodes in mammary gland development and cancer: Myc. Breast Cancer Research 11, 1 (2009).
Diesing, A.-K. et al. Mycotoxin deoxynivalenol (DON) mediates biphasic cellular response in intestinal porcine epithelial cell lines IPEC-1 and IPEC-J2. Toxicology letters 200, 8–18 (2011).
Reznikov, E. A., Comstock, S. S., Yi, C., Contractor, N. & Donovan, S. M. Dietary bovine lactoferrin increases intestinal cell proliferation in neonatal piglets. The Journal of nutrition 144, 1401–1408 (2014).
Babyatsky, M. & Podolsky, D. Growth and development of the gastrointestinal tract. Textbook of gastroenterology, 547–584 (1999).
Rubin, D. C. & Shaker, A. Small intestine: anatomy and structural anomalies. Yamada’s Textbook of Gastroenterology, 73–92 (2009).
Werling, R. W., Yaziji, H., Bacchi, C. E. & Gown, A. M. CDX2, a highly sensitive and specific marker of adenocarcinomas of intestinal origin: an immunohistochemical survey of 476 primary and metastatic carcinomas. The American journal of surgical pathology 27, 303–310 (2003).
Silberg, D. G., Swain, G. P., Suh, E. R. & Traber, P. G. Cdx1 and cdx2 expression during intestinal development. Gastroenterology 119, 961–971 (2000).
James, R., Erler, T. & Kazenwadel, J. Structure of the murine homeobox gene cdx-2. Expression in embryonic and adult intestinal epithelium. Journal of Biological Chemistry 269, 15229–15237 (1994).
Waga, S., Hannon, G. J., Beach, D. & Stillman, B. The p21 inhibitor of cyclin-dependent kinases controls DNA replication by interaction with PCNA. Nature 369, 574–578 (1994).
Hall, P. et al. Proliferating cell nuclear antigen (PCNA) immunolocalization in paraffin sections: An index of cell proliferation with evidence of deregulated expression in some, neoplasms. The Journal of pathology 162, 285–294 (1990).
Vanamala, J., Reddivari, L., Radhakrishnan, S. & Tarver, C. Resveratrol suppresses IGF-1 induced human colon cancer cell proliferation and elevates apoptosis via suppression of IGF-1R/Wnt and activation of p53 signaling pathways. BMC cancer 10, 238 (2010).
Levine, A. J., Momand, J. & Finlay, C. A. The p53 tumour suppressor gene. Nature 351, 453–456 (1991).
Kubbutat, M. H., Jones, S. N. & Vousden, K. H. Regulation of p53 stability by Mdm2. Nature 387, 299–303 (1997).
Haupt, Y., Maya, R., Kazaz, A. & Oren, M. Mdm2 promotes the rapid degradation of p53. Nature 387, 296–299 (1997).
Artavanis-Tsakonas, S., Rand, M. D. & Lake, R. J. Notch signaling: cell fate control and signal integration in development. Science 284, 770–776 (1999).
Jia, C. Y. et al. MiR-223 suppresses cell proliferation by targeting IGF-1R. PLoS ONE 6, e27008 (2011).
Creighton, C. J. et al. Molecular profiling uncovers a p53-associated role for microRNA-31 in inhibiting the proliferation of serous ovarian carcinomas and other cancers. Cancer research 70, 1906–1915 (2010).
He, L. et al. A microRNA component of the p53 tumour suppressor network. Nature 447, 1130–1134 (2007).
Afanasyeva, E. A. et al. MicroRNA miR-885-5p targets CDK2 and MCM5, activates p53 and inhibits proliferation and survival. Cell Death & Differentiation 18, 974–984 (2011).
Alexander, M. & O’Connell, R. Exosomal miRNAs regulate inflammatory responses (IRM11P. 624). The Journal of Immunology 194, 132.133–132.133 (2015).
Baumgart, S., Heinzelmann, J., Stoeckle, M., Ostenfeld, M. S. & Junker, K. Characterization of miRNA expression pattern from in-vitro obtained exosomes of different urinary bladder cancer cell lines. Cancer Research 75, 5182–5182 (2015).
Ding, G. et al. Pancreatic cancer-derived exosomes transfer miRNAs to dendritic cells and inhibit RFXAP expression via miR-212-3p. Oncotarget 6, 29877 (2015).
Baier, S. R., Nguyen, C., Xie, F., Wood, J. R. & Zempleni, J. MicroRNAs are absorbed in biologically meaningful amounts from nutritionally relevant doses of cow milk and affect gene expression in peripheral blood mononuclear cells, HEK-293 kidney cell cultures and mouse livers. The Journal of nutrition 144, 1495–1500 (2014).
Izumi, H. et al. Bovine milk exosomes contain microRNA and mRNA and are taken up by human macrophages. Journal of dairy science 98, 2920–2933 (2015).
Pieters, B. C. et al. Commercial cow milk contains physically stable extracellular vesicles expressing immunoregulatory TGF-β. PLoS one 10, e0121123 (2015).
Wolf, T., Baier, S. R. & Zempleni, J. The Intestinal Transport of Bovine Milk Exosomes Is Mediated by Endocytosis in Human Colon Carcinoma Caco-2 Cells and Rat Small Intestinal IEC-6 Cells. The Journal of nutrition, jn218586 (2015).
Kusuma, R., Friemel, T. & Zempleni, J. Human Endothelial Cell Transport Bovine Extracellular Vesicles. The FASEB Journal 29, LB357 (2015).
Howard, K. M. et al. Loss of miRNAs during processing and storage of cow’s (Bos taurus) milk. Journal of agricultural and food chemistry 63, 588–592 (2015).
Alsaweed, M., Lai, C. T., Hartmann, P. E., Geddes, D. T. & Kakulas, F. Human milk miRNAs primarily originate from the mammary gland resulting in unique miRNA profiles of fractionated milk. Scientific reports 6 (2016).
Izumi, H. et al. Bovine milk contains microRNA and messenger RNA that are stable under degradative conditions. Journal of dairy science 95, 4831–4841 (2012).
Arce, C., Ramirez-Boo, M., Lucena, C. & Garrido, J. Innate immune activation of swine intestinal epithelial cell lines (IPEC-J2 and IPI-2I) in response to LPS from Salmonella typhimurium. Comparative immunology, microbiology and infectious diseases 33, 161–174 (2010).
Ciapetti, G., Cenni, E., Pratelli, L. & Pizzoferrato, A. In vitro evaluation of cell/biomaterial interaction by MTT assay. Biomaterials 14, 359–364 (1993).
Cheung, W. M., Hui, W. S., Chu, P. W., Chiu, S. W. & Ip, N. Y. Ganoderma extract activates MAP kinases and induces the neuronal differentiation of rat pheochromocytoma PC12 cells. FEBS letters 486, 291–296 (2000).
Yu, X. et al. MicroRNA-10b promotes nucleus pulposus cell proliferation through RhoC-Akt pathway by targeting HOXD10 in intervetebral disc degeneration. PLoS ONE 8, e83080 (2013).
Sisdelli, L. et al. Incorporation of 5-ethynyl-2′-deoxyuridine (EdU) as a novel strategy for identification of the skewed X inactivation pattern in balanced and unbalanced X-rearrangements. Human genetics 135, 185–192 (2016).
Flomerfelt, F. A. & Gress, R. E. Analysis of cell proliferation and homeostasis using EdU labeling. T-Cell Development: Methods and Protocols 211–220 (2016).
He, Q., Man, L., Ji, Y. & Ding, F. Comparison in the biological characteristics between primary cultured sensory and motor Schwann cells. Neuroscience letters 521, 57–61 (2012).
Sun, Y. et al. Multicolor flow cytometry analysis of the proliferations of T‐lymphocyte subsets in vitro by EdU incorporation. Cytometry Part A 81, 901–909 (2012).
Feng, S. et al. MicroRNA-192 targeting retinoblastoma 1 inhibits cell proliferation and induces cell apoptosis in lung cancer cells. Nucleic acids research, gkr232 (2011).
Ye, R.-S. et al. Differentially expressed miRNAs after GnRH treatment and their potential roles in FSH regulation in porcine anterior pituitary cell. PLoS ONE 8, e57156 (2013).
Stoscheck, C. M. [6] Quantitation of protein. Methods in enzymology 182, 50–68 (1990).
Théry, C. et al. Proteomic analysis of dendritic cell-derived exosomes: a secreted subcellular compartment distinct from apoptotic vesicles. The Journal of Immunology 166, 7309–7318 (2001).
Liu, W. N. & Leung, K. N. The Immunomodulatory Activity of Jacaric Acid, a Conjugated Linolenic Acid Isomer, on Murine Peritoneal Macrophages. PLoS one 10 (2015).
Hochstim, C. J., Choi, J. Y., Lowe, D., Masood, R. & Rice, D. H. Biofilm detection with hematoxylin-eosin staining. Archives of Otolaryngology–Head & Neck Surgery 136, 453–456 (2010).
Cheng, X. et al. Critical role of miR-125b in lipogenesis by targeting stearoyl-CoA desaturase-1 (SCD-1). Journal of Animal Science 94, 65–76 (2016).
Acknowledgements
This work was supported by grants from the Key Project of Guangdong Provincial Nature Science Foundation (S2013020012766), National Basic Research Program of China (973 Program, 2011CB944200, 2009CB941600 and 2013CB127304), Natural Science Foundation of China program (31272529, 31072105 and 31472163) and The National Key Research and Development Program of China (2016YFD0500503). We thank the breeding farm of the Livestock Research Institute (Guangzhou, China) for providing milk samples.
Author information
Authors and Affiliations
Contributions
T.C. and M.-Y.X. carried out the in vitro and in vivo experiments and participated in drafting the manuscript. J.-J.S. and Q.-Y.X. carried out mRNA and proteins extraction, X.C. and R.-S.Y. carried out the Western blotting and q-PCR. R.-P.S. and L.-M.W. participated in the sample collections. M.L. and D.-L.L. performed the raw data analysis. Q.-Y.J. and Y.-L.Z. conceived of the study and participated in its design and coordination and helped to draft the manuscript. All authors read and approved the final manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Chen, T., Xie, MY., Sun, JJ. et al. Porcine milk-derived exosomes promote proliferation of intestinal epithelial cells. Sci Rep 6, 33862 (2016). https://doi.org/10.1038/srep33862
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep33862
This article is cited by
-
Maternal fiber-rich diet promotes early-life intestinal development in offspring through milk-derived extracellular vesicles carrying miR-146a-5p
Journal of Nanobiotechnology (2024)
-
Potential therapeutic effects of milk-derived exosomes on intestinal diseases
Journal of Nanobiotechnology (2023)
-
Regulation of adipogenesis by exosomal milk miRNA
Reviews in Endocrine and Metabolic Disorders (2023)
-
Expression profiles of hsa-miR-148a-3p and hsa-miR-125b-5p in human breast milk and infant formulae
International Breastfeeding Journal (2022)
-
Role of omega-3 polyunsaturated fatty acids, citrus pectin, and milk-derived exosomes on intestinal barrier integrity and immunity in animals
Journal of Animal Science and Biotechnology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.