Abstract
Forested tropical peatlands in Southeast Asia are being rapidly converted to agriculture or degraded into non-forest vegetation. Although large areas have been abandoned, there is little evidence for subsequent forest recovery. As part of a study of forest degradation and recovery, we used seed removal experiments and rodent surveys to investigate the potential role of post-dispersal seed predation in limiting the regeneration of woody plants. Two 14-day seed removal trials were done in deforested and forested peatland habitat in Central Kalimantan, Indonesia. Seeds of Nephelium lappaceum, Syzygium muelleri, Artocarpus heterophyllus (all animal-dispersed) and Combretocarpus rotundatus (wind-dispersed) were tested. Significantly more seeds (82.8%) were removed in forest than non-forest (38.1%) and Combretocarpus had the lowest removal in both habitats. Most handled seeds were eaten in situ and little caching was observed. Six species of rodents were captured in forest and five in non-forest. The most trapped taxa were three Maxomys spp. in forest (85.5% of individuals) and Rattus tiomanicus in non-forest (74.8%). Camera traps confirmed that rodents were responsible for seed removal. Seed predation in deforested areas, which have a much lower seed rain than forest, may contribute to the low density and diversity of regenerating forest.
Similar content being viewed by others
Introduction
Tropical peatlands cover around 250,000 km2 of SE Asia, but have been severely impacted over the last few decades by logging and conversion to agriculture1,2. Agricultural conversion of deep peat is difficult, however and vast areas have been abandoned after clearance. At least 18,000 km2 of the 160,000 km2 of peatlands found in Peninsula Malaysia and on the islands of Borneo and Sumatra, has now been severely degraded3, particularly in Riau and Jambi in Sumatra and in Central, East and South Kalimantan in Indonesian Borneo. Moreover remote sensing provides little evidence for extensive forest recovery after abandonment3.
Previous studies of natural regeneration in degraded tropical peatlands have shown that ferns, climbers, sedges and grasses dominate after disturbance and that these areas support fewer woody plant species than intact peat swamp forest ecosystems4,5. The seed rain into an extensive degraded area in Kalimantan was species-poor, with three-quarters of the seeds from only two wind-dispersed species6. This was attributed to a combination of isolation from forest seed sources and the dominance of the regrowth avifauna by bulbuls and other small passerines, while large-gaped birds and mammalian frugivores were absent7. Woody regrowth was also dominated by a few abundant wind-dispersed species, while most other species were potentially dispersed by bulbuls, but additional woody species had apparently sprouted from vegetative remnants of the previous forest cover8.
The contrast between the densities of the seed rain (mean 95 seeds m−2 yr−1 in the open and 1128 seeds m−2 yr−1 under trees) and woody regrowth (mean 0.09 individuals m−2, representing several years seed rain) in non-forest habitats in these studies6,8 suggests that post-dispersal seed predation may be a significant factor limiting forest recovery. Several tropical studies have suggested that seed predation is a filter limiting natural regeneration of woody plants in degraded areas9,10. Post-dispersal seed predators in SE Asia include rodents, pigs, ants and other insects, but rodents appear to be the dominant predators of seeds in the size range of the woody seed rain in degraded sites11.
This study therefore used seed removal experiments and rodent surveys to investigate post-dispersal seed removal in the same tropical peatland habitat in Central Kalimantan, Indonesia, where the seed rain and woody regeneration were previously studied6,8. Specifically, it aimed to: (1) determine whether levels of post-dispersal seed removal differed between forested and deforested habitats; (2) establish the species composition of the ground-feeding rodents in both habitats; (3) establish whether removal was secondary seed dispersal or predation; (4) assess the possible implications for forest recovery.
Methods
Study area
This study was carried out in the former Mega Rice Project (MRP) area in Central Kalimantan, Indonesia. The MRP was initiated in 1996 and aimed to develop one million hectares of peat swamp forest into land for rice cultivation. This involved widespread forest clearance and the excavation of canals to drain the naturally water-logged peat12. The project was cancelled in 1999, but by this time most of the area had already been logged and cleared13. Two habitat types were used in this study: (1) logged peat swamp forest (hereafter ‘forest’) and (2) non-forest regrowth (‘non-forest’), following other studies at this site6,7,8.
The forest was located in the Tuanan Study Area, part of the 300,000 ha Mawas Conservation Area in the former MRP (02°09′S; 114°26′E). This forest was not cleared during establishment of the MRP, but was selectively-logged in the 1990s and illegally logged subsequently14. All logging stopped in 200215. The non-forest was in Block A North-West (2°17′S; 114°31′E) of the former MRP. Tuanan and Block A NW are both located on the Mantangai peat dome, although they are now separated by the two primary canals. Block A NW covers approximately 45,000 ha and was subjected to forest clearance and widespread drainage. An elaborate grid system of 300 km of canals divides the area into compartments roughly 2.5 by 2.5 km16. The non-forest habitat was dominated by ferns, with smaller areas occupied by woody plants, bare ground and standing or fallen dead wood8.
Seed removal trials
Two pre-existing transects in the forest and two purposely cut transects in the non-forest were used in this study. Each transect was used for a single seed removal experimental trial lasting 14 days. The four trials took place between May and November 2011 and were not run concurrently due to logistical issues. All trials included seeds of one native animal-dispersed peat swamp species fruiting at the time of study, Nephelium lappaceum or Syzygium muelleri, seeds of Combretocarpus rotundatus (wind-dispersed), which is the dominant woody plant species in non-forest and seeds of locally bought Artocarpus heterophyllus (jackfruit). Mean seed sizes are shown in Table 1. Artocarpus has been used in other studies on seed predation17,18 since it seems to be a reasonable model for large seeds and was chosen because seeds of native species were not always available. Fruits of the native species were collected by climbing trees to retrieve ripe fruits. Seeds were removed from fruit and cleaned to remove all fleshy parts, except for Combretocarpus, which is a winged seed with no flesh. One metre of cotton thread ending in numbered flagging tape was attached to each seed with non-toxic glue so removed seeds could be tracked. Other seed removal studies have found no difference between the removal of marked and unmarked seeds19,20.
A set of nine seeds made up of three of each species (always Combretocarpus and Artocarpus, with the third either Nephelium or Syzygium) was laid out in a grid, with 50 cm between seeds. Each grid will be referred to as a seed station. Twenty seed stations were laid out at 50 m intervals along each transect, giving a total of 360 seeds (9 × 20 × 2) in each habitat type. Seed stations were at least 1 m from the main transect line. Where necessary, a 2 m2 area was cleared of above-ground vegetation to provide a substrate to place seeds on. Leaf litter and over-hanging vegetation were left intact.
Seed stations were checked daily for 14 days, with seeds classed as handled or not. Handled seeds were further classed as eaten at the station or removed. Where seeds were removed, the plan was to search up to 20 m radius, on the assumption that rodents rarely carry seeds further18, but in practice all were found within 5.5 m. Removed seeds were further classed as eaten if seed fragments were found and cached if seeds were found below the soil surface or below leaf litter. Cached seeds were checked for the remainder of the study. Seeds with evidence of seed coat damage (by insects or rodents) were also classed as preyed upon9 and not counted as surviving, except in Combretocarpus where damage to the wings was not counted as long as the seed remained intact.
Ten camera traps (Bushnell Trophy camera, 5 megapixel model, Bushnell, Inc.) were installed along one seed removal experimental trial transect in forest and one in non-forest. A camera trap was placed at every second seed station, 10–20 cm from the ground and with a view of the seeds. Cameras were set to take a 20 second video when the sensor was triggered.
Rodent surveys
Trapping was done along each transect to assess the composition of the ground-feeding rodent community. Surveys were carried out within a month of the seed removal trial on each transect, using Tomahawk collapsible live traps model 201 (Tomahawk Live Traps IIC) covered with locally available materials (fallen leaves, ferns, dead wood etc.) for camouflage and rain protection. On each transect, 90 traps were set at 10 m intervals on the ground, at least 2 m from the transect line. A seven day session (630 trap nights) was conducted along each transect. Traps were baited with a mixture of banana and peanut butter, opened at 16.30 and checked the following morning at 07.00. Animals were marked with Monel ear tags (National Band and Tag Co., USA), measured and then released. Species were identified using A Field Guide to the Mammals of Borneo21 with body, tail, ear and foot measurements taken. All captured animals were also photographed.
Data analysis
To compare the levels of post-dispersal seed removal in forest and non-forest, data were analysed as the proportion of seeds remaining at each seed station, because the survival of individual seeds at each seed station could not be considered as independent of one another. Data were pooled for each habitat, giving 40 stations for forest and 40 for non-forest. A Mann-Whitney U test was used to compare the numbers of seeds remaining with an intact seed coat after 14 days in forest and non-forest, with stations as replicates (SPSS. V16). To look at variation in levels of post-dispersal seed loss amongst species the cumulative percentage of seeds handled (i.e. eaten at the station or removed from it) over time was calculated for each species and habitat. To establish the overall fate of seeds in the removal trials, percentages of each seed fate were calculated for each habitat.
Results
Seed fates
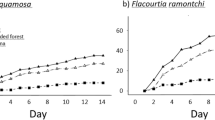
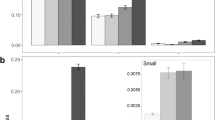
We were able to locate all tags (within 5.5 m of the seed station) and thus to establish the fates of all the seeds. Significantly more seeds (82.8%) were handled (i.e. eaten or removed) in forest than non-forest (38.1%) after 14 days (Mann Whitney U, P < 0.05) (Table 2). The mean number of seeds remaining with an intact seed coat at a seed station (out of a possible 9) was 1.6 (±1.1) in forest and 5.6 (±2.8) in non-forest. Combretocarpus had the highest proportion of seeds remaining at seed stations in both habitats, with more than half the seeds (51.7%) remaining in forest and more than three-quarters (75.8%) in non-forest. In contrast, no seeds of Nephelium and Syzygium and only one of Artocarpus (0.8%) remained in forest and 53.3%, 68.3% and 50.0% remained in non-forest (Fig. 1). In forest more seeds were handled than not handled, while in non-forest more seeds were not handled than handled. In both forest and non-forest most handled seeds were eaten at the seed station (191 and 118 respectively). More seeds were eaten away from the seed station in forest (99, 33% of handled seeds) than in non-forest (17, 12% of handled seeds) and more seeds were cached in forest (8, 0.027%) than non-forest (2, 0.015%). Of the cached seeds, only a single Artocarpus in forest remained cached and intact at the end of the study, 0.5 m from its original location.
Videos obtained by camera traps showed four instances of small mammals, consistent with the commonly trapped Rattus tiomanicus from their size, consuming seeds in the non-forest site and 12 instances in forest, one by a treeshrew and the others by rats, consistent with Maxomys spp., from their size and tail coloration. In forest there were also three instances of rats, consistent with Maxomys spp., removing seeds without consuming them.
Rodent surveys
A total of 354 different individual rodents of eight species were captured over 2520 trap-nights (Table 3). All were murids except for four Callosciurus notatus tree squirrels in the non-forest site. More individuals and more species were captured in forest (6 species, 205 individuals) than in non-forest (5 species, 147 individuals), although given the short timescale of the study and the large differences in the habitats, these counts should not be treated as estimates of relative abundance or diversity. The most trapped taxa were three Maxomys spp. in forest (85.5% of individuals) and Rattus tiomanicus in non-forest (74.8%). Two species of treeshrews (Tupaia spp.) were also trapped.
Discussion
The significantly higher level of post-dispersal seed removal in forest than non-forest is comparable with the results of other studies22,23,24. The rate of seed removal found for forest in this study was higher than that for lowland dipterocarp forests in SE Asia25,26, but not unusual for seed removal experiments in other parts of the tropics9,27. In contrast, the rate of seed removal in non-forest was lower than degraded landscapes in other parts of the tropics9,23,28. The animal-dispersed seeds, Artocarpus, Syzygium and Nephelium, all had very high levels of handling in forest (99–100%), but lower levels in non-forest (30–50%), while the wind-dispersed Combretocarpus had the lowest levels in both habitats. Low rates of seed handling have been reported for wind-dispersed species in other studies29.
The two habitats shared five ground-feeding rodent species, but with very different capture rates. Forest-specialist Maxomys spp. dominated the forest while the non-forest dominant, Rattus tiomanicus, was not recorded in forest. R. tiomanicus is typically found in scrubland, grassland and plantations but rarely in undisturbed forest21,30. The lack of trees explains the absence of arboreal rats (Lenothrix canus and Niviventer cremoriventer) in the degraded site. In other forest types in SE Asia, disturbance has caused a loss of native species and the invasion of generalists30,31, as happened in our sites, or the loss of rare species and the perseverance of common species32.
The camera trap videos showed that rodents were responsible for seed removal in both habitats, probably Maxomys spp. in forest and R. tiomanicus in non-forest. Other studies in SE Asia have also attributed seed removal to rodents. Video cameras were used to record seed removal agents in peat swamp forest in Sarawak, Malaysia and identified M. whiteheadi, Sundamys muelleri and Callosciurus notatus as the primary removal agents18, all of which were also captured in this study. While the limited use of camera traps did not record seed removal by all species captured, evidence from other studies shows they all consume some seeds18,26.
In both habitats only a tiny proportion of seeds was cached initially and only a single seed of Artocarpus in forest remained cached and intact by the end of the study. Rattus species are not known to scatter-hoard11 so the dominance of R. tiomanicus in non-forest may explain the absence of caching there. Maxomys species scatter-hoard elsewhere11, but very little caching of jackfruit seeds was reported in a peat swamp forest in Sarawak, Malaysia18. Tropical peatlands are typically water-logged and degraded areas often experience flooding, so the high water-table may make caching impractical. At these sites, therefore, seeds handled by rodents are almost all predated. Rodents swallow and disperse some very small seeds in their faeces11 and may contribute to the dispersal of small-seeded species, such as Melastoma malabathricum in the non-forest site, but this and other small-seeded species are also dispersed by the abundant bulbuls7.
While non-forest had a significantly lower level of seed predation than forest, non-forest sites had much lower seed rain densities, except under trees and three-quarters of the seeds were from two wind-dispersed species6. If the levels of predation of animal-dispersed seeds observed in non-forest areas in this study are typical, the impact on woody regrowth could be large: perhaps enough to contribute significantly to the low density and diversity of the regenerating forest. Future studies should use rodent-exclusion experiments to assess this further.
Additional Information
How to cite this article: Blackham, G. V. and Corlett, R. T. Post-dispersal seed removal by ground-feeding rodents in tropical peatlands, Central Kalimantan, Indonesia. Sci. Rep. 5, 14152; doi: 10.1038/srep14152 (2015).
References
Hirano, T., Kusin, K., Limin, S. & Osaki, M. Carbon dioxide emissions through oxidative peat decomposition on a burnt tropical peatland. Glob. Change Biol. 20, 555–565 (2014).
Page, S. E., Rieley, J. O. & Banks, C. J. Global and regional importance of the tropical peatland carbon pool. Glob. Change Biol. 17, 798–818 (2011).
Miettinen, J. & Liew, S. C. Degradation and development of peatlands in Peninsular Malaysia and in the islands of Sumatra and Borneo since 1990. Land Degrad. Develop. 21, 285–296 (2010).
Page, S. et al. Restoration ecology of lowland tropical peatlands in Southeast Asia: current knowledge and future research directions. Ecosystems 12, 888–905 (2009).
van Eijk, P., Leenman, P., Wibisono, I. T. C. & Giesen, W. Regeneration and restoration of degraded peat swamp forest in Berbak NP, Jambi, Sumatra, Indonesia. Malayan Nature J. 61, 223–241 (2009).
Blackham, G. V., Andri, T., Webb, E. L. & Corlett, R. T. Seed rain into a degraded tropical peatland in Central Kalimantan, Indonesia. Biol. Conserv. 167, 215–223 (2013).
Posa, M. R. C. Peat swamp forest avifauna of Central Kalimantan, Indonesia: Effects of habitat loss and degradation. Biol. Conserv. 144, 2548–2556 (2011).
Blackham, G. V., Webb, E. L. & Corlett, R. T. Natural regeneration in a degraded tropical peatland, Central Kalimantan, Indonesia: Implications for forest restoration. For. Ecol. Manage. 324, 8–15 (2014).
Holl, K. D. & Lulow, M. E. Effects of species, habitat and distance from edge on post-dispersal seed predation in a tropical rainforest. Biotropica 29, 459–468 (1997).
Nepstad, D. C., Uhl, C. & Serrão, E. A. S. Recuperation of a degraded Amazonian landscape - forest recovery and agricultural restoration. Ambio 20, 248–255 (1991).
Corlett, R. T. The ecology of tropical East Asia, second edition. Oxford University Press, Oxford, 2014.
Aldhous, P. Land remediation: Borneo is burning. Nature 432, 144–146 (2004).
McCarthy, J. F. Decentralisation and forest management in Kapuas district, Central Kalimantan. CIFOR, Bogor, Indonesia, 2001.
van Schaik, C. P., Wich, S. A., Utami, S. S. & Odom, K. A simple alternative to line transects of nests for estimating orangutan densities. Primates 46, 249–254 (2005).
Vogel, E. R., Haag, L., Mitra-Setia, T., van Schaik, C. P. & Dominy, N. J. Foraging and ranging behavior during a fallback episode: Hylobates albibarbis and Pongo pygmaeus wurmbii compared. Am. J. Phys. Anthropol. 140, 716–726 (2009).
Mawdsley, N. et al. Strategic peatland rehabilitation plan for Block A (North-West) in the Ex-Mega Rice Project area, Central Kalimantan. A report prepared for the Kalimantan Forests and Climate Partnership. Euroconsult Mott MacDonald/Deltares|Delft Hydraulics, 2009.
Sodhi, N. S. et al. Artificial nest and seed predation experiments on tropical Southeast Asian islands. Biodiv. Conserv. 12, 2415–2433 (2003).
Van der Meer, P. J., Kunne, P. L. B., Brunsting, A. M. H., Dibor, L. A. & Jansen, P. A. Evidence for scatter-hoarding in a tropical peat swamp forest in Malaysia. J. Trop. For. Sci. 20, 340–343 (2008).
Brewer, S. W. & Rejmánek, M. Small rodents as significant dispersers of tree seeds in a Neotropical forest. J. Vegetat. Sci. 10, 165–174 (1999).
Kitamura, S., Yumoto, T., Poonswad, P., Suzuki, S. & Wohandee, P. Rare seed-predating mammals determine seed fate of Canarium euphyllum, a large-seeded tree species in a moist evergreen forest, Thailand. Ecol. Res. 23, 169–177 (2008).
Payne, J. & Francis, C. M. A field guide to the mammals of Borneo. The Sabah Society, Kota Kinabalu, 2005.
Aide, T. M. & Cavelier, J. 1994. Barriers to lowland forest tropical restoration in the Sierra Nevada de Santa Marta, Colombia. Restor. Ecol. 2, 219–229 (1994).
Osunkoya, O. O. Postdispersal survivorship of north Queensland rainforest seeds and fruits: effects of forest, habitat and species. Austral. Ecol. 19, 52–64 (1994).
Hammond, D. S. Post-dispersal seed and seedling mortally of tropical dry forest trees after shifting agriculture, Chiapas, Mexiso. J. Trop. Ecol. 11, 295–313 (1995).
Blate, G. M., D. R. Peart & M. Leighton . Postdispersal predation on isolated seeds: a comparative study of 40 tree species in a Southeast Asian rainforest. Oikos 82, 522–538 (1998).
Wells, K. & Bagchi, R. Eat in or take away - Seed predation and removal by rats (Muridae) during a fruiting event in a dipterocarp rainforest. Raffles Bull. Zool. 53, 281–286 (2005).
Wenny, D. G. Seed dispersal, seed predation and seedling recruitment of a Neotropical montane tree. Ecol. Monogr. 70, 331–351 (2000).
Myster, R. W. (2003) Effects of species, density, patch-type and season on post-dispersal seed predation in a Puerto Rican pasture. Biotropica 35, 542–546 (2003).
Fornara, D. A. & Dalling, J. W. Seed bank dynamics in five Panamanian forests. J. Trop. Ecol. 21, 223–226 (2005).
Gibson, L. et al. Near-complete extinction of native small mammal fauna 25 years after forest fragmentation. Science, 341, 1508–1510 (2013).
Charles, J. K. & Ang, B. B. Non-volant small mammal community responses to fragmentation of kerangas forests in Brunei Darussalam. Biodiv. Conserv. 19, 543–561 (2010).
Wells, K., Kalko, E. K. V., Lakim, M. B. & Pfeiffer, M. Effects of rain forest logging on species richness and assemblage composition of small mammals in Southeast Asia. J. Biogeogr. 34, 1087–1099 (2007).
Acknowledgements
We would like to thank the Universitas Palangka Raya and the Borneo Orangutan Survival Foundation for their facilitation and logistical support of this research and Agus and Suwan for field assistance. Research permission was granted by the Indonesian Ministry of Research and Technology (RISTEK) (Research permit: No: 016/SIP/FRP/I/2011), Direktorat Jenderal PHKA, Balai Konservasi Sumber Daya Alam and BAPPEDA Palangka Raya and the BOS Scientific Advisory Board. J.M. Blackham and E.L. Webb provided useful comments and discussion. This study was supported by the Singapore-Delft Water Alliance peatland research programme (R 264-001-004-272) and publication was funded by the 1000 Talents Program (WQ20110491035).
Author information
Authors and Affiliations
Contributions
G.V.B. and R.T.C. designed the project and wrote the paper. G.V.B. did the fieldwork and analysed the data. Both authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Blackham, G., Corlett, R. Post-dispersal seed removal by ground-feeding rodents in tropical peatlands, Central Kalimantan, Indonesia. Sci Rep 5, 14152 (2015). https://doi.org/10.1038/srep14152
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep14152
This article is cited by
-
Secondary seed removal in a degraded forest habitat in Madagascar
Scientific Reports (2021)
-
Vertebrate frugivory on jackfruit Artocarpus heterophyllus Lam. (Moraceae) in its native and exotic ranges
Tropical Ecology (2021)
-
Neighbour effects do not always show consistent patterns, contrast of seed trait matters: evidence from a seed-rodent mutualism study
Behavioral Ecology and Sociobiology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.