Abstract
The visual magnocellular-dorsal (M-D) deficit theory of developmental dyslexia (DD) is still highly debated. Many researchers have made great efforts to investigate the relationship between M-D dysfunction and reading disability. Given that visual analysis plays an important role in Chinese reading, the present study tried to examine how the M-D dysfunction affected Chinese character recognition in Chinese children with DD. Sixteen DD children with M-D deficit, fifteen DD children with normal M-D function and twenty-seven age-matched typically developing children participated in this study. A global/local decision task was adopted, in which we manipulated the spatial frequency of target characters to separate an M-D condition from an unfiltered condition. Results of reaction times and error rates showed that in the M-D condition both M-D normal dyslexics and controls exhibited a significant global precedence effect, with faster responses and lower error rates in global decision than in local decision. In contrast, this global advantage was absent for the M-D impaired dyslexics. Accordingly, we propose that the M-D impairment present in some but not all dyslexics might influence global recognition of Chinese characters in this subgroup of children with DD, which might be implicated in their difficulties in learning to read.
Similar content being viewed by others
Introduction
Visual processing is the first step required in reading and written words need to be visually perceived precisely in order for their sound and meaning to be accessed1. The visual magnocellular-dorsal (M-D) pathway is one of the (mainly) parallel processing streams in the primate visual system. It starts from the magno-cells in the retina, from which axons project onto the magnocellular layers of the lateral geniculate nucleus (LGN), after which visual signals are passed to the primary visual cortex and other brain areas for cortical conduction including the visual motion areas (V5/MT), posterior parietal cortex (PPC) and orbitofrontal cortex2. The M-D stream is sensitive to visual stimuli with low spatial frequency, low contrast and high temporal frequency, such as stimuli with blurred contours or fast movement3. Studies of typically developing children have shown that visual M-D functions are closely associated with reading-related measures such as orthographic skills4, reading accuracy5, reading speed6 and reading comprehension7. Meanwhile, the visual M-D pathway is also responsible for visuo-spatial attention that contributes to visual processing of graphemes, the translation from graphemes to phonemes and phonological awareness8.
The visual magnocellular-dorsal deficit theory of developmental dyslexia (DD) proposes that the core cause of dyslexia is an impaired M-D stream9. Various studies have shown that individuals with developmental dyslexia exhibit M-D deficits10. It has been reported that individuals with dyslexia show poor performance11 and abnormal patterns of neural activity12 in response to fast changing/moving stimuli and to visual stimuli with low spatial frequency13. Because the M-D function controls visuo-spatial attention, relevant impairments of visuo-spatial attention have also been found in dyslexics, such as low scores in the visual search task8. Meanwhile, some results point in the direction of a causal role of the M-D dysfunction in dyslexia. A previous study14 demonstrated that visual spatial attention in pre-school children could predict their future reading-related skills in the 1st and 2nd grades. Dyslexic children have also been found to show lower M-D sensitivity than not only age-matched controls but also reading-level matched controls15. A recent research showed a possible genetic base of the M-D deficit16. The authors found that dyslexic children with a deletion in intron 2 of the DCDC2 gene (DCDC2d) exhibited higher thresholds of illusory visual motion perception than dyslexic children without DCDC2d, age-matched controls and reading-level controls; meanwhile the DCDC2d was also found to be associated with illusory motion perception in adult normal readers. These findings suggested that the M-D deficit was a potential neurobiological risk factor of DD rather than a simple effect of reading disorder16. Furthermore, it has been reported that training the visual attention skills of the dyslexics improved their reading speed, unveiling the causal role of attention in reading acquisition17. Given the visuo-spatial function controlled by the M-D stream8, the above finding seemed to provide direct evidence for the causal link between the M-D deficit and dyslexia. Accordingly, some researchers proposed a new remediation approach of perceptual learning for individuals with dyslexia (especially for the pre-reading children at DD risk)18. The authors believed that the low level training in perceptual learning could improve the general functionality of the M-D stream and further contribute to reading acquisition18. However, findings of some studies do not support the M-D deficit theory19. For example, some studies have found that children with dyslexia exhibited a general deficit in visual processing which was not limited to the M-D stream but also appeared in other visual streams such as the parvocellular-ventral (P-V) stream19. Some researchers have suggested that these inconsistent findings might be related with the heterogeneity of dyslexia20. Lovegrove et al. found about 75% individuals with dyslexia exhibited an impaired M-D stream21, whereas some researchers have pointed out that the prevalence of the dyslexics with M-D impairment might be overestimated11,22. A recent review23 reported that about 90% of studies specifically on the M-D deficit in dyslexics have shown that most of the dyslexics do suffer from mild M-D impairments, particularly displayed in tests employing low contrasts, high temporal frequencies and low spatial frequencies. Additionally, some researchers have indicated that dyslexia is not due to a single factor; instead, it is a multifactorial deficit24,25. One relevant study25 found that almost 92% (55/60) of the dyslexics exhibited more than two deficits, while only one dyslexic individual (60 dyslexic individuals in total) merely exhibited the M-D dysfunction, suggesting that the M-D deficit together with other risk factors lead to dyslexia. Although the visual M-D deficit theory of developmental dyslexia is still highly debated, it seems clear that at least some children with dyslexia do have impairments in M-D functions6,10.
How does the M-D dysfunction in dyslexics affect their reading? Researchers have made great efforts to determine what the relationship is between M-D stream deficiency and reading disability. Recently, a temporal sampling framework has been hypothesized based on a host of studies on neuronal oscillatory mechanisms related to the temporal sampling of speech. This framework offers a possible account of the causal role of the M-D deficit in reading impairments in dyslexia26,27,28. According to the temporal sampling hypothesis, visual coding of text requires a large population of neurons to be responding synchronously for a coherent percept of printed text to be formed27. During reading, a number of complex and interrelated components (e.g. letter/word recognition, visual search, guiding eye movement and attentional shifting) are mediated by the M-D stream which is implicated in synchronous gamma oscillations27. Therefore, it has been proposed that the M-D dysfunction might result in abnormal neural activity of gamma synchronization, which would further interrupt the coherent percepts of texts before letter and word recognition27,28. Although a great amount of research has been devoted to understanding synchronous oscillatory activity in dyslexia, most of the relevant studies of the temporal sampling framework have focused on the auditory domain (e.g. using speech perception tasks). However, reading is mainly a visual process and thus studies involving in visual components seemed to be more suitable to analyze the visual M-D deficit of dyslexia. Based on visual researches which found that low spatial frequency influencing M-D activity facilitated global processing in object identification2, some researchers have suggested that this M-D function can also exert an influence in the processing of printed text29. In the context of alphabetic scripts, Allen et al. found that participants made lexical decisions more quickly for lowercase English words (based on the whole word) than for mixed-case words (based on component letters) in the M-D condition29. The authors proposed a multistream model of visual word recognition to explain these results. This model postulates a race to a central processor by information in different visual streams. The low spatial frequency of a target word was sensitive to the M-D activity and a first-pass global image of the word was quickly generated which would facilitate the global recognition of this words29. Boden and Giaschi directly manipulated the spatial frequency of English words and did not find any advantage in the global recognition of low-spatial-frequency words30. However, in Boden et al.'s study, the accuracy of most participants was below 50% in the low-spatial-frequency condition while above 90% in other visual conditions (i.e. higher-spatial-frequency condition and unfiltered condition) and this task difficulty difference might interact with different spatial frequencies.
With a logographic writing system, the visual configuration of a Chinese character is markedly different from that of an alphabetically-written word and it has been suggested that visual processing played a more important role in Chinese reading31. Recognition of Chinese characters requires the reader to analyze the holistic information of character forms32. Previous studies using global/local decision tasks in adult readers with normal reading ability have found that the advantage of global recognition relative to local recognition for Chinese characters becomes more substantial as spatial frequency decreases, suggesting that the low-spatial-frequency sensitivity of the M-D stream might play a role in global recognition of Chinese characters33,34. Studies using the coherent motion detection task35,36, the temporal order judgement task37, the texture discrimination task38 and the moving grating task39 have yielded results indicating that Chinese children with dyslexia exhibit impairments in M-D functions. The study by Meng et al. indicated that 14 of 27 dyslexics (about 52%) were identified as individuals having M-D deficits35. However, to our knowledge, there has been no direct investigation of the relationship between M-D dysfunction of Chinese children with dyslexia and their ability in Chinese character recognition.
The aim of the present study was to examine whether M-D dysfunctions in Chinese children with developmental dyslexia affect Chinese character recognition. We used a global/local decision task and recruited three groups of participants: dyslexic children with M-D dysfunction, dyslexic children with normal M-D function and age-matched control children with normal M-D function and normal reading ability. Based on previous studies33,34, we investigated three issues: (i) if both the typically developing children and the dyslexics with normal M-D function show a significant global precedence effect in the M-D condition while the dyslexic children with M-D deficit do not, this would suggest that the abnormality in Chinese character recognition might be attributed to visual M-D dysfunction; (ii) if both dyslexic groups showed abnormal pattern of Chinese recognition in the M-D condition comparing with the age-matched control group, this would suggest that the disabled recognition of Chinese characters might not be specially due to the impaired M-D stream; (iii) if the two dyslexic groups exhibit global advantage in the M-D condition similar to that shown by the typically developing children, this would suggest that the impaired M-D stream in dyslexics might not have any negative influence on the global recognition of Chinese characters.
Results
Sixteen DD children with M-D deficit, fifteen DD children with normal M-D function and twenty-seven age-matched typically developing children participated in the present experiment. The dataset of one dyslexic child with M-D deficit was eliminated due to his higher error rate (>50%). Correct reaction times longer than three standard deviations away from the mean were excluded (68 observations, about 1.21% of the total). The remaining data were submitted to further analysis. Figure 1 showed the means of reaction times (RTs) and error rates (ERs) in each condition.
A three-way mixed ANOVA was conducted on RTs with group (typically developing children, M-D normal dyslexics, M-D impaired dyslexics) as a between-subject factor and decision type (global vs. local decision) and visual condition (M-D vs. unfiltered conditions) as two within-subject factors. The results of ANOVA showed a significant main effect of decision type [F(1,54) = 6.24, P = 0.02], with longer reaction time in local decision than in global decision. There was no significant main effect of visual condition [F(1,54) = 2.10, P = 0.15] nor of group [F(2,54) = 1.31, P = 0.28]. The interactions of decision type × group [F(2,54) = 2.22, P = 0.12], visual condition × group [F(2,54) = 0.40, P = 0.68] and decision type × visual condition [F(1,54) = 2.46, P = 0.12] were not significant. The triple interaction of group, decision type and visual condition was significant [F(2,54) = 4.30, P = 0.02]. Simple effect analyses showed that in the M-D condition, global decisions yielded shorter reaction times than local decision for both typically developing children [F(1,56) = 9.12, P = 0.004] and dyslexics with normal M-D function [F(1,56) = 6.22, P = 0.02], while there was no difference here for dyslexics with M-D deficit [F(1,56) = 0.54, P = 0.47]. In the unfiltered condition, typically developing children showed shorter reaction times in global decision than in local decision [marginally significant, F(1,56) = 3.59, P = 0.06], while there was no significant difference between global and local decision for the other two groups (M-D normal DD: [F(1,56) = 0.71, P = 0.40]; M-D impaired DD: [F(1,56) = 0.87, P = 0.36]). The group effect was non-significant for any condition (M-D condition & global decision: F(2,54) = 1.17, P = 0.32; M-D condition & local decision: F(2,54) = 0.84, P = 0.44; unfiltered condition & global decision: F(2,54) = 2.42, P = 0.10; unfiltered condition & local decision: F(2,54) = 0.87, P = 0.43). The visual condition effect was only significant in local decision for the typically developing children, with longer reaction times in the M-D condition than in the unfiltered condition [F(1,56) = 5.25, P = 0.03]; while non-significant in any other conditions (P > 0.1 for all).
The same ANOVA was conducted on ERs. The results showed a significant main effect of group [F(2,54) = 3.57, P = 0.04]. Post hoc analysis revealed that both groups of dyslexics exhibited higher error rates than typically developing children (P < 0.05 for both), while there was no significant difference between two dyslexic groups (P = 0.19). The main effect of decision type was significant [F(1,54) = 28.50, P < 0.001], with higher error rates for local response. The main effect of visual condition was also significant [F(1,54) = 94.08, P < 0.001], with higher error rates in the M-D condition than in the unfiltered condition. There were no significant two-way interactions (decision type × group: [F(2,54) = 2.57, P = 0.09], visual condition × group: [F(2,54) = 0.63, P = 0.54], decision type × visual condition: [F(1,54) = 1.20, P = 0.28]). The triple interaction of decision type, visual condition and group was significant [F(2,54) = 3.41, P = 0.04]. Simple effect analyses showed that each group had higher error rates in the M-D condition than in the unfiltered condition for both global decision and local decision (P < 0.05 for all). The group effect in the M-D condition was significant for local decision [F(2,54) = 4.69, P = 0.01] but not for global decision [F(2,54) = 0.25, P = 0.78] and post hoc analysis showed that dyslexics with normal M-D function made more errors than participants from the other two groups (P < 0.05 for both), with no difference between dyslexics with M-D deficit and typically developing children (P = 0.69). For the unfiltered condition, the group difference was also only significant in local response [F(2,54) = 4.01, P = 0.02]. Post hoc analysis showed higher error rates for dyslexics from each group comparing with the typically developing children (P < 0.05 for both) and no differences between the two dyslexic groups (P = 0.62). As to the effect of decision type, dyslexics with normal M-D function and typically developing children both showed significant effects in the M-D condition [M-D normal DD: F(1,56) = 18.03, P < 0.001; CA: F(1,56) = 3.10, P = 0.08, marginally significant] with higher error rates for local decision than for global decision, but not the dyslexics with M-D deficit [F(1,56) = 0.83, P = 0.37]. In the unfiltered condition, the two dyslexic groups exhibited lower error rates in global decision than in local decision (M-D normal DD: [F(1,56) = 6.28, P = 0.02]; M-D impaired DD: [F(1,56) = 9.66, P = 0.003]), while there was no difference here for the control group [F(1,56) = 1.82, P = 0.18].
Discussion
The present study explored whether impaired M-D function in dyslexics affects Chinese character recognition. In the M-D condition, age-matched control readers and dyslexic children with normal M-D function exhibited significant global advantage, but the M-impaired dyslexics did not. In the unfiltered condition, either results of RTs or ERs exhibited global advantage in all the groups, but the global precedence effect was smaller in the M-D condition than in the unfiltered condition for dyslexics with normal M-D function and typically developing children.
Age-matched typically developing children exhibited greater global precedence effects in the M-D condition comparing with the unfiltered condition. According to the multistream model of visual word recognition29, the M-D stream is responsible for passing on global information about visual stimuli whereas the P-V stream is responsible for passing on local information and the M-D stream usually processes information more quickly than the P-V stream. In the M-D condition, the M-D stream would be fully activated relatively to the unfiltered condition and therefore the difference between global and local recognition might be magnified, resulting in a larger global precedence effect2,29. The current findings were consistent with previous studies on adults33,34. These findings thus suggest that in Chinese children who are not reading-impaired the role of the M-D stream's low-spatial-frequency sensitivity in Chinese character recognition is just the same as it is in adults.
However, the present results differ from findings with alphabetic readers, where the global advantage is not found in studies of word recognition in adult readers with normal reading ability30. The inconsistency is presumably due to visual differences between alphabetic words and logographic characters, since Chinese characters are generally much more visually complex than words written alphabetically, so that visual processing has been thought to contribute more to processing of a logographic script such as Chinese as processing of alphabetic scripts31. Low spatial frequencies could provide information about general shape and location of written Chinese characters; visual spatial analysis and holistic processing play a critical role in Chinese-character processing32. Therefore, we propose that the role of the low-spatial-frequency sensitivity of the M-D stream in global recognition of print texts might be more important in Chinese reading than in alphabetic reading.
In the present study, dyslexic children with normal M-D function showed a larger global advantage in the M-D condition than in the unfiltered condition just as typically developing children did, while the dyslexics with M-D deficit did not, which supports the issue (i) that these dyslexics' M-D dysfunction impedes their global recognition of Chinese characters and this deficit is not a general consequence of dyslexia itself but instead is specifically due to the impaired M-D stream in this subgroup of Chinese children with dyslexia. As suggested by Allen et al.'s model29, the M-D dysfunction of these dyslexic children would retard the global processing of character forms and diminish the global precedence effect. Results of previous studies with readers of alphabetic scripts have shown that participants with higher M-D ability exhibited better performance in reading-related tasks, such as lexical decision, non-word reading, reading fluency and reading accuracy as compared to those with lower M-D ability40,41. Since one of the core symptoms for DD is difficulty in single word/character decoding which will further bring about deficits of sentence/text reading42,43, the present study focused on the global processing of single Chinese characters. It has been reported that the global recognition of Chinese characters plays a role in orthographic processing during reading32,44. Accordingly, we propose that the absence of the global precedence effect in Chinese dyslexic children with M-D deficits might have a negative impact on orthographic processing of Chinese characters and some other reading-related procedures. These findings may have practical implications for therapeutic intervention in dyslexia: In addition to improving linguistic ability, therapies should focus on deficits in visual processing skills to improve M-D functioning in the individuals with dyslexia who have impaired M-D functioning. In the context of alphabetic languages, it has been shown that a period of training of the dyslexics' visual attention skills, in which the attentional network is part of the M-D stream, can improve their reading fluency17. Moreover, some researchers have proposed the use of perceptual learning remediation in assisting individuals with dyslexia18. As to (some) Chinese children with dyslexia, future studies are urgently required to examine whether the basic training of visual M-D function could be helpful to the enhancement of reading ability.
Considering the reaction time and error rate results, all the participant groups showed global advantage when recognizing unfiltered characters. Previous studies using unfiltered visual stimuli have also found faster response in global processing than in local processing45,46, which agrees with the present results revealing the global precedence effect. In the unfiltered condition, visual stimuli would not fully activate the M-D stream47,48. Accordingly, although there were differences in M-D function across groups, it was assumed that group effect in global advantages would not be apparent due to insufficient activation of the M-D stream.
In the present study, dyslexics with normal M-D streams exhibited similar performance as typically developing children in Chinese character recognition in the M-D condition, while the dyslexic children with M-D deficit showed impaired recognition of Chinese characters in the M-D condition. These findings demonstrated that the impaired M-D stream might exert an influence on Chinese reading for a subset of dyslexics, i.e that the M-D deficit theory of dyslexia holds for some dyslexics though not for others.
A recent study49 has suggested that the M-D dysfunction in individuals with dyslexia is a consequence of limited reading experience i.e. is a consequence rather than a cause of reading disability. If that is so, in the present study, all dyslexics should show a deficiency in M-D function relative to age-matched typically developing children, because all would have had impoverished reading experience; yet we found a group of dyslexics who showed M-D functioning equivalent to that of nondyslexics. Therefore, our findings challenge the general idea that our dyslexics' M-D impairment was due to their impoverished reading experience.
As we have said, the existence of dyslexics with normal M-D function suggests that an M-D deficit cannot be a general explanation for dyslexia in Chinese, but could be the neural basis for one subtype of dyslexia in Chinese. The dyslexics with normal M-D function we studied would thus represent another subtype (or more than one other subtype) of dyslexia in Chinese; their dyslexia might for example be attributed to an abnormal neural mechanism for phonological processing50, abnormal morphological awareness which has been suggested to be one of the core deficits in Chinese children with dyslexia51, or to some other relevant impaired mechanisms52, but not of course to an M-D deficit. Exactly this appears also to be true for dyslexia in languages written alphabetically, since some but not all children with dyslexia who are readers of English show impaired M-D functioning and some do not40,41. These are of course not the only two subtypes of dyslexia seen in children who are readers of alphabetic scripts; Coltheart and Kohnen53 describe the characteristics of six such subtypes. Further work on Chinese children with dyslexia who do not have impaired M-D function might reveal a similar degree of heterogeneity i.e. multiple subtypes of dyslexia in Chinese.
Methods
Participants
Sixteen dyslexic children with M-D deficit (9 boys), fifteen dyslexic children with normal M-D function (11 boys) and twenty-seven age-matched typically developing children (13 boys) were selected from 592 forth-grade and fifth-grade students in four primary schools. All participants were right-handed as judged by the Handedness Inventory (Department of Neurology, Beijing Medical University Hospital) and had normal or corrected-to-normal vision without ophthalmologic or neurological abnormalities. No participant suffered from ADHD, judged by the Chinese Classification of Mental Disorder 3 (CCMD-3). Detailed information of each group is presented in Table 1. Informed consent was obtained from parents and teachers. The research project was approved by the Research Ethics Committee of the Institute of Psychology, Chinese Academy of Sciences. And the methods in the current study were carried out in accordance with the approved guidelines.
Psychometric tasks administered to identify dyslexics
In order to screen the dyslexics, three tasks which are widely used to identify children with developmental dyslexia in Mainland China35,36,37,38,39 were adopted:
-
a
Raven's Standard Progressive Matrices (RSPM)54, for testing nonverbal intelligence. RSPM is a standardized test of nonverbal intelligence with six sets of twelve items. For each item, children were shown a matrix with one missing part. The matrices comprised patterns and children were required to complete the patterns by selecting from six to eight alternatives. Difficulty increases as the test progresses. The raw score was the number of correct choices and the standardized score was converted from the raw score based on the Chinese norms established by Zhang and Wang54, as shown in Table 1.
-
b
A rapid digit naming test55. This test was used to measure participants' rapid automatized naming ability. In this task, five Arabic digits (2, 7, 4, 9 and 6) were presented from left to right on an A4 sheet in random order six times, with six rows and five digits per row. Participants were asked to read the 30 digits twice as quickly and accurately as possible from left to right, row by row. The time taken was recorded and the score was the average time of the two readings.
-
c
A standardized written vocabulary test56. This Chinese Character Recognition Test was adopted to examine participants' reading skill35,36,38,39. In this test, participants are required to write down a compound word based on a written target morpheme provided on the sheet. For example, the written target Chinese character might be “
 ” (pronounced/jian3/, meaning cut (the number here refers to tone); the child's task is to write another morpheme next to the target to form a real two-character word. Characters are divided into 10 groups based on reading difficulty (174 characters for 4th graders and 210 characters for 5th graders). A child responded correctly if he/she wrote, for example, “
” (pronounced/jian3/, meaning cut (the number here refers to tone); the child's task is to write another morpheme next to the target to form a real two-character word. Characters are divided into 10 groups based on reading difficulty (174 characters for 4th graders and 210 characters for 5th graders). A child responded correctly if he/she wrote, for example, “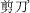 ” (/jian3 dao1/, which means scissors), or “
” (/jian3 dao1/, which means scissors), or “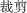 ” (/cai2 jian3/, which means cut out), or wrote other genuine word that includes the target morpheme “
” (/cai2 jian3/, which means cut out), or wrote other genuine word that includes the target morpheme “ ”. Each correct response was given one point. The score for each group of characters was calculated by multiplying the total points by the corresponding coefficient of difficulty. The final score for each participant was the sum of subscores for all 10 character groups.
”. Each correct response was given one point. The score for each group of characters was calculated by multiplying the total points by the corresponding coefficient of difficulty. The final score for each participant was the sum of subscores for all 10 character groups.
The inclusion criteria for the dyslexics were that nonverbal intelligence was in the normal range (i.e. the standard score of RSPM was greater than 85), while the score on the written vocabulary was at least 1.5 standard deviations below the average score of the same-grade children and the score on the rapid digit naming test was poorer than the mean score of age-matched control group. In this way, 31 children with dyslexia (prevalence of 5.2%) were identified. Table 1 presents the results of this psychometric testing.
Testing M-D and P-V functioning
Steady-pedestal and pulsed-pedestal paradigms designed by Porkney and Smith57 were used to assess the functioning of participants' M-D and P-V streams respectively, via measurement of spatial-frequency contrast thresholds. This paradigm can separate the M-D stream from the P-V stream based on differences in spatial-frequency contrast gains between the two visual pathways and accordingly it is regarded as one of the typical measurements of visual M-D/P-V functions57,58,59. Here visual stimuli consist of horizontal or vertical sinusoidal gratings with a visual angle of 5° × 5°. The computer monitor was viewed from 50 cm. The peak spatial frequency of gratings was 0.5 cycles per degree (cpd) in the M-D condition (steady-pedestal) and 5 cpd in the P-V condition (pulsed-pedestal)60. As in previous studies57,60, the luminance pedestal stayed on the black background in the M-D condition and only appeared together with the target grating in the P-V condition. Within each trial, a four-dot array was presented in the center of the screen for 1500 ms and then a target grating appeared for 500 ms. Participants were asked to determine the orientation of the target with different key presses, “z” for vertical and “n” for horizontal and the response window lasted for 3000 ms. A fixation array was displayed in the center of the screen in the random interval (from 1000 ms to 1500 ms) between two successive trials.
A two-yes-one-no staircase procedure was applied to estimate contrast thresholds, which terminated after 10 reversals. The minimum value of luminance contrast was 0.005. According to a pre-study, we set the step sizes thus: 0.03 for the first three reversals, 0.015 for the 4th-6th reversals and 0.005 for the last four reversals. The average contrast for the last six reversals was taken to estimate the contrast threshold (Table 1). The staircase started from the contrast value above the predictable contrast threshold (M-D condition: 0.1; P-V condition: 0.25) and the presenting procedure of staircase was programmed with Eprime 1.1.
Deviance analysis35,37 was adopted to select the individuals who had deficits in M-D stream but not in P-V stream. Individuals were classified with an M-D or a P-V deficit if their sensitivity scores on the relevant task (steady-pedestal or pulsed-pedestal) were poorer than the one tail 95% confidence limit of the control group (1.65 standard deviations) after the controls with extreme scores were removed from the sample. According to the above method35,37, we respectively calculated the cutoff for identifying M-D and P-V impaired individuals: 0.035 for the M-D deficit and 0.045 for the P-V deficit. Individuals with contrast thresholds higher than the cutoff values, which meant lower sensitivity, would be classified as having a deficit. Finally, sixteen of the 31 children with dyslexia (52%) were identified as having an impaired M-D stream and the remaining 15 children with dyslexia had a normal M-D stream. None of the participants, typically developing children or dyslexics, showed P-V dysfunction. Table 1 presents the results of the M-D and P-V testing.
Experimental task
This was a global/local decision task with two visual conditions: a filtered (i.e magnocellular-dorsal) condition and an unfiltered condition. The spatial frequency of target characters was 1.5 cpd in the M-D condition; these characters were generated by the software ImageJ 1.42q on a DELL laptop with a 12.1 inch monitor. In the unfiltered condition, the spatial frequency of target characters was not manipulated. There were 80 pseudo-characters with left-right structure as targets, 40 for each visual condition (see Figure 2 for examples of these). On each trial, the target character was followed by a probe stimulus. Forty pseudo-characters were used as probes in global decision, in which twenty of the probe stimuli were identical to the target characters. The other forty non-character radicals were used as probes in the local decision task, in which half of the probes were parts of the targets and half were not. In the local decision task, six radical probes were presented on the right side of the screen center and the other fourteen on the left side. The probe stimuli were never spatial-frequency filtered. The number of stimulus strokes was balanced between M-D and unfiltered conditions.
The presentation format of each trial in different conditions.
Trials (a) and (b) represent the magnocellular-dorsal condition. Trials (c) and (d) represent the unfiltered condition. Trials (a) and (c) required local decisions, as indicated by the rectangular cue; the cue is on the left in (a) and on the right in (c); the correct response for (a) is DIFFERENT and for (c) is SAME. Trials (b) and (d) required global decisions, as indicated by the square cue; the correct response for (b) is SAME and for (d) is DIFFERENT.
Participants were seated comfortably 50 cm from the computer monitor in a dark and sound-attenuated room, with a visual angle of 3.2° × 3.2°. The display resolution was set at 1024 × 768 and the refresh rate was 62.3 Hz.
In each trial, a 1000-ms fixation, a 500-ms target character, a 1000-ms cue and a probe stimulus were sequentially presented in the screen center (Figure 2). The probe disappeared upon response, or at the end of a 3000-ms response window. Between two adjacent trials, a fixation stayed in the screen for a variable interval ranging from 1000 ms to 1500 ms. The cue was presented following the target to inform participants about which decision (global or local decision) should be made. For global decisions, the cue was a square in the screen center (visual angle: 3.2° × 3.2°) and for local decisions the cue was a half-square in the screen center (visual angle: 3.2° × 1.6°) (see Figure 2). Participants were required to make a decision on whether the probe was the same as the target in global tasks or the probe was the same as the relative part of the target in local tasks. SAME decisions were indicated by pressing the Z key and DIFFERENT decisions by pressing the N key. This experiment was programmed with Eprime 1.1, reaction time and error rate were recorded.
There were two sessions in the formal experiment, one session for the M-D condition and the other for the unfiltered condition. Within each session, global and local decision randomly appeared with equal probability. There were ten practice trials before each session. The sequence of the two sessions was counterbalanced across participants.
References
Rauschecker, A. M. et al. Visual Feature-Tolerance in the Reading Network. Neuron 71, 941–953 (2011).
Bar, M. et al. Top-down facilitation of visual recognition. Proc. Natl Acad. Sci. USA 103, 449–454 (2006).
Stein, J. F. & Walsh, V. To see but not to read; the magnocellular theory of dyslexia. Trends. Neurosci. 20, 147–152 (1997).
Pammer, K. & Kevan, A. The contribution of visual sensitivity, phonological processing and non-verbal IQ to children's reading. Sci. Stud. Read. 11, 33–53 (2007).
Kevan, A. & Pammer, K. Predicting early reading skills from pre-reading measures of dorsal stream functioning. Neuropsychologia 47, 3174–3181 (2009).
Laycock, R. & Crewther, S. G. Towards an understanding of the role of the ‘magnocellular advantage’ in fluent reading. Neurosci. Biobehav. R. 32, 1494–1506 (2008).
Pammer, K. & Wheatley, C. Isolating the M(y)-cell response in dyslexia using the spatial frequency doubling illusion. Vis. Res. 41, 2139–2147 (2001).
Vidyasagar, T. R. & Pammer, K. Dyslexia: a deficit in visuo-spatial attention, not in phonological processing. Trends Cogn. Sci. 14, 57–63 (2010).
Harrar, V. et al. Multisensory Integration and Attention in Developmental Dyslexia. Curr. Biol. 24, 531–535 (2014).
Boden, C. & Giaschi, D. M-Stream Deficits and Reading-Related Visual Processesin Developmental Dyslexia. Psychol. Bull. 133, 346–366 (2007).
Conlon, E. G., Sanders, M. A. & Wright, C. M. Relationships between global motion and global form processing, practice, cognitive and visual processing in adults with dyslexia or visual discomfort. Neuropsychologia 47, 907–915 (2009).
Eden, G. F. et al. Abnormal processing of visual motion in dyslexia revealed by functional brain imaging. Nature 382, 66–69 (1996).
Livingstone, M., Rosen, G. D., Drislane, F. W. & Galaburda, A. M. Physiological and anatomical evidence for a magnocellular deficit in developmental dyslexia. Proc. Natl Acad. Sci. USA 88, 7943–7947 (1991).
Franceschini, S., Gori, S., Ruffino, M., Pedrolli, K. & Facoetti, A. A causal link between visual spatial attention and reading acquisition. Curr. Biol. 22, 814–819 (2012).
Gori, S., Cecchini, P., Bigoni, A., Molteni, M. & Facoetti, A. Magnocellular-Dorsal Pathway and Sub-Lexical Route in Developmental Dyslexia. Front. Hum. Neurosci. 8, 460 (2014).
Gori, S. et al. The DCDC2 intron 2 deletion impairs illusory motion perception unveiling the selective role of magnocellular-dorsal stream in reading (dis)ability. Cereb. Cortex, in press (2014).
Franceschini, S. et al. Action video games make dyslexic children read better. Curr. Biol. 23, 462–466 (2013).
Gori, S. & Facoetti, A. Perceptual learning as a possible new approach for remediation and prevention of developmental dyslexia. Vis. Res. 99, 78–87 (2014).
Farrag, A. F., Khedr, E. M. & Abel-Naser, W. Impaired parvocellular pathway in dyslexic children. Eur. J. Neurol. 9, 359–363 (2002).
Valdois, S., Bosse, M. L. & Ainturier, M. J. The cognitive deficits responsible for developmental dyslexia: Review of evidence for a selective visual attentional disorder. Dyslexia 10, 339–363 (2004).
Lovegrove, W., Slaghuis, W. L., Bowling, A. C., Nelson, P. & Geeves, E. Spatial frequency processing and the prediction of reading ability: a preliminary investigation. Percept. Psychophys. 40, 440–444 (1986).
Ramus, F. et al. Theories of developmental dyslexia: Insights from a multiple case study of dyslexic adults. Brain 126, 841–865 (2003).
Stein, J. Visual Contributions to Reading Difficulties: The Magnocellular Theory. Visual aspects of dyslexia. Stein, J. & Kapoula, Z. (eds.) 405–464 (Oxford University Press, Oxford, UK, 2012).
Bolzani, R. et al. Developmental dyslexia is a multifactor disorder: The neuropsychological approach. Perception 35, 91–92 (2006).
Menghini, D. et al. Different underlying neurocognitive deficits in developmental dyslexia: a comparative study. Neuropsychologia 48, 863–872 (2010).
Goswami, U. A temporal sampling framework for developmental dyslexia. Trends Cogn. Sci. 15, 3–10 (2011).
Pammer, K. Temporal sampling in vision and the implications for dyslexia. Front. Hum. Neurosci. 7, 933 (2013).
Vidyasagar, T. R. Reading into neuronal oscillations in the visual system: implications for developmental dyslexia. Front. Hum. Neurosci. 7, 811 (2013).
Allen, P. A., Smith, A. F., Lien, M. C., Kaut, K. P. & Canfield, A. A multistream model of visual word recognition. Atten. Percept. Psychophys. 71, 281–296 (2009).
Boden, C. & Giaschi, D. The role of low-spatial frequencies in lexical decision and masked priming. Brain. Cognition 69, 580–591 (2009).
McBride-Chang, C. et al. Early predictors of dyslexia in Chinese children: Familial history of dyslexia, language delay and cognitive profiles. J. Child Psychol. Psyc. 52, 204–211 (2011).
Tan, L. H., Laird, A. R., Li, K. & Fox, P. T. Neuroanatomical Correlates of Phonological Processing of Chinese Characters and Alphabetic Words: A Meta-Analysis. Hum. Brain Mapp. 25, 83–91 (2005).
Guo, X. C. Global precedence in the early perception of Chinese character recognition (in Chinese). Psychological Science (in Chinese), 23, 576–581 (2000).
Zhao, J., Bi, H. Y. & Qian, Y. The Influence of Visual Magnocellular Pathway on The Recognition of Chinese Character (in Chinese). Prog. Biochem. Biophys. (in Chinese), 40, 141–146 (2013).
Meng, X. Z., Cheng-Lai, A., Zeng, B., Stein, J. F. & Zhou, X. L. Dynamic visual perception and reading development in Chinese school children. Ann. Dyslexia 61, 161–176 (2011).
Qian, Y. & Bi, H. Y. The visual magnocellular deficit in Chinese-speaking children with developmental dyslexia. Front. Psychol. in press (2014).
Chung, K. K. H. et al. The role of visual and auditory temporal processing for Chinese children with developmental dyslexia. Ann. Dyslexia 58, 15–35 (2008).
Wang, Z. K. et al. A Perceptual Learning Deficit in Chinese Developmental Dyslexia as Revealed by Visual Texture Discrimination Training. Dyslexia in press (2014).
Wang, J. J., Bi, H. Y., Gao, L. Q. & Wydell, T. N. The visual magnocellular pathway in Chinese-speaking children with developmental dyslexia. Neuropsychologia 48, 3627–3633 (2010).
Levy, T., Walsh, V. & Lavidor, M. Dorsal stream modulation of visual word recognition in skilled readers. Vis. Res. 50, 883–888 (2010).
Sperling, A. J., Lu, Z. L., Manis, F. R. & Seidenberg, M. S. Selective magnocellular deficits in dyslexia: a “phantom contour” study. Neuropsychologia 41, 1422–1429 (2003).
Nation, K. & Snowling, M. J. Semantic processing and the development of word-recognition skills: Evidence from children with reading comprehension difficulties. J. Mem. Lang. 39, 85–101 (1998).
Yap, R. & Van Der Leij, A. Word processing in dyslexics. Read. Writ. 5, 261–279 (1993).
Kuo, W. J. et al. Orthographic and phonological processing of Chinese characters: an fMRI study. NeuroImage 21, 1721–1731 (2004).
Choi, E. J., Jang, K. M. & Kim, M. S. Electrophysiological correlates of local–global visual processing in college students with schizotypal traits: An event-related potential study. Biol. Psychol. 96, 158–165 (2004).
Poirel, N., Pineau, A. & Mellet, E. What does the nature of the stimuli tell us about the Global Precedence Effect? Acta Psychol. 127, 1–11 (2008).
Calderone, D. J. et al. Contributions of Low and High Spatial Frequency Processing to Impaired Object Recognition Circuitry in Schizophrenia. Cortex 23, 1849–1858 (2013).
Skottun, B. C. The magnocellular deficit theory of dyslexia: the evidence fromcontrast sensitivity. Vis. Res. 40, 111–127 (2000).
Olulade, O. A., Napoliello, E. M. & Eden, G. F. Abnormal visual motion processing is not a cause of dyslexia. Neuron 79, 1–11 (2013).
Boets, B. et al. Intact But Less Accessible Phonetic Representations in Adults with Dyslexia. Science 342, 1251–1254 (2013).
Shu, H., McBride-Chang, C., Wu, S. & Liu, H. Y. Understanding Chinese developmental dyslexia: Morphological awareness as a core cognitive construct. J. Educ. Psychol. 98, 122–133 (2006).
Butterworth, B. & Kovas, Y. Understanding Neurocognitive Developmental Disorders Can Improve Education for All. Science 340, 300–305 (2013).
Coltheart, M. & Kohnen, S. Acquired and developmental disorders of reading and spelling. Handbook of the Neuropsychology of Language. Faust, M. (ed.) (Blackwell, Oxford, UK, 2012).
Zhang, H. C. & Wang, X. P. Raven's Standard Progressive Matrices (in Chinese). (Beijing Normal University Press, Beijing, China, 1985).
Denckla, M. B. & Rudel, R. G. Rapid “automatized” naming (R.A.N): dyslexia differentiated from other learning disabilities. Neuropsychologia 14, 471–479 (1976).
Wang, X. L. & Tao, B. P. Chinese character recognition test battery and assessment scale for primary school children (in Chinese). (Shanghai Education Press, Shanghai, China, 1996).
Pokorny, J. & Smith, V. C. Psychophysical signatures associated with magnocellular and parvocellular pathway contrast gain. J. Opt. Soc. Am. 14, 2477–2486 (1997).
Pokorny, J. Review: steady and pulsed pedestals, the how and why of post-receptoral pathway separation. J. Vis. 11, 1–23 (2011).
Goodbourn, P. T. et al. Do different ‘magnocellular tasks’ probe the same neural substrates? Proc. R. Soc. B 279, 4263–4271 (2012).
Leonova, A., Pokorny, J. & Smith, V. C. Spatial frequency processing in inferred PC- and MC-pathways. Vis. Res. 43, 2133–2139 (2003).
Acknowledgements
This research was supported by Chinese Natural Science Foundation (31371044, 30970910) to Hongyan Bi.
Author information
Authors and Affiliations
Contributions
J.Z. designed and wrote the manuscript. Y.Q. performed the experiment and analyzed data. H.Y.B. supervised the project and wrote the manuscript. M.C. implemented the computerized experiment and wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Zhao, J., Qian, Y., Bi, HY. et al. The visual magnocellular-dorsal dysfunction in Chinese children with developmental dyslexia impedes Chinese character recognition. Sci Rep 4, 7068 (2014). https://doi.org/10.1038/srep07068
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep07068
This article is cited by
-
Enhanced reading abilities is modulated by faster visual spatial attention
Annals of Dyslexia (2022)
-
Magnocellular Based Visual Motion Training Improves Reading in Persian
Scientific Reports (2019)
-
Increased deficit of visual attention span with development in Chinese children with developmental dyslexia
Scientific Reports (2018)
-
The DCDC2 deletion is not a risk factor for dyslexia
Translational Psychiatry (2017)
-
On forward inferences of fast and slow readers. An eye movement study
Scientific Reports (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.





 ” (pronounced/jian3/, meaning cut (the number here refers to tone); the child's task is to write another morpheme next to the target to form a real two-character word. Characters are divided into 10 groups based on reading difficulty (174 characters for 4th graders and 210 characters for 5th graders). A child responded correctly if he/she wrote, for example, “
” (pronounced/jian3/, meaning cut (the number here refers to tone); the child's task is to write another morpheme next to the target to form a real two-character word. Characters are divided into 10 groups based on reading difficulty (174 characters for 4th graders and 210 characters for 5th graders). A child responded correctly if he/she wrote, for example, “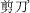 ” (/jian3 dao1/, which means scissors), or “
” (/jian3 dao1/, which means scissors), or “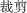 ” (/cai2 jian3/, which means cut out), or wrote other genuine word that includes the target morpheme “
” (/cai2 jian3/, which means cut out), or wrote other genuine word that includes the target morpheme “ ”. Each correct response was given one point. The score for each group of characters was calculated by multiplying the total points by the corresponding coefficient of difficulty. The final score for each participant was the sum of subscores for all 10 character groups.
”. Each correct response was given one point. The score for each group of characters was calculated by multiplying the total points by the corresponding coefficient of difficulty. The final score for each participant was the sum of subscores for all 10 character groups.