Abstract
The epithelial lining of the intestine is characterized by an immense cellular turn-over ascertaining an extensive regenerative capacity. Multiple reports suggest that besides the local intestinal stem cell pool, circulating cells of bone marrow origin (BMDCs) contribute to this process by fusing with the epithelial lineage. However, the functional relevance of these observations is unknown. In the present study we employ a model system in which we cannot only detect cell fusion but also examine the functional importance of this process in vivo. Our results indicate that fusion between BMDCs and intestinal epithelial cells is an extremely rare event under physiological conditions. More importantly, by employing a system in which fusion-derived cells can be specifically deleted after extensive tissue damage, we present evidence that cell fusion is not relevant for tissue regeneration. Our data decisively demonstrates that intestinal epithelial homeostasis and regeneration is not dependent on cell fusion involving BMDCs.
Similar content being viewed by others
Introduction
The intestinal epithelial layer is one of the most rapidly renewing tissues in the mammalian body1. A tightly regulated stem cell compartment located at the bottom region of the crypt regulates the generation of progenitor cells, which in turn give rise to the different lineages of differentiated epithelial cells, such as Goblet cells and enterocytes. Recent research has revealed that the intestinal stem cell population can be identified based on marker genes including Lgr5 and Ascl22,3. Especially in case of epithelial damage – for example due to inflammation or toxic substances – a great demand rests on these cells to regenerate the epithelial lining and maintain tissue integrity. Besides the local intestinal stem cell pool there has been speculation on the possibility that another source of cells may be involved in the regeneration of the intestinal epithelium after damage. These cells are believed to be of bone marrow origin and are referred to as bone marrow derived cells (BMDCs). A main hypothesis involving BMDCs is that they revitalize the local intestinal stem cell pool by fusing with epithelial cells, leading to an exchange of (damaged) genetic material and potentially installing an intestinal stem cell-like epigenetic program, mediated by factors in the cytoplasm4,5. In support of this theory there are several lines of evidence. The initial observations come from human female subjects that underwent sex-mismatch bone marrow (BM) transplantations as part of their leukaemia therapy, in these women Y-chromosomes could be detected in multiple tissues including the epithelial compartment of the intestine6,7,8,9. Similar results have been obtained in mouse models that involve sex-mismatch bone-marrow transplantation10. These finding always left open the possibility that it was mere transdifferentiation of BMDCs into the epithelial lineage more than cell fusion between epithelial cells and circulating hematopoietic cells. However, this issue is partially resolved by recent studies in which mouse models were employed that harbour genetic marker elements (e.g. GFP, Y-chromosome, β-galactosidase) in both the transplantation derived hematopoietic system as well as in the recipient tissues, including the epithelial lining of the gut5,11,12. In these models exchange of genetic elements is observed that could potentially be the result of cell fusion events as markers of both the donor and the recipient could be detected in individual epithelial cells. However, other explanations remain possible including the exchange of microvesicles or apoptotic bodies containing genetic material as well as inaccuracies during the detection procedure13. To date the exact nature of the BMDC involved in fusion remains unresolved however most evidence points to either mesenchymal stem cells or macrophages12.
Regardless of the hematopoietic cell type involved or the exact mechanism by which exchange of genetic material takes effect we wished to explore the functional consequences of the transfer of genetic material between BMDCs and epithelial cells in intestinal homeostasis. In order to do this we generated a mouse model system in which we could not only study the occurrence of transfer, but simultaneously the role of cell-fusion between BMDCs and tissue specific cells in a variety of organs. We confirmed the functionality of our model system in the liver in which fusion has been reported using a similar approach and at a similar frequency14,15. Surprisingly, we found that in our system cell fusion with- and transdifferentiation of, BMDCs into intestinal epithelial cells is extremely rare, if it occurs at all. Even in cases where extensive intestinal tissue damage was induced by irradiation or inflammation no fusion events could be detected. More importantly, we present functional evidence that strongly suggest that cell fusion is not involved in homeostasis or tissue repair in the intestine. We therefore conclude that the local intestinal stem cell pool is sufficient to maintain tissue integrity also during episodes of gross epithelial damage.
Results
Characteristics of a mouse model system to study cell fusion
To explore the occurrence of cell fusion in intestinal tissue renewal we devised a mouse model system that allows both for the detection of fusion-derived epithelium as well as selective eradication of these cells. Crucially, the latter allows investigation of the functional role of fusion between BMDCs and solid organ-specific cells. To this end we employed previously described mouse models that use the Cre-Lox recombination system16,17,18. Detection of fusion events involving BMDCs is achieved by transplanting mice that carry a LoxP controlled inducible Green Fluorescence Protein (iGFP)17, with BM isolated from CMV-Cre mice18 ( Fig. 1a ). Fusion of solid organ-specific cells with transplantation-derived hematopoietic cells results in the transfer of genetic material between Cre-expressing BMDCs and LoxP-controlled GFP containing cells. The result of such a fusion event will be detectable due to the recombination event on the iGFP locus, which is a permanent genetic mark and therefore also carried over in the offspring ( Fig. 1b ). This system has been successfully used before to detect in vivo cell fusion15. To be able to ablate the fusion derived cells we also crossed the iGFP mice into a strain of inducible-Diphtheria Toxin Receptor (iDTR) mice16 ( Fig. 1a ), giving rise to iDTR-GFP mice. Cre mediated recombination following a fusion event in this strain results in, next to GFP positivity, DTR expression from the ROSA26 locus, which allows for selective eradication of fusion-derived cells after injection of Diphtheria Toxin (DT).
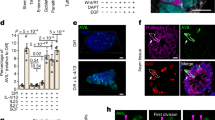
Mouse model system to study cell fusion.
(a) Schematic representation of the constructs the mice in this study harbour. The iGFP (also known as Z/EG) mice express constitutively β-galactosidase, however Cre mediated recombination involving the LoxP sites results in expression of enhanced GFP (EGFP). The iDTR construct, allows for Cre inducible DTR expression. In the CMV-Cre mouse Cre expression is driven by the constitutively active CMV promoter. (b) Depicted is a fusion event between a Cre expressing cell and an iDTR-GFP cell. Cre mediated recombination results in both GFP and DTR expression. (c) Efficient engraftment of donor bone marrow is demonstrated in iDTR mice transplanted with GFP+ bone marrow (derived from CMV-Cre x iGFP mice). Full blood is analyzed 6 weeks after transplantation. (d and e) GFP+ hepatocytes can be detected by IHC in the liver of both iDTR-GFPBM:Cre mice (d) and iDTRBM:GFP mice (e) >2 months after transplantation. (d and e) scale bars indicate 100 μm. (f) Quantification of GFP+ cells in the liver reveals that ∼1:4000 hepatocytes is fusion derived in iDTR-GFPBM:Cre mice 2–4 months after transplantation (n = 4). These cells can be efficiently ablated after intraperitoneal injection of DT (n = 3) as no GFP+ cells could be detected in up to ∼72,000 cells. # indicates no GFP+ could be detected in the indicated cumulative amount of cells from 3 mice. *** indicates p<0.001 as determined by one-sample t-test.
To confirm the efficiency of our BM transplantation protocol we used BM from a GFP+ strain (CMV-Cre mice crossed with iGFP) and transplanted it into GFP-, iDTR mice. Extensive engraftment (>93%) of GFP+ BM could be observed 6 weeks after transplantation ( Fig. 1c and Supplementary Fig. 1a ). Subsequently, we evaluated the validity of our system by confirming the presence of fusion-derived hepatocytes in iDTR-GFP mice transplanted with Cre-expressing BM (iDTR-GFPBM:Cre) ( Fig. 1d ). Also using iDTR mice transplanted with GFP+ BM (iDTRBM:GFP), GFP+ cells can be detected in the liver ( Fig. 1e ). Quantification of the GFP+ hepatocyte numbers indicates that fusion of BMDCs with liver cells occurs at similar rates to what has been reported before14 ( Fig. 1f ). Crucially, consistent with the coordinated expression of DTR and GFP in fused cells, DT administration in the iDTR-GFPBM:Cre results in rapid and efficient ablation of fusion-derived cells as no GFP+ cells could be detected up to ∼72.000 cells analyzed in multiple mice (n = 3) ( Fig. 1f ). This indicates that recombination events following cell fusion resulting in GFP and DTR expression occur at similar rate. More evidence for efficient eradication of DTR-expressing cells is obtained by incubating isolated splenocytes from iDTR mice crossed with CMV-Cre mice, i.e. expressing DTR constitutively in all tissues, with DT ( Supplementary Fig. S2 ). This resulted in a complete eradication of the DTR-expressing cells. Together these data confirm the functionality of the model to study the occurrence and relevance of cell fusion between BMDCs and solid organ specific cells in vivo.
Fusion of BMDCs with intestinal epithelial cells is a rare event
In order to investigate the presence of fusion-derived cells in the intestinal epithelial tissue we analyzed iDTR-GFP mice transplanted with Cre expressing BM (iDTR-GFPBM:Cre mice, Fig. 2a ) as well as the reverse experiment; CMV-Cre mice transplanted with BM from iDTR-GFP mice (CMV-CreBM:iDTR-GFP mice, data not shown). Fusion events in these mice can be detected by GFP expression as the fused cells, as well as their offspring, will be GFP+. We evaluated both the small intestine (SI) as well as the colon for GFP expression using immunohistochemistry (IHC). From each mouse (n = 12) multiple sections were evaluated, comprising a total of ∼8000 crypts ( Fig. 2a ). However, no fusion-derived GFP+ cells or crypt structures could be detected. Importantly, these same mice did demonstrate fusion events in the liver ( Fig. 1d ). To validate whether our model system is functional for investigating fusion events in the intestine, we firstly demonstrated that the iGFP transgenic locus is active in intestinal epithelial cells, as mice containing a recombined iGFP locus (iGFP x CMV-Cre mice) demonstrated widespread and high expression of GFP in a wide range of tissues including the intestine ( Fig. 2a ). Moreover, the active expression of Cre recombinase in intestinal epithelial cells in CMV-Cre mice was verified ( Supplementary Fig. S3 ) and the functional capability of the stem cell compartment was unaffected, as CMV-Cre mice were equally efficient as control mice in initiating intestinal organoid cultures ( Supplementary Fig. S4 ). Furthermore, Cre mediated recombination of the iGFP locus in intestinal epithelial cells occurs effectively, as transduction of iGFP organoid cultures with a lentiviral Cre expression vector results in rapid GFP expression ( Supplementary Fig. S5 ). Overall, these findings support the notion that the model we employ is, in principle, well suited for the detection of fusion events in the intestine.
IHC reveals lack of fusion in the intestine.
(a) IHC staining for GFP in colon and the SI. Left panel; positive control demonstrates widespread intense staining for GFP in the GFP+ mouse (CMV-Cre x iGFP). No GFP staining can be detected in the colon (middle panel) or SI (right panel) of iDTR-GFPBM:Cre mice. (b) MALT tissue displays extensive presence of donor derived GFP+ cells of hematopoietic origin in the intestine. (c) iDTRBM:GFP mice display widespread GFP staining in the stromal compartment of the colon. No GFP+ epithelial cells were detected. Similarly in SI tissue GFP+ in the stroma can be readily detected, however, no evidence for GFP+ epithelial cells is observed. (d) Immunofluorescence co-staining for GFP and EpCAM demonstrates no GFP+/EpCAM+ cells both in iDTR-GFPBM:Cre as well as in iDTRBM:GFP mice. (a–c) Representative examples of at least 3 separate mice are demonstrated. Scale bars indicate 50 μm.
To evaluate the existence of cell fusion in a more liberal system we transplanted iDTR mice with GFP+ BM (iDTRBM:GFP). Cellular fusion in this model can be detected in the intestinal epithelial compartment without the need for a recombination event. Importantly in these mice also transdifferentiation of BMDCs will result in GFP positivity of the epithelial cells in the gut. Using this model we observed efficient repopulation of the hematopoietic cells in the stromal compartment of the intestine (>60%) after injection of GFP+ BMDCs ( Supplementary Fig. S1b ). Also patches of mucosa associated lymphoid tissue (MALT) in the gut contain high amounts of GFP+ cells ( Fig. 2b ). However, also in this setting no GFP expression was detected in the epithelial compartment both in the colon ( Fig. 2c , left panel, n = 4) as well as in the SI ( Fig. 2c , right panel, n = 6). This finding was confirmed by lack of EpCAM/GFP double positive cells in an immunofluorescence staining ( Fig. 2d and Supplementary Fig. S6 ).
To rule out the possibility that GFP expression is below detection level by immunostaining or that the frequency of cell fusion is too rare to asses by IHC we proceeded by evaluating fusion occurrence by FACS on intestinal epithelial cell isolates. We confirmed that isolation of the epithelial compartment by EDTA dissociation result in a pure epithelial cell populations (>99%, Supplementary Fig. S7a and S7b ). Marked GFP expression in epithelial cells isolated from GFP+ mice was detected ( Fig. 3a , left panel). However, also with this detection method no evidence for fusion in the iDTR-GFPBM:Cre (n = 4) and CMV-CreBM:iDTR-GFP (n = 3) could be observed in both the colon and SI ( Fig. 3a and Supplementary Fig. S7c ). In iDTRBM:GFP mice a small fraction of GFP+ cells was present in the epithelial cell isolates, however backgating on the EpCAM channel establishes these are contaminating cells of non-epithelial origin ( Fig. 3b ). In multiple experiments (n = 6), we measured up to 80,000 EpCAM+ events, indicating that fusion-derived cells are less frequent than that number.
FACS demonstrates lack of fusion in the intestine also after full body radiation.
(a) Left panel demonstrates high GFP expression in the intestinal epithelial isolate of GFP+ mice. Middle panel; confirms lack of GFP expression in non-transplanted iDTR-GFP mice. Right panel reveals lack of fusion occurrence in iDTR-GFPBM:Cre mice up to 6 months after transplantation. More examples as well as CMV-CreBM:iDTR-GFP mice are depicted in Supplementary Fig. S7c . (b) Epithelial cell isolates demonstrate a small population of GFP+ cells in iDTRBM:GFP mice. Backgating of this population reveals these cells are EpCAM- and therefore do not represent epithelial cells. Representative example of 3 analyses is depicted. (c and d) Full body irradiation (8Gy) of both iDTR-GFPBM:Cre (c) and iDTRBM:GFP (d) mice does not result in increased fusion rate in the intestine (n = 2).
These data indicate that fusion of BMDCs with intestinal epithelial cells is an extremely rare event as detected by iGFP recombination. To evaluate the occurrence of fusion mediated recombination on the iDTR locus we used a semi-quantitative PCR approach. However, also with this technique we did not observe evidence of fusion which is more frequent than 1 in every 1000 cells in isolated crypt structures ( Supplementary Fig. S8a and S8b ). The original studies suggesting the occurrence of fusion in the intestine use FISH detection of the Y-chromosome in the gut in females that underwent sex-mismatch BM transplantations6,7,10. There are several limitations to this technique as it potentially both underestimates the number of cells containing a Y-chromosomes (due to sectioning of the nucleus) and on the other hand detection of Y-chromosomes in cells could be misleading in case nuclei overlay each other resulting in inadequate calls of Y-chromosome positive cells5. We carefully evaluated and quantified the presence of Y-chromosomes in the epithelial compartment of the gut in female mice that underwent BM transplantations from male mice ( Supplementary Fig. S8c and S8d ). Also with this technique no evidence for either significant fusion or transdifferentiation of BMDCs with-, or into, epithelial cells could be detected in our system.
Tissue damage in the intestine does not result in increased fusion events
Cell fusion between intestinal epithelial cells and BMDCs might serve a functional role during tissue damage repair. Indeed, multiple reports suggest that tissue damage in multiple tissues results in an increase in fusion events, also in the intestine. To evaluate these previous results we irradiated iDTR-GFPBM:Cre and iDTRBM:GFP mice with a single dose of 8Gy (n = 2). This is reported to be an adequate dose to cause extensive damage to rapidly dividing tissues including the epithelial lining of the gut19. Two weeks after irradiation we evaluated GFP expression both by FACS and by IHC, also in this setting no fusion-derived cell population in the gut could be detected ( Fig. 3c and 3d , IHC data are not shown).
Lack of functional relevance of cell fusion in tissue homeostasis and repair
Because we did not detect fusion in the intestine to the extent as reported before, it could either be that fusion indeed is a very rare event or alternatively that the fusion-derived cells only reside in the epithelial cell population for a very short time, but still fulfil an important physiological role. To evaluate this we induced intestinal tissue damage using dextran sulfate sodium (DSS), resulting in an extensive inflammatory response, disrupting intestinal epithelial integrity20. We speculate that in such a process fusion-mediated events can help to sustain the barrier function of the epithelial compartment in the gut. Indeed administration of DSS in the drinking water results in tissue damage and altered crypt morphology as well as a high hematopoietic cell influx ( Fig. 4a and 4b ). Moreover, mice lose weight in episodes following DSS administration to different extents depending on the DSS concentration ( Fig. 4c ). This reflects the intestinal damage that hampers nutrient uptake and the increased energy demand associated with the inflammatory response. To evaluate the role of cell fusion in regeneration of the intestine after DSS-induced damage we used iDTR mice transplanted with BM from GFP+ mice (iDTRBM:GFP; fusion events result in DTR expression) and we evaluated mouse weight following subsequently 1.5% and 3% of DSS administration in the drinking water for a 5 day period for each dose. In the DT-treated groups mice received DT intraperitoneally every 3 days to ablate the fusion-derived cells. In this way fusion-derived epithelial cells and their offspring, can not contribute to the process of tissue repair following DSS treatment. It appears that both the DT-treated, fusion knockout mice (n = 7) and the control mice (n = 6) react on DSS treatment in a similar fashion. Importantly the rate at which tissue damage is repaired after DSS administration is discontinued, as evidenced by increase in body weight, is comparable between both conditions ( Fig. 4c ). Scores of DSS-associated tissue damage in the gut including total intestinal weight and total intestinal length, both reflecting influx of hematopoietic cells are similar ( Fig. 4d ). Crucially, also histology of mice from the DT-treated, fusion knockout group and the control mice display no significant differences with respect to crypt size and sustained crypt loss ( Fig. 4e and 4f ). Importantly also DSS treatment did not appear to result in increased fusion rates ( Supplementary Fig. S9 ). Overall this demonstrates that no significant functional role for cell fusion involving BMDCs exists in the gut in a situation in which widespread tissue damage repair is taking place.
Cell fusion is dispensable for intestinal tissue repair.
(a) Treatment of mice with 3% DSS during a 5 day period results in widespread cell influx in the intestinal stroma and marked epithelial damage. (b) Increased influx of GFP+ BMDCs in iDTRBM:GFP mice can be detected after DSS treatment. (a and b) scale bars indicate 100 μm. (c) Graph depicts average mouse body weights during a period in which iDTRBM:GFP mice were subsequently exposed to 1,5% and 3% DSS in the drinking water. Mice in the DT treated group received DT injections intraperitoneally every three days to eradicate the fusion derived, DTR expressing cells. No significant difference between the two experimental groups is observed (error bars represent standard deviation, control group n = 6, DT treatment group n = 7). (d) Both weight (p = 0.44) and length (p = 0.81) of mouse intestines in both groups do not differ significantly. (e) Histological evaluation of colon tissue from iDTRBM:GFP treated with DT displays similar morphology and damage repair after DSS treatment as the non-DT treated control mice. Scale bars in upper figures indicate 500 μm, in high magnification figures they represent 100 μm. (f) Crypt length (p = 0.67) and fraction of crypt loss (p = 0.53) indicates similar extend of intestinal tissue repair in the experimental groups. (d and f) P-values are calculated using t-test statistics. Error bars represent standard deviations.
Discussion
Our results demonstrate that fusion in the intestinal lining between epithelial cells and BMDCs is very rare, if it occurs at all. These results are in sharp contrast with earlier studies on the role of BMDCs in intestinal epithelial tissue. A pressing question that remains is what the reason for the discrepancy is between some of these previous studies describing widespread fusion between BMDCs and epithelial cells with the findings we report here. In one of these earlier reports it is even suggested that fusion events in the intestine produce cells with stem cell properties, because genetic markers of both the recipient mice (β-galactosidase) and BMDC donor mice (GFP) can be detected in epithelial cells throughout the crypt villus-axis5.
Clearly, no such events could be detected in our system, even not after extensive damage is induced (8Gy radiation or DSS treatment). The inability to detect fusion events does not correspond to the functionality of our model system, as all the prerequisites for optimal detection of fusion events in the intestinal epithelial layer are met. In our model system we could successfully detect fusion-derived GFP+ hepatocytes at numbers comparable to those reported before14. Moreover, we extensively addressed the possibility that tissue specific effects in intestinal epithelial cells on functionality of the transgenes involved affect our readout ( Supplementary Figs. S3 , S4 and S5 ). We confirmed expression of the transgenes relevant for fusion detection, Cre and GFP, in both the epithelial compartment and in the BMDCs. Importantly, we excluded the possibility that Cre expression in our model has a toxic effect on the intestinal stem cells, which has been described before for other stem cell compartments21, making it an unlikely explanation for the inability to detect fusion events. Furthermore, we demonstrate that recombination of the iGFP (Z/EG) locus occurs effectively in intestinal epithelial cells in response to Cre expression.
Our main conclusion that fusion events in the intestine are very rare is further supported by experiments involving transplantation of GFP-expressing BM. In mice transplanted with GFP+ BM, GFP+ cells can be widely detected in the intestinal stromal compartment, but show no evidence for either widespread transdifferentiation or fusion. In some cases we do observe individual GFP+ cells intermingled with epithelial cells but these are most likely of hematopoietic origin and presumably resemble macrophages and intestinal lumen sampling dendritic cells and these cells do clearly not express the epithelial marker EpCAM ( Figs. 2c and 3b and data not shown). The notion that the EpCAM negative cells were previously misdetected as epithelial cells is strengthened by the inability to find significant levels of Y-chromosome cells in the intestinal epithelial after sex-mismatch BM transplantations. To conclude, the quantification of fusion associated, Cre-mediated recombination events, further fail to demonstrate fusion in significant quantities (<1 in every 1000 cells), supporting the notion that cell fusion is indeed a very rare event.
Our results, which point to the irrelevance of fusion events in homeostasis and regeneration of intestinal epithelium, are supported by recent studies on the biology of intestinal stem cells. Elegant novel mouse models demonstrate an astonishing potential of these cells in renewing and regenerating the intestinal epithelial layer. Tracking of single intestinal stem cells demonstrate that these cells are equipped with the potential to rapidly generate the whole intestinal epithelial lineage including the various lineages of differentiated cells that are present in the epithelial layer2,22,23. Single intestinal stem cells have even been shown to generate in vitro organoid cultures which can be propagated indefinitely and contain the crucial characteristics of intestinal epithelium including multiple lineages of differentiated cells and preservation of a stem cell compartment24. To conclude it appears that different intestinal stem cell compartments are involved in day-to-day refreshing of the epithelial lining in homeostasis on one hand and regeneration of damaged epithelium, including the normal stem cell pool, after extensive tissue damage on the other25,26,27. Our results, in combination with these novel findings, put forward there is no need for an additional process, such as cell fusion, to explain the unique properties of the intestinal epithelium. This directly suggests that the extensive regenerative capacity of the intestinal epithelial compartment is entirely due to the locally residing intestinal stem cell compartments in close association with the mesenchyme that supports them.
Methods
Mice
Mice experiments were performed in agreement with the animal ethical committee at our institution (Academical Medical Center, Amsterdam, The Netherlands). All mice in this study were generated on a BL6 background and described previously. The iGFP mouse is best known as Z/EG mice and was ordered from Charles River17. iDTR mice16 were kept homozygous. iDTR-GFP mice were bred homozygous for the iDTR construct and heterozygous for the iGFP construct. CMV-Cre mice were bred homozygous18.
Transplantation studies
For transplantation studies full bone marrow was isolated from 4–8 weeks old mice and 5·106 cells were injected in either the tail vein or intraperitoneally in lethally irradiated recipient mice (6–10 weeks old). Lethal irradiation was performed by irradiating the mice twice with 6Gy, four hours apart as previously described5. Transplantation efficiency was assessed 6 weeks after transplantation by analysis of whole blood by FACS (GFP). To ablate fusion derived, GFP+ hepatocytes, mice were injected on day 1 and day 2 with 150 ng Diphtheria Toxin (DT) (Sigma Aldrich) in 300 µl PBS and sacrificed on day 3. GFP+ hepatocytes were quantified in two mice in each experimental condition and >30 full liver sections of each mouse were scored, the fraction of GFP+ cells determined by normalizing using the average number of hepatocytes in one field.
DSS model
Mice were administered either 1.5% or 3% of dextran sulfate sodium (DSS) (TdB Consultancy AB, DB001) in the drinking water for a period of 5 days. To ablate the fusion derived, DTR+ cells, DT was injected intraperitoneally every three days (150 ng DT in 300 µl PBS).
Immunostaining
Intestinal tissue was obtained from mice directly after they were sacrificed. Swiss rolls were prepared and fixed in 4% formalin prior to paraffin embedding. Sections of 5 µm were prepared on a microtome. For GFP staining antigen retrieval was performed using sodium citrate 10 mM in which slides were boiled for 10 min. Slides were incubated overnight with a mouse IgG1 anti-GFP antibody (Roche, 1814460001) at a final concentration of 1.6 µg/ml. Staining was developed using the ARKkit (DAKO, K3954) according to manufacturers instructions. Blocking of endogenous biotin was performed using Biotinblock (Vector Laboratories, SP2001). Immunofluorescence staining was performed using anti-EpCAM antibody diluted 1:25 (Abcam E144/ab32392), Cre antiserum diluted 1:100 (clone 2D8, Millipore, MAB3120) and anti-GFP antibody at a final concentration of 16 µg/ml and incubated for 16 hours at 4°C. Aspecific staining was blocked using 10% goat serum for 30 min. at room temperature. Secondary antibodies used; for EpCAM staining goat anti-rabbit Alexa 546 labeled (Invitrogen A11010) at final concentration 8 µg/ml, for Cre staining after ARKkit application Streptavidin conjugated Alexa 546 was used at a final concentration of 8 µg/ml (Invitrogen, S11225) and for GFP staining also after ARKkit application Streptavidin conjugated Alexa 488 was used at a final concentration of 8 µg/ml (Invitrogen, S32354). Nuclei were counterstained with DAPI (Roche 10236276001).
FACS analysis
FACS analysis was performed using the BD FACS Canto®. For EpCAM staining we used the EpCAM-APC antibody (Bioscience, 17-5791-82) at a final concentration of 2 µg/ml, incubation was performed at 4°C for 20 min. Dead cells were excluded using propidium iodide. Epithelial cell fractions were obtained by incubating intestinal tissue fragments (2×2 mm) with EDTA (colon: 2 mM, small intestine: 25 mM) in PBS for 30 min at 4°C.
PCR analysis
Recombination specific PCR of iDTR; forward primer: 5′-CGTGATCTGCAACTCCAGTC-3′, reverse primer: 5′-TAGATCCAGTGGAAGGGTCC-3′. Annealing temperature 58°C, 35 cycles of 1 min. iDTR recombination was assessed in DNA derived from isolated crypt structures from iDTR-GFPBM:Cre mice. For the Cre expression PCR RNA was isolated using the RNeasy kit (Qiagen, 74104) and treated with DNase (Qiagen, 79254). Cre forward primer: 5′-TCTCACGTACTGACGGTGG-3′, reverse primer: 5′-ACCAGCTTGCATGATCTCC-3′. Annealing temperature 64°C, 35 cycles of 1 min. Primers and PCR conditions for genotyping of the mice are available upon request.
FISH analysis
Epitope retrieval was performed similar to IHC procedure and followed by 30 min. pepsin incubation (Sigma, P6887) at a pH 1.5 at 37°C. Afterwards slides were dehydrated and incubated with 5 µl denaturated probe (XMP Y-Red, Metasystem, D1421-050-TR) and incubated for 3 min. at 80°C, followed by overnight incubation at 37°C. Afterwards slides were washed with 0.4x saline sodium citrate (SSC) buffer at 70°C for 5 min, after which slides were rinsed with 2xSSC buffer containing 0.1% NP 40 solution (Fluka BioChemika 74385) then slides were dehydrated and evaluated using nuclear counterstaining with DAPI.
Organoid cultures
Organoids of the small intestine were established from freshly isolated glands (described above) from the indicated mice. The culture conditions and growth factors supplemented have been described elsewhere24. For evaluation of crypt forming capacity in Cre expressing intestinal epithelial structures, fragmented gland structures from the duodenum were plated and the fraction of budding organoid structures was determined after 10 days for the indicated mice. Transduction of iGFP organoid structures was performed as described recently28 using a CMV-Cre lentiviral vector that has been described elsewhere29.
Statistical analysis
A student's t-test was performed for statistical analysis using two-tailed nonequal variances and 95% CI using GraphPad Software. Mean ± standard deviation is represented on bar charts unless specified otherwise in the legend.
References
Medema, J. P. & Vermeulen, L. Microenvironmental regulation of stem cells in intestinal homeostasis and cancer. Nature 474(7351), 318 (2011).
Barker, N. et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449(7165), 1003 (2007).
van der Flier, L. G. et al. Transcription factor achaete scute-like 2 controls intestinal stem cell fate. Cell 136(5), 903 (2009).
Ogle, B. M., Cascalho, M. & Platt, J. L. Biological implications of cell fusion. Nat Rev Mol Cell Biol 6(7), 567 (2005).
Rizvi, A. Z. et al. Bone marrow-derived cells fuse with normal and transformed intestinal stem cells. Proc Natl Acad Sci U S A 103(16), 6321 (2006).
Korbling, M. et al. Hepatocytes and epithelial cells of donor origin in recipients of peripheral-blood stem cells. N Engl J Med 346(10), 738 (2002).
Matsumoto, T. et al. Increase of bone marrow-derived secretory lineage epithelial cells during regeneration in the human intestine. Gastroenterology 128(7), 1851 (2005).
Okamoto, R. et al. Damaged epithelia regenerated by bone marrow-derived cells in the human gastrointestinal tract. Nat Med 8(9), 1011 (2002).
Spyridonidis, A. et al. Epithelial tissue chimerism after human hematopoietic cell transplantation is a real phenomenon. Am J Pathol 164(4), 1147 (2004).
Krause, D. S. et al. Multi-organ, multi-lineage engraftment by a single bone marrow-derived stem cell. Cell 105(3), 369 (2001).
Davies, P. S. et al. Inflammation and proliferation act together to mediate intestinal cell fusion. PLoS One 4(8), e6530 (2009).
Powell, A. E. et al. Fusion between Intestinal epithelial cells and macrophages in a cancer context results in nuclear reprogramming. Cancer Res 71(4), 1497 (2011).
Waterhouse, M. et al. Horizontal DNA transfer from donor to host cells as an alternative mechanism of epithelial chimerism after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant 17(3), 319 (2011).
Nygren, J. M. et al. Myeloid and lymphoid contribution to non-haematopoietic lineages through irradiation-induced heterotypic cell fusion. Nat Cell Biol 10(5), 584 (2008).
Harris, R. G. et al. Lack of a fusion requirement for development of bone marrow-derived epithelia. Science 305(5680), 90 (2004).
Buch, T. et al. A Cre-inducible diphtheria toxin receptor mediates cell lineage ablation after toxin administration. Nat Methods 2(6), 419 (2005).
Novak, A. et al. Z/EG, a double reporter mouse line that expresses enhanced green fluorescent protein upon Cre-mediated excision. Genesis 28(3–4), 147 (2000).
Schwenk, F. Baron, U. & Rajewsky, K. A cre-transgenic mouse strain for the ubiquitous deletion of loxP-flanked gene segments including deletion in germ cells. Nucleic Acids Res 23(24), 5080 (1995).
Onal, C. et al. Protective effects of melatonin and octreotide against radiation-induced intestinal injury. Dig Dis Sci 56(2), 359 (2011).
Melgar, S., Karlsson, A. & Michaelsson, E. Acute colitis induced by dextran sulfate sodium progresses to chronicity in C57BL/6 but not in BALB/c mice: correlation between symptoms and inflammation. Am J Physiol Gastrointest Liver Physiol 288(6), G1328–G1338 (2005).
Schmidt-Supprian, M. & Rajewsky, K. Vagaries of conditional gene targeting. Nat Immunol 8(7), 665 (2007).
Lopez-Garcia, C. et al. Intestinal stem cell replacement follows a pattern of neutral drift. Science 330(6005), 822 (2010).
Snippert, H. J. et al. Intestinal crypt homeostasis results from neutral competition between symmetrically dividing Lgr5 stem cells. Cell 143(1), 134 (2010).
Sato, T. et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459(7244), 262 (2009).
Takeda, N. et al. Interconversion between intestinal stem cell populations in distinct niches. Science 334(6061), 1420 (2011).
Tian, H. et al. A reserve stem cell population in small intestine renders Lgr5-positive cells dispensable. Nature 478(7368), 255 (2011).
Montgomery, R. K. et al. Mouse telomerase reverse transcriptase (mTert) expression marks slowly cycling intestinal stem cells. Proc Natl Acad Sci U S A 108(1), 179 (2011).
Koo, B. K. et al. Controlled gene expression in primary Lgr5 organoid cultures. Nat Methods (2011).
de Vries, N. A. et al. Rapid and robust transgenic high-grade glioma mouse models for therapy intervention studies. Clin Cancer Res 16(13), 3431 (2010).
Acknowledgements
We wish to thank G.R. van den Brink for useful discussion, A.F. Pacha for technical assistance with the FISH experiments as well as the animal caretaker for their contribution to this work. The iDTR mice were a kind gift of A. Waisman. We thank O. van Tellingen for kindly providing the CMV-Cre containing lentivirus. JPM is supported by a VICI grant from the Dutch Science Organization (NWO). The authors report no conflict of interest.
Author information
Authors and Affiliations
Contributions
J.H.d.J., C.D.Z., H.M.R, F.D.S.e.M., V.L. and L.V.; performed experiments, D.J.R, J.P.M. and L.V.; planned research and analyzed data, L.V.; wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplemental figures
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareALike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
de Jong, J., Rodermond, H., Zimberlin, C. et al. Fusion of intestinal epithelial cells with bone marrow derived cells is dispensable for tissue homeostasis. Sci Rep 2, 271 (2012). https://doi.org/10.1038/srep00271
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep00271
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.