Key Points
-
Provides overview of possible aetiology of gingival overgrowth.
-
Discusses history and key clinical features which aid in diagnosis.
-
Discussion of systemic disease which may contribute to gingival overgrowth
Abstract
Most commonly, gingival overgrowth is a plaque-induced inflammatory process, which can be modified by systemic disease or medications. However, rare genetic conditions can result in gingival overgrowth with non-plaque-induced aetiology. It is also important to appreciate the potential differential diagnoses of other presentations of enlarged gingival tissues; some may be secondary to localised trauma or non-plaque-induced inflammation and, albeit rarely, others may be manifestations of more sinister diseases or lesions. A definitive diagnosis will then enable an appropriate management strategy. This paper aims to discuss clinical features and diagnoses for conditions presenting with gingival overgrowth and other enlargements of gingival tissues.
Similar content being viewed by others
Background
Gingival overgrowth describes a generalised or localised enlargement of the gingival tissues. This term has replaced gingival hyperplasia (increase in cell number) and gingival hypertrophy (increase in cell size) as these are histological diagnoses and do not accurately describe the varied pathological processes seen within the tissues. It is now understood that true enlargement involves changes in the cell size, cell multiplication, gingival vasculature and the extracellular matrix to varying degrees.1,2
A number of conditions present as swelling of the gingivae, rather than overgrowth per se, and these have been included in this article for completeness.
Aetiology
Gingival diseases including gingival overgrowth can be categorised as 'plaque-induced' and non-plaque-induced;2,3 however, often a more specific primary aetiology can be identified (Table 1).
There are several conditions which are not reliant on plaque induction, being genetic, systemic or infective in nature.4 In these conditions, associated plaque accumulation may exacerbate the clinical presentation.1
Within this paper, aetiology has been broadly classified into local and systemic.
Local factors
Inflammatory gingival overgrowth
Chronic inflammatory changes are common in cases of gingival overgrowth.5 This may be a result of prolonged exposure to dental plaque,6 localised trauma or a combination of factors as outlined in Table 1.
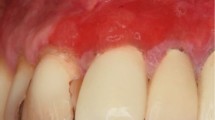
Clinical examination frequently reveals poor oral hygiene (Fig. 1). This may be secondary to tooth displacement, anatomical anomalies or dental work including prostheses (Fig. 2), poorly contoured restorations and orthodontic appliances, which favour the accumulation and retention of plaque.2,7
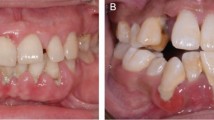
Gingival enlargement due to chronic inflammation has also been seen in mouth breathers and patients with incompetent lips (Figs 3a and b).8,9 Typically this appears as a red, oedematous lesion with a shiny surface.9 It is not fully understood why this occurs, it is theorised that it is due to chronic inflammation from long term surface dehydration.10 An association has also been made between obstructive sleep apnoea and increased periodontal disease, however, the causal affect is debatable.11
Localised gingival overgrowth may present as a fibrous epulis. It is considered that inflammation from local trauma can be exacerbated by poor plaque control and cause these lesions. Clinically, this appears as a firm, rubbery, pale pink swelling which may be sessile or pedunculated, often between two teeth. This is differentiated from the vascular pyogenic granuloma which presents as a diffuse swelling which is soft, shiny red-purple and bleeds readily (Fig. 4). There is likely to be plaque, calculus or food debris associated with these types of lesions.
Abscesses
Localised enlargement of the gingivae may also be attributed to abscesses related to dental or gingival tissues. Bacterial infection of an endodontic or periodontal origin may present as a buccal or palatal swelling, which may be at the gingival margin or closer to the sulcus if associated with periapical tissues.
These may have varying presentations but are often raised, fluctuant and erythematous as well as being tender to palpation. Pericoronitis around a partially erupted tooth may have a similar appearance.
Denture-induced fibrous inflammatory hyperplasia (FIH)
Denture-induced FIH occurs around the borders of an ill-fitting denture.12 Initially, a small ulcer may occur, which after chronic irritation from the flange can lead to inflammatory hyperplasia.12 It is often a raised, sessile mass in the form of folds with a smooth surface and normal mucosa colouring.
The lesions are benign and often asymptomatic; however, areas of chronic irritation and trauma may be predisposed to the development of neoplastic changes.13
False gingival enlargement
There may be an increase in the size of the gingival tissues due to the underlying hard tissues. Conditions that may cause this include benign bony tori or more sinister disease processes such as Paget's disease, fibrous dysplasia or cysts and tumours of odontogenic or other origins.10 The overlying tissues may appear normal or have coincidental inflammatory changes.10
Developmental gingival overgrowth involves the gingival tissues around erupting teeth.10 Bulky gingival tissues may occur overlying the unerupted/partially erupted dentition.10 This is usually self-limiting and will resolve once the junctional epithelium migrates to the cemento-enamel junction.10 If this does not happen, it is termed altered passive eruption and surgical intervention may be required to correct the clinical crown height. (Fig. 5).14
Systemic factors
Hormonal mediation of gingivitis
Pregnancy and puberty
Pregnancy can result in an increase in clinically significant gingival inflammation, which is seen even when there are no considerable differences in plaque scores.15 This is due to the modified host response to plaque.
Evidence demonstrates that an increase in circulating progesterone and oestrogen affect the composition of subgingival microflora, changing it to potentially more periodontally destructive species.15 Immunological changes during pregnancy cause individuals to be more susceptible to intra-cellular pathogens such as P. gingivalis, P. intermedia and A. actinomycetemcomitans, which avoid the host defences and are locally invasive.15 Sex hormones have an effect on the peripheral neutrophils, which reduce the effectiveness of phagocytosis and bactericidal mechanisms.15 They also have an effect on pro-inflammatory mediators such as prostaglandin E2, via endotoxin-stimulated monocytes.15 Finally, the sex hormones cause increased permeability of the blood vessels and reduce the keratinisation of the gingiva, which make the gingival tissue more susceptible to bacterial antigens.1
Pregnancy-related gingival enlargement may present as a generalised overgrowth (Fig. 6), or in the form of a localised pregnancy epulis (pyogenic granuloma) (Fig. 4).15 The pathogenesis of a pyogenic granuloma in pregnancy is not fully understood. It is thought to relate to the effect of the sex hormones stimulating an increased local synthesis of angiogenic factors, such as vascular endothelial growth factor.15 These lesions will not normally persist after pregnancy; however, occasionally surgical removal is indicated.15
During puberty, chronic gingival overgrowth may occur and it is usually self-limiting.10 The mechanism is similar to that of pregnancy, an altered level of sex hormones is associated with increased gingival inflammation, altered gingival microflora and increased presence of serum antibodies.16
Oral contraceptive pill
Oral contraceptives are one of the most commonly prescribed medications.17 Some clinical case reports have suggested gingival enlargement linked to the use of oral contraceptives.18 Preshaw et al. found no effect of modern low dose oral contraceptives on gingival tissues.19
Drug-influenced gingival overgrowth (DIGO)
Gingival overgrowth is a side effect of a number of medications,20,21 where the lesions are clinically and histologically indistinguishable from one another.21 If these medications are prescribed in combination there will often be a synergistic effect, worsening the clinical picture.22
Drug-induced gingival overgrowth tends to occur in the anterior gingivae,20 with onset typically within three months of starting the medication.23 It is not associated with attachment loss or tooth mobility, unless there is concurrent periodontal disease. Clinically lesions have a granular or pebbly surface that can coalesce to form lobules. This may obscure the clinical crown height or even cause tooth movement if enlargement is significant.24
The influence of plaque on the disease process is not fully understood;20 however, as with many inflammatory processes the severity of the lesion is affected by oral hygiene and erythema, oedema and bleeding on probing may be secondary.25
A recent review discusses a unified hypothesis of DIGO which involves a decrease in the active transport of folic acid in fibroblasts, and reduced cellular folate uptake.26 This leads to changes in matrix metalloproteinases and failure to activate collagenases, which decreases degradation of accumulated connective tissue.26 Further research is required to investigate whether folic acid supplements may have beneficial impact of DIGO.26
Anti-convulsants
Many patients diagnosed with epilepsy or with traumatic head injuries will be prescribed anti-convulsant medications. Of these, phenytoin has been implicated in inducing gingival overgrowth in up to 50% of patients (Fig. 7).27 The suggested theory is that genetically distinct populations of fibroblasts react to phenytoin, resulting in accumulation of connective tissues in susceptible individuals. There is also potentially a reduced catabolism of the collagen molecule within the gingival tissues.20
Phenytoin has largely been superseded by newer anti-convulsant drugs, and is no longer recommended as a first-line therapy in the treatment of epilepsy. As a result the prevalence of phenytoin-induced gingival overgrowth should be decreasing. Rarely, DIGO has also been reported in those taking other antiepileptic drugs such as valproic acid, phenobarbital and carbamazepine, however, these drugs appear to cause milder gingival enlargement.28
Calcium channel blockers
Calcium channel blocking drugs are used in the management of cardiovascular diseases, such as hypertension. It has been estimated that they are prescribed for 6% of the UK population over 40 years of age.24
It is reported that up to 20% of patients taking nifedipine and diltiazem experience gingival overgrowth (Fig. 8).24,25,29 Other hypertensive medications including amlodipine, felodipine and verapamil may also be implicated,20 however, research into this area largely pre-dates the widespread use of these drugs so the true association remains unknown.
The mechanism for gingival overgrowth in these cases has been attributed to stimulation of gingival fibroblasts which increases connective tissue matrix formation. In addition, it is suggested that there is reduced production of matrix metalloproteinases (reducing protein breakdown).24,30
Immunosuppressants
Immunosuppressant medications are often prescribed to patients receiving organ transplants in order to control the immune response to a foreign tissue.23 These include ciclosporin and tacrolimus.
Gingival overgrowth occurs in around 25–30% of patients taking the immunosuppressant ciclosporin (Fig. 9).20 The principal theory is that the main metabolite, hydroxyciclosporin, in combination with the parent compound, stimulates fibroblast proliferation.31 This increase in cell number and reduction in gingival connective tissue breakdown has been the speculated aetiology.20
Tacrolimus is now more frequently prescribed as an alternative immunosuppressant, however, it is not without complications.32,33Tacrolimus has also been reported to cause gingival overgrowth, although this is much less common than ciclosporin.20
Gingival overgrowth related to systemic disease
Leukaemia
Leukaemia is a group of malignant haematological disorders, involving the abnormal proliferation and development of leucocytes and their precursors in bone marrow.34 Normal haematopoeisis is suppressed and leukaemic white blood cells appear in the blood stream.34 Therefore, this results in a depressed blood cell count including mature white blood cells and platelets.34 Leukaemia is classified into acute lymphoblastic, acute myeloid, chronic lymphocytic and chronic myeloid leukaemias.34 The acute forms can lead to death in 26 months if left untreated.4,35
Clinically, the oral manifestations may include excessive bleeding and uncontrollable infections, which may occur from thrombocytopenia or neutropenia respectively.34 In acute leukaemia, 69% of patients were found to have oral signs including swelling, erythema, petechiae, ulceration and gingival bleeding and 33% had gingival enlargement (Figs 10a and b).4 In particular, gingival overgrowth is associated with acute myeloid leukaemia.4
Gingival changes may be the first signs presenting to a health professional, therefore, it is important to be diligent with diagnosis and immediate urgent referral if this is suspected. In an undiagnosed patient, there may be history of lethargy, night sweats and recent infections.36
Granulomatous diseases
Several orofacial granulomatous diseases are associated with oral changes including Crohn's disease, sarcoidosis and Melkersson-Rosenthal syndrome.4
Rarely, these diseases may present with gingival lesions characterised by swelling.4 Sarcoidosis occasionally causes fiery red granular gingival overgrowth, however, oral related features of sarcoidosis have only been reported in 68 patients.10,37
Wegener's syndrome is somewhat similar to sarcoidosis in that it is a systemic granulomatous disease that can involve the respiratory tract, kidneys, heart, nervous system and other areas.38 However, it classically presents with strawberry gingivae.39 It is an aggressive disease and follows a dramatic course of progression with a high mortality rate if not treated with corticosteroids and cytotoxic medications.38 If this is suspected referral should be made urgently.
Orofacial granulomatosis (OFG) is a rare inflammatory condition which causes persistent or recurrent enlargement of the oral and maxillary soft tissues including the gingivae (Fig. 11).40,41 There are several theories of pathogenesis including hereditary factors, allergy and infection, although the precise cause is still unknown. OFG has a wide presentation and is confirmed with histological examination consisting of non-caseating granulomas.40 OFG may be a manifestation of systemic disease including sarcoidosis and Crohn's disease, which must be considered in the management of OFG.41
Rarely primary tuberculosis has been reported to cause gingival overgrowth.42 In a recent case report, when the tuberculosis infection was treated, the gingival overgrowth resolved.42
Neurofibromatosis
Patients with neurofibromatosis may present with diffuse unilateral gingival enlargement of attached gingivae.43,44 Neurofibromatosis is a group of autosomal dominant conditions, which are characterised by numerous cutaneous lesions, and tumours of the central and peripheral nervous system.43,44 Type 1 affects 1 in 3,000 of the world population and 72% of these sufferers will have oral manifestations.43,44 These gingival swellings are fibrous and do not exhibit signs of inflammation. This may be associated with absent, impacted and/or malpositioned teeth. Surgical excision is advised if lesions are impairing form or function but multiple lesions may be difficult to eradicate completely and if involving other tissues there is a reported risk of neurosensory deficit and bleeding post operatively.
Sturge-Weber syndrome
Sturge-Weber syndrome is a rare congenital condition that is characterised by a venous angioma of leptomeninges over the cerebral cortex and ipsilateral angiomatous lesions of the face and sometimes the skull, jaws and oral soft tissues including gingival enlargement (Fig. 12a and b).45
Nutritional deficiency
Another reported systemically altered response to plaque is caused by a Vitamin C (ascorbic acid) deficiency (scurvy). This is hypothesised to modify the host response to plaque, via changes in immunological/inflammatory responses and the extracellular matrix.46 In frank scurvy, the gingivae may be bright red, swollen, ulcerated and susceptible to haemorrhaging.18 Scurvy is rare in the developed countries; however, certain low socio-economic groups may have restricted diets, and are at risk of developing these conditions.47
Other causes
Multiple pathological processes may present as gingival enlargement. The literature has reported gingival cysts, giant cell lesions (Fig. 13), neoplastic lesions and plasma cell gingivitis to name a few.10 The breadth of such presentations fall outside the scope of this article.
Malignant neoplasms
It is important to consider that malignant neoplasms may present as local enlargement affecting the gingival tissues. Approximately 5% of oral squamous cell carcinomas will present on the gingival tissues.48 Therefore, any concerning features, including persistent ulceration particularly with rolled margins, a granular base, any induration or fixation must be taken seriously.
Other tumours including malignant melanoma, non-hodgkin's lymphoma, plasma cytoid tumours (Fig. 14), sarcomas and metastatic tumours may occasionally present upon the gingival tissues.48 Concerning features include any lymphadenopathy, unexplained tooth mobility, paraesthesia, other sensory disturbances, irregular bone loss and unusual pigmentation.48 In the event of these signs an urgent referral to an oral and maxillofacial surgery unit is indicated.
Hereditary gingival fibromatosis
Hereditary gingival fibromatosis (HGF) involves a benign gingival enlargement, thought to be caused by an enhanced production of HGF fibroblasts, which favour accumulation of extracellular matrix (Figs 15a and b).49 Research has identified a specific genetic mutation with the 'son of sevenless1' (SOS1) gene associated with this condition.50 These lesions are independent of plaque although there may be secondary inflammation if plaque control is suboptimal. As the aetiology is non-modifiable, surgical management for HGF is often indicated.18
Clinically it is a slowly progressing enlargement which commences after tooth eruption. It is most commonly associated with the permanent dentition but can affect the primary teeth.51 The gingival enlargement progresses alongside the patient's normal development, typically affecting the maxillary tuberosities and labial aspects of mandibular molars (Figs 15a and b).51 HGF lesions present with dense, firm, resilient fibrous tissue extending over teeth surfaces. If severe enough to cover the occlusal surfaces, traumatic injury to the gingival tissues may occur.
Diagnosis
To determine aetiology a thorough history including medical and social information along with clinical examination is often sufficient.
History
Primarily when taking a history, the patient's presenting complaint should be established and in relation to gingival overgrowth, duration of onset and any associated pain, bleeding, tooth mobility or other symptoms noted. In addition to this, it is important to ascertain a dental history including any recent treatment, history of hygiene appointments and the patient's usual oral hygiene regime.
Social history may identify risk factors, including use of tobacco and alcohol. Family history may aid the diagnosis of conditions, which have a genetic element such as hereditary gingival fibromatosis and neurofibromatosis.
A medical history will also give information about any drugs being taken. The temporal relationship between beginning a course of medication and onset of gingival overgrowth may provide valuable diagnostic information regarding the aetiology, with DIGO presenting as early as one month after commencement of medication.52 Previous drug history may also be important in diagnosis if the patient has recently stopped taking drugs relating to DIGO, which are not immediately disclosed on drug histories.
When a rapid change has occurred, the patient has systemic signs of ill health or fails to respond to treatment; onward referral and further investigations should be considered.
Clinical examination
A thorough clinical examination should include assessment of plaque and periodontal indices.
Pocket depth alone is not a true marker of clinical attachment loss (CAL), due to the false pocketing associated with gingival overgrowth. However, true pocketing can be approximated using the estimated CEJ position. It also provides a useful baseline and can reveal concurrent periodontal disease and endodontic problems. (Figs 16a and b)
Key features
Gingival overgrowth may be described as localised or generalised. Disease initiation typically starts as a ballooning of the papillae, then progresses to involve the marginal gingiva and in more severe cases can cover the occlusal aspects of the dentition (Fig. 15b).18 A lesion may be generalised involving marginal, papillary and attached gingivae, or it may be localised with isolated sessile/pedunculated masses.10
A simple grading system can be used to describe the extent of the gingival overgrowth:53
-
0 = No signs of enlargement
-
1 = Confined to interdental papillae
-
2 = Involving papillae and marginal gingivae
-
3 = Covers ¾ or more of the crown.
The degree and nature of the inflammation may give clues to the aetiology. Fibrous, firm, stippled enlargement which is pink and homogenous indicates lack of inflammation. This is more often seen in patients with HGF or those with drug-induced gingival overgrowth and excellent oral hygiene (Figs 8, 15a and b). Where there is plaque-induced inflammation, either as a primary or secondary process, (Figs 1, 7, 16) the appearance is more likely to be smooth, shiny and reddened. Should the overgrowth be localised to a few teeth there are often plaque retentive factors present which should be assessed.
Histological examination
Tissue biopsy and histopathological examination can be used to deliver a definitive diagnosis. This is usually considered after an initial phase of management if resolution of the gingival overgrowth is not fully achieved. In some cases it is required at the outset if the history and examination identify cause for concern. In example, Figure 13 shows an unusual localised pigmented lesion which is not in keeping with typical plaque-induced inflammation. In addition, Figure 14 demonstrates a gingival enlargement that is localised with involvement beyond the mucogingival junction and its discrete isolation to the 14–17 with no dental cause is a concerning presentation requiring an urgent biopsy.
Conclusion
Commonly, gingival overgrowth is an inflammatory process related to plaque accumulation and trauma. Clinical presentation can be modified by medications or systemic condition. More rarely, there may be a genetic aetiology such as in hereditary gingival fibromatosis or an underlying systemic disease, which requires urgent medical intervention.
With thorough history-taking and clinical examination it should be possible to distinguish between those requiring urgent onward referral and dental management. In cases where this is not possible, early biopsy or medical investigations may be indicated. Sound diagnosis will aid effective management; the phases of which are discussed in the second article of this series.
References
Dannewitz B . Proliferation of the gingiva: aetiology, risk factors and treatment modalities for gingival enlargement. Perio 2007; 4: 83–91.
Lindhe J, Lang N P, Karring T . Clinical periodontology and implant dentistry. Blackwell Munksgaard, Ames, Iowa; Oxford, UK, 2008.
Armitage G C . Development of a classification system for periodontal diseases and conditions. Ann Perio 1999; 4: 1–6.
Holmstrup P . Non-plaque induced Inflammatory gingival Lesions. In Lindhe J (ed) Clinical periodontology and implant dentistry, Vol. 1. pp. 377–404. Blackwell Munksgaard; Oxford, 2008.
Stamm J W . Epidemiology of gingivitis. J Clin Perio 1986; 13: 360–366.
Research, Science and Therapy Committee of the American Academy of Periodontology. Treatment of plaque-induced gingivitis, chronic periodontitis, and other clinical conditions. J Periodontol 2005; 27: 202–211.
Travess H, Roberts-Harry D, Sandy J . Orthodontics. Part 6: Risks in orthodontic treatment. Br Dent J 2004; 196: 71–77.
Wagaiyu E G, Ashley F P . Mouthbreathing, lip seal and upper lip coverage and their relationship with gingival inflammation in 11–14 year-old schoolchildren. J Clin Perio 1991; 18: 698–702.
Jacobson L, Linder-Aronson S. Crowding and gingivitis: a comparison between mouthbreathers and nosebreathers. Scand J Dent Res 1972; 80: 500–504.
Carrenza F A, Hogan E L . Gingival enlargement. In Clinical Periodontology. pp. 373–390. Saunders Elsevier: Missouri, 2006.
Al-Jewair T.S, Al-Jasser R, Almas K . Periodontitis and obstructive sleep apnea's bidirectional relationship: a systematic review and meta-analysis. Sleep Breath 2015; 19: 1111–1120.
Coelho C.M, Zucoloto S, Lopes R A . Denture-induced fibrous inflammatory hyperplasia: a retrospective study in a school of dentistry. Int J Prosth 2000; 13: 148–151.
Feller L, Altini M, Lemmer J . Inflammation in the context of oral cancer. Oral Onc 2013; 49: 887.
Kalsi H.J, Hussain, Z, Darbar U . An update on crown lengthening. Part 1: Gingival tissue excess. Dent Update 2015; 42: 144–146, 149–150, 153.
Armitage G C . Bi-directional relationship between pregnancy and periodontal disease. Periodontol 2000 2013; 61: 160–176.
Nakagawa S, Fujii H, Machida Y, Okuda K . A longitudinal study from prepuberty to puberty of gingivitis. Correlation between the occurrence of Prevotella intermedia and sex hormones. J Clin Perio 1994; 21: 658–665.
Baker E, Roberts A P, Wilde K et al. Development of a core drug list towards improving prescribing education and reducing errors in the UK. Br J Clin Pharmacol 2011; 71: 190–198.
Mariotti A . Plaque-induced gingival diseases. In Lindhe J (ed) Clinical periodontology and implant dentistry. Vol. 1. pp. 405–419. Blackwell Munksgaard: Oxford, 2008.
Preshaw P.M, Knutsen M A, Mariotti A . Experimental gingivitis in women using oral contraceptives. J Dent Res 2001; 80: 2011–2015.
Hassell T M, Hefti A F . Drug-induced gingival overgrowth: old problem, new problem. Critical reviews in oral biology and medicine: an official publication of the American Association of Oral Biologists 1991; 2: 103–137.
Seymour R.A, Thomason J M, Ellis J S . The pathogenesis of drug-induced gingival overgrowth. J Clin Perio 23: 165–175.
Wilson R F, Morel A, Smith D et al. Contribution of individual drugs to gingival overgrowth in adult and juvenile renal transplant patients treated with multiple therapy. J Clin Perio 1998; 25: 457–464.
Seymour R A, Jacobs D J . Cyclosporin and the gingival tissues. J Clin Perio 1992; 19: 1–11.
Heasman P A, Hughes F J . Drugs, medications and periodontal disease. Br Dent J 2014; 217: 411–419.
Barclay S, Thomason J .M, Idle J R, Seymour R A . The incidence and severity of nifedipine-induced gingival overgrowth. J Clin Perio 1992; 19: 311–314.
Brown R S, Arany P R . Mechanism of drug-induced gingival overgrowth revisited: a unifying hypothesis. Oral Dis 2015; 21: e51–e61.
Angelopoulos A P, Goaz P W . Incidence of diphenylhydantoin gingival hyperplasia. Oral Surg Oral Med Oral Pathol 1972; 34: 898–906.
Dongari A, McDonnell H T, Langlais R P . Drug-induced gingival overgrowth. Oral Surg Oral Med Oral Pathol 1993; 76: 543–548.
Ellis J S, Seymour R A, Steele J G, Robertson P, Butler T J, Thomason J M . Prevalence of gingival overgrowth induced by calcium channel blockers: a community-based study. J Perio 1999; 70: 63–67.
Fu E, Nieh S, Hsiao C T, Hsieh Y D, Wikesjö U M, Shen E C . Nifedipine-induced gingival overgrowth in rats: brief review and experimental study. J Perio 1998; 69: 765–771.
Mariotti A, Hassell T, Jacobs D, Manning C J, Hefti A F . Cyclosporin A and hydroxycyclosporine (M17) affect the secretory phenotype of human gingival fibroblasts. J Oral Pathol Med 1998; 27: 260–266.
Mavrogiannis M, Ellis J .S, Thomason J M, Seymour R A . The management of drug-induced gingival overgrowth. J Clin Perio 2006; 33: 434–439.
Scott L.J, McKeage K, Keam S J, Plosker G L . Tacrolimus: a further update of its use in the management of organ transplantation. Drugs 2003; 63: 1247–1297.
Scully C, Cawson R A . Medical problems in dentistry. Churchill Livingstone: Edinburgh, 2005.
Oran B, Weisdorf D J . Survival for older patients with acute myeloid leukemia: a population-based study. Haematologica 2012; 97: 1916–1924.
Gallipoli P, Leach M . Gingival infiltration in acute monoblastic leukaemia. Br Dent J 2007; 203: 507–509.
Suresh L, Radfar L . Oral sarcoidosis: a review of literature: Oral sarcoidosis. Oral Dis 2005; 11: 138–145.
Soames J V, Southam J C . Oral pathology. Oxford University Press: Oxford, 2005.
Ruokonen H, Helve T, Arola J, Hietanen J, Lindqvist C, Hagstrom J . 'Strawberry like' gingivitis being the first sign of Wegener's granulomatosis. Eur J Intern Med 2009; 20: 651–653.
Grave B, McCullough M, Wiesenfeld D . Orofacial granulomatosisa 20-year review. Oral Dis 2009; 15: 46–51.
Troiano G, Dioguardi M, Giannatempo G et al. Orofacial granulomatosis: clinical signs of different pathologies. Med Principles Pract 2015; 24: 117–122.
Gill J S, Sandhu S, Gill S . Primary tuberculosis masquerading as gingival enlargement. Br Dent J 2010; 208: 343–345.
Jouhilahti EM, Visnapuu V, Soukka T et al. Oral soft tissue alterations in patients with neurofibromatosis. Clin Oral Investig 2012; 16: 551–558.
Cunha K S.G, Barboza E.P, Dias E P, Oliveira F M . Neurofibromatosis type I with periodontal manifestation. A case report and literature review. Br Dent J 2004; 196: 457–46.
Bhansali R.S, Yeltiwar R K, Agrawal A A . Periodontal management of gingival enlargement associated with Sturge-Weber syndrome. J Perio 2008; 79: 549–555.
Nishida M, Grossi S G, Dunford R G, Ho A W, Trevisan M, Genco R J . Dietary vitamin C and the risk for periodontal disease. J Perio 2000; 71: 1215–1223.
Oeffinger K C . Scurvy: more than historical relevance. Am Fam Physician 1993; 48: 609–613.
Scully C . Cancer. In Scully C (ed) Oral and Maxillofacial Medicine. Churchill Livingstone Elslever: Philadelphia, 2008.
Coletta R.D, Almeida O.P, Reynolds M A, Sauk J J . Alteration in expression of MMP1 and MMP2 but not TIMP1 and TIMP2 in hereditary gingival fibromatosis is mediated by TGF-beta 1 autocrine stimulation. J Perio Res 1999; 34: 457–463.
Hart T C, Zhang Y, Gorry M C et al. A mutation in the SOS1 gene causes hereditary gingival fibromatosis type 1. Am J Hum Genet 2002; 70: 943–954.
Coletta R D, Graner E . Hereditary gingival fibromatosis: a systematic review. J Perio 2006; 77: 753–764.
Harel-Raviv M, Eckler M, Lalani K, Raviv E, Gornitsky M . Nifedipine-induced gingival hyperplasia. Oral Surg Oral Med Oral Pathol Oral Radiol Endody 1995; 79: 715–722.
Bökenkamp A, Bohnhorst B, Beier C, Albers N, Offner G, Brodehl J . Nifedipine aggravates cyclosporine Ainduced gingival hyperplasia. Pediatr Nephrol 1994; 8: 181–185.
Author information
Authors and Affiliations
Corresponding author
Additional information
Refereed Paper
Rights and permissions
About this article
Cite this article
Beaumont, J., Chesterman, J., Kellett, M. et al. Gingival overgrowth: Part 1: aetiology and clinical diagnosis. Br Dent J 222, 85–91 (2017). https://doi.org/10.1038/sj.bdj.2017.71
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bdj.2017.71
This article is cited by
-
Systemic medicines taken by adult special care dental patients and implications for the management of their care
British Dental Journal (2021)