Abstract
Study design:
Case report.
Objectives:
Primary intramedullary spinal cord lymphoma is a rare entity. Studies have shown that there is a recent increase in the number of patients regardless of the status of the immunity. High index of suspicion should be kept in all patients with intramedullary tumors. Multidisciplinary approach at the earliest is required for best outcomes.
Setting:
Department of Neurosurgery, Medical Oncology, Pathology and Radiation oncology. Nizam’s Institute of Medical Sciences, Hyderabad, India.
Methods:
We describe the case of an 11-year-old boy who presented with paraparesis and sensory loss below T10 level. On imaging, the dorsal spine showed intramedullary lesion mimicking an astrocytoma.
Results:
Surgical decompression of the tumor was done and histopathology showed non-Hodgkin’s lymphoma, diffuse large B-cell type. There were no findings suggestive of congenital or acquired immunodeficiency. After complete staging evaluation, we instituted chemotherapy with modified DeAngelis protocol. At 2 years post treatment, he is in complete remission with near normal neurological status.
Conclusions:
Intramedullary spinal cord diffuse B-cell lymphoma in a pediatric age group is very rare and hence requires a high index of suspicion in patients presented with myelopathy. The outcomes are encouraging with current multidisciplinary approach.
Similar content being viewed by others
Introduction
Primary central nervous system (CNS) lymphoma is defined as lymphoma limited to CNS without systemic disease. Non-Hodgkin’s lymphomas (NHLs) account for only 0.3–1.5% of all CNS neoplasms1, 2 and 1% of all NHL.3 Primary spinal cord intramedullary lymphomas are very rare and <30 cases have been reported in the literature.4
Case report
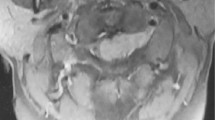
An 11-year-old boy presented with gradual onset, progressive weakness of the both lower limbs. No other abnormality was detected on general physical examination and other systemic examination. Magnetic resonance imaging (MRI) of the thoracic spine revealed an intramedullary lesion, hyperintense on T1W and iso- to hyperintense on T2W images extending from the upper border of T8 to upper border of T11 vertebrae. Gadolinium contrast images showed moderate enhancement of the lesion. (Figure 1). Blood counts and chemistry were normal and serology was negative for HIV.
Preoperative imaging. (1) TI W MRI of dorsal spine showing hyperintense intramedullary tumor extending from T8 to T12 with loss of anterior and posterior cerebrospinal fluid spaces and diffuse expansion of the cord. (2) T2 W MRI of dorsal spine showing iso- to hyperintense intramedullary tumor extending from T8 to T12. (3) Contrast MRI with fat saturation sagittal section and (4) axial section showing enhancement of the same lesion.
The patient was planned for surgery to decompress the spinal cord and obtain tissue for pathological diagnosis. He underwent T6-T12 laminectomy, midline myelotomy and subtotal tumor decompression. Intraoperatively, the lesion was greyish, soft, mildly vascular and suckable with no clear plane of cleavage from the surrounding cord. The power in both lower limbs improved in the immediate postoperative period.
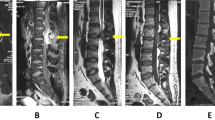
Hematoxylin and eosin-staining studies (Figure 2) showed diffuse infiltration of round cells in the nerve roots. The cells were large with hyperchromatic nucleus and scant cytoplasm. Mitosis was brisk. Immunohistochemistry (IHC) showed intense positivity for cluster differentiation (CD) 20 and the cells were negative for CD 3 and CD 99. The histomorphology and IHC were consistent with a diagnosis of diffuse large B-cell lymphoma.
Histopathology. (a and b) Showing diffuse infiltrates of the nerve roots by small round cells. (c) The cells are large with hyperchromatic nuclei and scant cytoplasm. Hematoxylin and eosin stain, × 400. (d and e) The cells showed diffuse cytoplasmic positivity for CD 20 (horseradish peroxidase (HRP) polymer CD 20) and (f) negativity for CD3. (HRP polymer, CD3).
Lymphoma staging workup was done. Contrast-enhanced computerized tomography scans of the neck, chest, abdomen and pelvis were normal. MRI of brain and whole spine was negative for other possible lesions. Bone marrow aspiration and biopsy was normal. High-resolution ultrasonography of testis was normal. Therefore, our final diagnosis was a primary spinal cord lymphoma.
Modified DeAngelis protocol was instituted.5, 6 Chemotherapy was administered for five cycles over a 10-week period. Each cycle consisted of intravenous methotrexate 2.5 g m−2 (3.5 g m−2 in previous DeAngelis5, 6) infused over 2–3 h and vincristine 1.4 mg m−2. In addition to methotrexate and vincristine, procarbazine 100 mg m−2 per day for 7 days was administered on cycles 1, 3 and 5. Intrathecal methotrexate 12 mg was administered for five doses scheduled for the week after each dose of intravenous methotrexate. Dexamethasone was administered on a standard schedule: 16 mg per day for week 1, 12 mg per day for week 2, 8 mg per day for week 3, 6 mg per day for week 4, 4 mg per day for week 5, and 2 mg per day for week 6 and was then discontinued. At completion of five courses of chemotherapy, he was treated with local radiation therapy to the spine from T6 to L2 level with 6 MV photons to a dose of 40 Gy given in 20 fractions at 2 Gy per fraction over 4 weeks. Following radiation therapy, he received two courses of high-dose cytarabine 3 g m−2 per day.
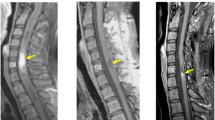
His follow-up spinal MRI after 6 months and 2 years (Figure 3) showed decreased enlargement and enhancement in the previous mass suggestive of remission. He, however, had abnormal dorsal kyphosis, for which deformity correction was performed. Screening with imaging of the whole spine and brain was free of any new pathologies. At 2 years following treatment, the boy was near normal in neurological status and was attending his school regularly.
Discussion
Flanagan et al.4 has published a series of 11 patients with spinal cord NHL with a median age of 62.5 years and conus cauda as the predominant location with a 2-year survival of 36%. Presentation in a pediatric age group is rare, as most of the previously reported cases are middle aged or elderly patients.4 Most intramedullary spinal cord lymphomas are present in the upper thoracic and lower cervical regions.2 As the incidence is raising both in immunocompromised and immunocompetent patients, one must keep into account the possibility of this entity in all intramedullary lesions.1 Enlargement of the spinal cord is seen in most of the cases of primary intramedullary spinal cord lymphoma with variable intensity in T1-weighted (W) images and hyperintensity in T2 W images.7 Surgical intervention is required not only for decompression but also for definitive histopathological examination.
Conventional corticosteroids and radiation resulted in remission of the lesion with poor survival rates.2 Previously, intrathecal and high-dose intravenous chemotherapy with methotrexate was used as a salvage therapy only after the recurrence of the previously irradiated brain or spinal lymphomas. Recent trials and literature on therapy with preradiation high-dose methotrexate and post-radiation Cytarabine according to DeAngelis protocol show promising results with improved outcomes compared with the previous regimes.3
Conclusion
Intramedullary spinal cord diffuse B-cell lymphoma in a pediatric age group is very rare and hence requires a high index of suspicion in patients presented with myelopathy. The outcomes are encouraging with current multidisciplinary approach.
References
Alves OL . Current management of primary cerebral lymphomas. Neurosurg. Quart. 1998; 8: 71–87.
Bekar A, Cordan T, Evrensel T, Tolunay S . A case of primary spinal intramedullary lymphoma. Surg Neurol 2001; 55: 261–264.
Sierra Del Rio M, Rousseau A, Soussain C, Ricard D, Hoang-Xuan K . Primary CNS lymphoma in immunocompetent patients. Oncologist 2009; 14: 526–539.
Flanagan EP, O’Neill BP, Porter AB, Lanzino G, Haberman TM, Keegan BM . Primary intramedullary spinal cord lymphoma. Neurology 2011; 77: 784–791.
DeAngelis LM, Seiferheld W, Schold SC, Fisher B, Schultz CJ, Radiation Therapy Oncology Group Study 93-10. Combination chemotherapy and radiotherapy for primary central nervous system lymphoma: Radiation Therapy Oncology Group Study 93-10. J Clin Oncol 2002; 20: 4643–4648.
Abrey LE, Yahalom J, DeAngelis LM . Treatment for primary CNS lymphoma: the next step. J Clin Oncol 2000; 18: 3144–3150.
Nakamizo T, Inoue H, Udaka F, Oda M, Kawai M, Uemura K et al. Magnetic resonance imaging of primary spinal intramedullary lymphoma. J Neuroimaging 2002; 12: 183–186.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Bhushanam, T., Rajesh, A., Linga, V. et al. Primary intramedullary non-Hodgkin’s lymphoma in an immunocompetent child. Spinal Cord 52 (Suppl 2), S21–S23 (2014). https://doi.org/10.1038/sc.2014.96
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2014.96