Abstract
Study design:
Retrospective study.
Objectives:
To investigate prognostic factors of sacral chordomas and provide theoretical foundation for an improvement of continuous disease-free survival (CDFS).
Methods:
Thirty-six patients underwent initial operation for sacral chordoma between 1992 and 2007. Data regarding age, gender, tumor size, tumor location, and type of surgery, surgical margins, surrounding muscle invasion, radiation therapy, and recurrences were reviewed and analyzed statistically.
Results:
The average duration of follow-up was 74.4 months (range, 16–182 months). Sixteen patients developed local recurrences at the time of final follow-up. The average recurrence time was 30 months (range, 3–84 months). The 5-year and 10-year actuarial CDFS were 59.5 and 42%, respectively. CDFS was found statistically longer in patients whose tumor highest level at or below S3 compared with above S3 (P=0.047). Patients who had invasion into the surrounding muscle had a shorter CDFS than those without surrounding muscle invasion (P=0.014). Statistic analysis showed that the type of surgery was the most valuable indicator of CDFS (P=0.001). The local recurrence rate was statistically higher when an inadequate margin has been achieved (P=0.021). The Cox multivariate regression analysis showed that surrounding muscle invasion (P=0.024) was an independent adverse prognostic factor for CDFS, whereas incomplete excision (P=0.056) achieved borderline significance.
Conclusions:
The higher level of tumor involvement, invasion into the surrounding muscle, incomplete excision, and inadequate surgical margins are poor prognostic factors. Resecting the tumor completely with wide surgical margins may provide a better prognosis for these patients.
Similar content being viewed by others
Introduction
Chordoma is a rare, low to intermediate-grade malignant bone tumor known to arise from the embryonic remnants of the notochord,1, 2 and recent studies suggest that chordomas can also arise from the precursor benign notochordal lesion.3 The majority of chordomas develop in the sacrococcygeal region (40–50%) or the base of the skull (35–40%), and only a small number are found in the mobile spine (15–20%).4, 5 Sacral chordomas generally grow slowly with insidious symptoms. The large size, the proximity to neurovascular structures, and the complex anatomy of the pelvis make complete or wide excision difficult, and these probably lead to poor local control. Achieving long-term survival remains a challenge and local recurrence is one of the most important predictors. In this study, we investigated the prognostic factors of sacral chordomas after surgical therapy and provide theoretical foundation for an improvement of continuous disease-free survival time (CDFS).
Materials and methods
A consecutive series of 36 patients with sacral chordoma were analyzed retrospectively. All patients were treated between 1992 and 2007 at the Department of Orthopedic Surgery, The First Affiliated Hospital of Soochow University. Patients who had performed earlier surgical treatment were excluded from the review. Follow-up included plain radiographs, computed tomography scan and magnetic resonance (MR) imaging evaluation at every 3 months in the first 2 years and at 6-month intervals after 3 years. Clinic visits were complemented by letters and questionnaires provided by our institution.
The information included in this report was retrieved from direct examination of the patient, evaluation of images, and patient interview. Data on the patient age, gender, symptoms, size of the tumor (largest diameter), location of the tumor (most proximal vertebral level involvement), with surrounding muscle invasion or not, type of surgery (intralesional, marginal, or wide), surgical margins (wide or inadequate), adjuvant treatment, and local recurrence were collected and analyzed for each patient. The histology of all patients was confirmed.
Preoperative images
All patients were examined with plain radiographs, computed tomography scan, and MR imaging. All tumors were osteolytic and sometimes difficult to detect on initial plain films. Patients were also evaluated with a whole-body bone scan and a computed tomography scan of the chest to ensure that no distant metastasis was present.
Treatment
Preoperatively, all patients underwent embolization of the tumor feeding arteries under digital subtraction angiography. We embolized the bilateral internal iliac artery, median sacral artery, and other tumor feeding arteries by broken pieces and striga of gelatin sponge.6 All cases were performed a posterior approach in an average of 1.5 days (range, 1–3 days) after the embolization. During the dissection, we tried to preserve bilateral S1–S2 nerve roots or at least one S3 nerve root to preserve the normal bowel and bladder function. If the first sacral vertebrae was excised, we would reconstruct the stability of pelvic ring by using the improved Galveston L-rod.7
Statistical analysis
Data were recorded in a Microsoft Excel 2003 spreadsheet and all statistical analysis were completed using SPSS 13.0 statistical software. CDFS was defined as the time interval from initial diagnosis to disease relapse. The following factors were evaluated: patient age, gender, tumor size, tumor location, surrounding muscle invasion, type of surgery, surgical margins, and adjuvant radiotherapy. The rates of CDFS were analyzed by Kaplan–Meier method. Risk factors for these end points were compared with use of the log-rank test. The multivariate analysis was performed by using the Cox proportional-hazards model. The level of significance was a probability value of <0.05.
Results
Of 36 patients, 16 were female and 20 were male. The mean age at diagnosis was 51.5 years (range, 18–77 years). Local pain was the chief presenting symptom in a majority of patients, and other presenting symptoms including bladder and bowel dysfunction, paresthesias, and sexual dysfunction were less frequently observed. The average duration of symptoms before diagnosis was 15.1 months (range, 2–60 months). At the time of presentation, all tumors were Stages IA–IB according to the system of the Musculoskeletal Tumor Society (MSTS). No patient developed metastases at the time of initial examination. Fifteen tumors were located in the low sacrum group (S3 or below), and 21 tumors were located in high sacrum group (above S3). The mean tumor size was 9.15 cm (range, 4.0–13.0 cm) in the greatest diameter. Thirteen patients were identified to have invasion of the surrounding muscle (piriformis or gluteus maximus).
A wide margin was defined as a cuff of normal tissue around the tumor except anteriorly, where the tumor was covered by the presacral fascia and was separated easily from the rectum without any evidence of infiltration. Marginal excision was defined as tumor that was not resected widely, and intralesional excision was defined as tumor was resected by partition or incomplete surgical excision. Radical wide excision was performed in 7 cases, marginal excision in 16 cases, and intralesional excision in the remaining 13 cases. A margin was considered adequate if it was wide according to intraoperative and histological assessments. All other margins including marginal and intralesional were considered to be inadequate. In our study, a wide margin was obtained in 7 patients (19.4%), an inadequate margin was obtained in 29 patients (80.6%). The mean operation time was 5.2 h, and the mean operation blood loss was 1026 ml (range, 400–3500 ml). Reconstruction was performed in only four cases whose tumor reached L5 and the total sacrum with substantial parts of both iliac wings being resected. In this series, radiation therapy was reserved for surgically contaminated margins or incomplete excision of the tumor. Overall, radiation therapy was administered to 15 (41.7%) from 36 patients, with an average dose of 50 Gy (range, 30–60 Gy). None of the patients received chemotherapy.
Outcome
Follow-up information was obtained in all cases. The average duration of follow-up was 74.4 months (range, 16–182 months). There were 16 (44.4%) patients who developed local recurrences. The average recurrence time was 30 months (range, 3–84 months). Five of them had a second local recurrence in 10–45 months after the first recurrence. Among these five patients, a 56-year-old male is still alive without disease 26 months after the second recurrence (Figures 1, 2, 3). For all patients in this study, the 5-year and 10-year actuarial CDFS were 59.3 and 42%, respectively (Figure 4). Of the 16 patients with local recurrences, 2 had no evidence of disease at last follow-up, 8 were alive with disease, and 6 died of tumor.
A 56-year-old man who presented with sacral pain and bowel dysfunction. A magnetic resonance imaging (MRI) made 35 months after the primary surgery. (a and b) A sagittal plane of T1-weighted MR image shows recurrent chordoma on the inner aspect of left pelvic cavity (white arrows). (c and d) A coronal plane of T1-weighted and fat-suppressed T2-weighted MR image shows the recurrence lesion invading the left sciatic nerve and piriformis muscle (black arrow).
Prognostic factors
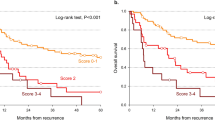
Univariate statistical analyses for prognostic factors are tabulated in Table 1. Significant adverse prognostic factors for CDFS were higher tumor location, with surrounding muscle invasion, incomplete excision, and inadequate surgical margins (P<0.05, Figure 5). All other factors analyzed including patient age, gender, tumor size, and adjuvant radiotherapy were not significant (P>0.05). The Cox multivariate regression analysis showed that surrounding muscle invasion (B=−1.300, P=0.024) was an independent adverse prognostic factor for CDFS, whereas incomplete excision (B=−1.669, P=0.056) achieved borderline significance (Figure 6). Results of cox regression analysis are listed in Table 2.
Discussion
Complete excision is necessary for sacral chordoma because of its poor sensitivity to radiotherapy and chemotherapy.5, 8 Aggressive techniques of surgery allow a more total resection, but local recurrence rate is still high.7, 8, 9 As many patients can survive despite the presence of symptoms, the use of CDFS time is potentially more valuable than the overall survival time. Therefore, we investigated the factors related with CDFS in sacral chordomas.
Until recently, age and sex were thought to be of no prognostic value in chordomas.10 However, O'Connell et al.11 reported that female sex was an independent predictors of shorter survival (P=0.004) for skull base chordoma. The same observation was reported by Thieblemont et al.12 in a series of 26 patients with chordoma. In univariate analysis, progression-free survival (PFS) was found significantly longer in males as compared with females (P=0.05), and patients under 60-years old had a longer progression-free survival (P=0.06) than older patients. However, in our series, CDFS was not found to be significantly different for patients with different age and sex group.
Cheng et al.13 reported that the highest level of tumor involvement was prognostically significant for CDFS and overall survival (P<0.05) in 23 patients. This was also observed in our study; patients in the high sacrum group had higher recurrence rates than those in the low sacrum group (P=0.04). We found that local infiltration was more likely to occur in the high sacrum group (47.6%) than in the low sacrum group (20%). When tumor involves to S2 or above, because of the anatomic characteristics or to preserve nerve roots, intralesional excision tends to be used. Therefore, careful evaluation of preoperative MR images should be performed to ascertain the extent of the tumor and plan operative scope.
The large tumor size at presentation has been consistently shown to be a negative prognostic factor. York et al.10 reported a series of 39 patients, and the recurrence rate of the big tumor group (>8 cm) was higher than the small tumor group (<8 cm) (P=0.053). We separated the patients into two groups on the basis of the average tumor size (9.15 cm), but in the univariate analysis, the difference of the median CDFS between different tumor size group had no significance (P=0.49). This may have occurred because it may be inversely correlated with the probability of achieving a wide excision in our series.
Extent of resection and surgical margins have consistently been shown to impact local control.4, 5, 14, 15 Similar to earlier studies, we found surgical type and surgical margins were intimately correlated to local recurrence. All patients with a wide margin had no recurrence. The majority of patients operated with intralesional margins developed recurrence. The risk of an inadequate procedure was mainly related to the extension of the disease in the soft tissue. In addition, intraoperative contamination or seeding of cells from tumor during operation frequently occurred in the intralesional excision group, and led to a high recurrence rate.
Hanna et al.16 suggested infiltration of the musculature adjacent to the sacrum and/or involvement of the sacroiliac joints increases the tendency of local recurrence, even after apparently successful en bloc resection of the tumor. All patients with invasion of the surrounding muscle had a higher recurrence rate (76.9%). Similar to Hanna et al., we found that presence of surrounding muscle invasion was a significant adverse prognostic factor for CDFS. This means invasion of the surrounding muscle in sacral chordoma may indicate the tumor is at an advanced stage. Excising parts of the piriformis and gluteus maximus may allow wider surgical margins posteriorly, which may result in better local disease control.
The role of traditional radiation remains controversial because chordomas are not very radiosensitive. Earlier studies have shown mixed results regarding the ability of radiotherapy to improve the prognosis when the surgical margin is positive.5, 17, 18 Some technologies such as protons or intensity modulated radiationtherapy have been used to improve the radiobiological effect.19 However, our results did not show that radiation therapy improved CDFS. Given the limitation of this series, the value of adjuvant radiation therapy needs to be addressed in future prospective multi-institutional studies.
In conclusion, the only factor positive in the Cox MVA in our series was the presence of surrounding muscle invasion. This may be related to more aggressive tumors on presentation that were more likely to recur despite aggressive therapy, or possibly this may be related to the inability to achieve maximal resection. This highlights the importance of early diagnosis, careful evaluation of preoperative MR images, and radical excision with wide surgical margins. Local failure is the major factor affecting postoperative outcome, but aggressive management of recurrences is obligatory to allow long-term survival.
Conflict of interest
The authors reported no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
References
Ribbert H . Uber die Ecchondosis physaliphora sphenooccipitalis. Centralbl Allg Pathol Anat 1894; 5: 457–461.
Samson IR, Springtleld DS, Suit HD, Mankin HJ . Operative treatment of sacrococcygeal chordoma. J Bone Joint Surg Am 1993; 75: 1476–1484.
Yamaguchi T, Yamato M, Saotome K . First histologically confirmed case of a classic chordoma arising in a precursor benign notochordal lesion: differential diagnosis of benign and malignant notochordal lesions. Skeletal Radiol 2002; 31: 413–418.
Bergh P, Kindblom LG, Gunterberg B, Remotti F, Ryd W, Meis-Kindblom JM et al. Prognostic factors in chordoma of the sacrum and mobile spine: a study of 39 patients. Cancer 2000; 88: 2122–2134.
Fuchs B, Dickey ID, Yaszemski MJ, Inwards CY, Sim FH . Operative management of sacral chordoma. J Bone Joint Surg Am 2005; 87: 2211–2216.
Yang HL, Zhu LF, Liu JY, Liu L, Lee A, Wang YW et al. Surgical treatment of sacral chordomas with preoperative arterial embolization and prognostic indicators of outcome. Spine J 2007; 7: 35S.
Gokaslan ZL, Romsdahl MM, Kroll SS, Walsh GL, Gillis TA, Wildrick DM et al. Total sacrectomy and Galveston L-rod reconstruction for malignant neoplasms. J Neurosurg 1997; 87: 781–787.
Hulen CA, Temple HT, Fox WP, Sama AA, Green BA, Eismont FJ et al. Oncologic and functional outcome following sacrectomy for sacral chordoma. J Bone Joint Surg Am 2006; 88: 1532–1539.
York JE, Kaczaraj A, Abi-Said D, Fuller GN, Skibber JM, Janjan NA et al. Sacral chordoma: 40-year experience at a major cancer center. Neurosurgery 1999; 44: 74–79.
Akmal S, Ole SN, Anne GJ, Johnny K, Ernst RW, Bjarne L et al. A retrospective clinicopathological study of 37 patients with chordoma: a Danish national series. Sarcoma 1997; 1: 161–165.
O'Connell JX, Renard LG, Liebsch NJ, Efird JT, Munzenrider JE, Rosenberg AE et al. Base of skull chordoma: a correlative study of histologic and clinical features of 62 cases. Cancer 1994; 74: 2261–2267.
Thieblement C, Biron P, Rocher F, Bouhour D, Bobin JY, Gerard JP et al. Prognostic factors in chordoma: role of postoperative radiotherapy. Eur J Cancer 1995; 31: 2255–2259.
Cheng EY, Ozerdemoglu RA, Transfeldt EE, Thompson RC . Lumbosacral chordoma: prognostic factors and treatment. Spine 1999; 24: 1639–1645.
Sundaresan N, Huvos AG, Krol G, Lane JM, Brennan M . Surgical treatment of spinal chordomas. Arch Surg 1987; 122: 1479–1482.
Kaiser TE, Pritchard DJ, Unni KK . Clinicopathologic study of sacrococcygeal chordoma. Cancer 1984; 53: 2574–2578.
Hanna SA, Aston WJS, Briggs TWR, Cannon SR, Saifuddin FA . Sacral chordoma: can local recurrence after sacrectomy be predicted? Clin Orthop Relat Res 2008; 466: 2217–2223.
Chandawarkar RY . Sacrococcygeal chordoma: review of 50 consecutive patients. World J Surg 1996; 20: 717–719.
Yonemoto T, Tatezaki SJ, Takenouchi T, Ishii T, Satoh T, Moriya H et al. The surgical management of sacrococcygeal chordoma. Cancer 1999; 85: 878–883.
Casali PG, Stacchiotti S, Sangalli C, Olmi P, Gronchi A . Chordoma. Curr Opin Oncol 2007; 19: 367–370.
Acknowledgements
We thank Professor Tiansi Tang, Dr Yiming Ji, Dr Genlin Wang, Dr Lifan Zhu, and Dr Christina Marry for their help and guidance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, K., Yang, H., Lu, J. et al. Prognostic factors of sacral chordoma after surgical therapy: a study of 36 patients. Spinal Cord 48, 166–171 (2010). https://doi.org/10.1038/sc.2009.95
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2009.95
Keywords
This article is cited by
-
Conditional survival and changing risk profile in patients with chordoma: a population-based longitudinal cohort study
Journal of Orthopaedic Surgery and Research (2019)
-
Recurrence or neurological loss? Resection mode selection for patients with large sacral chordoma: an analysis of prognostic factors and quality of life
Acta Neurochirurgica (2019)
-
Analysis of prognostic factors for survival in patients with primary spinal chordoma using the SEER Registry from 1973 to 2014
Journal of Orthopaedic Surgery and Research (2018)
-
Chordoma: a systematic review of the epidemiology and clinical prognostic factors predicting progression-free and overall survival
European Spine Journal (2018)
-
Cryosurgery in the excision of a giant local recurrent sacral chordoma: a case report and literature review
European Spine Journal (2018)