Abstract
Study design:
Randomized study.
Objectives:
To find out the incidence of deep vein thrombosis (DVT) in acute spinal cord injury (SCI) patients with and without therapeutic prophylaxis.
Setting:
Patients admitted in the department of Physical Medicine & Rehabilitation, SMS Medical College, Jaipur, India.
Methods:
All 297 patients received physical therapy measures and were randomly divided into two groups. 166 patients received prophylactic heparin, whereas 131 patients did not.
Results:
A total of three cases (1.8%) in study group and four cases (3%) in control group developed DVT. This difference was statistically insignificant (P>0.05).
Conclusion:
Incidence of DVT in SCI is low in our study.
Similar content being viewed by others
Introduction
Deep vein thrombosis (DVT) is one of the major complications after acute spinal cord injury (SCI)1 because of Virchow's triad, that is, stasis, endothelial cell injury and hypercoagulability.2 Thrombosis has been reported as early as 72 h after injury, with a peak between 7 and 10 days.3 This can cause pulmonary embolism, which is a common cause of death.1
Various studies on DVT in acute SCI without or with prophylaxis4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15 have reported incidence varying from 10 to 100% (Table 1a) and from 0 to 7% (Table 1b), respectively, using different diagnostic and preventive modalities. A study has been carried out in the Department of Physical Medicine & Rehabilitation and Rehabilitation Research Centre, Sawai Man Singh Medical College, Jaipur, with the aim of finding out
-
1
the incidence of DVT in SCI in the local population;
-
2
the role of clinical examination in the early detection of DVT; and
-
3
the role of heparin in DVT prophylaxis.
Materials and methods
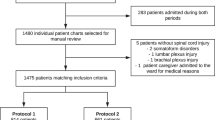
A total of 385 patients with acute SCI, admitted to the Physical Medicine & Rehabilitation department from 1 March 2004 to 30 November 2005, were included in this study. All these patients were evaluated for neurological examination and epidemiological profile. These patients were randomly divided into study and control groups. Patients in the study group were given 5000 IU of unfractionated low-dose heparin (ULDH), subcutaneously 12 hourly, from the day of admission until 3 months after injury, free of cost from government supply (Cost of ULDH is US $ 69/euros 53 for an average of 80 days prophylaxis), and the patients in the control group were not given heparin. Physical therapy measures, such as passive range of motion exercise and gentle effleurage, were advised in both the groups. Their calf girths and body temperature were recorded daily and an increase of more than 2 cm in girth was considered as significant for DVT. Color Doppler study was carried out in all the cases on admission and at 3 months follow-up.
A total of 88 patients dropped out from the study because of various reasons (Table 2). The remaining 297 cases were included in the study. There were 166 (56%) cases in the study group and 131 (44%) cases in the control group.
We have followed all applicable institutional and governmental regulations regarding the ethical use of human volunteers during the course of this study.
Results
A majority of the patients entered into our study within 10 days after injury (study group—132 cases, 80%, control group—101 cases, 77%) with a mean of 8 days ranging from 3 to 40 days. The mean age of these patients was 32 years, with a range from 10 to 80 years. The average monthly family income of these patients is US $ 67/euros 52, on which an average of five family members depend for their 1-month livelihood. The majority of the patients in both groups (study group—144 cases, 87%, control group—97 cases, 74%) were males. There were more quadriplegics with American Spinal Injury Association (ASIA) grade A and flaccid paralysis in both groups (Table 3). One-fifth of the patients in both the groups (study group—32 cases, 20%, control group—26 cases, 20%) had fever. There was an increase in calf girth in <10% of the patients in both groups (study group—9 cases, 5%, control group—10 cases, 8%).
Deep vein thrombosis was noted and confirmed by color Doppler in three patients (1.8%) in the study group within 6–10 days after injury; two were paraplegics, one quadriplegic, two in ASIA grade A, one in ASIA grade D, all without spasticity and two having fever with an increase in calf girth.
In the control group, DVT developed in four patients (3%) within 5–28 days after injury, two were quadriplegics and two paraplegics, all in ASIA grade A, three without spasticity, one with grade 1 spasticity in lower limbs and two patients having fever with an increase in calf girth.
Those patients who developed DVT were managed by 15 000 IU of subcutaneous heparin,16 started with a 12 hourly dosage schedule, which was, if needed, increased to an 8 hourly schedule, till activated partial thromboplastin time (APTT) was 1.5 times baseline. Thereafter, warfarin was given (2 mg) simultaneously with heparin till INR was 1.5–2 times. Later, heparin was withdrawn and the patient was maintained on warfarin until first follow-up.
Data were analyzed using a non-parametric test (χ2 with Yate's correction as some data have <5 value).
Heparin prophylaxis had no significant correlation with the incidence of DVT (P<0.05). Significant correlation between occurrence of DVT and various demographic factors was not found (P<0.05). The level of injury, ASIA grading, spasticity in lower limbs and day of admission after injury were not related with the occurrence of DVT (P<0.05). Fever had high significant correlation (P<0.01) and increase in calf girth had very high significant correlation (P<0.001) with occurrence of DVT. When we take both clinical signs, namely, fever as well as increase in calf girth into account, they had very high significant correlation with the occurrence of DVT (P<0.001).
Discussion
The literature reveals that the incidence of DVT varies from population to population and from country to country. Watson,5, 7 at two different times, using the same diagnostic modality in the same population, revealed different incidence (17%, 14%). Silver8 and Watson,7 at the same time using the same diagnostic method, but at two different places in the UK, revealed a different incidence of DVT (25 and 17%, respectively). Silver8 and Watson,5, 7 in the UK and Todd et al.12 and Rossi et al.11 in USA revealed different incidence. This indicates that there is wide variation in the incidence of DVT. Whether this difference is because of the difference in the patient sample or patient care is not clear.6
Perkash and colleagues6 studied that the level of injury and spasticity have no correlation with thromboembolism. Our study was also in concurrence with these findings. Watson5, 7 reported a higher incidence of thromboembolism in complete lesions and in thoracic lesions. Our study was at variance with these findings as the level of injury and ASIA grade were not found to be statistically related with the occurrence of DVT
Clinical signs, that is, increase in calf girth5, 7, 8, 9, 13, 17 and fever,18, 19, 20 have been supported in various studies for diagnosing DVT. We found that if the patient had both fever as well as an increase in calf girth, then they had highly significant correlation with the occurrence of DVT. This is in concordance with the study carried out by Swarczinski.17
We have treated patients of DVT with subcutaneous heparin and warfarin, which was found to be safe for those cases in which continuous monitoring of APTT and international normalized ratio (INR) is not possible.
There is no statistically significant difference in the incidence of DVT in the study (1.8%) and control groups (3%) in our study, and the cost incurred on heparin prophylaxis is almost equal to 1-month livelihood of a five-member family.
Chahal21 was of the opinion that thrombotic complications rarely occur in India. A recent study by Saraf et al.4 showed a low incidence of DVT (10%) and pulmonary embolism (zero percent) in acute SCI patients without any prophylaxis. Their incidence is relatively more than that in our study, as they have not used physical methods like effleurage and range of movement exercises from the day of admission as suggested by Hove,9 which we have used. The reasons for the low incidence of DVT in Indian patients could be genetic factors, such as an inherited resistance to thrombosis formation, environmental factors or the low socio-economic status of our rural patients, leading to a lower consumption of a fat-rich diet and warmer climatic conditions and so on.4, 21
On considering the high cost of chemoprophylaxis, we recommend that routine DVT prophylaxis is not needed; it increases the treatment cost, which is very important in a developing country such as India where its cost is equal to 1-month livelihood of a five-member family of the patient. Therefore, all centers involved in the management of acute SCI patients must find out the incidence of DVT in their population, and if the incidence of DVT in their population is statistically significant, chemoprophylaxis must be carried out. However, if the incidence is not statistically significant, then physical methods as suggested by Hove9 must be used to achieve 0% incidence of DVT.
Conclusion
To conclude, day of admission, level of injury, ASIA grading and spasticity have no correlation with the incidence of DVT. Clinical signs, namely, fever and increase in calf girth, are highly correlated with DVT. Heparin prophylaxis has no role in reducing the incidence of DVT to a statistically significant point. Physical therapy measures, such as range of motion exercise and gentle effleurage, should be encouraged. If a clinical examination reveals an increase in calf girth and rise in temperature, DVT should be suspected and the patient must be investigated to exclude DVT.
We recommend that studies on the incidence of DVT in SCI should be carried out at centers where such patients of acute SCI are managed. If incidence is statistically significant in that population, then prophylaxis must be carried out.
References
Aito S, Pieri A, Andrea MD, Marcelli F, Cominelli E . Primary prevention of DVT and PE in acute SCI patients. Spinal Cord 2002; 40: 300–303.
Miranda AR, Haussoni HI . Mechanism of thrombosis in SCI. Haematol Oncol Clin North Am 2000; 14: 401–416.
Merli GJ, Crabbe S, Paluzzi RG, Fritz D . Etiology, incidence and prevention of DVT in acute SCI. Arch PMR 1993; 74: 1199–1205.
Saraf SK, Rana RJB, Sharma OP . Venous thromboembolism in acute spinal cord injury patients. Indian J Orthop 2007; 41: 194–197.
Watson N . Anticoagulant therapy in the prevention of venous thrombosis & PE in the SCI. Paraplegia 1978; 16: 265–269.
Perkash A, Perkash V, Perkash I . Experience with management of thromboembolism in patients with SCI: part I incidence, diagnosis & role of some risk factors. Paraplegia 1978; 16: 322–331.
Watson N . Anticoagulant therapy in the treatment of venous thrombosis & PE in acute SCI. Paraplegia 1974; 12: 197–201.
Silver JR . The prophylactic use of anticoagulant therapy in the patients of PE in 100 conservative spinal injury patients. Paraplegia 1974; 12: 188–196.
Hove EV . Prevention of thrombophlebitis in spinal injury patients. Paraplegia 1978; 16: 332–335.
Merli GJ, Herbison GJ, Ditunno JF, Weitz HH, Henzes JH, Park CH et al. Deep vein thrombosis : prophylaxis in acute spinal cord injury patients. Arch PMR 1988; 69: 661–664.
Rossi EC, Green D, Rosen JS, Spies SM, Yao JST . Sequential changes in factor VIII & platelet, preceding DVT in patients with SCI. Br J Haematol 1980; 45: 143–151.
Todd JW, Frisbie JH, Rossier AB, Adams DF, Als AV, Armenia RJ et al. DVT in acute SCI: a comparison of I125 fibrinogen leg scanning, IPG & venography. Paraplegia 1976; 14: 50–57.
Casas ER, Sanchez MP, Arias CR, Masip JP . Prophylaxis of venous thrombosis & PE in patients with acute traumatic spinal cord lesions. Paraplegia 1977; 15: 209–214.
Green D, Lee MY, Ito VY, Cohn T, Press J, Filbrandt PR et al. Prevention of thromboembolism after SCI using LMWH. Ann Int Med 1990; 113: 571–574.
Green D, Lee MY, Lim AC, Chmiel JS, Vetter M, Pang T et al. Fixed vs adjusted dose heparin in the prophylaxis and thromboembolism in SCI. JAMA 1988; 260: 1255–1258.
Doyle DJ, Turpie AG, Hirsh J, Best C, Kinch D, Levine MN et al. Adjusted SC heparin or IV heparin in patients with acute DVT. Ann Int Med 1987; 107: 441–445.
Swarczinski C . The value of serial calf girth measurements for monitoring DVT in SCI. J Neurosci Nurs 1991; 23: 306–314.
Watson N . Venous thrombosis & PE in SCI. Paraplegia 1968; 6: 113–121.
Walsh JJ, Tribe C . Phlebothrombosis & PE in paraplegia. Paraplegia 1965; 30: 209–213.
Beraldo PSS, Neves EGC, Alves CMF, Khan P, Cirilo ACM, Alencar MRB . Pyrexia in hospitalized SCI patients. Paraplegia 1993; 31: 186–191.
Chahal A . Proceedings of the annual scientific meeting the International Medical Society of Paraplegia discussing. Int J Paraplegia 1974; 12: 202 quoted from Casas ER, Sanchez MP, Arias CR, Masip JP. Prophylaxis of venous thrombosis & PE in patients with acute traumatic spinal cord lesions. Paraplegia, 1977–78; 15: 209–214.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Agarwal, N., Mathur, N. Deep vein thrombosis in acute spinal cord injury. Spinal Cord 47, 769–772 (2009). https://doi.org/10.1038/sc.2009.37
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2009.37
Keywords
This article is cited by
-
Comparison of two pharmacological prophylaxis strategies for venous thromboembolism in spinal cord injury patients: a retrospective study
Spinal Cord (2019)
-
Effect of and satisfaction with www.elearnSCI.org for training of nurse students: a submodule pilot study
Spinal Cord (2014)
-
Pharmacological prophylaxis for deep vein thrombosis in acute spinal cord injury: an Indian perspective
Spinal Cord (2014)
-
Heparin for venous thromboembolism prophylaxis in patients with acute spinal cord injury: a systematic review and meta-analysis
Spinal Cord (2013)
-
Physical therapy versus heparin for prevention of deep venous thrombosis
Spinal Cord (2011)