Abstract
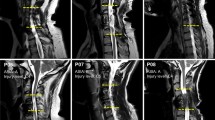
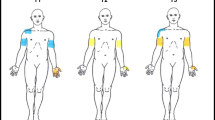
We questioned whether deafferentation following SCI would result in an increase in somatic sensitivity possibly due to cortical reorganization. Dysesthetic pain syndrome (DPS) below the level of a spinal cord injury (SCI) is a common complication. We hypothesized that DPS patients would show increased cortical reorganization because of high levels of sensory stimulation following injury. Sixteen dysesthetic pain SCI patients, 15 SCI patients without pain, and 16 control subjects were examined for two-point discrimination thresholds (2PDT) of the forearm, neck, and spine. The SCI pain group had significantly smaller 2PDTs than either SCI no pain or control groups, particularly over the neck and spine. The SCI pain group had a significant inverse correlation between perceived degree of pain (visual analogue scale) and 2PDT in the spinal skin area. The findings indicate that SCI patients with severe DPS have a higher sensitivity to somatosensory stimuli, particularly in skin areas with projections to primary somatosensory cortex areas adjacent to the deafferentated region. The increase in 2PDT may be due to an increase in the size of the somatosensory cortical areas allotted to the corresponding skin areas.
Similar content being viewed by others
Article PDF
References
Davidoff G, Roth E, Guarracini M, Sliwa J, Yarkony G (1987) Function limiting dysesthetic pain syndrome among traumatic spinal cord injury patients: a cross-sectional study. Pain 29: 39–48.
White J C, Sweet W H (1969) Pain of spinal origin. In: Thomas CC, editor. Pain and Neurosurgeon, A Forty Year Experience. Springfield, Illinois: 435–479.
Lenz F A, Tasker R R, Dostrovsky J O, Kwan H C, Gorecki J, Hirayama T, Murphy J T (1987) Abnormal single-unit activity recorded in the somatosensory thalamus of a quadriplegic patient with central pain. Pain 31: 225–236.
Lenz F A, Kwan H C, Dostrovsky J O, Tasker R R (1989) Characteristics of the bursting pattern of action potentials that occurs in the thalamus of patients with central pain. Brain Res 496: 357–360.
Melzack R (1991) The gate control theory 25 years later: new perspectives on phantom limb pain. In: Bond MR, Charlton JE, Woolf CJ, editors. Proc Vlth World Congress on Pain. Elsevier, Amsterdam: 9–21.
Melzack R, Loeser J D (1978) Phantom body pain in paraplegics: evidence for a central ‘pattern generating mechanism’ for pain. Pain 4: 195–210.
Cesaro P, Mann M W, Moretti J L, Defer G, Roualdes B, Nguyen J P, et al (1991) Central pain and thalamic hyperactivity: a single photon emission computerized tomographic study. Pain 47: 329–336.
Cohen L G, Topka H, Cole R A, Hallett M (1991) Leg paresthesias induced by magnetic brain stimulation in patients with thoracic spinal cord injury. Neurology 41: 1283–1288.
Guilbaud G, Peschanski M, Besson J M (1989) Experimental data related to nociception and pain at the supraspinal level. In: PD Wall, R Melzack, editors. Textbook of Pain. Churchill Livingstone, New York: 141–153.
Marshall J (1951) Sensory disturbances in cortical wounds with special reference to pain. J Neurol Neurosurg Psychiatry 14: 187–204.
White J C, Sweet W H (1969) Cerebral operations for relief of pain, In: CC Thomas, editor. Pain and Neurosurgeon, A Forty Year Experience. Springfield, Illinois: 773–842.
Sweet W H (1982) Cerebral localization of pain, In: RA Thompson, JR Green, editors. New Perspectives in Cerebral Localization. Raven Press, New York: 205–242.
Jones A K P, Qi L Y, Fujirawa T, Luthra S K, Ashburner J, Bloomfield P et al (1991) In vivo distribution of opioid receptors in man in relation to the cortical projections of the medial and lateral pain systems measured with positron emission tomography, Neurosci Lett 126: 25–28.
Chulder E H, Anton F, Dubner R, Kenshalo D R Jr (1990) Responses of nociceptive SI neurons in monkeys and pain sensation in humans elicited by noxious thermal stimulation: effect of interstimulus interval. J Neurophysiol 63: 559–569.
Talbot J D, Marritt S, Evans A C, Meyer E, Bushnell M C, Duncan G H (1991) Multiple representions of pain in human cerebral cortex. Science 251: 1355–1358.
Kenshalo D R Jr, Isensee O (1983) Responses of primate SI cortical neurons to noxious stimuli. J Neurophysiol 50: 1476–1496.
Kenshalo D R Jr, Chudler E H, Anton F, Dubner R (1988) SI nociceptive neurons participate in the encoding process by which monkeys perceive the intensity of noxious thermal stimulation. Brain Res 454: 378–382.
Cervero F, Shouenborg J, Sjolund B H, Waddell P J (1984) Cutaneous inputs to dorsal horn neurons in adult rats treated at birth with capsaicin. Brain Res 301: 47–57.
Cook A J, Woolf C J, Wall P D, McMahon S B (1987) Dynamic receptive field plasticity in rat spinal cord dorsal horn following C-primary afferent input. Nature 325: 151–153.
Hoheisel U, Mense S (1989) Long-term changes in discharge behaviour of cat dorsal horn neurons following noxious stimulation of deep tissues. Pain 36: 239–247.
Hylden J L K, Nahin R L, Traub R J, Dubner R (1989) Expansion of receptive fields of spinal lamina I projection neurons in rats with unilateral adjuvant-induced inflammation: the contribution of dorsal horn mechanisms. Pain 37: 229–243.
Laird J M A, Cervero F (1989) A comparative study of the changes in receptive-field properties of multireceptive and nociceptive rat dorsal horn neurons following noxious mechanical stimulation, J Neurophysiol 62: 854–863.
Woolf C J, King A E (1990) Dynamic alterations in the cutaneous mechanoreceptive fields of dorsal horn neurons in the rat spinal cord. J Neurosci 10: 2717–2726.
Ovelmen-Levitt J, Johnson B, Bedenbaugh P, Nashold B S Jr (1984) Dorsal root rhizotomy and avulsion in the cat: A comparison of long term effects on dorsal horn neuronal activity. Neurosurgery 15: 921–927.
Kjerulf T D, Loeser J D (1973) Neuronal hyperactivity following deafferentation of the lateral cuneate nucleus. Exp Neurol 39: 70–85.
Nashold B S Jr (1988) Deafferentation pain in man and animals as it relates to the DREZ operation. Can J Neurol Sci 15: 5–9.
Kaas J H, Merzenich M M, Killackey H P (1983) The reorganization of somatosensory cortex following peripheral nerve damage in adult and developing mammals. Ann Rev Neurosci 6: 325–326.
Kelahan A M, Doetsch G S (1984) Time-dependent changes in the functional organization of somatosensory cerebral cortex following digit amputation in adult raccoons. Somatosens Res 2: 49–81.
Merzenich M M, Nelson R J, Stryker M P, Cynader M S, Schoppmann A, Zook J M (1984) Somatosensory cortical map changes following digit amputation in adult monkeys. J Comp Neurol 224: 591–605.
Wall J T, Kaas J T, Sur M, Nelson R J, Fellman D J, Merzenich M M (1986) Functional reorganization in somatosensory cortical areas 3b and 1 of adult monkeys after median nerve repair: possible relationships to sensory recovery in human. J Neurosci 6: 218–233.
Haber W B (1958) Reactions to loss of a limb: Physiological and psychological aspects. Ann N Y Acad Sci 74: 14–24.
Moberg E (1962) Criticism and study of methods for examining sensibility in the hand. Neurology 12: 8.
Moberg E (1990) Two-point discrimination test. Scand J Rehabil Med 22: 127–134.
McLeod J G, Lance J W (1983) Neurological examination. In: JG McLeod, JW Lance, editors. Introductory Neurology. Melbourne: Blackwell Scientific Publications: 30.
Dellon A L, Mackinnon S E, Crosby P M (1987) Reliability of two-point discrimination measurements. J Hand Surg 12A: 693–696.
Dellon A L, Kallman C H (1983) Evaluation of functional sensation in the hand. J Hand Surg 8: 865–870.
Goldstein E B (1984) Perceiving touch, temperature, and pain. In: EB Goldstein, editor. Sensation and Perception. Wadsworth, Belmont, California: 85–108.
Bell-Krotoski J A, Buford W L (1988) The force/time relationship of clinically used sensory testing instruments. J Hand Ther 1: 76–85.
American Spinal Injury Association (1982) Standards of Neurological Classification of Spinal Injury Patients. Chicago, IL: 2–14.
Peters M L, Schmidt A J M (1991) A comparison of two-point discrimination threshold of tactual, non-painful stimuli between chronic low back pain patients and controls. Pain 44: 57–60.
Seltzer S F, Seltzer J L (1986) Tactual sensitivity of chronic pain patients to non-painful stimuli. Pain 27: 291–295.
Dubner R (1991) Neuronal plasticity and pain following peripheral tissue inflammation or nerve injury. In: MR Bond, JE Charlton, CG Woolf, editors. Proc Vlth World Congress on Pain. Elsevier, Amsterdam: 263–276.
Dubner R (1992) Hyperalgesia and expanded receptive fields. Pain 48: 3–4.
Jenkins W M, Merzenich M M, Ochs M T, Allard T, Guic-Robles E (1990) Functional reorganization of primary somatosensory cortex in adult owl monkeys after behavioral controlled tactile stimulation. J Neurophysiol 63: 82–104.
Clark S A, Allard T, Jenkins W M, Merzenich M M (1988) Receptive fields in the body-surface map in adult cortex defined by temporally correlated inputs. Nature 332: 444–445.
Levy W J, Amassian V E, Traad M, Cadwell J (1990) Focal magnetic coil stimulation reveals motor cortical system reorganized in humans after traumatic quadriplegia. Brain Res 510: 130–134.
Topka H, Cohen L G, Cole R A, Hallett M (1991) Reorganization of corticospinal pathways following spinal cord injury. Neurology 41: 1276–1283.
Katz J, Melzack R (1990) Pain ‘memories' in phantom limbs: review and clinical observations. Pain 43: 319–336.
Goldberger M E, Murray M, Tessler A (1993) Sprouting and regeneration in the spinal cord: their roles in recovery of function after spinal injury. In: A Gorio, editor. Neuroregeneration. Raven Press, New York: 241–264.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Song, Z., Cohen, M., Ament, P. et al. Two-point discrimination thresholds in spinal cord injured patients with dysesthetic pain. Spinal Cord 31, 485–493 (1993). https://doi.org/10.1038/sc.1993.79
Issue Date:
DOI: https://doi.org/10.1038/sc.1993.79
Keywords
This article is cited by
-
Upper cervical two-point discrimination thresholds in migraine patients and headache-free controls
The Journal of Headache and Pain (2018)