Abstract
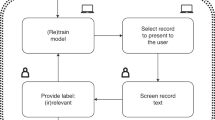
Scientific publications in mental health are rapidly growing in number and complexity, making curation of abstracts for systematic reviews increasingly time consuming and challenging. As a result, systematic reviews on broad topics have also complicated human review due, in part, to variations and lack of objectivity among multiple human reviewers. Resolving these complexities can be time consuming and impact the accuracy and breadth of systematic reviews. To address these challenges, we propose and evaluate multiple machine-learning-based approaches to capture inclusion and exclusion criteria and automate the abstract selection process. We fine-tuned or trained models on psychiatry abstracts from four systematic-review topic areas. The models were then applied to abstracts derived from an independently curated oncology literature database. Transformer-based machine-learning models outperformed trained human reviewers in abstract screening for three out of four topic areas with accuracies ranging from –4% to 17.7%. Such approaches may facilitate the sharing and synthesis of research expertise across disciplines.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$59.00 per year
only $4.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Source data are provided with this paper. All papers used as source data for abstracts are available at Embase, Web of Science, PsycInfo, CINAHL, PubMed, and NCI. All datasets, including abstracts, used for machine learning and evaluation of results (human reviewers and automated) are clearly identified and publicly available at https://github.com/MetaAnalysisPipeline/MetaAnalysisPipeline.
Code availability
The code used for text preprocessing and machine-learning pipelines for SciBERT, BERT, and Naïve Bayes are available at https://github.com/MetaAnalysisPipeline/MetaAnalysisPipeline.
References
Levels of Evidence and Grades for Recommendations for Developers of Clinical Practice Guidelines (NHMRC, 2009).
Hoffmann, T., Bennett, S. & Mar, C. D. Evidence-Based Practice Across the Health Professions (Churchill Livingstone, 2014).
Kendall, S. Evidence-based resources simplified. Can. Fam. Physician 54, 241–243 (2008).
Davidson, M. & Iles, R. in Research Methods in Health: Foundations for Evidence-Based Practice (ed. Liamputtong, P.) 285–300 (Oxford Univ. Press, 2010).
Cook, D. J., Mulrow, C. D. & Haynes, R. B. Systematic reviews: synthesis of best evidence for clinical decisions. Ann. Intern. Med. 126, 376–380 (1997).
Glass, G. V. Primary, secondary, and meta-analysis of research. Educ. Res. 5, 3–8 (1976).
Greco, T., Zangrillo, A., Biondi-Zoccai, G. & Landoni, G. Meta-analysis: pitfalls and hints. Heart Lung Vessels 5, 219–225 (2013).
Michelson, M. & Reuter, K. The significant cost of systematic reviews and meta-analyses: a call for greater involvement of machine learning to assess the promise of clinical trials. Contemp. Clin. Trials Commun. 16, 100443 (2019).
Allen, I. E. Estimating time to conduct a meta-analysis from number of citations retrieved. JAMA 282, 634–635 (1999).
Polanczyk, G., de Lima, M. S., Horta, B. L., Biederman, J. & Rohde, L. A. The worldwide prevalence of ADHD: a systematic review and metaregression analysis. Am. J. Psychiatry 164, 942–948 (2007).
Kennis, M. et al. Prospective biomarkers of major depressive disorder: a systematic review and meta-analysis. Mol. Psychiatry 25, 321–338 (2020).
Broyd, S. J. et al. Default-mode brain dysfunction in mental disorders: a systematic review. Neurosci. Biobehav. Rev. 33, 279–296 (2009).
Dowlati, Y. et al. A meta-analysis of cytokines in major depression. Biol. Psychiatry 67, 446–457 (2010).
Cipriani, A. et al. Comparative efficacy and acceptability of 21 antidepressant drugs for the acute treatment of adults with major depressive disorder: a systematic review and network meta-analysis. Lancet 391, 1357–1366 (2018).
Moore, T. H. M. et al. Cannabis use and risk of psychotic or affective mental health outcomes: a systematic review. Lancet 370, 319–328 (2007).
Xiong, J. et al. Impact of COVID-19 pandemic on mental health in the general population: a systematic review. J. Affect. Disord. 277, 55–64 (2020).
Leucht, S. et al. Comparative efficacy and tolerability of 15 antipsychotic drugs in schizophrenia: a multiple-treatments meta-analysis. Lancet 382, 951–962 (2013).
Bown, M. J. & Sutton, A. J. Quality control in systematic reviews and meta-analyses. Eur. J. Vasc. Endovasc. Surg. 40, 669–677 (2010).
Gurevitch, J., Koricheva, J., Nakagawa, S. & Stewart, G. Meta-analysis and the science of research synthesis. Nature. 555, 175–182 (2018).
Elliott, J. H. et al. Living systematic reviews: an emerging opportunity to narrow the evidence–practice gap. PLoS Med. 11, e1001603 (2014).
Lerner, I., Créquit, P., Ravaud, P. & Atal, I. Automatic screening using word embeddings achieved high sensitivity and workload reduction for updating living network meta-analyses. J. Clin. Epidemiol. 108, 86–94 (2019).
Bao, Y. et al. Using machine learning and natural language processing to review and classify the medical literature on cancer susceptibility genes. JCO Clin. Cancer Inform. https://doi.org/10.1200/CCI.19.00042 (2019).
Bannach-Brown, A. et al. Machine learning algorithms for systematic review: reducing workload in a preclinical review of animal studies and reducing human screening error. Syst. Rev. 8, 23 (2019).
Lange, T., Schwarzer, G., Datzmann, T. & Binder, H. Machine learning for identifying relevant publications in updates of systematic reviews of diagnostic test studies. Res. Synth. Methods https://doi.org/10.1002/jrsm.1486 (2021).
Khalil, H., Ameen, D. & Zarnegar, A. Tools to support the automation of systematic reviews: a scoping review. J. Clin. Epidemiol. 144, 22–42 (2022).
Ouzzani, M., Hammady, H., Fedorowicz, Z. & Elmagarmid, A. Rayyan-a web and mobile app for systematic reviews. Syst. Rev. 5, 210 (2016).
Gates, A. et al. Performance and Usability of Machine Learning for Screening in Systematic Reviews: A Comparative Evaluation of Three Tools (Agency for Healthcare Research and Quality, 2019); http://www.ncbi.nlm.nih.gov/books/NBK550175/
Orgeolet, L. et al. Can artificial intelligence replace manual search for systematic literature? Review on cutaneous manifestations in primary Sjögren’s syndrome. Rheumatology (Oxford) 59, 811–819 (2020).
Xiong, Z. et al. A machine learning aided systematic review and meta-analysis of the relative risk of atrial fibrillation in patients with diabetes mellitus. Front. Physiol. 9, 835 (2018).
Tshitoyan, V. et al. Unsupervised word embeddings capture latent knowledge from materials science literature. Nature. 571, 95–98 (2019).
Olier, I. et al. Transformational machine learning: learning how to learn from many related scientific problems. Proc. Natl Acad. Sci. USA 118, e2108013118 (2021).
Nichols, J. D., Oli, M. K., Kendall, W. L. & Boomer, G. S. Opinion: a better approach for dealing with reproducibility and replicability in science. Proc Natl Acad. Sci. USA 118, e2100769118 (2021).
Patel, B. N. et al. Human–machine partnership with artificial intelligence for chest radiograph diagnosis. NPJ Digit. Med. 2, 111 (2019).
Marshall, I. J., Noel-Storr, A., Kuiper, J., Thomas, J. & Wallace, B. C. Machine learning for identifying randomized controlled trials: an evaluation and practitioner’s guide. Res. Synth. Methods. 9, 602–614 (2018).
Wolf, T. et al. HuggingFace’s transformers: state-of-the-art natural language processing. ArXiv http://arxiv.org/abs/1910.03771 (2020).
Norman, C. R., Leeflang, M. M. G., Porcher, R. & Névéol, A. Measuring the impact of screening automation on meta-analyses of diagnostic test accuracy. Syst. Rev. 8, 243 (2019).
Frénay, B. & Kabán, A. A comprehensive introduction to label noise. In Proc. European Symposium on Artificial Neural Networks, Computational Intelligence and Machine Learning 267–276 (ESANN, 2014).
Delgado-Rodriguez, M. Bias. J. Epidemiol. Community Health. 58, 635–641 (2004).
Song, H., Kim, M., Park, D. & Lee, J. G. Learning from noisy labels with deep neural networks: a survey. ArXiv http://arxiv.org/abs/2007.08199 (2020).
Beltagy, I., Lo, K. & Cohan, A. SciBERT: a pretrained language model for scientific text. ArXiv http://arxiv.org/abs/1903.10676 (2019).
Devlin, J., Chang, M. W., Lee, K. & Toutanova, K. BERT: pre-training of deep bidirectional transformers for language understanding. ArXiv http://arxiv.org/abs/1810.04805 (2019).
Pedregosa, F. et al. Scikit-learn: machine learning in Python. J. Mach. Learn. Res. 12, 2825–2830 (2011).
Vaswani, A. et al. Attention is all you need. ArXiv http://arxiv.org/abs/1706.03762 (2017).
Berlin, J. A. & Golub, R. M. Meta-analysis as evidence: building a better pyramid. JAMA. 312, 603–605 (2014).
Oremus, M., Oremus, C., Hall, G. B. C. & McKinnon, M. C. ECT & cognition systematic review team. Inter-rater and test–retest reliability of quality assessments by novice student raters using the Jadad and Newcastle–Ottawa Scales. BMJ Open 2, e001368 (2012).
Atkinson, D. & Murray, M. Improving Interrater Reliability (ERIC, 1987); https://eric.ed.gov/?id=ED287175.
Linder, S. K., Kamath, G. R., Pratt, G. F., Saraykar, S. S. & Volk, R. J. Citation searches are more sensitive than keyword searches to identify studies using specific measurement instruments. J. Clin. Epidemiol. 68, 412–417 (2015).
Kanaris, I., Kanaris, K., Houvardas, I. & Stamatatos, E. Words vs. character n-grams for anti-spam filtering. Int. J. Artif. Intel.l 20, 1–20 (2006).
Chen, P. H., Zafar, H., Galperin-Aizenberg, M. & Cook, T. Integrating natural language processing and machine learning algorithms to categorize oncologic response in radiology reports. J. Digit. Imaging. 31, 178–184 (2018).
Yamamoto, S., Lauscher, A., Ponzetto, S. P., Glavaš, G. & Morishima, S. Self-supervised learning for visual summary identification in scientific publications. ArXiv http://arxiv.org/abs/2012.11213 (2021).
Bisong, E. In Building Machine Learning and Deep Learning Models on Google Cloud Platform: A Comprehensive Guide for Beginners 1st edn (ed. Bisong, E.) 59–64 (Apress, 2019); https://doi.org/10.1007/978-1-4842-4470-8_7.
McKinney, W. Data Structures for Statistical Computing in Python (SciPy, 2010); https://doi.org/10.25080/Majora-92bf1922-00a.
Harris, C. R. et al. Array programming with NumPy. Nature 585, 357–362 (2020).
Abadi, M. et al. TensorFlow: Large-Scale Machine Learning on Heterogeneous Distributed Systems. ArXiv https://arxiv.org/abs/1603.04467 (2016).
Paszke, A. et al. PyTorch: An Imperative Style, High-Performance Deep Learning Library. ArXiv https://arxiv.org/abs/1912.01703 (2019).
Van Rossum, G. & Drake, F. L. Python 3 Reference Manual (CreateSpace, 2009).
Acknowledgments
We express appreciation to the members of our Pediatric Emotion and Resilience Lab (PEARL) for their contributions to this work. We have no funding to disclose related to this work.
Author information
Authors and Affiliations
Contributions
A.J.G., M.G.G., K.K.R., and M.K.S. conceptualized and executed the study protocol. A.J.G., M.G.G., K.K.R., and M.K.S. contributed to the analyses. A.J.G., M.G.G., M.K.S., K.K.R., V.P., and A.N. contributed to writing of the manuscript. A.J.G. and M.G.G. contributed to visualizations. K.K.R., A.N., A.F.N., V.P., S.R.K., T.P., M.L., S.S., and M.K.S. contributed to the manual tagging of abstracts.
Corresponding author
Ethics declarations
Competing interests
M.K.S. has received research support from Stanford’s Maternal Child Health Research Institute and Stanford’s Department of Psychiatry and Behavioral Sciences, National Institute of Mental Health, National Institute of Aging, Patient Centered Outcomes Research Institute, Johnson and Johnson, and the Brain and Behavior Research Foundation. She is on the advisory board for Sunovion and Skyland Trail and is a consultant for Johnson and Johnson, Alkermes, and Neumora. She has previously consulted for X, moonshot factory, Alphabet Inc., and Limbix Health. She receives honoraria from the American Academy of Child and Adolescent Psychiatry and royalties from American Psychiatric Association Publishing and Thrive Global. K.K.R. receives support from The Permanente Medical Group’s Physician Researcher Program. No other authors report any biomedical financial interests or potential conflicts of interest.
Peer review
Peer review information
Nature Mental Health thanks Federica Colombo and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–10, Tables 1–14, and Methods.
Supplementary Data
Source data for Supplementary Figs. 1–5, 7, and 8 and Supplementary Tables 11–13.
Source data
Source Data Fig. 2
Source data for Fig. 2.
Source Data Fig. 3
Source data for Fig. 3.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gorelik, A.J., Gorelik, M.G., Ridout, K.K. et al. Evaluating efficiency and accuracy of deep-learning-based approaches on study selection for psychiatry systematic reviews. Nat. Mental Health 1, 623–632 (2023). https://doi.org/10.1038/s44220-023-00109-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44220-023-00109-w