Abstract
Resilience is the cornerstone to mental health, and entails multiple biological and psychological mechanistic processes. However, the interplay of the psychobiological processes in shaping resilience is unclear. Here we report the results of testing whether an acute cortisol response and positive affectivity traits moderate the relationship between participants’ five-year major life stress and current psychological symptoms. The participants comprised 147 individuals (93 females and 54 males, age = 24–45 years) without clinical diagnosis. Acute stress was induced using the Trier Social Stress Task. We found that both the cortisol response to anticipatory acute stress and positive affectivity moderated the stress–symptom relationship. Specifically, a positive relationship between life stress and current symptoms was only observed at low, but not high, levels of cortisol response and positive affectivity. Moreover, the moderating effect of cortisol response was only observed at a low level of trait positive affectivity. These results unravel how the biological and emotional processes of the stress response interact to shape resilience to major life stress.
Similar content being viewed by others
Main
Chronic stress causes overarching adverse consequences for mental health1. Past studies have suggested that major life stress predicted subsequent major depression2, and associated closely with the onset of anxiety disorders, addiction and suicide3,4. On the other hand, individuals with high stress resilience are, by definition, more immune to negative stress-related mental health consequences5. Accumulating evidence suggests that resilience has both biological and psychological markers that could help identify individuals with differential vulnerability to chronic stress6,7. For example, individual differences in cortisol responses to chronic stress predicted depressive and anxiety symptoms3.
Stress triggers multi-faceted biological responses, primarily by activating the hypothalamus–pituitary–adrenal (HPA) axis, which induces secretion of cortisol as part of the adaptation process to environmental challenges8. Cortisol triggers widespread downstream effects on biological and emotional systems9. For example, cortisol exerts important regulatory functions on the amygdala, hippocampus and prefrontal cortex, which may mediate its acute and long-term effects on affective response and regulation5. Importantly, existing evidence indicates that more pronounced cortisol responses to acute stress are associated with, and prospectively predicted, resilience to chronic life stress over a four-year period10. Moreover, the offspring of mothers with post-traumatic stress disorder (PTSD), who may possess increased risk for psychopathology, exhibited a reduced cortisol response to acute stress compared to offspring of non-PTSD mothers11. These findings collectively indicate cortisol response to acute stress is a biological marker of resilience to chronic stress7.
On the psychological level, past evidence suggests that better mental health outcomes following chronic stress are associated with more positive affectivity12, which refers to the trait of being joyful, interested and contented in life. Positive affectivity involves positive affect and positive emotion regulation strategies. A higher level of positive affect was previously found to ‘buffer’ the association between chronic life stress and affective symptoms among young adults, such that the positive relationship between chronic stress and affective symptoms was only observed among individuals with low positive affect, but not among those with high positive affect13. On the other hand, negative affect showed no significant moderating effect, suggesting that positive affect may be particularly important for maintaining good mental health under chronic life stress5. Furthermore, trait positive affect prospectively predicted fewer psychological symptoms six months later among adolescents coping with type-1 diabetes14.
One positive emotion regulation strategy is positive reappraisal, or reframing, which entails the tendency to reinterpret adverse situations in positive or meaningful manners15. A greater tendency towards positive reappraisal prospectively predicted reduced lifetime likelihood of developing PTSD in combat veterans16, and was associated with better psychosomatic adjustment after surviving a natural disaster17. Furthermore, among women newly diagnosed with cancer, both positive affect and positive reappraisal were associated with better self-reported quality of life18.
Another positive emotion regulation strategy is positive refocusing, which involves steering one’s attention away from the adverse event towards other positive stimuli19. A higher tendency towards positive refocusing was previously found to protect against both depression and anxiety symptoms20. Among patients with hypertension, positive refocusing at baseline predicted lower levels of depressive symptoms six months later21. Among young adults, greater use of positive refocusing was associated with better psychological adaptation to life adversity22. Therefore, positive affect, positive reappraisal and positive refocusing are strong candidate psychological markers for resilience to chronic stress. Although these traits are related, they are conceptually distinct from each other, as positive affect refers to one’s chronic emotional status, whereas positive reappraisal and refocusing are two different emotion regulation strategies, respectively involving cognitive reframing of events and redirecting one’s attentions.
Although existing evidence indicates that both the acute cortisol response and trait positive affectivity are candidate stress resilience markers, no study has investigated how they interact in relation to resilience to life stress. This is particularly important given that stress responses are multisystemic in nature and entail interacting, coordinated psychobiological processes23. To achieve this aim, we induced acute stress using the well-established Trier Social Stress Test (TSST)24 on a sample of adults without major clinical diagnoses, to boost the generalizability of our findings to wider populations. Resilience was defined in relation to mental health outcome following major life stress, and individuals who maintained good mental health (fewer symptoms) despite experiencing life stress were considered resilient. Following this, variables that reduced the positive relationship between life stress and mental health symptoms were considered resilience markers13. Our primary hypothesis was that higher cortisol responses to acute stress and trait positive affectivity would reduce the positive relationship between major life stress and mental health symptoms. Our secondary hypothesis was that the moderating effect of cortisol response on the life stress–symptom relationship would further depend on trait positive affectivity.
Results
Descriptive demographic analyses
The data analyses included 147 participants (93 females and 54 males; see Supplementary Section 3 for the participant exclusion flow diagram). The mean age was 30.2 years (range = 24–45 years, standard deviation (s.d.) = 4.6 years). Participants reported 1.1 major life stress events on average (range = 0–7, s.d. = 1.3). Their mean Symptom Checklist-90 (SCL-90) total score was 37.8 (s.d. = 34.7). Age (Spearman’s ρ = 0.03 and 0.04, P > 0.66) and sex (Mann–Whitney U test Z = 1.60 and 1.71, P > 0.08) had no significant association with the number of major life stress events, or with SCL-90 score.
Descriptive correlation analyses
Cortisol and positive affectivity correlations
See Supplementary Section 4 for the participants’ mean and individual cortisol response trajectory. Controlling for the pre-TSST (T2) cortisol level, anticipatory (T3 minus T2) and peak (T4 minus T2) cortisol responses showed no significant correlation with trait positive affectivity, including positive affect, positive reappraisal and positive refocusing (|Spearman’s ρ| < 0.08, P > 0.28 and |Spearman’s ρ| < 0.11, P > 0.22, respectively). The three trait measures showed significant correlations with each other: positive affect and positive reappraisal, Spearman’s ρ = 0.46, P < 0.001; positive affect and positive refocusing, Spearman’s ρ = 0.20, P = 0.02; positive reappraisal and positive refocusing, Spearman’s ρ = 0.37, P < 0.001.
Life stress and SCL-90 correlation
As expected, major life stress (Spearman’s ρ = 0.37, P < 0.001) was associated significantly with the total SCL-90 score, even after controlling for daily hassle score (Spearman’s ρ = 0.30, P < 0.001). Major life stress and daily hassle score correlated significantly with each other (Spearman’s ρ = 0.24, P = 0.004).
Cortisol, positive affectivity and SCL-90 correlation
Controlling for the pre-TSST (T2) cortisol level, anticipatory and peak cortisol responses showed no significant correlation with SCL-90 score (P > 0.06). Of the psychological variables of interest, positive affect negatively correlated with SCL-90 total score (Spearman’s ρ = −0.36, P < 0.001). Positive reappraisal showed a trend of negatively correlating with the SCL-90 score (Spearman’s ρ = −0.14, P = 0.08). Positive refocusing showed no correlation with the SCL-90 score (P > 0.85).
TSST effect
Cortisol
Linear mixed-effect analysis revealed a significant main effect of time (F2, 288 = 41.90, P < 0.001). A post hoc paired-sample t-test revealed a significant increase of salivary cortisol level from pre-TSST (T2) to immediately after TSST (T3) (t144 = 8.65, P < 0.001), which then showed a trend of increase again at 20 min after TSST (T4) (t144 = 1.91, P = 0.06).
Profile of mood states
The total mood disturbance score computed from the Profile of Mood States (POMS) subscales showed a significant main effect of time (F1, 146 = 41.25, P < 0.001), indicating a significant increase of negative mood after TSST compared to before TSST.
Moderating the stress–symptom relationship
Cortisol
We found that the anticipatory cortisol response (T3 minus T2) significantly and negatively moderated the relationship between major life stress and SCL-90 score (F1, 139 = 5.12, bootstrapped confidence interval (CI) = −10.6462 to −0.9843, P = 0.04), while controlling for pre-TSST (T2) cortisol level and daily hassle. Follow-up analysis revealed that, at a lower increase of cortisol during anticipatory stress, major life stress correlated positively with SCL-90 score (Spearman’s ρ = 0.47, P = 0.001). This relationship became a positive trend at an intermediate level of cortisol increase (Spearman’s ρ = 0.26, P = 0.07), and was insignificant at a high level of cortisol increase (P = 0.82) (Fig. 1). The peak cortisol response (T4 minus T2) did not significantly moderate the relationship between major life stress and SCL-90 score (bootstrapped P > 0.1).
This moderating analysis was conducted using the PROCESS macro implemented in SPSS (pre-set Model No. 1). The test was two-sided. We conducted a Holm–Bonferroni correction procedure to adjust for the two types of cortisol response (anticipatory and peak) analysed. The anticipatory cortisol response (T3 minus T2) significantly and negatively moderated the relationship between major life stress and SCL-90 score (F1,139 = 5.12, bootstrapped CI = −10.6462 to −0.9843, P = 0.04), while controlling for pre-TSST (T2) cortisol level and daily hassle. At a lower cortisol response, major life stress correlated positively with SCL-90 score (Spearman’s ρ = 0.47, P = 0.001). This relationship became a positive trend at an intermediate level of cortisol increase (Spearman’s ρ = 0.26, P = 0.07), and insignificant at a high level of cortisol increase (P = 0.82). The relationship is plotted separately for participants with low, medium and high levels of anticipatory cortisol response, divided according to 33% and 67% percentiles. Note that the x and y axes include negative values due to the intercept and nuisance variables being regressed out from the dependent and independent variables in the partial correlation analysis. ***P < 0.001; **P < 0.01; *P < 0.05; NS, not significant.
Positive affectivity
Positive affect negatively moderated the relationship between major life stress and SCL-90 score (F1, 139 = 2.71, bootstrapped CI = −1.4000 to −0.0075, P = 0.05), while controlling for daily hassle. Follow-up analysis revealed that, at a low level of positive affect, major life stress correlated positively with SCL-90 score (Spearman’s ρ = 0.33, P = 0.02). This relationship became a positive trend at an intermediate level of positive affect (Spearman’s ρ = 0.26, P = 0.09), and insignificant at a high level of positive affect (P = 0.71) (Fig. 2).
This moderating analysis was conducted using the PROCESS macro implemented in SPSS (pre-set Model No. 1). The test was two-sided. As we analysed only one measure of positive affect, no multiple-testing correction was conducted. Positive affect negatively moderated the relationship between major life stress and SCL-90 score (F1, 139 = 2.71, bootstrapped CI = −1.4000 to −0.0075, P = 0.05), while controlling for daily hassle. At a low level of positive affect, major life stress correlated positively with SCL-90 score (Spearman’s ρ = 0.33, P = 0.02). This relationship became a positive trend at an intermediate level of positive affect (Spearman’s ρ = 0.26, P = 0.09), and was insignificant at a high level of positive affect (P = 0.71). The relationship is plotted separately for participants with low, medium and high levels of positive affect, divided according to 33% and 67% percentiles. Note that the x and y axes include negative values due to the intercept and nuisance variables being regressed out from the dependent and independent variables in partial correlation analysis. ***P < 0.001; **P < 0.01; *P < 0.05; NS, not significant.
A similar negative moderating effect was observed for positive refocusing (F1, 139 = 3.69, bootstrapped CI = −5.3073 to −0.5542, P = 0.04), while controlling for daily hassle. Follow-up analysis revealed that, at a low level of positive refocusing, major life stress correlated positively with SCL-90 score (Spearman’s ρ = 0.52, P = 0.01). This relationship became a positive trend at an intermediate level of positive refocusing (Spearman’s ρ = 0.19, P = 0.08), and insignificant at a high level of positive refocusing (P = 0.29) (Fig. 3).
This moderating analysis was conducted using the PROCESS macro implemented in SPSS (pre-set Model No. 1). The test was two-sided. We conducted a Holm–Bonferroni correction procedure to adjust for the two types of positive emotion regulation strategy (positive reappraisal and refocusing) analysed. Positive refocusing significantly and negatively moderated the relationship between major life stress and SCL-90 score (F1, 139 = 3.69, bootstrapped CI = −5.3073 to −0.5542, P = 0.04), while controlling for daily hassle. At a low level of positive refocusing, major life stress correlated positively with SCL-90 score (Spearman’s ρ = 0.52, P = 0.01). This relationship became a positive trend at an intermediate level of positive refocusing (Spearman’s ρ = 0.19, P = 0.08), and insignificant at a high level of positive refocusing (P = 0.29). The relationship is plotted separately for participants with low, medium and high levels of positive refocusing, divided according to 33% and 67% percentiles. Note that the x and y axes include negative values due to the intercept and nuisance variables being regressed out from the dependent and independent variables in partial correlation analysis. ***P < 0.001; **P < 0.01; *P < 0.05.
Positive reappraisal did not significantly moderate the relationship between major life stress and SCL-90 score (bootstrapped P > 0.34).
Moderated moderating effect
Positive affect
We further tested whether the moderating effect of cortisol on the life stress–symptom relationship was in turn moderated by trait positive affectivity. Moderated moderating analyses revealed that the moderating effect of cortisol was further moderated by positive affect (F1, 135 = 3.97, bootstrapped CI = 0.0204 to 2.0364, P = 0.05). Specifically, the negative moderating effect of cortisol was only observed at a low level of positive affect (F1, 47 = 7.57, bootstrapped CI = −21.5529 to −5.0381, P = 0.005), but not at an intermediate or high level of positive affect (bootstrapped P > 0.35) (Fig. 4a). Follow-up analyses revealed that at a low level of positive affect, major life stress correlated positively with SCL-90 score (Spearman’s ρ = 0.63, P = 0.01) among those with a low cortisol response, showing a trend of correlating with the SCL-90 score (Spearman’s ρ = 0.39, P = 0.06) among those with a medium cortisol response, and insignificantly among those with a high cortisol response (Spearman’s ρ = −0.18, P = 0.59).
a,b, The trait positive affect (a) and positive reappraisal (b) moderated the moderating effect of anticipatory cortisol response on the relationship between life stress and SCL-90. This moderated moderating analysis was conducted using the PROCESS macro implemented in SPSS (pre-set Model No. 3). The test was two-sided. We conducted a Holm–Bonferroni correction procedure to adjust for the two types of positive emotion regulation strategy analysed. In a, the moderating effect of cortisol is seen to be further moderated by positive affect (F1, 135 = 3.97, bootstrapped CI = 0.0204 to 2.0364, P = 0.05). The negative moderating effect of cortisol was only observed at a low level of positive affect (F1, 47 = 7.57, bootstrapped CI = −21.5529 to −5.0381, P = 0.005), but not at an intermediate or high level of positive affect (bootstrapped P > 0.35). In b, the moderating effect of cortisol was also moderated by positive reappraisal (F1, 135 = 5.28, bootstrapped CI = 0.5133 to 8.0221, P = 0.04). The negative moderating effect of cortisol was observed at a low level (F1, 38 = 5.50, bootstrapped CI = −21.5859 to −1.3569, P = 0.04) and intermediate level (F1, 55 = 4.85, bootstrapped CI = −19.7838 to −0.3268, P = 0.04) of positive reappraisal, but not at a high level (bootstrapped P = 0.45). ***P < 0.001; **P < 0.01; *P < 0.05.
Positive reappraisal
In addition, the moderating effect of cortisol was further moderated by positive reappraisal (F1, 135 = 5.28, bootstrapped CI = 0.5133 to 8.0221, P = 0.04). Specifically, the negative moderating effect of cortisol was observed at a low level (F1, 38 = 5.50, bootstrapped CI = −21.5859 to −1.3569, P = 0.04) and an intermediate level (F1, 55 = 4.85, bootstrapped CI = −19.7838 to −0.3268, P = 0.04) of positive reappraisal, but not at a high level (bootstrapped P = 0.45) (Fig. 4b). Follow-up analyses revealed that, at a low level of positive reappraisal, major life stress correlated positively with SCL-90 score (Spearman’s ρ = 0.704, P = 0.003) among those with a low cortisol response, insignificantly (Spearman’s ρ = 0.35, P = 0.20) among those with a medium cortisol response, and negatively with SCL-90 score among those with a high cortisol response (Spearman’s ρ = −0.58, P = 0.03). At an intermediate level of positive reappraisal, major life stress showed a trend of correlating positively with SCL-90 score (Spearman’s ρ = 0.48, P = 0.09) among those with a low cortisol response, and insignificantly among those with a medium or high level of cortisol response (P > 0.32).
Positive refocusing
The moderating effect of cortisol was not significantly moderated by positive refocusing (F1, 135 = 0.82, bootstrapped P > 0.36).
Discussion
In this Article we have revealed the effects of anticipatory cortisol response and trait positive affectivity on resilience to major life stress. Specifically, major life stress was positively correlated with participants’ current psychological symptoms, but only among those with a low cortisol response to anticipatory stress, or with low trait positive affectivity. On the contrary, for individuals with medium or high levels of cortisol response or trait positive affectivity, the stress–symptom association reduced in a dose-dependent manner. We additionally discovered that the effect of cortisol response was further dependent on the level of trait positive affectivity. Specifically, positive association between major life stress and current symptoms was only observed at low levels of both cortisol response and positive affectivity. These results have unravelled the intricate interplay between the biological and trait affective processes in relation to major life stress resilience.
Emerging evidence indicates an association between individuals’ anticipatory cortisol response to acute stress and their psychobiological outcomes following chronic stress. For example, one study found that the anticipatory cortisol response to the TSST mediated the effect of perceived stress and oxidative stress damage among women who cared for demented spouses, implicating a key role for the anticipatory cortisol system in underpinning the accumulative biological effects of chronic stress25. Moreover, the offspring of PTSD mothers, who were considered more vulnerable to developing stress-related psychological illnesses, exhibited a ‘flattened’ anticipatory stress response compared to offspring of non-PTSD mothers11. However, neither of those studies explicitly quantified the participants’ resilience to life stress. Our results demonstrate that a high anticipatory cortisol response nullified the pattern of increased psychological symptoms after experiencing greater levels of major life stress, indicating a protective role of the high-reactive cortisol system when the individual prepares for encountering acute stress. It is known that anticipation prior to actual occurrence of the stressor effectively activates the HPA axis, which in turn induces coordinated biological and affective responses and allows the individual to better adjust to the stressful environment26. During acute stress, the elevation in cortisol levels increases alertness27 and regulates energy metabolism and output28, preparing the body to engage in an adaptive ‘fight-or-flight’ response and achieve better performance29. In the longer term, this better adaptation to acute stress due to a greater anticipatory cortisol increase was found to dampen the effect of peer victimization in increasing rumination and depressive symptoms one year later30. On the contrary, the blunted anticipatory cortisol response was associated with major depression31, increased genetic risk for major depression32, and schizophrenia33. Thus, converging evidence suggests that a potent anticipatory cortisol response to acute stress represents a biologically adaptive system that protects an individual from mental health damage following major life stress.
We did not find a significant effect of peak cortisol response on resilience. Past findings on this association were inconsistent. Peak cortisol change was not associated with oxidative stress damage among chronically stressed women25, and was not related to a questionnaire measure of resilience among male students34. However, the peak cortisol change predicted a four-year trajectory of resilience among police officers10. Although multiple discrepancies in participant characteristics and resilience measurement preclude conclusions based on previous findings, our current results suggest that anticipatory and peak cortisol responses may have differential correlates with resilience to major life stress. This is consistent with the previously proposed separation of indirect activation of the HPA axis, which delivers stress anticipation signals, and direction activation of the HPA axis, which delivers stress reaction signals35.
Consistent with existing evidence indicating the protective effects of positive affectivity trait on mental health following major life stress13,21, we observed that both positive affect and positive refocusing ‘buffered’ the relationship between life stress and psychological symptoms. It has long been recognized that the ability to maintain a positive prospect during chronic stress is key to resilience36. In our study, positive affect and positive refocusing correlated positively with each other. On the one hand, the strategy of shifting one’s attention away from aversive events towards other positive aspects of life may boost resilience by enhancing general positive emotions. On the other hand, based on past studies showing an association between trait positive affect and attention control37, it could also be that individuals with higher trait positive affect are more able to (re)direct their attention to maintain focus on positive aspects, even under major life stress. Regardless, converging evidence suggests that positive affectivity, which is related to greater sensitivity to rewards and better capacity of attending to positive aspects, is a core characteristic of resilience to chronic stress.
We did not find a significant moderating effect of positive reappraisal on the stress–symptom relationship, despite previous studies reporting such a link16,18. Many previous studies reporting a relationship between positive reappraisal/reframing and resilience were conducted on special populations, such as combat veterans16, victims of a natural disaster17 and newly diagnosed cancer patients18. It could be that better cognitive reappraisal/reframing ability is particularly important for mental health following certain types of life stress. The different mental health outcome measures may also account for the discrepancy. Although past studies assessed PTSD onset, psychosomatic disturbance and quality of life as outcomes, we comprehensively measured participants’ psychological symptoms. It remains to be determined by future studies whether cognitive reappraisal/reframing may be differentially linked to resilience depending on the type of life stress and nature of health outcome.
We did not find significant correlation between positive affect and acute cortisol responses, contrary to the findings of previous studies38. Several discrepancies in the positive affect measure and nature of the acute stress task may explain the finding difference. The direct association between anticipatory cortisol response and SCL-90 score also did not reach the level of significance. This result was consistent with a previous study with a similar sample size, which found no significant relationship between mood symptoms and cortisol reactivity to the TSST39.
The core results of our study are that the association between cortisol response and resilience is further dependent on trait positive affectivity. To the best of our knowledge, such an interactive effect of stress-related biological and trait affective processes on resilience has never been studied before, but is highly pertinent to understanding the integrated psychobiological systems underpinning resilience. In animals and humans, cortisol release following acute stress acts on glucocorticoid receptors in the brain, exerting a regulatory influence on cellular functions that may last beyond the timescale of the stressor40. The glucocorticoid receptors are densely located in the brain limbic circuitries involved in emotion regulation41. In addition, cortisol exerts an indirect influence on the brain reward system via modulating dopaminergic circuitries5,42. Therefore, a blunted cortisol response may cause maladaptive emotion processing and regulation functions within key neural circuitries. However, such deficiencies may be compensated by a high positive affectivity trait, possibly mediated by direct enhancement of dopaminergic functions, as well as functional interactions between the dopaminergic circuitries and limbic networks43. The reason we did not observe a significant moderating effect of positive refocusing on the cortisol–resilience relationship is unclear. It could be that affective attention has a separate and parallel biological mechanism to the cortisol system. For example, a recent study showed that pharmacological challenge to the cholinergic system altered participants’ affective attention function44.
One recent review highlighted various confounding factors that may affect psychobiological responses to the TSST, including the acclimation period before TSST, time of day, mood measure (subjective versus objective) and TSST panel sex composition45. In this study, we adhered to the most standard TSST protocol by (1) having participants physically rest for over an hour after arriving at the laboratory, (2) delivering TSST at a mostly fixed time (15:45 to 16:00) across all participants, and (3) having both male and female panel members for each participant. Although we collected no objective affective response data, the expected mood changes in response to the TSST supported the validity of the self-reported mood measure.
Several limitations need to be noted. First, the current findings may not fully generalize to individuals who developed clinical conditions following major life stress. The averaged major life stress reported by participants was relatively low, which was expected for a non-clinical, relatively young sample, and the number of events may be higher for clinical samples. Second, we did not collect data about female participants’ menstrual cycle or participants’ body mass index, which could have impacted on participants’ cortisol response profiles. Future studies may replicate our results while controlling for these variables. Third, in this study we tested the cortisol response and trait positive affectivity markers of resilience to major life stress. However, due to the cross-sectional nature of data, we could not be certain of the direction of influence, as it could also be interpreted as the psychological symptoms causing the difference in cortisol response39. Future prospective studies are necessary to test whether baseline cortisol response and trait affectivity may predict future mental health outcomes following major life stress. Fourth, our cortisol sampling timepoints may be too sparse to capture the detailed post-task cortisol change profile, although this was partly intended to reduce the effect of frequent cortisol sampling on mood state. Given our focus on anticipatory and peak cortisol responses, we also did not include cortisol sampling during the recovery phase. Future studies may replicate the current findings with more cortisol measurements, and with ‘peak’ timepoints tailored to the response profile of individual participants. Finally, our life stress questionnaire included relatively heterogeneous types of events; this was to capture participants’ major life stress experiences as completely as possible. Future studies may focus on more specific types of event (for example, acute versus chronic) to investigate stress resilience at a finer scale.
In conclusion, we found that a higher anticipatory cortisol increase to acute stress, as well as trait positive affectivity, were associated with better mental health outcome following major life stress. Importantly, the relationship between cortisol response and resilience further depended on the level of trait positive affectivity. These results have implications for the identification of individuals with high psychobiological vulnerability to major life stress, and intervention for those individuals.
Methods
Participants
This study was approved by the Institutional Review Board of the University of Hong Kong. All participants provided written informed consent. One hundred and sixty-five participants were recruited via advertisement, printed and social media, and from the FAMILY Cohort46, which is a participant registry containing ~30,000 local citizens. All participants had to be aged 24–45 years with at least a secondary level of education. Exclusion criteria included (1) current or past major physical or neurological conditions; (2) major psychological illnesses such as mood and anxiety disorders, schizophrenia and addiction; (3) any medication or other treatment received within two weeks before the study that might affect the endocrinological system; (4) (for females) pregnancy or breastfeeding. Psychological illnesses were assessed with the Structured Clinical Interview for DSM-5 Disorders, Clinician Version (SCID-CV). To rule out the potential confounding effect of intensive childhood trauma, we asked each participant to report any directly exposed or witnessed traumatic events in her/his lifetime, including those happening in childhood. Any participant who self-reported symptoms that met the diagnostic criteria for PTSD was excluded.
After stage-2 screening, 16 participants were screened out. One other participant was screened out due to abnormal cortisol change (>4 s.d. from total mean), and another was excluded due to a very high SCL-90 score of 210 (>4 s.d. from total mean), leaving 147 participants in the data analysis (aged 24–45 years, 93 females, 54 males). Two participants’ cortisol samples were used for protocol testing purposes, leaving 145 participants in the current cortisol-related analyses. This sample size is sufficient according to several previous studies investigating the association between cortisol response, positive affectivity and stress-related mental outcomes11,14,34. Post hoc power analysis based on ref. 11 revealed that our sample size achieved good power (>0.8).
Further details about the participants are included in Supplementary Section 2.
Procedure
Eligible participants were invited to join the experiment, which was always held in the afternoon between 14:00 and 18:00. Before the experiment, participants were reminded to (1) avoid food or beverage consumption within the past hour; (2) avoid intense physical activities or teeth-brushing within 2 h, (3) avoid caffeine intake or smoking on the day of the experiment; (4) avoid alcohol consumption within 24 h of the experiment.
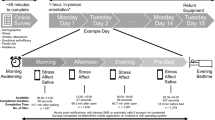
Upon arrival at the laboratory, each participant completed the screening and demographic information-collection procedures, then provided the first saliva sample (T1). They then rested for 30 min in a quiet room (room A), where they were provided with reading materials containing emotionally neutral content. This allowed the cortisol level to reach a resting baseline. Following the resting phase, the participant completed the first POMS and provided a second saliva sample (T2), then was taken to another room (room B) for the TSST. Upon completion of the TSST, the participant was taken back to room A. The third saliva sample was collected (T3) and the second POMS was completed. The participant then rested for 20 min before the fourth saliva sample was collected (T4). After that, the participant completed the Chinese Affect Scale (CAS), Brief Cognitive Emotion Regulation Questionnaire (CERQ) and Brief COPE questionnaires, as well as the Symptom Checklist-90 (SCL-90), Daily Hassle Scale (DHS) and Life Stress Index (LSI) questionnaires. Following study completion, participants were debriefed, thanked and paid HKD$600 for time and travel compensation. Further details about questionnaire administration timing are included in Fig. 5 and Supplementary Section 2.
Task and materials
The Trier Social Stress Test
The TSST is a widely used laboratory paradigm to induce acute stress to participants9,24. The TSST consists of three successive phases: (1) anticipation/preparation, (2) speech and (3) mental arithmetic, each lasting for 5 min. Before the anticipation phase, the participant was instructed to imagine s/he was attending an interview for her/his ‘ideal job’, during which they would need to deliver a 5-min free speech to demonstrate to a panel of ‘expert examiners’ that s/he was the best candidate for the job. The anticipatory/preparatory phase started immediately after the instruction, during which the participant prepared for the speech alone. In the subsequent speech phase, the participant delivered the speech to a panel of three ‘judges’ (always one male and two females, all dressed in white laboratory gowns) who remained emotionless and speechless unless the participant was unable to speak for the full 5 min. If the latter happened, the ‘chief’ judge would ask one or several prompt questions (for example, ‘What do you consider to be your main merit for this job?’). Dummy camera and audio devices were pre-installed in the room to enhance the perceived vividness of the job interview. Following the speech phase, the participant was asked to perform a mental arithmetic task involving sequentially subtracting 17 from 2,023. The participant needed to speak out the answer loudly after every calculation, and had to start over again from the beginning if they made a mistake.
Salivary cortisol
Salivary cortisol samples were collected using the Salivette Cortisol Kit (Sarstedt, cat. no. 51.1534.500). For each sample collection, the participant was instructed to chew a cotton swab for 60 s to fully soak the swab with saliva. The swab was then placed into the Salivette tube, and saliva samples were retrieved from the tube by centrifugation at 3,000g for 5 min. Quantification of cortisol concentration was performed with liquid chromatography-tandem mass spectrometry analysis (LC-MS/MS), which is considered a highly accurate and sensitive method of analysing salivary cortisol47.
Salivary cortisol was collected at four timepoints before and after the TSST. The first sample (T1) was collected shortly after the participant completed the screening and demographic information-collection procedures. Half an hour later, the second sample was collected immediately before the participant received instructions for the TSST (T2); this was considered to be the baseline level. The third sample was collected immediately after completion of the TSST (T3). The fourth sample was collected 20 min after completion of the TSST (T4) (Fig. 5). Because the salivary cortisol response to acute stress has a typical delay of around 15–20 min (ref. 48), we considered the contrast T3 minus T2 as reflecting anticipatory stress (corresponding to the instruction/preparatory phase), whereas the contrast T4 minus T2 was considered the peak stress response25. Based on recent reviews and meta-analyses23,45,48, we placed T3 and T4 at +0 and +20 min post-TSST to capture both anticipatory and peak cortisol responses. All salivary cortisol collections happened between 15:00 and 16:30, to minimize the confounding effect of diurnal cortisol variations.
Questionnaires
The LSI assesses the experience of major life events, derived from the Life Stress Assessment49 and the Life Events Checklist for DSM-5 (ref. 50) (Supplementary Section 1). Participants indicated whether they had personally experienced the event or witnessed the event happening to others, in the past five years. Because witnessing is generally considered as causing weaker stress-related mental damage than experiencing oneself51,52, we gave a score of 1 for every personal experience of a life event, and 0.5 for every witnessing of the event. For each event, the participant was also asked to provide details about the event’s frequency and duration, as well as a brief description of the event. For witnessing events, only those happening to the participant’s close friend or family member counted. All answers were independently reviewed by a panel of five judges (one clinical psychologist with more than 30 years of experience, two junior professorial-level academicians with more than 5 years of experience in stress-related research, one occupational therapist with more than 5 years of experience, and one junior research assistant in charge of collecting responses from the participant) to evaluate whether the event fulfilled the criteria for major life stress. The evaluation was based on multiple types of information about the event, such as duration, intensity, recurring frequency, relationship of involved other parties with the participant, and detailed nature of the event. Any discrepancy in rating was resolved through discussion. An event would score only if all panel members agreed on it. Further details about life stress assessment are included in Supplementary Section 2.
The DHS contains 63 items that assess daily stress and annoyance over the past month53. Because minor hassles and stress in daily life may also induce temporary psychological symptoms, we also incorporated this variable in the study54. Participants indicated the frequency of occurrence for each item. This scale had two subscales: the covert hassles subscale, containing 42 items (for example, inner concerns, internal consistency = 0.88), and the overt hassles factor, containing 21 items (for example, environmental hassles, internal consistency = 0.80) (ref. 53). The two subscales were pooled together to produce a total score.
The SCL-90 (ref. 55) has 90 items that measure psychological symptoms over the past week across ten dimensions: somatization, obsessive-compulsive, interpersonal sensitivity, depression, anxiety, hostility, phobic anxiety, paranoid ideation, psychoticism and additional. This scale performed well in detecting patients with mental disorders56 and in measuring subclinical psychological symptoms among community samples57. The Cronbach’s alpha in our sample was 0.936. A total score was calculated using the sum of all subscales to quantify mental symptoms. Further details are included in Supplementary Section 2.
The CAS assesses trait positive and negative affects58. All items are rated on a five-point Likert scale (1 = very slightly or not at all, 5 = extremely). In this study, we focused on the positive affect subscale, which contains ten items and has an internal consistency of 0.9 (ref. 58).
The CERQ assesses nine strategies of cognitive control over emotion59. All items are rated on a five-point Likert scale (1 = (almost) never, 5 = (almost) always). In this study, we focused on the positive refocusing subscale, which measures the tendency to think about positive experiences rather than the actual negative event. The internal consistency of the positive refocusing subscale is 0.86 (ref. 59).
The COPE measures 14 problem- and emotion-focused strategies when coping with adversity60. All items are rated on a four-point Likert scale (0 = I haven’t been doing this at all, 3 = I’ve been doing this a lot). In this study, we focused on the positive reframing subscale, which refers to the tendency to reappraise the negative situation more positively, through changing perspectives or digging positive aspects (that is, positive reappraisal). The positive reappraisal subscale has a reasonable internal consistency of 0.64 (ref. 60).
The POMS assesses five transient negative emotion states (tension, anger, fatigue, confusion and depression) and two positive emotion states (vigour and esteem)61. All items were rated on a five-point Likert scale (0 = not at all, 4 = extremely). The mean internal consistency among the subscales is 0.942 (ref. 62). A total mood disturbance score was computed by summing the scores for the positive and negative emotion states, and subtracting the former from the latter.
The timeframes of CAS, CERQ and Brief COPE were all set to be ‘In general’ to specifically capture the trait characteristics in those domains.
Statistical analysis
Data analyses were performed using SPSS v.26. Data normality was checked using the Kolmogorov–Smirnov test. Cortisol level changes and daily hassle score were non-normal, and a natural logarithm (ln) transformation was applied to those measures. To correct for residual deviation from non-normality and outlier effect, we conducted Spearman (rather than Pearson’s) correlation analysis, which computed ρ (rather than r) values as the correlation coefficient63.
A data analysis pipeline is included in Fig. 6. Descriptive correlation analyses were conducted among cortisol response, major life stress, SCL-90 total score, trait positive affectivity (positive affect, positive reappraisal and positive refocusing) and daily hassle score. For correlation analyses involving cortisol response, the baseline pre-TSST (T2) cortisol level was controlled for as a covariate, because baseline level may affect the amplitude of subsequent cortisol level changes. For correlation analyses involving major life stress, daily hassle score was controlled for as a covariate. The purpose of the correlation analyses was to provide an overview of relationships among key variables.
To test whether the TSST caused significant cortisol responses and mood changes, we performed linear mixed modelling analyses on cortisol level and POMS score before and after the TSST, with time as the within-subject factor (cortisol: T2, T3 and T4; mood: T2 and T3). The significant main effect of time was further evaluated using a post hoc paired-sample t-test.
To test whether a higher cortisol response reduced the relationship between major life stress and SCL-90 score, we conducted a moderating analysis using the PROCESS macro implemented in SPSS (pre-set Model No.1), utilizing a bias-corrected bootstrapping (5,000 times) approach64. The independent variable was life stress, the outcome variable was SCL-90 score, and the moderator was cortisol response (anticipatory or peak), controlling for pre-TSST (T2) cortisol level and daily hassle score.
To test whether greater positive affectivity (positive affect, positive reappraisal, positive refocusing) reduced the relationship between major life stress and SCL-90 score, we similarly conducted a moderating analysis as above, except that the moderator was positive affectivity traits.
All significant moderating effects were further evaluated using simple-effect Spearman’s full partial correlation analyses, where nuisance variables (T2 cortisol, daily hassle) and intercept were regressed out from both dependent and independent variables before computing the correlation. During follow-up analyses and graph plotting, we split the continuous moderating variables into low, medium and high groups based on 33.33% quantiles.
Finally, to test whether the moderating effect of cortisol change was also dependent on positive affectivity trait, we conducted moderated moderation analyses using PROCESS (pre-set Model No.3). The model set-up was identical to that used for testing the simple moderating effect of cortisol, except that a second moderator (that is, positive affectivity trait) was added, and we tested whether it had a second-order moderating effect on the cortisol moderating effect.
All statistical thresholds were set at P < 0.05 (two-tailed). As we analysed two types of cortisol response (anticipatory and peak) and two positive emotion regulation strategies (positive reappraisal and positive refocusing), we conducted a Holm–Bonferroni correction procedure within these categories of testing for the key moderation and moderated moderation analyses.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this Article.
Data availability
Data included in this work are provided in the Supplementary Information. Source data are provided with this paper.
References
Toussaint, L., Shields, G. S., Dorn, G. & Slavich, G. M. Effects of lifetime stress exposure on mental and physical health in young adulthood: how stress degrades and forgiveness protects health. J. Health Psychol. 21, 1004–1014 (2016).
Tennant, C. Life events, stress and depression: a review of recent findings. Aust. N. Z. J. Psychiatry 36, 173–182 (2002).
Chesnut, M. et al. Stress markers for mental states and biotypes of depression and anxiety: a scoping review and preliminary illustrative analysis. Chronic Stress https://doi.org/10.1177/24705470211000338 (2021).
Algren, M. H. et al. Associations between perceived stress, socioeconomic status, and health-risk behaviour in deprived neighbourhoods in Denmark: a cross-sectional study. BMC Public Health 18, 250 (2018).
Rutten, B. P. et al. Resilience in mental health: linking psychological and neurobiological perspectives. Acta Psychiatr. Scand. 128, 3–20 (2013).
Haglund, M. E., Nestadt, P. S., Cooper, N. S., Southwick, S. M. & Charney, D. S. Psychobiological mechanisms of resilience: relevance to prevention and treatment of stress-related psychopathology. Dev. Psychopathol. 19, 889–920 (2007).
Walker, F. R., Pfingst, K., Carnevali, L., Sgoifo, A. & Nalivaiko, E. In the search for integrative biomarker of resilience to psychological stress. Neurosci. Biobehav. Rev. 74, 310–320 (2017).
Feder, A., Nestler, E. J. & Charney, D. S. Psychobiology and molecular genetics of resilience. Nat. Rev. Neurosci. 10, 446–457 (2009).
Allen, A. P., Kennedy, P. J., Cryan, J. F., Dinan, T. G. & Clarke, G. Biological and psychological markers of stress in humans: focus on the Trier Social Stress Test. Neurosci. Biobehav. Rev. 38, 94–124 (2014).
Galatzer-Levy, I. R. et al. Cortisol response to an experimental stress paradigm prospectively predicts long-term distress and resilience trajectories in response to active police service. J. Psychiatric Res. 56, 36–42 (2014).
Danielson, C. K., Hankin, B. L. & Badanes, L. S. Youth offspring of mothers with posttraumatic stress disorder have altered stress reactivity in response to a laboratory stressor. Psychoneuroendocrinology 53, 170–178 (2015).
Yehuda, R., Flory, J. D., Southwick, S. & Charney, D. S. Developing an agenda for translational studies of resilience and vulnerability following trauma exposure. Ann. N. Y. Acad. Sci. 1071, 379–396 (2006).
Sewart, A. R. et al. Positive affect as a buffer between chronic stress and symptom severity of emotional disorders. Clin. Psychol. Sci. 7, 914–927 (2019).
Lord, J. H., Rumburg, T. M. & Jaser, S. S. Staying positive: positive affect as a predictor of resilience in adolescents with type 1 diabetes. J. Pediatr. Psychol. 40, 968–977 (2015).
Folkman, S. & Moskowitz, J. T. Coping: pitfalls and promise. Annu. Rev. Psychol. 55, 745–774 (2004).
Aldwin, C. M., Levenson, M. R. & Spiro, A. Vulnerability and resilience to combat exposure: can stress have lifelong effects? Psychol. Aging 9, 34–44 (1994).
Dollinger, S. J. The need for meaning following disaster: attributions and emotional upset. Pers. Soc. Psychol. Bull. 12, 300–310 (1986).
Manne, S. L. et al. Holding back, intimacy, and psychological and relationship outcomes among couples coping with prostate cancer. J. Family Psychol. 29, 708–719 (2015).
Garnefski, N. & Kraaij, V. Cognitive emotion regulation questionnaire—development of a short 18-item version (CERQ-short). Pers. Individ. Differ. 41, 1045–1053 (2006).
Min, J. A., Yu, J. J., Lee, C. U. & Chae, J. H. Cognitive emotion regulation strategies contributing to resilience in patients with depression and/or anxiety disorders. Compr. Psychiatry 54, 1190–1197 (2013).
Xiao, J. et al. A prospective study of cognitive emotion regulation strategies and depressive symptoms in patients with essential hypertension. Clin. Exp. Hypertens. 33, 63–68 (2011).
Kim, S. R. & Lee, S. M. Resilient college students in school-to-work transition. Int. J. Stress Manag. 25, 195–207 (2018).
Man, I. S. C. et al. Multi-systemic evaluation of biological and emotional responses to the Trier Social Stress Test: a meta-analysis and systematic review. Front. Neuroendocrinol. 68, 101050 (2022).
Kirschbaum, C., Pirke, K. M. & Hellhammer, D. H. The ‘Trier Social Stress Test’—a tool for investigating psychobiological stress responses in a laboratory setting. Neuropsychobiology 28, 76–81 (1993).
Aschbacher, K. et al. Good stress, bad stress and oxidative stress: insights from anticipatory cortisol reactivity. Psychoneuroendocrinology 38, 1698–1708 (2013).
Pulópulos Tripiana, M. M., Hidalgo Calvo, V., Puig Pérez, S. & Salvador Fernández-Montejo, O. A. Psychophysiological response to social stressors: relevance of sex and age. Psicothema 30, 171–176 (2018).
Nicolaides, N. C., Kyratzi, E., Lamprokostopoulou, A., Chrousos, G. P. & Charmandari, E. Stress, the stress system and the role of glucocorticoids. Neuroimmunomodulation 22, 6–19 (2015).
Thau, L., Gandhi, J. & Sharma, S. Physiology, Cortisol (StatPearls Publishing, 2022).
van Paridon, K. N., Timmis, M. A., Nevison, C. M. & Bristow, M. The anticipatory stress response to sport competition: a systematic review with meta-analysis of cortisol reactivity. BMJ Open Sport Exerc. Med. 3, e000261 (2017).
Rudolph, K. D., Troop-Gordon, W. & Granger, D. A. Individual differences in biological stress responses moderate the contribution of early peer victimization to subsequent depressive symptoms. Psychopharmacology 214, 209–219 (2011).
Burke, H. M., Davis, M. C., Otte, C. & Mohr, D. C. Depression and cortisol responses to psychological stress: a meta-analysis. Psychoneuroendocrinology 30, 846–856 (2005).
Avery, B. M. & Vrshek-Schallhorn, S. Nonsynonymous HTR2C polymorphism predicts cortisol response to psychosocial stress: effects in males and females. Psychoneuroendocrinology 70, 134–141 (2016).
Zorn, J. V. et al. Cortisol stress reactivity across psychiatric disorders: a systematic review and meta-analysis. Psychoneuroendocrinology 77, 25–36 (2017).
Mikolajczak, M., Roy, E., Luminet, O. & De Timary, P. Resilience and hypothalamic-pituitary-adrenal axis reactivity under acute stress in young men. Stress 11, 477–482 (2008).
Herman, J. P. et al. Central mechanisms of stress integration: hierarchical circuitry controlling hypothalamo-pituitary-adrenocortical responsiveness. Front. Neuroendocrinol. 24, 151–180 (2003).
Charney, D. S. Psychobiological mechanisms of resilience and vulnerability: implications for successful adaptation to extreme stress. Am. J. Psychiatry 161, 195–216 (2004).
Vasey, M. W., Harbaugh, C. N., Mikolich, M., Firestone, A. & Bijttebier, P. Positive affectivity and attentional control moderate the link between negative affectivity and depressed mood. Pers. Individ. Differ. 54, 802–807 (2013).
Bostock, S., Hamer, M., Wawrzyniak, A. J., Mitchell, E. S. & Steptoe, A. Positive emotional style and subjective, cardiovascular and cortisol responses to acute laboratory stress. Psychoneuroendocrinology 36, 1175–1183 (2011).
Fiksdal, A. et al. Associations between symptoms of depression and anxiety and cortisol responses to and recovery from acute stress. Psychoneuroendocrinology 102, 44–52 (2019).
Russo, S. J., Murrough, J. W., Han, M. H., Charney, D. S. & Nestler, E. J. Neurobiology of resilience. Nat. Neurosci. 15, 1475–1484 (2012).
Urry, H. L. et al. Amygdala and ventromedial prefrontal cortex are inversely coupled during regulation of negative affect and predict the diurnal pattern of cortisol secretion among older adults. J. Neurosci. 26, 4415–4425 (2006).
Pizzagalli, D. A. Depression, stress and anhedonia: toward a synthesis and integrated model. Annu. Rev. Clin. Psychol. 10, 393–423 (2014).
Chaudhury, D. et al. Rapid regulation of depression-related behaviours by control of midbrain dopamine neurons. Nature 493, 532–536 (2013).
Nabulsi, L. et al. Normalization of impaired emotion inhibition in bipolar disorder mediated by cholinergic neurotransmission in the cingulate cortex. Neuropsychopharmacology 47, 1643–1651 (2022).
Linares, N. N., Charron, V., Ouimet, A. J., Labelle, P. R. & Plamondon, H. A systematic review of the Trier Social Stress Test methodology: issues in promoting study comparison and replicable research. Neurobiol. Stress 13, 100235 (2020).
Leung, G. M. et al. Cohort profile: FAMILY cohort. Int. J. Epidemiol. 46, e1 (2017).
van den Ouweland, J. M. & Kema, I. P. The role of liquid chromatography-tandem mass spectrometry in the clinical laboratory. J. Chromatogr. B 883, 18–32 (2012).
Liu, J. J. et al. Sex differences in salivary cortisol reactivity to the Trier Social Stress Test (TSST): a meta-analysis. Psychoneuroendocrinology 82, 26–37 (2017).
Nan, H. et al. Depressive symptoms in people with chronic physical conditions: prevalence and risk factors in a Hong Kong community sample. BMC Psychiatry 12, 198 (2012).
Weathers, F. W. et al. The Life Events Checklist for DSM-5 (LEC-5) (US Department of Veterans Affairs, 2013); https://www.ptsd.va.gov/
Gray, M. J., Litz, B. T., Hsu, J. L. & Lombardo, T. W. Psychometric properties of the life events checklist. Assessment 11, 330–341 (2004).
Harbertson, J., Ziajko, L. & Watrous, J. Examining the development of PTSD symptoms in individuals who witness acute stress reaction on the battlefield. Br. J. Psychiatry Open 7, e74 (2021).
Holm, J. E. & Holroyd, K. A. The daily hassles scale (revised): does it measure stress or symptoms? Behav. Assess. 14, 465–482 (1992).
Bouteyre, E., Maurel, M. & Bernaud, J. L. Daily hassles and depressive symptoms among first year psychology students in France: the role of coping and social support. Stress Health 23, 93–99 (2007).
Zhou, J., Zhou, Y. & Qiu, J. Study of item text in the Chinese symptom checklist-90. Medicine 100, e24841 (2021).
Schmitz, N., Kruse, J., Heckrath, C., Alberti, L. & Tress, W. Diagnosing mental disorders in primary care: the General Health Questionnaire (GHQ) and the Symptom Check List (SCL-90-R) as screening instruments. Soc. Psychiatry Psychiatric Epidemiol. 34, 360–366 (1999).
Urbán, R. et al. Bifactor structural model of symptom checklists: SCL-90-R and Brief Symptom Inventory (BSI) in a non-clinical community sample. Psychiatry Res. 216, 146–154 (2014).
Hamid, P. N. & Cheng, S. T. The development and validation of an index of emotional disposition and mood state: the Chinese Affect Scale. Educ. Psychol. Meas. 56, 995–1014 (1996).
Zhu, X. et al. Psychometric properties of the cognitive emotion regulation questionnaire: Chinese version. Cogn. Emot. 22, 288–307 (2008).
Bai, S., Liu, J., Bo, Y. & Zhang, Z. Reliability and validity of the brief COPE scale among offenders in China. In Psychology and Law: Research for Practice (eds Novo, M. & Seijo, D.) 7–18 (Sciendo, 2020).
Grove, J. R. & Prapavessis, H. Preliminary evidence for the reliability and validity of an abbreviated profile of mood states. Int. J. Sport Psychol. 23, 93–109 (1992).
Cheung, S. Y. & Lam, E. T. An innovative shortened bilingual version of the Profile of Mood States (POMS-SBV). School Psychol. Int. 26, 121–128 (2005).
Pernet, C. R., Wilcox, R. R. & Rousselet, G. A. Robust correlation analyses: false positive and power validation using a new open source MATLAB toolbox. Front. Psychol. 3, 606 (2013).
Hayes, A. F. Introduction to Mediation, Moderation and Conditional Process Analysis: A Regression-Based Approach (Guilford, 2017).
Acknowledgements
This project was funded by the Hong Kong Research Grant Council Collaborative Research Fund (C7069-19GF) awarded to T.M.C.L. We acknowledge the assistance of the University of Hong Kong Li Ka Shing Faculty of Medicine Centre for PanorOmic Sciences Proteomics and Metabolomics Core.
Author information
Authors and Affiliations
Contributions
R.S. contributed to study design, data analysis and interpretation, and drafting and revision of the manuscript. I.S.C.M. contributed to data collection, data analysis and interpretation, and drafting and revision of the manuscript. C.L. contributed to data collection, analysis and manuscript revision. S.-Y.Y., W.K.H., S.X.L., F.Y.L. and Y.K.W. contributed to study conceptualization and manuscript revision. T.M.C.L. contributed to study conceptualization, study design, data interpretation and manuscript revision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Mental Health thanks Amy Sewart, Wei Zhang, Aljoscha Dreisoerner and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Section 1 (Life Stress Index), Supplement Section 2 (detailed methods), Supplement Section 3 (Supplementary Fig. 1), Supplementary Section 4 (Supplementary Fig. 2).
Supplementary Data 1
Source Data for Supplementary 4 (Supplementary Fig. 2).
Source data
Source Data Fig. 1
Statistical Source Data for plotting Fig. 1.
Source Data Fig. 2
Statistical Source Data for plotting Fig. 2.
Source Data Fig. 3
Statistical Source Data for plotting Fig. 3.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shao, R., Man, I.S.C., Yau, SY. et al. The interplay of acute cortisol response and trait affectivity in associating with stress resilience. Nat. Mental Health 1, 114–123 (2023). https://doi.org/10.1038/s44220-023-00016-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44220-023-00016-0