Abstract
Suffering due to mental health problems is rising inexorably in all regions of the world. One major reason is that our understanding of the causation, prevention and treatment of mental health problems has been hindered by an over-reliance on diagnostic categories. Yet there is growing evidence for alternative approaches from across multiple disciplines, including neuroscience, which elucidates neural regions and networks underlying specific mental health experiences; cognitive science, which identifies cognitive functions and impairments relevant to mental health; clinical science, which identifies symptom patterns associated with cognitive dysfunctions; developmental science, which identifies environmental influences on brain development in early life; social science, which identifies sociocultural influences on mental health; intervention science, which identifies the ‘active ingredients’ of psychological interventions; and implementation science, which designs scalable interventions to effectively deliver these active ingredients. We propose an integrative model that converges these diverse disciplinary perspectives, from neural circuits to interventions, that can be delivered at scale, with a potential for higher coverage, greater personalization and greater efficacy than traditional diagnostic approaches. This integrative approach can lead to a radical opportunity to shift the needle on mental health-related suffering globally.
Similar content being viewed by others
Main
Mental health problems are among the leading causes of global disease ‘burden’ (the term ‘burden’ is based on a binary model of mental illness that is explicitly rejected herein, but which is the basis of the majority of clinical research over the past 50 years), and are associated with premature mortality (particularly in young people). Their prevalence and impact are rising in all regions of the world, leading to massive personal, family and societal costs1. Despite tens of billions of dollars of research funding over the past four decades, we have only a shallow understanding of these conditions, with no signature pathology or biomarkers for any condition, and only marginal progress regarding prevention or therapeutics. One of the major reasons—perhaps the single most important one—for this state of affairs is the adoption of diagnostic categories, exemplified by the two major classification systems in use (the International Classification of Diseases (ICD)2 and the Diagnostic and Statistical Manual of Mental Disorders (DSM)3). Diagnostic categorization, which is eminently applicable to infectious diseases, where there is a clear distinction between individuals infected with a specific pathogen and those who are not, is ill-suited for mental health problems, for which there is rarely a comparable boundary between ‘normality’ and ‘illness’. Consequently, diagnostic categories are plagued with considerable overlap and ill-defined boundaries4.
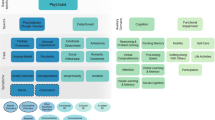
A number of scientific endeavours have sought to address mental health problems without relying on diagnostic categories. For example, the National Institute of Mental Health Research Domain Criteria initiative (RDoC) embraces dimensional approaches for identifying biological and cognitive processes that explain mental health and illness5,6. The RDoC focuses on six major domains of functioning (for example, positive valence systems), and constructs and subconstructs (for example, reward responsiveness and reward anticipation) within each domain, each of which is measurable across an array of ‘units of analysis’, including genes, molecules and cells, neural circuits, physiology, behavior and self-report. The dimensionality of constructs (from ‘normal’ to ‘abnormal’) is a fundamental principle, consistent with robust evidence for dimensional models of mental health that have emerged from analyses of symptom structure independent of neuroscience, such as the Hierarchical Taxonomy of Psychopathology (HiTOP) model4,5,7. Inspired by these dimensional models, we propose a dynamic convergence across disciplines to bridge neural circuits, cognitive function, symptom profiles, developmental phases, sociocultural influences and targeted and personalized interventions that can be fashioned for delivery through scalable platforms such as community health workers or digital tools. Our ‘circuits-to-communities’ framework not only aligns with the RDoC emphasis on neuroscience and dimensional models of psychopathology, but extends beyond the RDoC by linking dimensional models to clinical interventions that can be translated to population-level impact through innovative global mental health practices.
In the forthcoming sections, we review advances in each discipline (neurocognitive science, clinical science, developmental-social science, intervention science and implementation science), providing the building blocks for our integrative model. We provide examples relevant to (1) symptoms of fear/anxiety and depression, the most common mental health experiences, although the model is applicable to many other forms of psychopathology (for example, obsessive–compulsive, trauma-related, somatic symptoms, eating and substance use), (2) active ingredients of interventions and their personalization and (3) their adaptation for delivery in communities on a global scale. We conclude with future research directions and pathways to full adoption of a circuits-to-communities framework.
Neural and cognitive systems relevant to mental health
The acceleration of neurocognitive science over the past few decades has led to major advances in identifying brain regions implicated in mental health problems, including the most common conditions of depression and anxiety8,9. Early focus on abnormalities in specific areas of the brain has expanded to abnormalities in the structural and functional connections between neural regions10,11. Elucidating the neural systems and cognitive functions relevant to mental health may inform more precisely targeted and more efficient treatments—treatments that address the neural and cognitive functions that are most relevant for an individual’s particular symptom profile.
The cognitive functions relevant to mental health (for example, threat detection, reward learning, emotion regulation) have been mostly investigated using paradigms that measure a single principal construct with well-demarcated neural circuits, usually derived from validated animal behavioral paradigms. Examples of paradigms relevant to anxiety and depression include Pavlovian fear learning and extinction, predictable and unpredictable threat tests, face matching, reward anticipation and attainment, and reinforcement learning. These paradigms probe circuits such as the cortico-amygdala circuit, which supports vigilance for, and responses to, threatening stimuli12,13,14. The amygdala is engaged by a range of threat-relevant cues and defensive behaviors8,15, whereas the ventromedial prefrontal cortex is involved in fear extinction and extinction recall16,17,18. The cortico-striatal circuit is involved in processing the hedonic value of stimuli (reward processing)19, reinforcement learning20 and motivation21,22. This circuit involves projections from midbrain neuromodulator nuclei (that is, dopaminergic, serotoninergic and noradrenergic) to subcortical areas within the basal ganglia (for example, ventral striatum) and cortical target regions (for example, orbitofrontal cortex).
Neural and cognitive features have been associated with mental health across hundreds of studies. These include changes in neural and cognitive indices of attention and threat detection in relation to anxiety23, and reward responsivity and reward learning in relation to depression9,24,25. In terms of connectivity across neural regions, reduced structural connectivity within the uncinate fasciculus tract that connects the ventromedial prefrontal cortex and amygdala26 has been associated with poor regulation of negative emotions and elevated depression27,28 and anxiety29,30. Functional connectivity between the orbitofrontal cortex and ventral striatum has been associated with depression, and, in particular, anhedonia9,31. In some cases, neural patterns have been shown to not only correlate but to predict the subsequent development of clinically important symptoms of anxiety32 and depression33, suggesting a contributory role to the development of mental health problems. (Conversely, as we describe in more detail later, mental health symptoms probably influence neural processes and cognitive functions.) Box 1 provides exemplars of circuit–function–symptom relations for fear learning and reward processing.
Treatments that specifically target circuit-cognitive changes already exist. Examples include exposure therapies (that is, systematic, repeated facing of feared objects or situations) that specifically target extinction-related mechanisms34, cognitive training programs that specifically target attentional bias for anxiety35, imagery training for depression36, response inhibition training for intrusive images37, and behavioral treatments that specifically target reward hyposensitivity38,39. Thus, there is already enough evidence to scale up cognitively targeted interventions for community implementation.
In terms of neurocognitive advances, neural processes are now recognized as larger in scale than serial connections or regions of interest11,40,41. The emerging field of network neuroscience provides a conceptual framework and statistical toolkit for this scale and complexity42. In the same vein, cognitive functions themselves (for example, reward, semantic and emotional) are better understood within network models versus as independent entities, and clustering of these functions may provide better correspondence with neural functioning43,44,45. Neural and cognitive network models are likely to elucidate brain involvement in cognitive functions beyond what is described in Box 1, and yield even more targeted and more efficient treatments.
In terms of clinical advances, the majority of studies compare neurocognitive circuits and functions across diagnostically classified groups and healthy controls. However, the true value of neuroscience lies in elucidating cognitive functions associated with symptom dimensions or profiles rather than diagnostic categories. Dimensional models offer greater precision in treatment targets and greater treatment personalization. As an example, symptoms of anhedonia combined with psychomotor retardation may benefit more from behavioral interventions that differ from interventions most suited to symptoms of sadness and agitation, even though all are criterion symptoms for the diagnosis of major depression.
Developmental science and sociocultural influences
Both animal and human research indicate that the brain is highly sensitive to stress and adversity during the early months to years of life. This adversity sensitizes cells in brain regions that respond to threatening stimuli, including the amygdala, the anterior cingulate cortex and the insula46. For example, individuals who grew up in adversity or who have a history of childhood maltreatment display structural alterations in the amygdala that sustain into adulthood and show heightened amygdala reactivity to threatening stimuli47,48,49. Early life adversity also affects the development of the cortico-striatal reward circuit and is associated with sustained deficits in reward processing50,51,52,53. Preliminary research suggests that even stress during the prenatal period, as measured by placental gene expression and epigenetic marks (that is, DNA methylation), can generate risk for mental and physical health problems across the lifespan52,53. Collectively, this suggests that social determinants, including poverty, discriminatory experiences and marginalization, generate risk for mental health problems in part through affecting neural and genomic signalling early in life54.
Despite the effect of early life adversity on the developing brain, many forms of mental health problems, including depression and some variants of anxiety, do not emerge until adolescence55. This suggests that stress and adversity incubate in the developing brain until a developmental inflection point associated with the onset of a particular mental health problem is reached. Multiple biological and psychosocial factors converge during adolescence, making it ‘an age of risk’ for the onset of symptoms. Adolescence is characterized by heightened neuronal sensitivity during which the amygdala, the ventral striatum and the prefrontal cortex go through normative growth spurts56,57,58. Longitudinal research indicates both the volume and activity of the amygdala and ventral striatum are heightened in adolescents relative to both children and adults59,60. The prefrontal cortex, in turn, has a delayed and prolonged development, and the regulatory processes it supports improve in a linear manner through early adulthood61. Adolescence may thus be a period during which the brain’s threat and reward systems are particularly sensitive to stress, and stress exposure during adolescence may compound the effects of early adversity on the brain. Neural circuits involving the amygdala, the ventral striatum and the prefrontal cortex are also very responsive to social stressors during adolescence50,56,62,63, which dramatically increase during this period as the individual establishes their own identity, completes education, seeks a livelihood and forms intimate relationships62,64.
Collectively, the evidence suggests that (1) childhood is an important period for prevention strategies to lower the risk for symptoms, and for the identification of biomarkers to identify at-risk individuals, (2) adolescence is a critical point for intervening, and (3) interventions should be attuned to sociocultural influences. In our model, we consider some of these components by examining circuit–function–symptom relations in the context of developmental and sociocultural factors to generate active ingredients of interventions. For example, early childhood exposure to trauma, living in a warzone or the sequelae of the COVID-19 pandemic could translate to heightened responses to threat stimuli in related neural circuits (amygdala, ventral striatum, prefrontal cortex) that could be targeted through emotion regulation interventions, such as problem solving. Problem-solving skills may be especially suited to adolescents given their unique neurodevelopmental transitions. Our circuits-to-communities model integrates developmental and social influences with neural and cognitive circuits, which together inform interventions.
Clinical science: symptom dimensions
Diagnostic classifications (ICD and DSM) are beleaguered by high levels of cross-diagnostic comorbidity and within-diagnosis heterogeneity, which together impede personalized mental health care. Dimensional hierarchical models, such as the HiTop model4,5 or the depression and anxiety Tri-level model65,66, have been gaining support as alternatives to categorical diagnoses. These factor analytic models evaluate patterns of covariation among symptoms across different levels of generality and specificity. They also identify symptom clusters that are common to multiple groupings or conditions and disentangle the different sources of variance within symptom clusters. For example, the Tri-level model portions variance attributable to broad general distress/negative affect (common to depression and anxiety) from variance attributable to fears (more specific to anxiety) and to anhedonia-apprehension (more specific to depression)65,66. Such portioning has already proven useful when mapping symptoms to cognitive functions and neural underpinnings67,68.
Moreover, just as with networks of brain regions and networks of cognitive functions, symptoms per se are increasingly understood as networks of interactions, with one symptom contributing to another symptom (for example, inactivity leads to apathy, which leads to hopelessness)69. From the symptom network perspective, mental disorders emerge when groups of tightly coupled symptoms actively maintain each other, leading to clusters of symptoms that become self-sustaining. Inherent to this model is that symptoms may persist in a sustained activating cycle even after the initial cause has dissipated.
At first glance, the symptom network approach may appear at odds with a circuit–function–symptom model, which could seem to imply directionality from an underlying biological cause to a symptom outcome. Yet with the emergence of network approaches at the circuit and function levels, the circuit–function–symptom model can easily accommodate multi-directional influences, wherein symptom expression itself can evoke changes in underlying neural systems and cognitive functions, a topic we explore in more detail in the following. Hence, even though the initial underlying causes of a symptom network may have dissipated, and even though symptoms are driven at least in part by other symptoms, our model highlights the importance of linking symptom networks to circuit–function networks that can be targeted in treatment, with treatment itself having reciprocally positive effects upon symptoms as well as circuit–functions (that is, symptom reduction produces neural changes, and neural changes produce symptom reduction).
Intervening anywhere in the symptom network could have ripple effects to other symptoms in the network. Furthermore, a symptom network model can identify symptoms that are most influential on the spread of symptoms (that is, centrality), which in turn may highlight the underlying network of circuits–functions that is most essential as a treatment target. This logic parallels recent developments in network neuroscience that identify drivers or hubs in the brain that when modified have a particularly strong effect on other brain systems and symptoms11,40.
Notably, symptom dimension approaches provide greater precision for understanding sociocultural influences on mental health than do diagnostic categorizations70. For example, a systematic review of 138 studies of the experience of depression across many world regions observed that several features of depression that were reported with high frequency were not included in diagnostic criteria, whereas others which were in the diagnostic criteria were rarely reported71. These results suggest that symptom checklists may more fully capture depression in all of its geographic heterogeneity than do diagnostic criteria.
Intervention science: targeted and personalized active ingredients
A circuit–function–symptom model is consistent with a targeted and personalized active ingredients approach to mental health interventions. Active ingredients are the elements within a treatment shown to be responsible for therapeutic change; targeted active ingredients are designed to treat underlying cognitive or behavioral patterns (either directly or through compensatory mechanisms)72, and personalized active ingredients are the ones that are selected for an individual from an available set of active ingredients, based on prediction of the strongest response. A targeted and personalized active ingredients approach differs from standard therapeutics, often composed of multiple elements in a ‘one-size-fits-all’ approach for diagnosed disorders, and differs from a ‘common elements’ approach, wherein contents of ‘winning’ evidence-supported treatment manuals are ‘distilled’ or separated into distinct techniques but are not mechanistically determined73. Targeted active ingredients are targeted at specific cognitive or behavioral features that have been elucidated through neuroscience and cognitive science, are relevant to mental health and are developmentally informed. Examples of targeted active ingredients of psychological therapies include exposure therapy to target avoidance and fear extinction74, concreteness training to target overgeneralization of depressive cognition75, attentional bias training and interpretation bias training to target attention and interpretation biases76, response inhibition training to target intrusive memories37 and imagery training to counteract depressive future thinking36. In Box 2 we provide more detailed examples of extinction-based active ingredients for fear symptoms and reward processing-based active ingredients for anhedonia symptoms.
Each active ingredient involves a purported mechanism, targeted by a specific strategy that is derived from cognitive neuroscience. No one particular mechanism, be it cognitive (for example, distorted thinking style), behavioral (for example, experiential avoidance) or biological (for example, neural patterns), takes precedence as an explanatory factor for psychopathology. Rather, the approach is driven by evidence for any or all of a broad array of possible mechanisms. By being so targeted, these intervention ingredients are efficient relative to standard therapies. Even more targeted active ingredients may evolve alongside advances in neuroscience and cognitive science, and advances in network neuroscience may identify control hubs in the brain that can be targeted to drive symptom change40.
Although most evidence-based therapies comprise active ingredients, they are usually combined into one package, which prohibits ascertainment of which ingredient is responsible for change and prohibits personalization of care (or selection of a particular ingredient for a given individual based on expected response). (Note that, herein, we are emphasizing personalized mental health care, consistent with the notion of precision medicine, and different from the concept of person-centred care, which applies to all individuals equally.) Moreover, the few studies of individual active ingredients utilize diagnostically classified samples, rather than circuit–function classified individuals, which may have limited their effectiveness. As an example, the active ingredient of attention bias modification training produces relatively small effect sizes for individuals with anxiety disorders77, perhaps because only some individuals with anxiety disorders exhibit an attentional bias to threat78,79. Symptoms of general anxiety may link more strongly with attentional bias towards threat, whereas phobic symptoms may link more strongly with avoidance of threat80. Thus, the effect size of attentional bias modification is probably dampened when applied to the entire diagnostic group, and amplified when applied to anxious individuals with attentional bias towards threat. As another example, some individuals with post-traumatic stress disorder exhibit amygdala hyperactivation and prefrontal cortex hypoactivation in response to trauma reminders, whereas others show the reverse pattern, and these neural differences have been linked to symptom profiles of emotional reactivity versus numbing and avoidance81. Consequently, active ingredients that target amygdala hyperactivation and prefrontal cortex hypoactivation may be more effective for those exhibiting emotional reactivity, whereas other active ingredients may benefit those with numbing and avoidance. A circuit–function–symptom active ingredient approach facilitates a more efficacious personalized approach to mental health care.
Implementation science: task-sharing of brief psychosocial interventions
A large body of evidence has demonstrated the effectiveness of psychosocial interventions, typically complex multi-ingredient treatment packages designed to treat diagnostic categories of mental health problems. These interventions are recommended as the first-line treatment for a wide range of mental health problems, in particular during childhood and adolescence (such as autism, emotional and behavioral disorders), and for anxiety- and stress-related disorders across the life course. They are the best initial treatment choice for mood disorders (for which alternative pharmacological interventions are also widely used)82,83 and enhance the effectiveness of pharmacological interventions for psychoses. The evidence, however, is not without limitations, such as the absence of long-term outcomes from the majority of implementation studies, although recent findings are emerging to suggest general persistence of benefits over lengthy time periods84,85. Adverse outcomes typically are not reported, although it remains unclear as to whether this is because they were absent or they were not assessed during or beyond the duration of the study. Furthermore, there have been few effectiveness trials in public health settings with regular health workers performing multiple functions.
Moreover, there are a number of notable limitations of the multi-ingredient packages: their one-size-fits-all approach, akin to pharmacotherapy, which treats a disorder rather than an individual and, in doing so, perpetuates the categorical diagnosis-driven approach to mental health problems; their complexity, which requires a large number of sessions to deliver to protocol and a considerable number of skills to be mastered by the patient; and the proliferation of hundreds of packages of psychosocial interventions, which differ marginally from one another, sharing a number of ‘active ingredients’, leading to a fragmentation of therapeutic approaches for mental health problems. The arduous training and supervision requirements to deliver these interventions with fidelity have effectively restricted their delivery to highly trained, and expensive, mental health professionals, who are in short supply globally. These challenges have greatly hampered the scale-up of these therapies.
These challenges have been addressed head-on by implementation scientists working in low-resource contexts globally, including in the most resourced countries in the world, such as the United States. In doing so, these scientists have designed briefer interventions that target hypothesized mediators of mental health problems (for example, interventions comprising only the active ingredient of behavioral activation to target low-response contingent positive reinforcement, hypothesized to contribute to the onset and maintenance of depression) and are thus easier to learn and implement with fidelity (and therefore be delivered by non-specialized providers, enhancing accessibility and reducing cost) and easier for patients to understand and master (therefore enhancing acceptability). These pared-down interventions represent the first stage of a circuit–function–symptom–communities approach, in which continuous research advances lead to even more targeted and personalized interventions that are scalable. Importantly, treatment need has been typically identified on the basis of symptom presentation (assessed using brief symptom questionnaires), eschewing the requirement for a diagnosis (which needs a more intensive assessment by a highly trained provider)86. The research agenda of the circuits-to-communities model will go beyond existing pared-down programs and eventuate in active ingredients that target specific cognitive functions and are selected to suit an individual’s symptom profile in combination with their developmental stage and relevant sociocultural influences (thus both targeted and personalized).
Over the past two decades, the effectiveness of these pared-down psychosocial interventions delivered through task-sharing with a wide range of non-specialist providers (for example, community health workers, nurses and lay counsellors) in routine, real-world, settings (for example, in primary care, community settings and schools) has been repeatedly demonstrated in randomized controlled trials (more than 100 so far). Furthermore, a number of recent systematic reviews have concluded that this approach, combining a parsimonious, active-ingredient intervention with a pragmatic delivery model, is the most cost-effective strategy to shift the needle on the crisis of unmet needs for quality care for mental health problems87,88,89. Delivery of active ingredients can be greatly facilitated by digital technologies, as described in more detail in the following.
The parallels between the use of such ‘simpler’ targeted interventions designed around a single, or a few, active ingredients and the mechanistically informed active ingredients described as a key approach of intervention science are obvious. That notwithstanding, there remains a gap between existing implementations of pared-down interventions and the rapid advancements in neurocognitive science and clinical science that are leading to novel active ingredients and, importantly, ways to match active ingredients to each individual.
An integrative multidisciplinary framework for global mental health
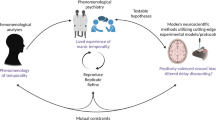
Our circuits-to-communities framework is not a theory of change per se, nor is it a model of system changes needed to implement new approaches to mental health care. Rather, our model is a call for a new approach to treatments that are targeted, personalized and scalable. We link and converge the highly specialized and segregated disciplines of neuroscience, cognitive science, developmental science, clinical science, intervention science and implementation science to inform the design and delivery of targeted and personalized active ingredients of treatment for mental health problems. By examining networks and circuit–function–symptom chains in individuals that take sociocultural and developmental influences into account, we move beyond average-based inferences and one-size-fit-all treatment modalities. We argue that the emerging selection of specific active ingredients can be delivered through low-cost strategies, such as task-sharing or digital therapies. Our model is not based on any of the individual disciplines in discussion, but rather on their integration to produce personalized therapeutic active ingredients that can be applied at scale, using data-driven algorithms.
Alongside interactions within networks of neural processes, cognitive functions and symptoms, our integrative circuits-to-symptoms framework emphasizes dynamic relationships across neural circuits, the cognitive functions supported by those neural circuits, and symptom expression. To illustrate the reciprocity, as much as neural systems underpin cognitive and behavioral responding (for example, elevated activation of the amygdala is related to pronounced detection and avoidance of threat)90,91, behavioral responding in turn can exert neural effects. In animal models, for example, neural changes emerge as a function of behavioral ‘learned helplessness training’ on the one hand (for example, excessive serotonin is released in projection regions of the dorsal raphe nucleus) and behavioral ‘control training’ on the other hand (for example, the medial prefrontal cortex inhibits the serotonergic dorsal raphe nucleus)92. There are multiple corresponding examples for behavioral training in humans to influence neural responses (for example, attention bias modification training influences neural correlates of response monitoring93, exposure therapy influences neural response to conditional stimuli94, and behavioral reward training influences physiological response to reward). Conversely, symptom reduction from psychological therapies (albeit multi-ingredient) is associated with changes in neural circuits for anxiety95,96, depression and anhedonia97,98. Hence, in our integrative framework, neurocognitive circuits and functions influence symptoms, and targeted interventions, including behavioral ones, influence both symptomatic and neurocognitive effects.
A core assumption of the circuit-to-communities model is that targeted active ingredients will be more efficient and more effective when personalized. This is because, within a package of therapeutic elements, some elements are likely to be ineffective for a given individual, increasing the risk of iatrogenic effects, inefficiency and treatment dropout. Personalization methodologies already have been demonstrated to improve treatment response, such as the Personalized Advantage Index for choosing between psychotherapy packages99. Evidence is beginning to emerge in support of prescriptive matching to specific treatment ingredients from symptom network models100.
Links to symptom profiles will be the key pin for large-scale implementation, wherein symptom profiles themselves will become the proxy for underlying neural and cognitive dysfunctions (whether related to distal mechanisms, compensatory mechanisms or symptoms themselves). In alignment with our circuit–function–symptom model, active ingredients would be selected on the basis of the central symptom cluster within a symptom network model, because central clusters have most influence on the spread of symptoms and thereby represent the underlying network of circuits–functions that is most essential as a treatment target. This will require the generation of circuit–function–symptom networks, as we have described, calibrated or adjusted to account for sociocultural and developmental influences upon symptom expression (Box 3).
Future research directions
Greater precision in neural systems: neural network models and precision imaging
There are two developments in structural and functional brain imaging that have implications for understanding circuit–function–symptom pathways and the development of personalized interventions. The first pertains to the emerging field of network neuroscience42. Historically, structural and functional neuroimaging studies have examined brain regions in isolation of each other or examined the associations between just a handful of brain regions. Network neuroscience builds on a branch of mathematics called graph theory to model the connections between hundreds to thousands of regions of interests across the brain101,102. Variations in these network metrics have been associated with individual differences in emotion, cognition and attention103,104, as well as mental health, including depression105,106. Newer variants of network neuroscience (for example, network control theory) examine how the activation of one node in a network affects the rest of that network107. This work has the potential to generate new insights into neural architecture and identify prominent drivers or hubs in the brain that can be targeted by person-centred interventions.
Another important development is precision imaging108. The majority of research on mental health focuses on group-based statistics, examining how diverse groups differ on some outcome variable, or how one treatment compares to another. Group comparisons, however, do not capture the heterogeneity of biological and psychological characteristics across any given disorder109,110. Furthermore, the personalized interventions proposed in this Perspective are about understanding and affecting the lives of individuals, not groups. Thus, ultimately, we will need to model the individual at both biological and psychological levels of analysis. Precision functional magnetic resonance imaging (fMRI) approaches, for example, use extended data acquisition and forward-thinking analyses of the functional connections in the brain to provide reliable and stable individual measures of brain organization111,112. Early reports indicate that precision fMRI is more sensitive to individual differences and clinical symptoms than standard group-based analyses, and can increase the association between fMRI measures and behavior113,114,115. Future research is needed to examine whether precision imaging (1) generates more individualized prognostic markers of risk, (2) facilitates the development of personalized interventions and (3) provide personalized targets in the brain for behavioral therapies.
Greater precision in symptom profiling and integration with neural networks
In parallel to the greater precision in understanding neural and cognitive systems, there is a need for greater precision in symptom profiling. Although dimensional models (for example, HiTOP, tri-level model) have many advantages over diagnostic categories, they are limited to traditional symptom questionnaire measures that represent explicit retrospective judgements of oneself that, although relevant in and of themselves, are biased (for example, depressive recall of negative over positive self-referent information)116. Ecological momentary assessments of subjective experience capture affective dynamics in daily life that are less biased than traditional questionnaires that estimate status over lengthy intervals, but remain limited to self reporting117.
A challenge for the field is achieving greater precision in individualized symptom assessment that is not fully reliant on language, self-awareness and retrospection, although in nascent stages, digital phenotyping of behaviors and peripheral physiology through smartphones and wearable sensors can record features such as movement, sleep, social interaction, heart rate variability, vocal and facial features118 that continuously snapshot mental health-relevant features. Traditional questionnaire symptom ratings combined with moment-to-moment dynamic trajectories in self-report plus continuous physiological and behavioral expressions may lead to more precise measurement needed for precision mapping onto neurobiology. The digital divide will remain an obstacle, however, for global implementation of these high-frequency naturalistic measurements.
Individually targeted care embedded in social interventions
The emphasis throughout has been on translating research on circuit–function–symptom pathways into personalized interventions that can be distributed on a global scale. Therefore, our focus has been on scaling upwards from circuits to communities, with feedback regarding their effectiveness. Yet, as discussed, the links between circuits and symptoms are influenced by developmental and sociocultural factors, including maltreatment and neglect47,49. This work has inspired family-centred interventions designed to increase parental warmth and responsiveness that have a positive effect on brain development and help protect youth from the onset of stress-related mental and physical health problems119. There is growing evidence, however, that the society or larger community in which a person develops also affects the brain and body120. Early exposure to community stressors such as poverty or neighbourhood violence alter brain systems involved in a variety of cognitive and affective processes121,122. Theory and research suggest that these alterations are one possible mechanism through which social determinants of health such as poverty and structural inequality get under the skin to heighten risk for mental health problems, as well as various physical health conditions123. This work also suggests that social policies designed to address structural inequities in society may help reduce health inequities in part through having a positive effect on brain development124. In line with this Perspective, Troller-Renfree et al. reported that a modest monthly cash transfer to low-income families had a causal impact on infant brain activity associated with cognitive and emotional development125. A full integration of social determinants of health and circuit–function–symptom pathways is beyond the scope of this Perspective. However, an important future direction will be to integrate research examining the mechanisms through which social inequities like poverty affect the brain and body with the work of social scientists and policy makers investigating the structural foundation of inequality and economic determinants of health disparities126. This could help synergize the development of targeted and personalized interventions with societal policies and social programs to optimize mental and physical health and well-being.
Pathways to global implementation
Large-scale implementation of the circuit-to-communities model will require digital tools that automate selection of the most fitting active ingredients for a given individual (which are based on the research to date linking circuits to functions and symptoms) and include data-driven feedback systems for updating the selection algorithms and the active ingredients themselves based on the response of the individual. In this way, the research agenda continuously informs global implementation, which in turn continuously feeds back to inform the research.
As an example, the community or frontline health worker might have an app in which a person enters symptom ratings, preferably over repeated occasions to permit symptom network modelling and identification of central drivers of symptom extension, as well as their developmental stage, early adversity and ongoing stress exposure. These person-specific symptom and contextual data would be fed into the circuit–function–symptom data corpus, from which an automated algorithmic output indicates the active ingredient(s) most likely to be effective for the individual. The selected active ingredient(s) would be implemented by the community health worker, with repeated symptom measurement to index treatment response. In this continuous cycle of measurement-based care, efficacy of a targeted active ingredient will be assessed along with broad reassessment of symptom nodes to determine adjuvant or subsequent active ingredients to implement. Ongoing circuit–function–symptom research, taking sociocultural and developmental factors into account, will inform refinements to the active ingredients and the selection algorithms. Ongoing data collection regarding the effectiveness of the personalized active ingredients will feed back to inform refinements to the active ingredients and the selection algorithms, ensuring calibration across different sample characteristics and settings. In this way, sociocultural and developmental influences are incorporated into the data corpus that guides intervention selection as well as into ongoing refinement of selection tools and of interventions themselves to suit different developmental phases and cultural factors.
Aside from guiding the selection and evaluating the efficacy of active ingredients, digital technologies offer two more pathways for global implementation. The first is training and fidelity assurance of health-care workers. Digital platforms can be used for competency-based training and quality assurance in the delivery of specific active ingredients, as is already being rolled out in rural India127. The second is actual delivery of the active ingredients. Although health workers will be better able to deliver active ingredients than complex psychological treatments86, digital technologies will greatly facilitate the delivery of selected targeted active ingredient therapies with fidelity, especially given the scope of an ever-growing set of active ingredients. The diverse opportunities offered by digital technologies targeting the person with or without the aid of non-specialist providers dramatically opens up access to care, with evidence of their efficacy in diverse contexts already accruing128.
Summary and synthesis
The integrative framework we propose offers a pathway for circuit–function–symptom dynamic relationships to inform targeted and efficient psychological treatments that can be scaled up in diverse global contexts. Our framework utilizes the logic of network theory within and between circuits, functions and symptoms to identify central, yet modifiable, targets for interventions in the context of developmental and sociocultural factors. Inspired by the RDoC taxonomy, the framework traverses different disciplines by evaluating circuit–function–symptom changes rather than diagnoses, while implementation science opens a window for scaling up personalized interventions using task-sharing and digital technologies. Our integrative framework builds on and complements the RDoC taxonomy in a number of ways. First, it moves beyond serial linkage between units of analysis (circuits), domains (function) and constructs (symptoms) to highlight multidimensional and dynamic relationships within and across neural networks, cognitive functions and symptom networks. Second, it completes the cycle with personalized and targeted clinical interventions and their scalable implementation at the population-level impact through innovative global mental health practices. Third, our framework emphasizes the importance of placing circuit–function–symptom profiles within a developmental, environmental and sociocultural context. Transformations in research approaches, paradigms and multidisciplinary collaborations are imperative for the successful implementation of our framework. It is the alignment across disciplines that can lead to sizable advancement in our understanding of the causation, prevention and treatment of mental health problems.
References
Patel, V. et al. The Lancet Commission on global mental health and sustainable development. Lancet 392, 1553–1598 (2018).
International Statistical Classification of Diseases and Related Health Problems 11th edn (World Health Organization, 2019).
Diagnostic and Statistical Manual of Mental Disorders: DSM-5 (American Psychiatric Association, 2013).
Krueger, R. F. et al. Progress in achieving quantitative classification of psychopathology. World Psychiatry 17, 282–293 (2018).
Kotov, R. et al. The hierarchical taxonomy of psychopathology (HiTOP): a quantitative nosology based on consensus of evidence. Annu. Rev. Clin. Psychol. 17, 83–108 (2021).
Sanislow, C. A., Morris, S. E., Pacheco, J. & Cuthbert, B. N. in The New Oxford Textbook of Psychiatry 3rd edn. (eds. Geddes, J. R. et al.) Ch. 8 (Oxford Univ. Press, 2020). This is a description of the National Institutes of Mental Health Research Domain Criteria Initiative, which promotes a dimensional model for understanding psychopathology, integrating genes, molecules and cells, neural circuits, physiology, behavior and self-reporting.
Michelini, G., Palumbo, I. M., DeYoung, C. G., Latzman, R. D. & Kotov, R. Linking RDoC and HiTOP: a new interface for advancing psychiatric nosology and neuroscience. Clin. Psychol. Rev. 86, 102025 (2021).
Fox, A. S. & Shackman, A. J. The central extended amygdala in fear and anxiety: closing the gap between mechanistic and neuroimaging research. Neurosci. Lett. 693, 58–67 (2019).
Nusslock, R. & Alloy, L. B. Reward processing and mood-related symptoms: an RDoC and translational neuroscience perspective. J. Affect. Disord. 216, 3–16 (2017).
Menon, V. Large-scale brain networks and psychopathology: a unifying triple network model. Trends Cogn. Sci. 15, 483–506 (2011).
Bassett, D. S., Xia, C. H. & Satterthwaite, T. D. Understanding the emergence of neuropsychiatric disorders with network neuroscience. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 3, 742–753 (2018). Circuit (network)–disorder: this article links network neuroscience in the context of functional connectivity with the expression of symptoms of psychopathology.
Janak, P. H. & Tye, K. M. From circuits to behaviour in the amygdala. Nature 517, 284–292 (2015).
Paré, D. & Quirk, G. J. When scientific paradigms lead to tunnel vision: lessons from the study of fear. NPJ Sci. Learn. 2, 6 (2017). This is an example of traditional connectivity analyses that examine the association between psychiatric symptoms and a neural circuit between two brain regions.
Shackman, A. J. & Fox, A. S. Contributions of the central extended amygdala to fear and anxiety. J. Neurosci. 36, 8050–8063 (2016).
Headley, D. B., Kanta, V., Kyriazi, P. & Paré, D. Embracing complexity in defensive networks. Neuron 103, 189–201 (2019). (Complex) circuit–function: this concoction of empirical findings challenges prevailing views of ‘neurons of interest’ and ‘region of interest’ to establish the circuit–function link.
Bukalo, O. et al. Prefrontal inputs to the amygdala instruct fear extinction memory formation. Sci. Adv. https://doi.org/10.1126/sciadv.1500251 (2015).
Delgado, M. R., Nearing, K. I., Ledoux, J. E. & Phelps, E. A. Neural circuitry underlying the regulation of conditioned fear and its relation to extinction. Neuron 59, 829–838 (2008).
Milad, M. R. et al. Recall of fear extinction in humans activates the ventromedial prefrontal cortex and hippocampus in concert. Biol. Psychiatry 62, 446–454 (2007).
Delgado, M. R., Nystrom, L. E., Fissell, C., Noll, D. C. & Fiez, J. A. Tracking the hemodynamic responses to reward and punishment in the striatum. J. Neurophysiol. 84, 3072–3077 (2000).
Delgado, M. R., Miller, M. M., Inati, S. & Phelps, E. A. An fMRI study of reward-related probability learning. Neuroimage. 24, 862–873 (2005).
Adcock, R. A., Thangavel, A., Whitfield-Gabrieli, S., Knutson, B. & Gabrieli, J. D. Reward-motivated learning: mesolimbic activation precedes memory formation. Neuron 50, 507–517 (2006).
Haber, S. N. & Knutson, B. The reward circuit: linking primate anatomy and human imaging. Neuropsychopharmacology 35, 4–26 (2010). Circuit–function: this is the most comprehensive integration of structure–function in the reward circuitry, with clear implications for circuit–function–symptom inferences.
Craske, M. G. et al. Anxiety disorders. Nat. Rev. Dis. Primers. 3, 17024 (2017).
Herzallah, M. M. et al. Depression reduces accuracy while parkinsonism slows response time for processing positive feedback in patients with Parkinson’s disease with comorbid major depressive disorder tested on a probabilistic category-learning task. Front. Psychiatry 8, 84 (2017). Computation–function–disorder (neurology and psychiatry): this experimental account traverses the computational neural heuristics, cognitive and disorder domains, across neurology and psychiatry, to investigate major depressive disorder in a standalone setting versus a comorbidity setting.
Herzallah, M. M. et al. Depression impairs learning, whereas the selective serotonin reuptake inhibitor, paroxetine, impairs generalization in patients with major depressive disorder. J. Affective Disord. 151, 484–492 (2013).
Von Der Heide, R., Vyas, G. & Olson, I. The social network-network: size is predicted by brain structure and function in the amygdala and paralimbic regions. Social Cogn. Affective Neurosci. https://doi.org/10.1093/scan/nsu009 (2014).
Aghajani, M. et al. Altered white-matter architecture in treatment-naive adolescents with clinical depression. Psychol. Med. 44, 2287–2298 (2014).
Tromp, D. P. M. et al. Altered uncinate fasciculus microstructure in childhood anxiety disorders in boys but not girls. Am. J. Psychiatry. 176, 208–216 (2019).
Kim, M. J. & Whalen, P. J. The structural integrity of an amygdala-prefrontal pathway predicts trait anxiety. J. Neurosci. 29, 11614–11618 (2009). Circuit–disorder: this is an empirical account of the role of the structural integrity of the amygdala–prefrontal circuits and anxiety psychopathology.
Phan, K. L. et al. Preliminary evidence of white matter abnormality in the uncinate fasciculus in generalized social anxiety disorder. Biol. Psychiatry 66, 691–694 (2009).
Young, C. B. et al. Anhedonia and general distress show dissociable ventromedial prefrontal cortex connectivity in major depressive disorder. Transl. Psychiatry. 6, e810 (2016).
Craske, M. G., Hermans, D. & Vervliet, B. State-of-the-art and future directions for extinction as a translational model for fear and anxiety. Phil. Trans. R. Soc. B Biol. Sci. https://doi.org/10.1098/rstb.2017.0025 (2018). This is an illustration of how advances in neuroscience and cognitive science around fear learning inform targeted and efficient interventions for anxiety disorders.
Nusslock, R. et al. Cognitive vulnerability and frontal brain asymmetry: common predictors of first prospective depressive episode. J. Abnormal Psychol. 120, 497–503 (2011).
Craske, M. G., Treanor, M., Zbozinek, T. D. & Vervliet, B. Optimizing exposure therapy with an inhibitory retrieval approach and the OptEx Nexus. Behav. Res. Ther. 152, 104069 (2022).
Hang, Y., Zhang, G., Wang, C., Zhang, N. & Liu, G. Attention bias modification for anxiety disorders in children and adolescents: a systematic review and meta-analysis. Psychiatry Res. 300, 113896 (2021).
Murphy, S. E. et al. Imagining a brighter future: the effect of positive imagery training on mood, prospective mental imagery and emotional bias in older adults. Psychiatry Res. 230, 36–43 (2015).
Iyadurai, L. et al. Intrusive memories of trauma: a target for research bridging cognitive science and its clinical application. Clin. Psychol. Rev. 69, 67–82 (2019).
Cernasov, P. et al. Multilevel growth curve analyses of behavioral activation for anhedonia (BATA) and mindfulness-based cognitive therapy effects on anhedonia and resting-state functional connectivity: interim results of a randomized trial. J. Affective Disord. 292, 161–171 (2021).
Craske, M. G. et al. Positive affect treatment for depression and anxiety: a randomized clinical trial for a core feature of anhedonia. J. Consult. Clin. Psychol. 87, 457–471 (2019).
Braun, U. et al. From maps to multi-dimensional network mechanisms of mental disorders. Neuron 97, 14–31 (2018). This paper describes the application of network neuroscience and large-scale brain networks to the elucidation of risk and resilience for psychiatric symptoms.
Stam, C. J. Modern network science of neurological disorders. Nat. Rev. Neurosci. 15, 683–695 (2014).
Bassett, D. S., Zurn, P. & Gold, J. I. On the nature and use of models in network neuroscience. Nat. Rev. Neurosci. 19, 566–578 (2018). This is a primer on network neuroscience that moves beyond examining two or just a few brain regions to modeling large-scale and distributed brain networks.
Halford, G. S., Wilson, W. H., Andrews, G. & Phillips, S. Categorizing Cognition: Toward Conceptual Coherence in the Foundations of Psychology (MIT Press, 2014).
Laird, A. R. et al. Behavioral interpretations of intrinsic connectivity networks. J. Cogn. Neurosci. 23, 4022–4037 (2011).
Mancuso, L. et al. Tasks activating the default mode network map multiple functional systems. Brain Struct. Funct. https://doi.org/10.1007/s00429-022-02467-0 (2022).
Nusslock, R. & Miller, G. E. Early-life adversity and physical and emotional health across the lifespan: a neuroimmune network hypothesis. Biol. Psychiatry 80, 23–32 (2016).
Teicher, M. H., Samson, J. A., Anderson, C. M. & Ohashi, K. The effects of childhood maltreatment on brain structure, function and connectivity. Nat. Rev. Neurosci. 17, 652–666 (2016).
Ho, T. C. & King, L. S. Mechanisms of neuroplasticity linking early adversity to depression: developmental considerations. Transl. Psychiatry 11, 1–13 (2021).
McLaughlin, K. A., Weissman, D. & Bitrán, D. Childhood adversity and neural development: a systematic review. Annu. Rev. Dev. Psychol. 1, 277–312 (2019).
Luby, J. et al. The effects of poverty on childhood brain development: the mediating effect of caregiving and stressful life events. JAMA Pediatr. 167, 1135–1142 (2013).
Dillon, D. G. et al. Childhood adversity is associated with left basal ganglia dysfunction during reward anticipation in adulthood. Biol. Psychiatry 66, 206–213 (2009).
Cao-Lei, L. et al. Prenatal stress and epigenetics. Neurosci. Biobehav. Rev. 117, 198–210 (2020).
Miller, G. E. et al. Maternal socioeconomic disadvantage is associated with transcriptional indications of greater immune activation and slower tissue maturation in placental biopsies and newborn cord blood. Brain Behav. Immun. 64, 276–284 (2017).
Alegría, M., NeMoyer, A., Falgàs Bagué, I., Wang, Y. & Alvarez, K. Social determinants of mental health: where we are and where we need to go. Curr. Psychiatry Rep. 20, 95 (2018).
Avenevoli, S., Swendsen, J., He, J. P., Burstein, M. & Merikangas, K. R. Major depression in the national comorbidity survey-adolescent supplement: prevalence, correlates and treatment. J. Am. Acad. Child Adolesc. Psychiatry 54, 37–44 (2015).
Scherf, K. S., Smyth, J. M. & Delgado, M. R. The amygdala: an agent of change in adolescent neural networks. Horm. Behav. 64, 298–313 (2013).
Somerville, L. H. & Casey, B. J. Developmental neurobiology of cognitive control and motivational systems. Curr. Opin. Neurobiol. 20, 236–241 (2010).
Arain, M. et al. Maturation of the adolescent brain. Neuropsychiatr. Dis. Treat. 9, 449–461 (2013).
Hu, S., Pruessner, J. C., Coupé, P. & Collins, D. L. Volumetric analysis of medial temporal lobe structures in brain development from childhood to adolescence. NeuroImage 74, 276–287 (2013).
Pfeifer, J. H. et al. Entering adolescence: resistance to peer influence, risky behavior and neural changes in emotion reactivity. Neuron 69, 1029–1036 (2011).
Kolk, S. M. & Rakic, P. Development of prefrontal cortex. Neuropsychopharmacology 47, 41–57 (2022).
Crone, E. A. & Dahl, R. E. Understanding adolescence as a period of social-affective engagement and goal flexibility. Nat. Rev. Neurosci. 13, 636–650 (2012).
Hanson, J. L. et al. Structural variations in prefrontal cortex mediate the relationship between early childhood stress and spatial working memory. J. Neurosci. 32, 7917–7925 (2012).
Nelson, E. E., Leibenluft, E., McClure, E. B. & Pine, D. S. The social re-orientation of adolescence: a neuroscience perspective on the process and its relation to psychopathology. Psychol. Med. 35, 163–174 (2005).
Naragon-Gainey, K., Prenoveau, J. M., Brown, T. A. & Zinbarg, R. E. A comparison and integration of structural models of depression and anxiety in a clinical sample: support for and validation of the tri-level model. J. Abnormal Psychol. 125, 853–867 (2016).
Prenoveau, J. M. et al. Testing a hierarchical model of anxiety and depression in adolescents: a tri-level model. J. Anxiety Disord. 24, 334–344 (2010).
Young, K. S. et al. Dysregulation of threat neurocircuitry during fear extinction: the role of anhedonia. Neuropsychopharmacology 46, 1650–1657 (2021).
Peng, Y. et al. Threat neurocircuitry predicts the development of anxiety and depression symptoms in a longitudinal study. Biol. Psychiatry Cogn. Neurosci. Neuroimaging https://doi.org/10.1016/j.bpsc.2021.12.013 (2022).
Borsboom, D. A network theory of mental disorders. World Psychiatry 16, 5–13 (2017).
Lewis-Fernández, R. & Kirmayer, L. J. Cultural concepts of distress and psychiatric disorders: understanding symptom experience and expression in context. Transcult. Psychiatry 56, 786–803 (2019).
Haroz, E. E. et al. How is depression experienced around the world? A systematic review of qualitative literature. Social Sci. Med. 183, 151–162 (2017).
Holmes, E. A. et al. The Lancet Psychiatry Commission on psychological treatments research in tomorrow’s science. Lancet Psychiatry 5, 237–286 (2018).
Boustani, M. M. et al. Common elements of childhood universal mental health programming. Adm. Policy Ment. Health 47, 475–486 (2020).
Craske, M. G., Treanor, M., Zbozinek, T. D. & Vervliet, B. Optimizing exposure therapy with an inhibitory retrieval approach and the OptEx Nexus. Behav. Res. Ther. 152, 104069 (2022).
Watkins, E. et al. Implementing multifactorial psychotherapy research in online virtual environments (IMPROVE-2): study protocol for a phase III trial of the MOST randomized component selection method for internet cognitive-behavioural therapy for depression. BMC Psychiatry 16, 345 (2016).
Fodor, L. A. et al. Efficacy of cognitive bias modification interventions in anxiety and depressive disorders: a systematic review and network meta-analysis. Lancet Psychiatry 7, 506–514 (2020).
Cristea, I. A., Kok, R. N. & Cuijpers, P. Efficacy of cognitive bias modification interventions in anxiety and depression: meta-analysis. Br. J. Psychiatry 206, 7–16 (2015).
Barry, T. J., Vervliet, B. & Hermans, D. An integrative review of attention biases and their contribution to treatment for anxiety disorders. Front. Psychol. 6, 968 (2015).
Fox, E., Zougkou, K., Ashwin, C. & Cahill, S. Investigating the efficacy of attention bias modification in reducing high spider fear: the role of individual differences in initial bias. J. Behav. Ther. Exp. Psychiatry 49, 84–93 (2015).
Waters, A. M. & Craske, M. G. Towards a cognitive-learning formulation of youth anxiety: a narrative review of theory and evidence and implications for treatment. Clin. Psychol. Rev. 50, 50–66 (2016).
Lanius, R. A. et al. Emotion modulation in PTSD: clinical and neurobiological evidence for a dissociative subtype. Am. J. Psychiatry 167, 640–647 (2010).
Cuijpers, P. et al. A network meta‐analysis of the effects of psychotherapies, pharmacotherapies and their combination in the treatment of adult depression. World Psychiatry 19, 92–107 (2020).
Furukawa, T. A. et al. Initial treatment choices to achieve sustained response in major depression: a systematic review and network meta‐analysis. World Psychiatry 20, 387–396 (2021).
Bhat, B. et al. (eds.) The Long-run Effects of Psychotherapy on Depression, Beliefs and Economic Outcomes. CEPR Press discussion paper no. 17309 (2022); https://cepr.org/publications/dp17309
Bhalotra, S., Baranov, V. & Maselko, J. The long-term impact of treating maternal depression: evidence from a randomized controlled trial in Pakistan. https://paa.confex.com/paa/2016/mediafile/ExtendedAbstract/Paper7956/maternal_draft1.pdf (2016).
Singla, D. R. et al. Psychological treatments for the world: lessons from low-and middle-income countries. Annu. Rev. Clin. Psychol. 13, 149–181 (2017).
Cuijpers, P., Karyotaki, E., Reijnders, M., Purgato, M. & Barbui, C. Psychotherapies for depression in low‐and middle‐income countries: a meta‐analysis. World Psychiatry 17, 90–101 (2018). This meta-analysis shows the effectiveness of psychotherapies for depression in low- to middle-income countries, with larger effect sizes in non-Western countries than in Western countries.
Patel, V. Scale up task-sharing of psychological therapies. Lancet 399, 343–345 (2022).
van Ginneken, N. et al. Primary‐level worker interventions for the care of people living with mental disorders and distress in low‐and middle‐income countries. Cochrane Database Syst. Rev. 8, CD009149 (2021).
Schlund, M. W. & Cataldo, M. F. Amygdala involvement in human avoidance, escape and approach behavior. NeuroImage 53, 769–776 (2010).
Suslow, T. et al. Amygdala activation during masked presentation of emotional faces predicts conscious detection of threat-related faces. Brain Cogn. 61, 243–248 (2006).
Maier, S. F. & Seligman, M. E. Learned helplessness at fifty: insights from neuroscience. Psychol. Rev. 123, 349–367 (2016).
Nelson, B. D., Jackson, F., Amir, N. & Hajcak, G. Attention bias modification reduces neural correlates of response monitoring. Biol. Psychol. 129, 103–110 (2017).
Kircher, T. et al. Effect of cognitive-behavioral therapy on neural correlates of fear conditioning in panic disorder. Biol. Psychiatry 73, 93–101 (2013).
Sandman, C. F. et al. Changes in functional connectivity with cognitive behavioral therapy for social anxiety disorder predict outcomes at follow-up. Behav. Res. Ther. 129, 103612 (2020).
Young, K. S. et al. Treatment for social anxiety disorder alters functional connectivity in emotion regulation neural circuitry. Psychiatry Res. Neuroimaging 261, 44–51 (2017).
Mori, A. et al. Behavioral activation can normalize neural hypoactivation in subthreshold depression during a monetary incentive delay task. J. Affective Disord. 189, 254–262 (2016).
Nagy, G. A. et al. Reward network modulation as a mechanism of change in behavioral activation. Behav. Modification 44, 186–213 (2020).
Cohen, Z. D. & DeRubeis, R. J. Treatment selection in depression. Annu. Rev. Clin. Psychol. 14, 209–236 (2018).
Fernandez, K. C., Fisher, A. J. & Chi, C. Development and initial implementation of the Dynamic Assessment Treatment Algorithm (DATA). PLoS One 12, e0178806 (2017).
Bollobás, B., & Thomason, A. in North-Holland Mathematics Studies Vol. 118 (eds. Karoński, M. & Ruciński, A.) 47–97 (1985).
Bullmore, E. & Sporns, O. Complex brain networks: graph theoretical analysis of structural and functional systems. Nat. Rev. Neurosci. 10, 186–198 (2009).
Braun, U. et al. Dynamic reconfiguration of frontal brain networks during executive cognition in humans. Proc. Natl Acad. Sci. USA 112, 11678–11683 (2015).
Shine, J. M. et al. The dynamics of functional brain networks: integrated network states during cognitive task performance. Neuron 92, 544–554 (2016).
Lord, A., Horn, D., Breakspear, M. & Walter, M. Changes in community structure of resting state functional connectivity in unipolar depression. PLoS ONE 7, e41282 (2012).
Ye, M. et al. Changes of functional brain networks in major depressive disorder: a graph theoretical analysis of resting-state fMRI. PLoS ONE 10, e0133775 (2015).
Liu, Y. Y., Slotine, J. J. & Barabási, A. L. Controllability of complex networks. Nature 473, 167–173 (2011). This paper introduces network control theory, which is suited to establishing causality in a neural network and identifying hubs and targets for treatment.
Gratton, C. et al. Defining individual-specific functional neuroanatomy for precision psychiatry. Biol. Psychiatry 88, 28–39 (2020). This paper discusses strategies for modeling neural networks at the individual-subject level, which is necessary for developing personalized and precision-based interventions.
Etkin, A., Gyurak, A. & O’Hara, R. A neurobiological approach to the cognitive deficits of psychiatric disorders. Dialogues Clin. Neurosci. 15, 419–429 (2013).
Insel, T. R. & Cuthbert, B. N. Medicine. Brain disorders? Precisely. Science 348, 499–500 (2015).
Seitzman, B. A. et al. Trait-like variants in human functional brain networks. Proc. Natl Acad. Sci. USA 116, 22851–22861 (2019).
Laumann, T. O. et al. Functional system and areal organization of a highly sampled individual human brain. Neuron 87, 657–670 (2015).
Finn, E. S. et al. Functional connectome fingerprinting: identifying individuals using patterns of brain connectivity. Nat. Neurosci. 18, 1664–1671 (2015).
Gordon, E. M. et al. High-fidelity measures of whole-brain functional connectivity and white matter integrity mediate relationships between traumatic brain injury and post-traumatic stress disorder symptoms. J. Neurotrauma 35, 767–779 (2018).
Kong, R. et al. Spatial topography of individual-specific cortical networks predicts human cognition, personality and emotion. Cereb. Cortex 29, 2533–2551 (2019).
Romero, N., Sanchez, A. & Vazquez, C. Memory biases in remitted depression: the role of negative cognitions at explicit and automatic processing levels. J. Behav. Ther. Exp. Psychiatry. 45, 128–135 (2014).
Colombo, D. et al. Current state and future directions of technology-based ecological momentary assessment and intervention for major depressive disorder: a systematic review. J. Clin. Med. https://doi.org/10.3390/jcm8040465 (2019).
De Angel, V. et al. Digital health tools for the passive monitoring of depression: a systematic review of methods. NPJ Digit. Med. 5, 1–14 (2022).
Brody, G. H. et al. Protective prevention effects on the association of poverty with brain development. JAMA Pediatr. 171, 46–52 (2017).
Lund, C. et al. Social determinants of mental disorders and the Sustainable Development Goals: a systematic review of reviews. Lancet Psychiatry 5, 357–369 (2018).
Brito, N. H. & Noble, K. G. Socioeconomic status and structural brain development. Front. Neurosci. 8, 276 (2014).
Farah, M. J. The neuroscience of socioeconomic status: correlates, causes and consequences. Neuron 96, 56–71 (2017).
Noble, K. G. et al. Family income, parental education and brain structure in children and adolescents. Nat. Neurosci. 18, 773–778 (2015).
Nusslock, R. & Farah, M. J. The affective neuroscience of poverty. J. Cogn. Neurosci. 34, 1806–1809 (2022).
Troller-Renfree, S. V. et al. The impact of a poverty reduction intervention on infant brain activity. Proc. Natl Acad. Sci. USA 119, e2115649119 (2022).
Ridley, M., Rao, G., Schilbach, F. & Patel, V. Poverty, depression and anxiety: causal evidence and mechanisms. Science 370, eaay0214 (2020).
Vikram, P. et al. EMPOWER: toward the global dissemination of psychosocial interventions. Focus 20, 301–306 (2022).
Fu, Z., Burger, H., Arjadi, R. & Bockting, C. L. Effectiveness of digital psychological interventions for mental health problems in low-income and middle-income countries: a systematic review and meta-analysis. Lancet Psychiatry 7, 851–864 (2020). This meta-analysis shows the moderate effectiveness of digital interventions for mental health problems in low- and middle-income countries, although there is considerable heterogeneity and need for further research.
Bouton, M. E., Maren, S. & McNally, G. P. Behavioral and neurobiological mechanisms of Pavlovian and instrumental extinction learning. Physiol. Rev. 101, 611–681 (2021).
Fullana, M. A. et al. Amygdala where art thou? Neurosci. Biobehav. Rev. 102, 430–431 (2019).
Cooper, S. E., Grillon, C. & Lissek, S. Impaired discriminative fear conditioning during later training trials differentiates generalized anxiety disorder, but not panic disorder, from healthy control participants. Comprehensive Psychiatry 85, 84–93 (2018).
Duits, P. et al. Updated meta-analysis of classical fear conditioning in the anxiety disorders. Depress. Anxiety 32, 239–253 (2015).
Craske, M. G. et al. Is aversive learning a marker of risk for anxiety disorders in children? Behav. Res. Ther. 46, 954–967 (2008).
Der-Avakian, A. & Markou, A. The neurobiology of anhedonia and other reward-related deficits. Trends Neurosci. 35, 68–77 (2012).
Berridge, K. C. & Kringelbach, M. L. Pleasure systems in the brain. Neuron 86, 646–664 (2015).
Admon, R. et al. Dopaminergic enhancement of striatal response to reward in major depression. Am. J. Psychiatry 174, 378–386 (2017).
Liu, W. H. et al. Anhedonia is associated with blunted reward sensitivity in first-degree relatives of patients with major depression. J. Affective Disord. 190, 640–648 (2016).
Vrieze, E. et al. Reduced reward learning predicts outcome in major depressive disorder. Biol. Psychiatry 73, 639–645 (2013). Function–disorder–treatment: this experimental paper connects reward learning, major depressive disorder, and response to treatment, in a clear illustration of the cognition–disorder–treatment axis.
Argyelan, M. et al. Dopamine modulates striatal response to reward and punishment in patients with Parkinson’s disease: a pharmacological challenge fMRI study. Neuroreport 29, 532–540 (2018).
Mattfeld, A. T., Gluck, M. A. & Stark, C. E. Functional specialization within the striatum along both the dorsal/ventral and anterior/posterior axes during associative learning via reward and punishment. Learn. Mem. 18, 703–711 (2011).
Wang, S., Leri, F. & Rizvi, S. J. Anhedonia as a central factor in depression: neural mechanisms revealed from preclinical to clinical evidence. Prog. Neuropsychopharmacol. Biol. Psychiatry 110, 110289 (2021). This article illustrates advances in neuroscience and cognitive science relevant to reward processing and symptoms of anhedonia.
Auerbach, R. P., Pagliaccio, D. & Pizzagalli, D. A. Toward an improved understanding of anhedonia. JAMA Psychiatry 76, 571–573 (2019).
Nelson, B. D., Perlman, G., Klein, D. N., Kotov, R. & Hajcak, G. Blunted neural response to rewards as a prospective predictor of the development of depression in adolescent girls. Am. J. Psychiatry 173, 1223–1230 (2016).
Mackin, D. M. et al. Reward processing and future life stress: stress generation pathway to depression. J. Abnormal Psychol. 128, 305–314 (2019).
Rizvi, S. J., Pizzagalli, D. A., Sproule, B. A. & Kennedy, S. H. Assessing anhedonia in depression: potentials and pitfalls. Neurosci. Biobehav. Rev. 65, 21–35 (2016).
Kalisch, R., Gerlicher, A. M. & Duvarci, S. A dopaminergic basis for fear extinction. Trends Cogn. Sci. 23, 274–277 (2019).
Moustafa, A. A. et al. A model of amygdala-hippocampal-prefrontal interaction in fear conditioning and extinction in animals. Brain Cogn. 81, 29–43 (2013).
Forcadell, E. et al. Does fear extinction in the laboratory predict outcomes of exposure therapy? A treatment analog study. Int. J. Psychophysiol. 121, 63–71 (2017).
Lange, I. et al. Neural responses during extinction learning predict exposure therapy outcome in phobia: results from a randomized-controlled trial. Neuropsychopharmacology 45, 534–541 (2020).
Waters, A. M. & Pine, D. S. Evaluating differences in Pavlovian fear acquisition and extinction as predictors of outcome from cognitive behavioural therapy for anxious children. J. Child Psychol. Psychiatry 57, 869–876 (2016).
Pittig, A. et al. Change of threat expectancy as mechanism of exposure-based psychotherapy for anxiety disorders: evidence from 8,484 exposure exercises of 605 patients. Clin. Psychol. Sci. https://doi.org/10.1177/21677026221101379 (2022).
Dunn, B. D. et al. Bringing light into darkness: a multiple baseline mixed methods case series evaluation of Augmented Depression Therapy (ADepT). Behav. Res. Ther. https://doi.org/10.1016/j.brat.2019.103418 (2019).
Taylor, C. T., Lyubomirsky, S. & Stein, M. B. Upregulating the positive affect system in anxiety and depression: outcomes of a positive activity intervention. Depress. Anxiety 34, 267–280 (2017).
Kryza-Lacombe, M. et al. Changes in neural reward processing following Amplification of Positivity treatment for depression and anxiety: preliminary findings from a randomized waitlist controlled trial. Behav. Res. Ther. 142, 103860 (2021).
Acknowledgements
We acknowledge grants and sponsorship related to the subject of this paper from the National Institute of Mental Health, the Fogarty International Center at the US National Institutes of Health, the Wellcome Trust, the Surgo Foundation, Grand Challenges Canada, the Lone Star Depression Challenge, the Manton Foundation, Arab Palestinian Investment Company (APIC), Hikma Pharmaceuticals, GMS Holdings, and F. Ghandour and S. Mouasher.
Author information
Authors and Affiliations
Contributions
M.G.C. wrote the first draft of the manuscript. M.G.C. and M.M.H. prepared the figures. M.M.H. and V.P. developed the conceptual framework of the manuscript. V.P. drafted the introductory and global health sections. R.N. and M.M.H. drafted the circuit and function sections. M.G.C. drafted the clinical science and intervention science sections. All authors edited, reviewed and refined all versions of the manuscript.
Corresponding author
Ethics declarations
Competing interests
M.C., R.N. and M.H. declare no competing interests. V.P. has received consulting fees from Modern Health, Janssen and Google in the past 12 months.
Peer review
Peer review information
Nature Mental Health thanks Zulfiqar Bhutta, Shekhar Seshadri and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Craske, M.G., Herzallah, M.M., Nusslock, R. et al. From neural circuits to communities: an integrative multidisciplinary roadmap for global mental health. Nat. Mental Health 1, 12–24 (2023). https://doi.org/10.1038/s44220-022-00012-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44220-022-00012-w
This article is cited by
-
Time to synergize mental health with brain health
Nature Mental Health (2023)