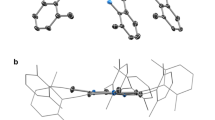
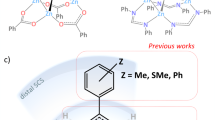
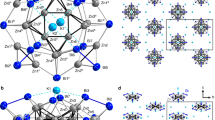
Abstract
The deployment of metalloclusters in applications such as catalysis and materials synthesis requires robust methods for site-differentiation: the conversion of clusters with symmetric ligand spheres to those with unsymmetrical ligand spheres. However, imparting precise patterns of site-differentiation is challenging because, compared with mononuclear complexes, the ligands bound to clusters exert limited spatial and electronic influence on one another. Here, we report a method that uses sterically encumbering ligands to bind to only a subset of a cluster’s coordination sites. Specifically, we show that homoleptic, phosphine-ligated Fe–S clusters undergo ligand substitution with N-heterocyclic carbenes to give heteroleptic clusters in which the resultant clusters’ site-differentiation patterns are encoded by the steric profile of the incoming N-heterocyclic carbene. This method affords access to every site-differentiation pattern for cuboidal [Fe4S4] clusters and can be extended to other cluster types, particularly in the stereoselective synthesis of site-differentiated Chevrel-type [Fe6S8] clusters.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All characterization data, computational data and experimental protocols are provided in the Supplementary Information. Crystallographic data for the structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2214357–2214363. Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures. Coordinates of optimized structures are included as Supplementary Information.
References
Muetterties, E. L. Metal clusters in catalysis III.—Clusters as models for chemisorption processes and heterogeneous. Catalysis. Bull. des Sociétés Chim. Belges 84, 959–986 (1975).
Lee, S. C. & Holm, R. H. Nonmolecular metal chalcogenide/halide solids and their molecular cluster analogues. Angew. Chemie Int. Ed. 29, 840–856 (1990).
Beinert, H., Holm, R. H. & Munck, E. Iron-sulfur clusters: nature’s modular, multipurpose structures. Science 277, 653–659 (1997).
Sokolov, M. N., Fedin, V. P. & Sykes, A. G. In Comprehensive Coordination Chemistry II Vol. 4 (eds McCleverty, J. A., Meyer, T. J. & Wedd, A. G.) 761–823 (Elsevier Pergamon, 2003).
Degroot, M. W. & Corrigan, J. F. In Comprehensive Coordination Chemistry II Vol. 7 (eds McCleverty, J. A., Meyer, T. J., Fujita, M., Powell, A. K. & Creutz, C. A.) 57–123 (Elsevier Pergamon, 2003).
Jena, P. & Sun, Q. Super atomic clusters: design rules and potential for building blocks of materials. Chem. Rev. 118, 5755–5870 (2018).
Doud, E. A. et al. Superatoms in materials science. Nat. Rev. Mater. 5, 371–387 (2020).
Cesari, C., Shon, J. H., Zacchini, S. & Berben, L. A. Metal carbonyl clusters of groups 8–10: synthesis and catalysis. Chem. Soc. Rev. 50, 9503–9539 (2021).
Venkateswara Rao, P. & Holm, R. H. Synthetic analogues of the active sites of iron-sulfur proteins. Chem. Rev. 104, 527–560 (2004).
Lee, S. C., Lo, W. & Holm, R. H. Developments in the biomimetic chemistry of cubane-type and higher nuclearity iron–sulfur clusters. Chem. Rev. 114, 3579–3600 (2014).
Rathnayaka, S. C. & Mankad, N. P. Coordination chemistry of the CuZ site in nitrous oxide reductase and its synthetic mimics. Coord. Chem. Rev. 429, 213718 (2021).
Bigness, A., Vaddypally, S., Zdilla, M. J. & Mendoza-Cortes, J. L. Ubiquity of cubanes in bioinorganic relevant compounds. Coord. Chem. Rev. 450, 214168 (2022).
Bag, S., Trikalitis, P. N., Chupas, P. J., Armatas, G. S. & Kanatzidis, M. G. Porous semiconducting gels and aerogels from chalcogenide clusters. Science 317, 490–493 (2007).
Claridge, S. A. et al. Cluster-assembled materials. ACS Nano 3, 244–255 (2009).
Roy, X. et al. Nanoscale atoms in solid-state chemistry. Science 341, 157–160 (2013).
Tomalia, D. A. & Khanna, S. N. A. Systematic framework and nanoperiodic concept for unifying nanoscience: hard/soft nanoelements, superatoms, meta-atoms, new emerging properties, periodic property patterns, and predictive mendeleev-like nanoperiodic tables. Chem. Rev. 116, 2705–2774 (2016).
Horwitz, N. E. et al. Redox-active 1D coordination polymers of iron–sulfur clusters. J. Am. Chem. Soc. 141, 3940–3951 (2019).
Zhang, J., Bu, X., Feng, P. & Wu, T. Metal chalcogenide supertetrahedral clusters: synthetic control over assembly, dispersibility, and their functional applications. Acc. Chem. Res. 53, 2261–2272 (2020).
Gillen, J. H. et al. Synthesis and disassembly of an organometallic polymer comprising redox-active Co4S4 clusters and janus biscarbene linkers. Chem. Commun. 58, 4885–4888 (2022).
Bartholomew, A. K. et al. Superatom regiochemistry dictates the assembly and surface reactivity of a two-dimensional material. J. Am. Chem. Soc. 144, 1119–1124 (2022).
Kephart, J. A., Mitchell, B. S., Kaminsky, W. & Velian, A. Multi-active site dynamics on a molecular Cr/Co/Se cluster catalyst. J. Am. Chem. Soc. 144, 9206–9211 (2022).
McCool, N. S., Robinson, D. M., Sheats, J. E. & Dismukes, G. C. A Co4O4 cubane water oxidation catalyst inspired by photosynthesis. J. Am. Chem. Soc. 133, 11446–11449 (2011).
Li, G. & Jin, R. Atomically precise gold nanoclusters as new model catalysts. Acc. Chem. Res. 46, 1749–1758 (2013).
Pombeiro, A. J. L. & Kukushkin, V. Y. In Comprehensive Coordination Chemistry II Vol. 1 (eds McCleverty, J. A., Meyer, T. J. & Lever, A. B. P.) 585–594 (Elsevier Pergamon, 2003).
Stack, T. D. P. & Holm, R. H. Subsite-specific functionalization of the [4Fe-4S]2+ analog of iron-sulfur protein clusters. J. Am. Chem. Spc. 109, 2546–2547 (1987).
Walsdorff, C., Saak, W. & Pohl, S. A new preorganized tridentate ligand bearing three indolethiolategroups. Preparation of 3∶1 subsite-differentiated Fe4S4 clusters. J. Chem. Soc. Dalt. Trans. 11, 1857–1862 (1997).
Ohki, Y. et al. Synthetic analogues of [Fe4S4(Cys)3(His)] in hydrogenases and [Fe4S4(Cys)4] in HiPIP derived from all-ferric [Fe4S4{N(SiMe3)2}4]. Proc. Natl Acad. Sci. 108, 12635–12640 (2011).
Kanady, J. S., Tsui, E. Y., Day, M. W. & Agapie, T. A synthetic model of the mn3ca subsite of the oxygen-evolving complex in photosystem II. Science 333, 733–736 (2011).
Terada, T. et al. Tridentate thiolate ligands: application to the synthesis of the site-differentiated [4Fe-4S] cluster having a hydrosulfide ligand at the unique iron center. Chem. Asian J 7, 920–929 (2012).
McSkimming, A. & Suess, D. L. M. Selective synthesis of site-differentiated Fe4S4 and Fe6S6 clusters. Inorg. Chem. 57, 14904–14912 (2018).
Ye, M., Thompson, N. B., Brown, A. C. & Suess, D. L. M. A synthetic model of enzymatic [Fe4S4]–Alkyl intermediates. J. Am. Chem. Soc. 141, 13330–13335 (2019).
Brown, A. C. & Suess, D. L. M. Controlling substrate binding to Fe4S4 clusters through remote steric effects. Inorg. Chem. 58, 5273–5280 (2019).
McSkimming, A., Sridharan, A., Thompson, N. B., Müller, P. & Suess, D. L. M. An [Fe4S4]3+-alkyl cluster stabilized by an expanded scorpionate ligand. J. Am. Chem. Soc. 142, 14314–14323 (2020).
Long, J. R., Williamson, A. S. & Holm, R. H. Dimensional reduction of Re6Se8Cl2: sheets, chains, and discrete clusters composed of chloride‐terminated [Re6Q8]2+ (Q = S, Se) cores. Angew. Chemie Int. Ed. 34, 226–229 (1995).
Zheng, Z., Gray, T. G. & Holm, R. H. Synthesis and structures of solvated monoclusters and bridged Di- and triclusters based on the cubic building block [Re6(μ3-Se)8]2+. Inorg. Chem. 38, 4888–4895 (1999).
Champsaur, A. M. et al. Weaving nanoscale cloth through electrostatic templating. J. Am. Chem. Soc. 139, 11718–11721 (2017).
Jin, S., Adamchuk, J., Xiang, B. & DiSalvo, F. J. The Dean−Evans relation in 31P NMR spectroscopy and its application to the chemistry of octahedral tungsten sulfide clusters. J. Am. Chem. Soc. 124, 9229–9240 (2002).
Que, L., Bobrik, M. A., Ibers, J. A. & Holm, R. H. Synthetic analogs of the active sites of iron-sulfur proteins. VII. Ligand substitution reactions of the tetranuclear clusters [Fe4S4(SR)4]2− and the structure of [(CH3)4N]2[Fe4S4(SC6H5)4]. J. Am. Chem. Soc. 96, 4168–4178 (1974).
Willer, M. W., Long, J. R., McLauchlan, C. C. & Holm, R. H. Ligand substitution reactions of [Re6S8Br6]4–: a basis set of Re6S8 clusters for building multicluster assemblies. Inorg. Chem. 37, 328–333 (1998).
Reed, D. A. et al. Controlling ligand coordination spheres and cluster fusion in superatoms. J. Am. Chem. Soc. 144, 306–313 (2022).
Zhou, H.-C. & Holm, R. H. Synthesis and reactions of cubane-type iron−sulfur−phosphine clusters, including soluble clusters of nuclearities 8 and 16. Inorg. Chem. 42, 11–21 (2003).
Zheng, Z., Long, J. R. & Holm, R. H. A basis set of Re6Se8 cluster building blocks and demonstration of their linking capability: directed synthesis of an Re12Se16 dicluster. J. Am. Chem. Soc. 119, 2163–2171 (1997).
Harmjanz, M., Saak, W., Haase, D. & Pohl, S. Aryl isonitrile binding to [Fe4S4] clusters: formation of [Fe4S4]+ and [{Fe4S4}2]2+ cores. Chem. Commun. (1997) 951–952 (1997).
Champsaur, A. M. et al. Building diatomic and triatomic superatom molecules. Nano Lett. 16, 5273–5277 (2016).
Steigerwald, M. L. et al. Effect of diverse ligands on the course of a molecules-to-solids process and properties of its intermediates. Inorg. Chem. 33, 3389–3395 (1994).
Goh, C., Segal, B. M., Huang, J., Long, J. R. & Holm, R. H. Polycubane clusters: synthesis of [Fe4S4(PR3)4]1+,0 (R = But, Cy, Pri) and [Fe4S4]0 core aggregation upon loss of phosphine. J. Am. Chem. Soc. 118, 11844–11853 (1996).
Fuhr, O., Dehnen, S. & Fenske, D. Chalcogenide clusters of copper and silver from silylated chalcogenide sources chalcogenide clusters of copper and silver from silylated chalcogenide sources. Chem. Soc. Rev. 42, 1871 (2013).
Drance, M. J. et al. Controlled expansion of a strong-field iron nitride cluster: multi-site ligand substitution as a strategy for activating interstitial nitride nucleophilicity. Angew. Chemie Int. Ed. 57, 13057–13061 (2018).
Loewen, N. D., Pattanayak, S., Herber, R., Fettinger, J. C. & Berben, L. A. Quantification of the electrostatic effect on redox potential by positive charges in a catalyst microenvironment. J. Phys. Chem. Lett. 12, 3066–3073 (2021).
Cammack, R. In Advances in Inorganic Chemistry: Iron Sulfur Proteins Vol. 38 (eds Sykes, A. G. & Cammack, R.) 281–322 (Elsevier, 1992).
Rees, D. C. & Howard, J. B. The interface between the biological and inorganic worlds: iron-sulfur metalloclusters. Science 300, 929–931 (2003).
Rouault, T. A. Iron-sulfur proteins hiding in plain sight. Nat. Chem. Biol. 11, 442–445 (2015).
Bista, D. et al. High-spin superatom stabilized by dual subshell filling. J. Am. Chem. Soc. 144, 5172–5179 (2022).
Deng, L. & Holm, R. H. Stabilization of fully reduced iron− sulfur clusters by carbene ligation: the [FenSn]0 Oxidation Levels (n = 4, 8). J. Am. Chem. Soc. 130, 9878–9886 (2008).
Díez-González, S. & Nolan, S. P. Stereoelectronic parameters associated with N-heterocyclic carbene (NHC) ligands: a quest for understanding. Coord. Chem. Rev. 251, 874–883 (2007).
Huang, J., Stevens, E. D., Nolan, S. P. & Petersen, J. L. Olefin metathesis-active ruthenium complexes bearing a nucleophilic carbene ligand. J. Am. Chem. Soc. 121, 2674–2678 (1999).
Weskamp, T., Schattenmann, W. C., Spiegler, M. & Herrmann, W. A. A novel class of ruthenium catalysts for olefin metathesis. Angew. Chemie Int. Ed. 37, 2490–2493 (1998).
Brown, A. C. & Suess, D. L. M. Reversible formation of alkyl radicals at [Fe4S4] clusters and its implications for selectivity in radical SAM enzymes. J. Am. Chem. Soc. 142, 14240–14248 (2020).
Sridharan, A., Brown, A. C. & Suess, D. L. M. A terminal imido complex of an iron–sulfur cluster. Angew. Chemie Int. Ed. 60, 12802–12806 (2021).
Brown, A. C., Thompson, N. B. & Suess, D. L. M. Evidence for low-valent electronic configurations in iron-sulfur clusters. J. Am. Chem. Soc. 144, 9066–9073 (2022).
Ye, M., Brown, A. C. & Suess, D. L. M. Reversible alkyl-group migration between iron and sulfur in [Fe4S4] clusters. J. Am. Chem. Soc. 144, 13184–13195 (2022).
Brown, A. C. & Suess, D. L. M. Valence localization in alkyne and alkene adducts of synthetic [Fe4S4]+ clusters. Inorg. Chem. 62, 1911–1918 (2023).
Kim, Y., Sridharan, A. & Suess, D. L. M. The elusive mononitrosylated [Fe4S4] cluster in three redox states. Angew. Chemie Int. Ed. 61, e202213032 (2022).
Tan, L. L., Holm, R. H. & Lee, S. C. Structural analysis of cubane-type iron clusters. Polyhedron 58, 206–217 (2013).
Guzei, I. A. & Wendt, M. An improved method for the computation of ligand steric effects based on solid angles. Dalt. Trans. 33, 3991–3999 (2006).
Falivene, L. et al. Towards the online computer-aided design of catalytic pockets. Nat. Chem. 11, 872–879 (2019).
Goddard, C. A., Long, J. R. & Holm, R. H. Synthesis and characterization of four consecutive members of the five-member [Fe6S8(PEt3)6]n+ (n = 0−4) cluster electron transfer series. Inorg. Chem. 35, 4347–4354 (1996).
Huang, J., Schanz, H. J., Stevens, E. D. & Nolan, S. P. Stereoelectronic effects characterizing nucleophilic carbene ligands bound to the Cp*RuCl (Cp* = η5-C5Me5) moiety: a structural and thermochemical investigation. Organometallics 18, 2370–2375 (1999).
Nelson, D. J. & Nolan, S. P. Quantifying and understanding the electronic properties of N-heterocyclic carbenes. Chem. Soc. Rev. 42, 6723 (2013).
Durham, J. L., Wilson, W. B., Huh, D. N., Mcdonald, R. & Szczepura, L. F. Organometallic rhenium(III) chalcogenide clusters: coordination of N-heterocyclic carbenes. Chem. Commun. 51, 10536–10538 (2015).
Acknowledgements
The authors thank N. B. Lewis and M. Kumar for their assistance with mass spectrometry experiments and P. Müller for assistance with XRD experiments. This work was supported by the National Institute of General Medical Sciences of the National Institutes of Health under award number R01GM136882 (D.L.M.S.) and by seed funding from the American Chemical Society Petroleum Research Fund under award number 60568-DNI3 (D.L.M.S.). A.C.B. acknowledges fellowships from MathWorks, the National Science Foundation (Graduate Research Fellowship no. 1122374) and the Fannie and John Hertz Foundation.
Author information
Authors and Affiliations
Contributions
T.M.B., A.C.B. and A.S. performed the experiments. T.M.B., A.C.B., A.S. and D.L.M.S. analysed the data and contributed to the manuscript writing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editor: A. Groves, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Synthetic procedures and supplemental data.
Supplementary Data 1
Crystallographic data for 2; CCDC 2214363.
Supplementary Data 2
Crystallographic data for 3; CCDC 2214362.
Supplementary Data 3
Crystallographic data for 4; CCDC 2214361.
Supplementary Data 4
Crystallographic data for 5; CCDC 2214359.
Supplementary Data 5
Crystallographic data for 7; CCDC 2214360.
Supplementary Data 6
Crystallographic data for 9; CCDC 2214357.
Supplementary Data 7
Crystallographic data for 10; CCDC 2214358.
Supplementary Data 8
Coordinate files of optimized mononuclear complexes.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bostelaar, T.M., Brown, A.C., Sridharan, A. et al. A general method for metallocluster site-differentiation. Nat. Synth 2, 740–748 (2023). https://doi.org/10.1038/s44160-023-00286-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44160-023-00286-7
This article is cited by
-
Controlled cluster compositions
Nature Synthesis (2023)