Abstract
Artificial photosynthesis systems are proposed as an efficient alternative route to capture CO2 to produce additional food for growing global demand. Here a two-step CO2 electrolyser system was developed to produce a highly concentrated acetate stream with a 57% carbon selectivity (CO2 to acetate), allowing its direct use for the heterotrophic cultivation of yeast, mushroom-producing fungus and a photosynthetic green alga, in the dark without inputs from biological photosynthesis. An evaluation of nine crop plants found that carbon from exogenously supplied acetate incorporates into biomass through major metabolic pathways. Coupling this approach to existing photovoltaic systems could increase solar-to-food energy conversion efficiency by about fourfold over biological photosynthesis, reducing the solar footprint required. This technology allows for a reimagination of how food can be produced in controlled environments.
Similar content being viewed by others
Main
Food demand is growing globally, but food production is ultimately constrained by the energy conversion efficiency of photosynthesis. Most crop plants can convert sunlight and CO2 into plant biomass at an energy conversion efficiency of only ~1% or less1. Large tracts of land are thus required for crop cultivation to capture the requisite solar energy to provide food for humanity. Recent breeding and genetic engineering efforts to increase photosynthetic efficiency have yielded only select gains in a limited number of food crops2,3,4. Increasing the energy efficiency of food production (solar-to-biomass conversion) would allow for more food to be produced using less resources.
Artificial photosynthesis seeks to overcome the limitations of biological photosynthesis, including low efficiency of solar energy capture and poor carbon dioxide reduction, and could provide an alternative route for food production. Recent studies have demonstrated systems that convert CO2 and H2O into reduced species, such as CO, formate, methanol and H2, through electrolysis processes. CO2, CO and H2 can be upgraded to fuels and chemicals through gas-phase fermentation by select bacteria5,6,7; however, gas–liquid mass transfer limits the volumetric efficiency and results in uneconomic fermentation systems. The use of formate or methanol as a carbon source for fermentation is limited because formaldehyde, a toxic intermediate, is formed during biological metabolism of these substrates8,9,10. To date, electrochemically derived substrates cannot support the growth of most food-producing organisms11. However, acetate is a soluble, two-carbon substrate that can be electrochemically produced12 and is more readily metabolized by a broad range of organisms. The use of acetate produced from CO2 electrolysis to cultivate food-producing organisms could allow food production independent of biological photosynthesis but has not yet been demonstrated.
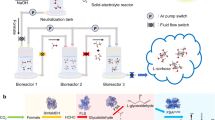
Here we describe the development of a hybrid inorganic–biological system for food production. A two-step electrochemical process converts CO2 to acetate, which serves as a carbon and energy source for algae, yeast, mushroom-producing fungus, lettuce, rice, cowpea, green pea, canola, tomato, pepper, tobacco and Arabidopsis (A. thaliana) (Fig. 1). Coupling this system of carbon fixation to photovoltaics offers an alternative, more energy-efficient approach to food production.
a, CO2 electrolysis uses electricity (generated by photovoltaics) to convert CO2 and H2O into O2 and acetate. This process was optimized to produce an effluent output ideal for supporting the growth of food-producing organisms. b, Chlamydomonas, Saccharomyces, mushroom-producing fungus and a variety of vascular crop plants were grown using the electrolyser-produced effluent. c, The organisms grown using the electrolyser-produced effluent serve as food or food products. This system is capable of making food independent of photosynthesis, using CO2, H2O and solar energy.
Results
Acetate production from CO2 electrolysis
To provide a carbon and energy source independent of biological photosynthesis that can sustain the growth of food-producing organisms, we developed an electrocatalytic process to produce acetate (as either sodium or potassium acetate depending on the electrolyte salt) from CO2. Acetate produced directly from electrochemical CO2 reduction using a copper catalyst has a less than 15% carbon selectivity, which is defined as the amount of carbon in the end product(s) divided by the total amount of carbon reduced in the system13,14,15,16. However, recent studies on CO reduction have demonstrated that acetate can be produced at industrially relevant reaction rates with a carbon selectivity of greater than 50% using a nanostructured copper catalyst12,17,18. To achieve maximum selectivity and production of acetate from a direct CO2 feed, a two-step electrolyser system was demonstrated to convert CO2 to CO and then CO to acetate through a tandem process (Fig. 2a and Extended Data Fig. 1a). More specifically, CO2 is fed to the cathode of the first electrolyser, which utilizes a commercial silver catalyst supported on a gas diffusion layer (that is, a carbon paper) and produces a gaseous product stream containing CO, H2 and trace CO2. The gas diffusion electrode improves the gaseous CO2 transport to the electrocatalyst, achieving higher currents towards CO2 reduction products compared with dissolved CO2 in a typical batch reactor16,19,20,21. A solution of 1 M KHCO3 in deionized water was used as the electrolyte on the anode side (that is, anolyte) for the CO2 electrolyser and recirculated through the anode compartment to maintain the ionic conductivity. The presence of an aqueous electrolyte has been shown to reduce the total cell potential for anion exchange membrane-based CO2 electrolysers13,22. An IrO2 anode was utilized for CO2 reduction, due to its stability in neutral pH. The gas product stream was then fed to the cathode chamber of the CO electrolyser, which contains a commercial copper catalyst for CO reduction, 1 M KOH as the anolyte and a NiFeOx anode. This design is similar to a tandem system previously reported by Romero Cuellar et al.23. By maximizing the conversion of the first and second electrolysers, as well as specifically targeting acetate over other multi-carbon products, this system was able to achieve a single-pass conversion of CO2 to acetate of 25%, a large improvement over the <1% conversion previously reported (Supplementary Table 1). Effluents, the liquid products of electrolysis containing acetate and other by-products (Extended Data Fig. 1c,d), were evaluated as carbon and energy sources for the cultivation of food-producing organisms. Early experiments found that effluents with an acetate-to-electrolyte ratio below 0.4 did not support the growth of algae (Supplementary Note). Maximizing the ratio of acetate to electrolyte was therefore crucial for integrating these carbon products with biological food production.
a–f, Early (a–c) and optimized (d–f) two-step electrolysis systems for the production of acetate. Panels a and d show overviews of the schematics. Effluent containing acetate is collected in the anode compartment of the CO electrolyser. In b and e, carbon selectivities towards specific products and area-normalized production rates are shown. Acetate, propionate, n-propanol and ethanol were collected and quantified in effluent, ethylene was quantified continuously over the course of the experiment and the average value is presented. In c and f, the electrolyser voltage stabilities of CO2 electrolyser (gold) and CO electrolyser (silver) are shown. CO2 electrolyser operated for the entire experiment using 1 M KHCO3 electrolyte, a silver nanoparticle cathode and an iridium oxide anode. CO electrolyser operation began after 30 minutes using 1 M KOH electrolyte, a copper nanoparticle cathode and a nickel iron oxide anode. CO2 flow was held constant at 7 ml min−1. Oscillations in potential were caused by fluctuations in the back-pressure controller. Panel c shows a voltage increase over the time observed. Fresh KOH was fed to recover the electrolyser voltage (arrow) to determine whether the electrolyser voltage increase was due to the acidification of electrolyte. The CO2 scrubber contained 5 M NaOH to capture residual CO2.
Operating parameters of the tandem CO2 electrolysis system were identified that maximized the conversion of CO2 feed to acetate. For the production of CO, the operating current density of the CO2 electrolyser was 100 mA cm−2 at an inlet flow rate of 7 ml min−1 CO2, which maximized CO2 conversion to CO at 43% and maintained a high level of performance (Extended Data Fig. 1e–h). For the CO electrolyser, the operating current density of 150 mA cm−2 was selected to achieve greater than 80% conversion of electrochemically produced CO into C2+ products over the entire duration of electrolysis (Extended Data Fig. 1g,h). Initial experiments found that trace amounts of CO2 negatively affected acetate selectivity in the CO electrolyser and led to a rapid increase in CO electrolyser voltage (Fig. 2b,c). When a 5 M NaOH scrubber was introduced between the two electrolysers, acetate selectivity was increased by over a factor of 3, because the scrubber prevented the unreacted CO2 from the first reactor from reaching the CO electrolyser (Fig. 2d–f and Extended Data Fig. 1b). Overall, 57% of reacted CO2 formed acetate at a production rate of 0.7 g d−1 cm−2, representing the highest conversion of CO2 feed to acetate reported to date (Fig. 2e and Supplementary Table 1).
The tandem CO2 electrolysis system operated stably while producing effluent with high acetate-to-electrolyte ratios. The electrolyser voltage for the CO2 electrolyser remained constant near 2.95 V (Fig. 2f) with less than a 60 mV increase in voltage over the six-hour experiment. The CO reactor operated at 2.22 V with a voltage increase of 160 mV over the course of the experiment (Fig. 2f), which is attributed to the pH shift in the electrolyte from 13.7 to 13.4 as acetate accumulated. Both catholyte (through the use of a cold-trap) and anolyte were collected and analysed for product quantification. As >99% of the produced acetate was collected in the anolyte, this was the primary effluent used for food production. The final effluent produced contained 0.75 M acetate and an acetate-to-electrolyte salt ratio of 0.75, representing the highest recorded acetate-to-electrolyte product stream to date (Supplementary Table 1).
Using CO2 electrolysis to grow food heterotrophically
We used the photosynthetic alga Chlamydomonas (C. reinhardtii), which can grow heterotrophically on acetate in the dark, as a model to determine whether the improved effluent produced by the electrolysers could support the growth of food-producing organisms. Chlamydomonas is added to processed foods for protein fortification and has been shown to have a positive effect on human gastrointestinal health24,25,26,27,28. Algae can produce large amounts of starch, protein and oil. Chlamydomonas grown heterotrophically can produce over 1 g of starch, 1 g of protein and 0.7 g of lipid per litre per day28,29. To evaluate effluent as a carbon source, Chlamydomonas was grown heterotrophically in the dark with effluent diluted to match the acetate concentration of a typical liquid heterotrophic growth medium (17.5 mM acetate in Tris–acetate–phosphate (TAP) medium) and adjusted to pH 7.2. Effluent-derived acetate served as the sole carbon and energy source. Effluents from different electrolysis experiments used throughout this manuscript are noted by the acetate-to-electrolyte concentration ratio (acetate concentration: electrolyte concentration), with more details provided in Supplementary Table 2. All effluents evaluated with an acetate-to-electrolyte ratio of greater than 0.4 M acetate: 1 M electrolyte enabled the growth of Chlamydomonas in the dark (Supplementary Note, Fig. 3a–c, Extended Data Figs. 2 and 3a–e, and Supplementary Table 3).
a, Image of Chlamydomonas cultures taken after four days of growth in darkness; the cultures were grown with effluent (0.691 M acetate: 1 M KOH) or with no effluent. b,c, Growth of Chlamydomonas cultures grown in the dark, shaking, at 30 °C, with effluent (0.75 M acetate: 1 M KOH), with acetate (control) or with neither (no acetate or effluent). Panel b shows daily cell counts for eight days of growth, and c shows dry weight after two days of growth. Chlamydomonas cultures were grown in TAP media with acetate, without acetate or with effluent in place of acetate to match the acetate concentration of a typical liquid heterotrophic growth medium (17.5 mM). All Chlamydomonas media were adjusted to pH 7.2. d,e, Saccharomyces cerevisiae cultures were grown at 30 °C, shaking, in YPD media with glucose (3.45 g l−1), without glucose (0 g l−1) or with effluent (0.75 M acetate: 1 M KOH) in place of glucose with an acetate concentration to match the energetic equivalent of 3.45 g glucose per l (53.36 kJ l−1). All Saccharomyces media were adjusted to pH 6.0. Panel d shows optical density (OD) (600 nm) over 96 hours. Panel e shows dry weight 24 hours after inoculation. In b–e, each data point represents three biological replicates. The error bars represent standard deviations. In c and e, two-tailed unpaired t-tests showed no significant difference between Chlamydomonas growth in acetate and in effluent media (P = 0.1045) and no significant difference between Saccharomyces growth in glucose and in effluent media (P = 0.1857). NS, not significant. f, Images of pearl oyster, enokitake, elm oyster and blue oyster mushroom mycelium from colonization of a solid vermiculite substrate soaked with YPD media containing simulated effluent in place of glucose (0.0691 M acetate: 1 M KOH) to reach 0.5% (w/w) acetate as the primary carbon and energy source. The images were taken 24 days after inoculation. Scale bar, 20 mm. g, Growth seven days after inoculation of pearl oyster mushroom mycelium (white) on solid vermiculite substrate soaked with liquid YPD media containing simulated effluent in place of glucose (0.0691 M acetate: 1 M KOH) as indicated. Scale bar, 20 mm.
Chlamydomonas grown on an effluent with an improved acetate-to-electrolyte salt ratio (0.75 M acetate: 1 M KOH) had a yield of 0.28 g algae per g acetate (Fig. 3b,c), which is comparable to yields reported from non-effluent medias29. Chlamydomonas utilized over 99% of acetate in the media (Extended Data Fig. 3f). No products of photosynthesis (such as carbohydrates) or ancient photosynthesis (such as petroleum-derived carbon sources) were required for growth. Hence, our cultivation of a photosynthetic organism using carbon fixed through electrolysis is fully decoupled from biological photosynthesis.
The nutritional yeast Saccharomyces cerevisiae is used as a food source in single-cell protein spreads and in the production of breads and fermented beverages30,31. Yeast is a heterotrophic organism most commonly grown with glucose derived from photosynthesis (that is, starch-derived) as a carbon and energy source. To cultivate yeast without inputs derived from photosynthesis, we replaced the primary carbon source, glucose, in the yeast–peptone–dextrose (YPD) media with effluent from electrolysis. All media were adjusted to pH 6.0. Electrolyser-produced effluent (0.75 M acetate: 1 M KOH) supported the growth of yeast, enabling a yield of 0.19 g yeast biomass per g acetate as well as a ninefold increase in OD600 and a twofold increase in dry weight compared with yeast grown without acetate or glucose (Fig. 3d,e). After successfully decoupling the production of yeast from photosynthesis-derived carbon and energy, we sought to achieve the same results in mushroom-producing fungus. Mushrooms are widely consumed as a food, and fungal mycelium has emerged as a high-protein meat analogue32. These fungi are typically cultivated on solid substrates composed of photosynthesis-derived carbohydrates, such as cellulose or rice flour. To cultivate fungal mycelium without these carbohydrates, we developed a solid-state fermentation approach that used effluent as the primary carbon and energy source. Due to the amounts needed, simulated effluents were used here, made up of the same components as electrolyser-produced effluents. Simulated effluent (0.691 M acetate: 1 M KOH) was added to YPD media without glucose to reach 0.5% (w/w) acetate; this substrate supported the growth of pearl oyster, blue oyster, elm oyster, coral tooth and enokitake mycelium (Fig. 3f and Extended Data Fig. 3g–i). The majority of the substrate was fully colonized by all five species, indicating that effluent can serve as a carbon source for the cultivation of a variety of mushroom-producing fungus mycelia. Higher levels of effluent inhibited mycelium growth (Fig. 3g and Extended Data Fig. 3g), probably due to the higher levels of effluent by-products, specifically propionate, which is used as an antifungal food preservative33. The successful growth of mycelium indicates the ability to produce mycelium-based foods and potentially mushrooms independent of carbon and energy derived from photosynthesis.
Acetate can be metabolized by crop plants
To further evaluate whether carbon fixed through CO2 electrolysis could be used to produce plant-based food, we examined the potential of acetate to serve as a carbon and energy source for crops. We tracked acetate incorporation into plant biomass using heavy-isotope 13C-acetate labelled at both carbon atoms to evaluate whether exogenous acetate can be metabolized by crops. Previous acetate incorporation studies have primarily focused on metabolites involved in lipid biosynthesis in only a few plant species. In these studies, acetate was used in low concentrations as a tracer, not as a carbon source (Supplementary Table 4)34,35,36. To investigate acetate utilization as a carbon source, we grew undifferentiated lettuce tissue (callus) (to avoid the carbon and energy stored in seeds) in the dark in liquid media containing effluent (0.691 M acetate: 1 M KOH) and added in 13C-acetate to enable tracking of carbon incorporation. Calli showed extensive 13C-labelling in multiple pathways, confirming that lettuce tissue metabolizes acetate as a carbon and energy source (Fig. 4a). Labelling of citrate/isocitrate, succinate, α-ketoglutarate and malate indicated that exogenous acetate was converted to biologically active acetyl-CoA and entered the tricarboxylic acid cycle (TCA) cycle to produce energy (GTP, NADH and FADH2) (Fig. 4a). Amino acids were also labelled with 13C, indicating that carbon from acetate can be used to build proteins. Products and intermediates of gluconeogenesis were also labelled, indicating that carbon from acetate can be used for carbohydrate biosynthesis (Fig. 4a and Extended Data Fig. 4). Carbon incorporation of 13C-acetate into amino acids and sugars through the TCA cycle, glycolysis and gluconeogenesis provides strong evidence that exogenous acetate can be readily incorporated into the bulk biomass of lettuce tissue grown in dark conditions and could be a carbon and energy source for other crops.
a, Heat maps showing the log2 fold enrichment of 13C between treated and untreated whole lettuce plants and lettuce callus (undifferentiated lettuce). Whole lettuce plants were grown in the light and treated with no acetate or with 13C-acetate (n = 3; each replicate was a mixture of tissue from ten plants). Lettuce callus was grown in the dark and treated with electrolyser-produced effluent (0.691 M acetate: 1 M KOH) (n = 4) and 13C-acetate (n = 3). A variety of crop species were treated with 13C-acetate, and incorporation was measured. 2/3-PG, 2/3-phosphoglycerate. Representative no-acetate control replicates for whole lettuce plant, lettuce callus, cowpea and Arabidopsis are included for comparison. There was broad 13C incorporation into crop metabolism, indicating that exogenously supplied acetate can be used for energy and biomass production in these plants. b, Schematic of exogenous acetate metabolism by a typical plant cell. c, 13C incorporation into iso/citrate and malate at multiple carbon positions in whole lettuce plants. We calculated the ratio of iso/citrate and malate molecules that were detected with 13C incorporation compared with 12C. M + X denotes the number of labelled carbons in the molecule. The error bars represent the standard deviation between the biological replicates (n = 3).
To test acetate uptake and utilization by whole plants, we grew lettuce plants in the light with 13C-acetate added to the plant growth agar medium at 2 mM. In vegetative leaf tissue, we observed that amino acids, sugars and intermediates from the TCA cycle, glycolysis and gluconeogenesis were labelled with 13C at multiple carbon positions (Fig. 4a–c). Finding labelled metabolites in leaf tissue indicates that carbon from acetate assimilated by the roots can be distributed throughout the plant. In addition to lettuce, we discovered that acetate can be incorporated into a broad variety of crops. Rice, green pea, jalapeño pepper, canola, tomato, cowpea, tobacco and Arabidopsis seedlings grown in the light on solid agar containing 13C-acetate all showed similar 13C-labelling of amino acids, carbohydrates and TCA cycle intermediates (Fig. 4a and Extended Data Fig. 5) as was observed in lettuce and lettuce callus. This metabolite labelling pattern suggests a conserved mechanism of acetate utilization that exists across diverse plant species.
Lettuce seeds germinated normally on all concentrations of acetate evaluated (up to 10 mM) (Extended Data Fig. 6a,b). However, plant growth was largely inhibited by acetate at concentrations that would have measurably increased plant biomass, although some growth parameters such as roots showed increased growth (Extended Data Figs. 6 and 7). Lettuce plants grown with electrolyser-produced effluent (0.691 M acetate: 1 M KOH or 0.648 M acetate: 1 M KHCO3) added to reach a final media concentration of 1.0 mM acetate did not show additional growth inhibition in plant weight or leaf number from secondary electrolysis products (Extended Data Fig. 7b,d). Plant tolerance and consumption of acetate as a heterotrophic energy source will need to be increased to fully decouple plants from biological photosynthesis. Together, these analyses explore acetate as a heterotrophic carbon source for crops and illustrate that many crop species can process acetate into a usable biological form and incorporate it into major energy-production cycles through TCA and biomass in the form of sugars and amino acids.
Discussion
We demonstrated a hybrid inorganic–biological system that can produce food from carbon dioxide and electricity, independent of biological photosynthesis. CO2 electrolysis for acetate production was significantly improved for the purpose of biological integration. Using a two-step process, we achieved electrochemical reduction of CO2 to acetate with a 57% carbon selectivity towards acetate, the highest published value to date. The electrolysis system was further engineered to produce improved effluents with acetate-to-electrolyte salt ratios as high as 0.75, well above the ratio determined necessary to support biological growth. Electrochemically derived acetate was incorporated into a diverse variety of organisms grown for food, including algae, fungi and crop plants. This includes the cultivation of a photosynthetic organism, Chlamydomonas, utilizing carbon fixed through electrolysis, independent of biological photosynthesis. 13C-labelling experiments showed that a broad range of crops can utilize exogenous acetate for energy and biomass production, suggesting that acetate has the capacity to support crop growth with further optimization.
By powering electrolysis with photovoltaics, the conversion of sunlight and CO2 to food in our system (photovoltaics to electrolysis to acetate to yeast) is almost 18 times more solar-to-biomass energy-conversion-efficient than typical food production, which relies on biological photosynthesis (photosynthesis to glucose to yeast) (Extended Data Fig. 8). For algae production, our process (photovoltaics to electrolysis to acetate to algae) is almost four times more solar-to-biomass energy-conversion-efficient than biological photosynthesis of crop plants (photosynthesis to crop plants) and is equivalent to or higher than the instantaneous energy efficiency of outdoor algae production37 (Fig. 5 and Extended Data Fig. 8). There is potential for continued improvement of the system through advances in photovoltaics, electrolysis and acetate utilization in food-producing organisms. Our approach to food production is ideally suited for applications where high energy efficiencies and low physical space usage are desired, such as on space flight missions or in controlled environments on Earth. Widespread adoption of this approach in conjunction with readily available solar energy could allow for the production of more food or animal feed for a given solar footprint, which will help meet the rising demand for food without the expansion of agricultural lands.
Sankey diagrams of solar energy to plant- and algae-based food production compare the efficiencies of artificial and biological photosynthesis. Losses during energy conversion from sunlight (100% solar energy) to food are represented by arrows; the width of each arrow is proportional to the energy lost, and the percentage of the total energy lost at each step is indicated. The green arrows indicate the solar energy that can be found in biomass grown from artificial and biological photosynthesis; the width of each arrow is proportional to the energy, and the percentage of the total solar energy found in the biomass is indicated. The values for biological photosynthesis are from Zhu et al.57. For artificial photosynthesis, the values for electrochemical CO2 reduction to acetate and heterotrophic cultivation on acetate were determined in this work. The value for photovoltaic losses is based on a commercially available silicon solar cell49,50.
Methods
Electrolysis methods
Cathode preparations
For the CO2 reduction electrodes, commercial silver (Ag) catalysts (nanopowder with <100 nm particle size, 99.5%) were purchased from Sigma Aldrich. Catalyst ink was prepared by suspending 100 mg of Ag nanopowder in 20 ml of 2:1 (v:v) isopropanol/deionized water. The suspension was sonicated for five minutes to allow complete dispersion of Ag particles. Afterwards, Sustanion XA-9 ionomer (5% (w/w) in ethanol, Dioxide Materials) was added until the ionomer reached 10% (w/w) in solution (metal basis). The resulting mixture was further sonicated for an additional 30 minutes to allow complete dispersion. To prepare the electrodes, a 25 cm2 piece of Sigracet 39BB gas carbon paper (Fuel Cell Store), used as a gas diffusion layer, was heated on a hot plate to 100 °C. Catalyst ink was airbrushed onto the carbon paper until a loading of 1.4 mg cm−2 was reached. The electrode was dried at 70 °C overnight to ensure the complete evaporation of solvents.
For the CO reduction electrodes, commercial copper (Cu) catalyst (nanopowder with 25 nm particle size (TEM)) was purchased from Sigma Aldrich. Catalyst ink was prepared by suspending 100 mg of Cu nanopowder in 20 ml of 2:1 (v:v) isopropanol/deionized water. The suspension was sonicated for five minutes to allow complete dispersion of Cu particles. Afterwards, Sustanion XA-9 ionomer (5% (w/w) in ethanol, Dioxide Materials) was added until the ionomer reached 20% (w/w) in solution (metal basis). The resulting mixture was further sonicated for an additional 30 minutes to allow complete dispersion. To prepare the electrodes, a 5 cm2 piece of Sigracet 39BB carbon paper (Fuel Cell Store) was heated on a hot plate to 100 °C. Catalyst ink was drop-casted onto the carbon paper until a loading of 2.0 mg cm−2 was reached. The electrode was dried at 70 °C overnight to ensure the complete evaporation of solvents.
Anode preparations
For experiments using 1 M KHCO3, IrO2 catalysts were prepared via a previously reported method38. In a typical preparation, titanium felt (Fuel Cell Store) was degreased using acetone and etched in boiling 0.5 M oxalic acid (98%, Sigma Aldrich). The titanium felt was then dip-coated in a solution of 10 ml of isopropanol, 10 vol% concentrated HCl (ACS reagent, Sigma Aldrich), containing 30 mg dissolved IrCl3·xH2O (99.8%, Alfa Aesar). This was followed by drying at 100 °C for ten minutes and calcination at 500 °C for ten minutes. This procedure was repeated until a loading of 3 mg cm−2 was achieved.
For experiments conducted in alkaline conditions, NiFeOx anodes prepared by a previously reported method39 were used. In a typical preparation, Ni foam (>99.99%, MTI Corporation) was sonicated in a 5 M HCl solution for 30 minutes to remove the NiOx layer on the surface. The foam was then rinsed using deionized water and ethanol and dried in air. Electrodeposition was carried out in a standard three-electrode electrochemical reactor containing the nickel foam as the working electrode, a platinum wire counter electrode and a Ag/AgCl (Pine Research) reference electrode. The electrolyte bath contained 3 mM Ni(NO3)2·6H2O and 3 mM Fe(NO3)3·9H2O. A constant potential of −1.0 V versus Ag/AgCl was applied for 300 seconds. The electrode was then removed and rinsed with ethanol and deionized water, followed by drying overnight at 70 °C to fully remove solvents. A fresh cathode was used for each experiment, whereas the anodes were reused multiple times.
Flow electrolyser
Both the CO2 and CO electrolysers were constructed as 5 cm2 membrane electrode assemblies with serpentine flow patterns. The cathode end plates and the CO2 electrolyser anode end plate were gold-plated stainless steel. The CO electrolyser anode end plate was not gold coated, as stainless steel is stable in alkaline conditions. The CO2 gas flow rate into the electrolysers was controlled via a Brooks GF040 and held at 7 ml min−1 for the two-step experiments. The back pressure was controlled using a back-pressure controller (Cole-Palmer). The anolyte flow rates for both electrolysers were controlled via a peristaltic pump, with flow rates ranging between 0.5 and 1 ml min−1. A cold trap chilled using ice was placed between the gas outlet stream of the CO electrolyser and the back-pressure controller to capture any vaporized liquid products that might exit through the gas stream.
For the CO2 electrolyser, the Ag cathode and the IrO2 anode were pressed into a Sustanion 37-50 Anion Exchange Membrane (Dioxide Materials) at 20 lb-in torque. Laser-cut Teflon (McMaster Carr) was used as a gasket to ensure that the electrolyser was gas-tight. 1 M KHCO3 was used as the anolyte for all CO2 reduction experiments. A constant current was applied using a power source (National Instruments). For the CO electrolyser, the Cu cathode and the IrO2 or NiFeOx anode were pressed against an FAA-3-50 anion exchange membrane (Fumatech) at 20 lb-in torque. 1 M KHCO3 or 2 M KOH was recirculated through the anode chamber. Liquid products were accumulated in the anolyte of the CO electrolyser until a target concentration was reached. The quantification of liquid products was conducted at the end of the experiment. For the CO electrolyser, the applied current was controlled using an Autolab PG128N potentiostat (Metrohm).
Gas products were quantified using a Multiple Gas Analyzer No. 5 gas chromatography system (SRI Instruments) equipped with a HaySep D and Molsieve 5 A column connected to a thermal conductivity detector. All gas products were quantified using the thermal conductivity detector data. Liquid products were quantified using a Bruker AVIII 600 MHz NMR spectrometer. Typically, 20 to 100 μl of collected electrolyte exiting the reactor was diluted to 500 μl, and then 100 μl D2O containing 20 ppm (m/m) dimethyl sulfoxide (≥99.9%, Alfa Aesar) was added. One-dimensional 1H spectrum was measured with water suppression using a pre-saturation method.
Effluents containing all liquid products were used for the growth of food-producing organisms. Electrolyser-produced effluents were used when possible, but in some cases simulated effluents of the same makeup were used.
Algae methods
Chlamydomonas reinhardtii (strain 21gr+, CC-1690 from the Chlamydomonas Resource Center) was cultivated on TAP media40 where the source of acetate was a commercial supplier (Sigma 64-19-7), simulated effluent, electrolyser-produced effluent or none at all. When effluent (electrolyser produced or simulated) was the acetate source, it was added until the desired final acetate concentration was reached (17.5 mM). Ethanol (Koptec 64-17-5), propionic acid (Sigma 79-09-4) and n-propanol (Sigma 71-23-8) were added as indicated in Supplementary Table 3. All media were adjusted to pH 7.2 with 5 M HCl. Effluents and media concentrations are shown in Supplementary Tables 2 and 3.
Chlamydomonas was grown in 50 ml of media in 250 ml flasks in the dark. The flasks were stationary at 22 °C except for the experiment using 0.75 M acetate: 1 M KOH (data in Fig. 3b,c), where the flasks were at 30 °C with a shaking speed of 150 r.p.m.29. Aliquots were taken in a darkened biosafety cabinet and used for OD measurements at wavelength 750 nm (QuickDrop Spectrophotometer, Molecular Devices), chlorophyll extraction and quantification41, and cell counts using a haemocytometer or the Bio-Rad TC20 Automated Cell Counter. For dry cell weights, the entire culture was centrifuged, washed two times with deionized water to remove residual salts, dried overnight at 100 °C and then weighed. Images were taken with a Nikon 7500 DSLR camera.
Yeast and mushroom methods
Saccharomyces cerevisiae was cultured in a base medium of yeast extract (10 g l−1) and peptone (20 g l−1) with glucose, effluent or no additional primary carbon source42. Standardized comparisons of acetate and glucose were made on the basis of energy content, and a variety of glucose and effluent concentrations were tested, as shown in Supplementary Table 5. The highest concentration is based on 2% sodium acetate42. Media with a carbon source with the energetic equivalent of 53.36 kJ l−1 (0.061 M glucose or 0.019 M acetate) were determined to be the most efficient and thus were used in the yeast experiments shown in Fig. 3d,e. All media were adjusted to pH 6.0 with 5 M HCl. Cells were grown in 5 ml of media in culture tubes at 30 °C and 251 r.p.m. Growth was monitored by measuring OD at 600 nm and dry cell weight at 96 hours. For dry weights, the pellet was washed with deionized water to remove residual salts, dried overnight at 100 °C and then weighed. Effluents and media concentrations are shown in Supplementary Tables 2 and 3.
For the cultivation experiments with mushroom-producing fungi, the following five species were used: pearl oyster (Pleurotus ostreatus), elm oyster (Hypsizygus ulmarius), blue oyster (Pleurotus ostreatus var. columbinus), enokitake (Flammulina velutipes) and coral tooth (Hericium coralloides). The strains were purchased from liquidfungi.com and maintained on a liquid medium of glucose, yeast extract and peptone. Fungal mycelia were grown in a solid-state fermentation approach that roughly followed the PF-Tek methodology43; however, the carbon sources typically added, such as starch (rice flour), peat moss and coconut fibre, were omitted. A solid substrate of fine-grade vermiculite (30 g) was mixed with gypsum (0.5 g) and added to a 10 oz wide-mouth mason jar (Kamota). Liquid growth media (90 ml) were added to each jar, which soaked into the vermiculite. Media composition was the same as the maintenance media except that the carbon source was glucose (20 g l−1), acetate (as indicated), effluent (as indicated), no carbon source or a combination of these. Media pH was adjusted to 6.0 with HCl. The jars were closed with lids with four drilled holes (12.7 mm in diameter) covered with a synthetic filter disk (0.3 µm pore size) to allow gas exchange. The jars were autoclaved for 45 minutes at 121 °C. Liquid mycelium cultures were centrifuged, washed with sterilized media, resuspended and then used to inoculate the sterilized jars.
Plant methods
Plant material, media and growth conditions
The following nine plants were used: lettuce (Lactuca sativa L. cv. ‘Black Seeded Simpson’), rice (Oryza sativa ssp. japonica cv. ‘Kitaake’), green pea (Pisum sativum), tomato (Solanum lycopersicum cv. ‘Micro-Tom’), jalapeño pepper (Capsicum annuum cv. ‘Jalapeño’), canola (Brassica napus), cowpea (Vigna unguiculata L. cv. CB46), thale cress (A. thaliana var. ‘Columbia’) and tobacco (Nicotiana tabacum cv. Xanthi).
Plants were cultivated on 50 ml of basal medium of 1/2 Murashige and Skoog salts44 (Caisson Labs) and Gamborg’s vitamins45 (Phytotech Labs), unless otherwise noted. Solid media were made with 0.7% in vitro growth-grade agarose (Caisson Labs). The following were added as indicated: sucrose (2%), acetic acid, 13C2-acetate (Sigma Aldrich) or electrolyser-produced effluent. All media were adjusted to pH 5.8 using 1 M KOH or HCl. The plants were grown in 16:8-hour light–dark cycles under fluorescent lighting (100 µmol m−2 s−1) at 22 °C, unless otherwise noted.
Acetate feeding experiment
Lettuce was germinated in soil for 11 days. The plants were clipped at the base of the stem and were further trimmed so that all lettuce stems were the same length for the start of the experiment. Each plant was transferred to an 8 ml glass vial containing deionized water with acetate dissolved at various concentrations (nine plantlets per treatment: 0, 0.1, 0.3, 0.6, 1, 3, 6 and 10 mM acetate). After 29 days, the plantlets were imaged (using a Nikon 7500 DSLR with an AF-S VR Micro-Nikkor 105 mm f/2.8 G IF-ED lens) and removed from the solution for measurement of leaf number, stem length, root length and fresh weight. Stem length was measured from the first lateral root to the tip of the stem, and roots were measured on the basis of the longest root on the plantlet.
Experimental setup for plant germination and 13C exposure
For the lettuce germination experiments, seeds were sterilized using a 15% bleach solution and a drop of Tween for every 50 ml of sterilizing solution. The seeds were incubated in the sterilizing solution for 15 minutes while shaking and then washed five times with sterile water, five minutes for each wash while shaking. Ten lettuce seeds were placed on agar basal media (25 ml) supplemented with acetate (0.1, 0.3, 0.6, 1, 2, 3, 6 or 10 mM), with 2 mM labelled 13C2-acetate or with no additions to the base medium. They were allowed to germinate and grow for 28 days and were then imaged and removed to measure stem length, leaf number and fresh weight (not including the roots). Tissue samples for the 13C-labelling experiments and the untreated controls were frozen in liquid nitrogen and stored at −80 °C until they were used for metabolomic analysis. For the other plant species, the same sterilization procedure was used for the seeds, and the plants were grown until there was at least a few hundred milligrams of tissue for metabolomic analysis (canola, 14 days; rice, 14 days; Arabidopsis, 18 days; Micro-Tom tomato, 22 days; green pea, 22 days; Nicotiana benthamiana, 32 days; cowpea, 32 days).
Lettuce callus in liquid culture
Undifferentiated lettuce callus was generated by plating pieces of lettuce leaf tissue on callus-inducing media (1/2 Murashige and Skoog salts, 0.05 mg l−1 α-naphthaleneacetic acid, 0.4 mg l−1 6-benzyl aminopurine and Gamborg’s vitamins45) (Phytotech Labs). The calli were cut to the desired size and weighed to ensure that an equal amount of tissue was added to each flask. Each flask of calli was incubated in liquid 1/2 Murashige and Skoog media supplemented with acetate, 13C2-acetate or sucrose (2%) as indicated and to the specified concentrations. All cultures were grown at 22 °C in the dark at 100 r.p.m. Tissue samples for the 13C-labelling experiment and control samples were frozen in liquid nitrogen and stored at −80 °C until they were used for metabolomic analysis.
Metabolomic analysis
Sample preparation
Plant tissue was freeze-dried, and then approximately 10 mg was weighed into a 2 ml tube and homogenized using a bead mill, using three 2.8 mm beads per tube. To each sample, 750 µl of 1:2 water:methanol was added, and the samples were then vortexed for 60 min at 4 °C. Then, 500 µl of chloroform was added, and the samples were vortexed at 4 °C for an additional 15 min. After centrifugation for 10 min (16,000 g at 4 °C), the top, polar layer was transferred to a glass vial and analysed by liquid chromatography–mass spectrometry (LC–MS).
LC–MS
LC–MS metabolomics analysis was performed at the University of California, Riverside Metabolomics Core Facility. The analysis was performed on a Synapt G2-Si quadrupole time-of-flight mass spectrometer (Waters). Metabolite separations were carried out on an I-class UPLC system (Waters) using a ZIC-pHILIC column (2.1 mm × 150 mm, 5 µM) (EMD Millipore). The two mobile phases used were (A) water with 15 mM ammonium bicarbonate adjusted to pH 9.6 with ammonium hydroxide and (B) acetonitrile. The column was held at 20 °C, and the flow rate was 150 µl min−1. The sample injection volume was 2 µl. The following gradient was performed: 0 min, 10% A, 90% B; 1.5 min, 10% A, 90% B; 16 min, 80% A, 20% B; 29 min, 80% A, 20% B; 31 min, 10% A, 90% B; 32 min, 10% A, 90% B.
The mass spectrometer was operated in negative ion mode (50 to 1200 m/z) with a 100 ms scan time. Tandem MS was acquired in a data-dependent fashion. The source temperature was 150 °C, and the desolvation temperature was 600 °C. Nitrogen was used as a desolvation gas (1,100 l h−1) and cone gas (150 l h−1). The collision gas used was argon. The capillary voltage was 2 kV. Leucine enkephalin was infused and used for mass correction.
Data processing and analysis
Data processing was performed with the open-source Skyline software46. Metabolites were identified by MS (less than 5 ppm) and tandem MS using the Metlin database47. Data for isocitrate and citrate and for 2-phosphoglycerate and 3-phosphoglycerate are included as cumulative values (iso/citrate and 2/3-phosphoglycerate) because they are not distinguishable through the LC–MS methodology used. The log2 13C enrichment was calculated for the heat maps using the equation:
where M is the area under the curve measured by LC–MS of molecules made up of 12C atoms only, and M + X is the area under the curve measured by LC–MS of molecules with 13C-isotope atoms incorporated into the molecule, X being the number of 13C-isotope atoms incorporated. When multiple biological replicates were available, they were averaged before dividing treatment by control. The untreated control replicates shown are a single representative replicate normalized to the average of all replicates, which helps visualize any variation between controls.
Energy efficiency calculations
Efficiency calculations for electrocatalysis
The following equations were used to determine the efficiency of the electrolysis process. Faradaic efficiency (FE) from gas chromatography was calculated as
where n is the number of electrons transferred, F is Faraday’s constant, x is the mole fraction of the product, V is the total molar flow rate of the gas and jTot is the total current. Liquid Faradaic efficiency was calculated using quantitative 1H-NMR.
Carbon selectivity was calculated as
where ni is the number of electrons transferred to product i, ji is the partial current towards product i and Ci is the number of carbons in product i. This value represents the percentage of CO2 reacted towards C2+ products found in a given product or the molar selectivity of a given product scaled to the number of carbons contained in it.
To determine the overall efficiency of the electrolysis process, we calculated the theoretical energy required to produce the C2+ products and divided that by the actual amount of energy it took to produce those C2+ products. The C2+ product distribution is shown in Supplementary Table 6, where we assume 1 g of the C2+ products.
Theoretical potentials were calculated using the following equation:
where E0 is the theoretical potential, n is the number of electrons and ΔG0 is the Gibbs free energy of reaction.
Theoretical energy was calculated as follows:
where \(E_i^0\) is the theoretical cell potential for species i, mi is the mass produced of product i and MWi is the molecular weight of product i. The values for E0 and m can be found in Supplementary Table 6.
The actual energy input to produce 1 g of C2+ products is calculated using the following equation:
where mAcO is the mass of acetate produced to produce a total of 1 g of C2+ products at the Faradaic efficiencies calculated and listed in Supplementary Table 6, nAcO is the number of moles of electrons passed per mole of acetate produced, ECO is the measured cell potential for the CO electrolyser, MWAcO is the molecular weight of acetate and FEAcO is the measured Faradaic efficiency of acetate. For the CO2 electrolyser:
where NCO is the moles of carbon monoxide necessary to produce a total of 1 g of C2+ products at the 92% (measured) conversion in the CO electrolyser, nCO is the number of electrons passed per mol of carbon monoxide produced, \(E_{{\mathrm{CO}}_2}\) is the measured cell potential for the CO2 electrolyser and FECO is the measured Faradaic efficiency of carbon monoxide.
Using the values listed in Supplementary Table 6, the energetic efficiency was calculated by taking the ratio of the theoretical energy and the actual energy input per 1 g of product:
Our tandem electrolysis process has an energy efficiency of 35.62% for the production of all C2+ products, which accounts for losses due to selectivity, conversion and overpotential. If you consider ethylene as a complete loss and consider only products in the effluent, the energy efficiency of C2+ product production is 24.26%. We next calculated the energy efficiency of the production of just acetate, as this is what is used as the energy and carbon source for algal and yeast growth. To calculate acetate efficiency, we did not include losses due to selectivity because in a commercial setting, the other C2+ products (for example, ethylene) would be considered co-products of the process. The efficiency numbers including ethylene as a co-product were used to calculate the energy efficiency of the whole system. More details are shown below:
Efficiency calculations for food production
The efficiency values reported here are based on the statistical averages of at least three biological replicates. These calculations were conducted similarly to the approaches in Blankenship et al.1 and Nangle et al.48.
Energy efficiency of the heterotrophic cultivation of Chlamydomonas without inputs from biological photosynthesis
The energy efficiency of biological photosynthesis is defined as the energy content of the biomass that can be harvested annually divided by the annual solar irradiance over the same area1. To be able to compare our process to biological photosynthesis, we calculated the conversion efficiency of sunlight to biomass using photovoltaics to power our process of the electrolytic production of acetate followed by the heterotrophic cultivation of Chlamydomonas in the dark. We define the energy efficiency as the increase in biomass energy content divided by the required solar energy input. The increase in biomass energy content is calculated as
where ΔXbiomass is the gain of algal biomass and \(\Delta H_{{\mathrm{biomass}}}^{{\circ}}\) is the enthalpy of combustion of algal biomass, which was determined experimentally using an oxygen bomb calorimeter. The required solar energy input is calculated as
where CAcO is the acetate concentration in the algal media, MWAcO is the molecular weight of acetate, wconsumed is the fraction of acetate consumed by the algae, EAcO is the energy required to generate acetate in the electrolyser (calculated above), ηAM1.5 is the maximum power conversion efficiency (peak solar intensity, AM1.5 spectral distribution) of a commercially available silicon solar cell (from Canadian Solar49,50) and ηannual is the photovoltaic annual efficiency, which is about 95% of the maximum power conversion efficiency value due to the changing solar zenith angle throughout the day and year1. The energy efficiency is then calculated as
Comparison of this number with the ~1% annual efficiency for most crop plants1 shows that our approach can be almost four times more energy efficient than biological photosynthesis for the cultivation of photosynthetic organisms. Improvements in photovoltaic maximum power conversion efficiency would increase the efficiency of our approach. For example, multi-junction solar cells have been shown to reach efficiencies as high as 47.1%49, which coupled to our system would bring the overall energy efficiency of sunlight to food to ~9%.
For Fig. 5 and Extended Data Fig. 8, we calculated the energy efficiencies at each step within the system as follows:
where Yalgae/acetate is the yield of algae grown with acetate effluent media, and \(\Delta H_{\mathrm{AcO}}^{\circ}\) is the enthalpy of combustion of acetate.
Energy efficiency of the cultivation of yeast
Nutritional yeast is heterotrophic and is typically cultivated with glucose derived from biological photosynthesis as the primary carbon and energy source. In our process, electrolysis-derived acetate is substituted for glucose. To compare these two ways to produce yeast, we calculated the amount of yeast that could be produced per area of land in Illinois, a large corn-producing state in the United States. To calculate the amount of yeast that could be produced by our process independent of biological photosynthesis, we used the annual average solar irradiance in Illinois to calculate the electricity that could be generated by photovoltaics to synthesize acetate in the CO2 electrolyser and subsequently used to cultivate yeast, as follows:
where Ee is the average annual solar irradiance from the North America Land Data Assimilation System Daily Sunlight (kJ m−2) dataset for Illinois from 2000 to 201151; ηAM1.5 is the maximum power conversion efficiency (peak solar intensity, AM1.5 spectral distribution) of a commercially available silicon solar cell49; ηannual is the photovoltaic annual efficiency, which is about 95% of the maximum power conversion efficiency value due to the changing solar zenith angle throughout the day and year2; EAcO is the energy required to generate acetate in the electrolyser (calculated above); and Yyeast/acetate is the yield of yeast grown with acetate effluent media.
To calculate the amount of yeast that could be produced per area of land with biological photosynthesis, we used the annual corn harvest data from Illinois to determine the glucose that could be generated by photosynthesis and subsequently used as the primary carbon and energy source to cultivate yeast, as follows:
where \(Y_{{\mathrm{corn}}\;{\mathrm{per}}\;{\mathrm{m}}^2}\) is the average corn kernel production in Illinois per square meter52, Yglucose/corn is the glucose produced from a bushel of corn kernels53 and Yyeast/glucose is the yield of yeast grown with glucose as the carbon source54. Using our artificial photosynthesis approach, almost 18 times more yeast could be produced per area of land.
For Extended Data Fig. 8, we calculated the energy efficiencies at each step within the systems as follows:
where \(\Delta H_{\mathrm{biomass}}^{\circ}\) is the enthalpy of combustion of yeast biomass55, \(\Delta H_{\mathrm{kernel}}^{\circ}\) is the enthalpy of combustion of corn kernels56, wglucose/corn is the mass fraction of glucose found in a corn kernel53 and \(\Delta H_{\mathrm{glucose}}^{\circ}\) is the enthalpy of combustion of glucose.
There are many ways in which the energy of biological photosynthesis can be improved on, such as increasing CO2 concentrations and metabolic engineering. We have included numbers for the theoretical maxima of both systems in Extended Data Fig. 8b (refs. 48,49,50,51,52,53,54,55,56,57,58,59).
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
The metabolomics data (Fig. 4a and Extended Data Figs. 4 and 5b) and all source data can be found at https://doi.org/10.6086/D1VT2V in the Dryad Digital Repository.
References
Blankenship, R. E. et al. Comparing photosynthetic and photovoltaic efficiencies and recognizing the potential for improvement. Science 332, 805–809 (2011).
Ort, D. R. et al. Redesigning photosynthesis to sustainably meet global food and bioenergy demand. Proc. Natl Acad. Sci. USA 112, 8529–8536 (2015).
Kromdijk, J. et al. Improving photosynthesis and crop productivity by accelerating recovery from photoprotection. Science 354, 857–861 (2016).
South, P. F., Cavanagh, A. P., Liu, H. W. & Ort, D. R. Synthetic glycolate metabolism pathways stimulate crop growth and productivity in the field. Science 363, eaat9077 (2019).
Liu, C., Colón, B. C., Ziesack, M., Silver, P. A. & Nocera, D. G. Water splitting–biosynthetic system with CO2 reduction efficiencies exceeding photosynthesis. Science 352, 1210–1213 (2016).
Torella, J. P. et al. Efficient solar-to-fuels production from a hybrid microbial–water-splitting catalyst system. Proc. Natl Acad. Sci. USA 112, 2337–2342 (2015).
Haas, T., Krause, R., Weber, R., Demler, M. & Schmid, G. Technical photosynthesis involving CO2 electrolysis and fermentation. Nat. Catal. 1, 32–39 (2018).
Yishai, O., Lindner, S. N., Gonzalez de la Cruz, J., Tenenboim, H. & Bar-Even, A. The formate bio-economy. Curr. Opin. Chem. Biol. 35, 1–9 (2016).
Cai, T. et al. Cell-free chemoenzymatic starch synthesis from carbon dioxide. Science 373, 1523–1527 (2021).
Woolston, B. M., King, J. R., Reiter, M., Van Hove, B. & Stephanopoulos, G. Improving formaldehyde consumption drives methanol assimilation in engineered E. coli. Nat. Commun. 9, 2387 (2018).
Li, R., Moore, M., Bonham-Smith, P. C. & King, J. Overexpression of formate dehydrogenase in Arabidopsis thaliana resulted in plants tolerant to high concentrations of formate. J. Plant Physiol. 159, 1069–1076 (2002).
Luc, W. et al. Two-dimensional copper nanosheets for electrochemical reduction of carbon monoxide to acetate. Nat. Catal. 2, 423–430 (2019).
Gabardo, C. M. et al. Continuous carbon dioxide electroreduction to concentrated multi-carbon products using a membrane electrode assembly. Joule 3, 2777–2791 (2019).
Lv, J.-J. et al. A highly porous copper electrocatalyst for carbon dioxide reduction. Adv. Mater. 30, e1803111 (2018).
Li, C. W. & Kanan, M. W. CO2 reduction at low overpotential on Cu electrodes resulting from the reduction of thick Cu2O films. J. Am. Chem. Soc. 134, 7231–7234 (2012).
de Arquer, F. P. G. et al. CO2 electrolysis to multicarbon products at activities greater than 1 A cm−2. Science 367, 661–666 (2020).
Jouny, M., Luc, W. & Jiao, F. High-rate electroreduction of carbon monoxide to multi-carbon products. Nat. Catal. 1, 748–755 (2018).
Ripatti, D. S., Veltman, T. R. & Kanan, M. W. Carbon monoxide gas diffusion electrolysis that produces concentrated C2 products with high single-pass conversion. Joule 3, 240–256 (2019).
Weekes, D. M., Salvatore, D. A., Reyes, A., Huang, A. & Berlinguette, C. P. Electrolytic CO2 reduction in a flow cell. Acc. Chem. Res. 51, 910–918 (2018).
Endrődi, B. et al. Multilayer electrolyzer stack converts carbon dioxide to gas products at high pressure with high efficiency. ACS Energy Lett. 4, 1770–1777 (2019).
Ozden, A. et al. Cascade CO2 electroreduction enables efficient carbonate-free production of ethylene. Joule 5, 706–719 (2021).
Liu, Z., Yang, H., Kutz, R. & Masel, R. I. CO2 electrolysis to CO and O2 at high selectivity, stability and efficiency using sustainion membranes. J. Electrochem. Soc. 165, J3371 (2018).
Romero Cuellar, N. S. et al. Two-step electrochemical reduction of CO2 towards multi-carbon products at high current densities. J. CO2 Util. 36, 263–275 (2020).
Murbach, T. S. et al. A toxicological evaluation of Chlamydomonas reinhardtii, a green algae. Int. J. Toxicol. 37, 53–62 (2018).
Kiataramgul, A. et al. An oral delivery system for controlling white spot syndrome virus infection in shrimp using transgenic microalgae. Aquaculture 521, 735022 (2020).
Khemiri, S. et al. Microalgae biomass as an additional ingredient of gluten-free bread: dough rheology, texture quality and nutritional properties. Algal Res. 50, 101998 (2020).
Fields, F. J. et al. Effects of the microalgae Chlamydomonas on gastrointestinal health. J. Funct. Foods 65, 103738 (2020).
Torres-Tiji, Y., Fields, F. J. & Mayfield, S. P. Microalgae as a future food source. Biotechnol. Adv. 41, 107536 (2020).
Zhang, Z. et al. Efficient heterotrophic cultivation of Chlamydomonas reinhardtii. J. Appl. Phycol. 31, 1545–1554 (2019).
Ugalde, U. O. & Castrillo, J. I. in Applied Mycology and Biotechnology (eds Khachatourians, G. G. & Arora, D. K.) Vol. 2, 123–149 (Elsevier, 2002).
Pérez-Torrado, R. et al. Yeast biomass, an optimised product with myriad applications in the food industry. Trends Food Sci. Technol. 46, 167–175 (2015).
Stephan, A., Ahlborn, J., Zajul, M. & Zorn, H. Edible mushroom mycelia of Pleurotus sapidus as novel protein sources in a vegan boiled sausage analog system: functionality and sensory tests in comparison to commercial proteins and meat sausages. Eur. Food Res. Technol. 244, 913–924 (2018).
Brock, M. & Buckel, W. On the mechanism of action of the antifungal agent propionate. Eur. J. Biochem. 271, 3227–3241 (2004).
Wang, P. et al. A 13C isotope labeling method for the measurement of lignin metabolic flux in Arabidopsis stems. Plant Methods 14, 51 (2018).
Dethloff, F., Orf, I. & Kopka, J. Rapid in situ 13C tracing of sucrose utilization in Arabidopsis sink and source leaves. Plant Methods 13, 87 (2017).
Allen, D. K., Bates, P. D. & Tjellström, H. Tracking the metabolic pulse of plant lipid production with isotopic labeling and flux analyses: past, present and future. Prog. Lipid Res. 58, 97–120 (2015).
Melis, A. Solar energy conversion efficiencies in photosynthesis: minimizing the chlorophyll antennae to maximize efficiency. Plant Sci. 177, 272–280 (2009).
Luc, W., Rosen, J. & Jiao, F. An Ir-based anode for a practical CO2 electrolyzer. Catal. Today 288, 79–84 (2017).
Lu, X. & Zhao, C. Electrodeposition of hierarchically structured three-dimensional nickel–iron electrodes for efficient oxygen evolution at high current densities. Nat. Commun. 6, 6616 (2015).
Gorman, D. S. & Levine, R. P. Cytochrome f and plastocyanin: their sequence in the photosynthetic electron transport chain of Chlamydomonas reinhardi. Proc. Natl Acad. Sci. USA 54, 1665–1669 (1965).
Harris, E. H., Stern, D. B. & Witman, G. B. The Chlamydomonas Sourcebook Vol. 2 (Academic Press San Diego, 1989).
Minard, K. I. & McAlister-Henn, L. Redox responses in yeast to acetate as the carbon source. Arch. Biochem. Biophys. 483, 136–143 (2009).
Yachaj. Mushroom cultivation: from falconer to fanaticus and beyond. Entheogen Rev. 10, 127–139 (2001).
Murashige, T. & Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 15, 473–497 (1962).
Gamborg, O. L., Murashige, T., Thorpe, T. A. & Vasil, I. K. Plant tissue culture media. In Vitro 12, 473–478 (1976).
MacLean, B. et al. Skyline: an open source document editor for creating and analyzing targeted proteomics experiments. Bioinformatics 26, 966–968 (2010).
Guijas, C. et al. METLIN: a technology platform for identifying knowns and unknowns. Anal. Chem. 90, 3156–3164 (2018).
Nangle, S. N. et al. Valorization of CO2 through lithoautotrophic production of sustainable chemicals in Cupriavidus necator. Metab. Eng. 62, 207–220 (2020).
Green, M. A. et al. Solar cell efficiency tables (version 55). Prog. Photovoltaics Res. Appl. 28, 3–15 (2020).
Job, E. Canadian Solar Sets a 22.80% Conversion Efficiency World Record for P-Type Large Area Multi-crystalline Silicon Solar Cell (Canadian Solar, 2019); https://investors.canadiansolar.com/news-releases/news-release-details/canadian-solar-sets-2280-conversion-efficiency-world-record-p
North America Land Data Assimilation System (NLDAS) Daily Sunlight (KJ/m2) (1979–2011) Request (CDC WONDER Online Database, 2013); https://wonder.cdc.gov/NASA-INSOLAR.html
Schleusener, M. & Roemer, H. Illinois Agricultural Statistics 2016 Annual Bulletin. (USDA, 2016); https://www.nass.usda.gov/Statistics_by_State/Illinois/Publications/Annual_Statistical_Bulletin/2016/2016-IL_Annual_Bulletin_Entire_Report.pdf
Clifford, C. B. Composition of Corn and Yield of Ethanol from Corn (Penn State Univ., accessed December 10, 2020;) https://www.e-education.psu.edu/egee439/node/672
Verduyn, C., Stouthamer, A. H., Scheffers, W. A. & van Dijken, J. P. A theoretical evaluation of growth yields of yeasts. Antonie Van Leeuwenhoek 59, 49–63 (1991).
NIST Chemistry WebBook, SRD 69 (National Institute of Standards and Technology, 2018); https://www.nist.gov/
Spieser, H. Burning Shelled Corn as a Heating Fuel (Ontario Ministry of Agriculture, Food, and Rural Affairs, 2011); http://www.omafra.gov.on.ca/english/engineer/facts/11-021.htm
Zhu, X.-G., Long, S. P. & Ort, D. R. Improving photosynthetic efficiency for greater yield. Annu. Rev. Plant Biol. 61, 235–261 (2010).
Mongiovi, P. Heat of combustion of S. cerevisiae grown in batch culture on glucose. (BioNumbers, accessed December 10, 2020) https://bionumbers.hms.harvard.edu/bionumber.aspx?&id=101698&ver=0
Standard Weight per Bushel for Agricultural Commodities (Illinois General Assembly, accessed December 10, 2020) https://www.ilga.gov/commission/jcar/admincode/008/00800600ZZ9998bR.html
Waskom, M. seaborn.boxplot—seaborn 0.10.1 documentation. Accessed December 10, 2020; https://seaborn.pydata.org/generated/seaborn.boxplot.html
Acknowledgements
We thank J. Kirkwood (University of California, Riverside (UCR)) and the Institute of Integrative Genome Biology Metabolomics Core Facility at UCR for help with metabolomics analysis; the UCR Plant Transformation Research Center, where all plant experiments were conducted; H. Blanch (UCR) for advice on the efficiency calculations; Y. Li (UCR), S. Xu (UCR) and S. Wu (UCR) for advice and reagents for the yeast experiments; J. Hoover (UCR) for his early efforts towards acetate isolation; C. Mendoza (UCR) for help early on with the algae experiments; and M. Jouny (University of Delaware) for his efforts in developing the early concept of the two-step process. We thank T. Xiang (University of North Carolina, Charlotte), B. Velazquez Benitez (UCR), S. Frey (UCR) and J. Russo (UCR) for providing feedback on the manuscript. The following funding supported this work: Translational Research Institute for Space Health (TRISH) through NASA grant no. NNX16AO69A (R.E.J., E.H., M.H.-D., M.L.O.-C. and A.N.), Foundation for Food & Agriculture Research grant no. FF-NIA20–000000009 (R.E.J.), National Science Foundation grant no. DBI-1922642 (M.H.-D.), a Link Foundation Energy Fellowship (E.H.), Department of Energy grant no. DE-FE0029868 (F.J. and S.O.) and National Science Foundation grant no. CBET-1803200 (F.J. and S.O.). The content of this publication is solely the responsibility of the authors and does not necessarily represent the official views of the Foundation for Food & Agriculture Research (FFAR).
Author information
Authors and Affiliations
Contributions
R.E.J. and F.J. conceived the experiments. S.O. performed the CO2 electrolysis experiments. E.C.H. and D.N.L. performed the algae experiments. E.C.H. performed the yeast experiments. R.E.J. performed the mushroom experiments. M.H.-D., A.F.N. and M.L.O.-C. helped conceive the plant experiments, performed them and analysed the data for them. S.O. calculated the efficiencies for electrocatalysis and R.E.J. and E.C.H. calculated the efficiencies for food production. R.E.J., F.J., S.O., E.C.H. and M.H.-D. analysed the data and wrote the manuscript. All authors edited and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Food thanks Daniel Ducat, Jinlong Gong and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Tandem CO2 electrochemical system for the production of acetate.
a,b, Schematic of the two-step electrolysis system without (a) and with (b) 5 M NaOH scrubber. Oxygen was allowed to vent to the atmosphere from anolytes. Primary products were used as effluents for growth. c, Electrolyser voltage of direct feed CO electrolyser using 2 M KOH and 1 M KHCO3. 1 M KHCO3 was produced in two separate effluents. Electrolyte was changed at 380 minutes, causing the large spike in potential. 0.476 M acetate: 1 M KHCO3 was produced in the first half of the KHCO3 experiment, 0.648 M acetate: 1 M KHCO3 in the second half, and 0.691 M acetate: 1 M KOH was produced in the 2 M KOH experiment. d, Normalized production rate of dissolved CO reduction products collected in the effluent. Liquid product production rate from KHCO3 is cumulative from both experiments, and represents the average over the entire experiment. Data presented represents one experiment for each electrolyte. e, Outlet molar fractions of gas products and feeds from the two-step electrolyser system (left axis). Flow rate (right axis) was measured continuously and is indicated by the arrow. The 5 M NaOH scrubber introduced at 30 minutes completely removed CO2. The CO electrolyser operated from 45 to 345 minutes. The increase in flow rate is attributed to the increase in H2 production over the course of experiment in the CO2 electrolyser. f, Conversion of CO2 and CO during the reaction. The CO2 electrolyser was run without the scrubber at first (grey), a 5 M NaOH scrubber was introduced at 45 minutes (green), the CO electrolyser began operation at 60 minutes (blue). No CO2 was detected on gas chromatogram after the 5 M NaOH scrubber was introduced. CO conversion is based on the average CO flow rate measured before CO electrolyser operation. g, Outlet molar concentrations of gas products and feeds (left axis) for CO2 electrolyser and corresponding Faradaic efficiencies towards CO and H2 (right axis) at varied inlet CO2 flow rates. The arrow indicates the red and black scatter belong to the Faradaic efficiency of the system. h, Electrolyser voltage over the course of the flow rate experiment. Oscillations in cell voltage are due to oscillations in the back pressure controller. Step changes were caused by a decrease in inlet CO2 flow rate.
Extended Data Fig. 2 Chlamydomonas growth varies depending on salinity and acetate:electrolyte ratio of media.
a, The fraction of growth compared to growth in standard acetate media as electrolyte salt concentrations increased. Fraction of growth was calculated as cells per ml of culture grown in effluent media divided by cells per ml of culture grown in acetate positive control media on the last day of growth. Growth was not observed for electrolyte salt concentrations above 80 mM. Media made with effluents that have lower electrolyte concentrations support higher amounts of growth. Standard acetate media is represented as 0 M. Line is a linear regression (R2 = 0.969). b, The same data as in a but plotted against acetate: electrolyte salt ratio. Media made with effluents with higher acetate-to-electrolyte salt ratio support higher amounts of growth. The threshold for growth is between 0.2 and 0.4 acetate-to-electrolyte salt ratio. Standard Tris-acetate-phosphate (TAP) media is arbitrarily set to a ratio of 0.8 since it contains no electrolyte. These graphs do not include growth of Chlamydomonas with the most optimized effluents, as seen in Fig. 3. Chlamydomonas cultures were grown in TAP media with effluent in place of acetate to match the acetate concentration of a typical liquid heterotrophic growth medium (17.5 mM). All media was adjusted to pH 7.2.
Extended Data Fig. 3 Chlamydomonas and mushroom mycelium can grow heterotrophically with electrolyser produced effluent as the sole carbon and energy source.
a, b, c, d, e, Chlamydomonas grown in the dark with electrolyser produced effluents (0.691 M acetate: 1 M KOH, 0.476 M acetate: 1 M KHCO3, and 0.648 M acetate: 1 M KHCO3), acetate, and no acetate. (a) Images taken on day 0 and 4, (b) cell counts, (c) optical density (OD) (750 nm), (d) chlorophyll concentration, (e) and dry weight after 16 days of growth. f, Percentage of acetate in media utilized by Chlamydomonas growth determined by comparing NMR measured acetate concentration of media before and after growth. Cultures were grown in Tris-acetate-phosphate (TAP) media with acetate, without acetate (TP), or with effluent in place of acetate to match the acetate concentration of a typical liquid heterotrophic growth medium (17.5 mM). All media was adjusted to pH 7.2. Each data point represents three biological replicates. Error bars indicate standard deviations. Images are representative of all replicates. g, Mushroom mycelium’s ability to colonize vermiculite substrate soaked with YPD media with no glucose (0 g l-1) and increasing levels of effluent as the carbon source. Full colonization (+++) represents 75% to 100% of substrate colonized, partial colonization (++) represents 15% to 75%, limited colonization (+) represents 1% to 15% of substrate colonized. Effluent added to reach 0.5% (w/w) acetate could support the growth of all fungal species evaluated. h, i, Images of pearl oyster (h) and enokitake (i) mushroom mycelium colonization of a solid vermiculite substrate soaked with YPD media containing glucose (20 g l-1), glucose (20 g l-1) and effluent (0.0691 M acetate: 1 M KOH) added to reach 0.5% (w/w) acetate, or only effluent (0.0691 M acetate: 1 M KOH) in place of glucose (0 g l-1) to reach 0.5% (w/w) acetate as the primary carbon and energy source. Images were taken 24 days post inoculation and are representative of at least 3 replicates. Substrates were fully colonized for all three media. The morphology of the pearl oyster mycelium on top of the effluent containing substrate was different and not as ‘fluffy’ as compared to the glucose containing media. Images are representative of at least 3 replicates. Scale bars: 20 mm.
Extended Data Fig. 4 Heat map of metabolites that had 13C-labeling in undifferentiated lettuce cells (calli).
Heat map of all the replicates of the lettuce callus samples treated with no acetate (n = 3), 2 mM 13C-acetate (n = 3), 5 mM 13C-acetate + 5 mM acetate from KHCO3 effluent (0.648 M acetate: 1 M KHCO3, n = 3), or 5 mM 13C-acetate + 5 mM acetate from KOH effluent (0.691 M acetate: 1 M KOH, n = 4). Number after treatment corresponds to the replicate number. Log2 fold enrichment of 13C between treated and untreated samples, see methods. All samples were grown in the dark showing the ability of plant cells to incorporate 13C from acetate into biomass without light. M+1, 2, 3 or 4 denotes the additional mass of a molecule, which corresponds to the number of carbons in a molecule that are labeled with carbon isotopes.
Extended Data Fig. 5 Heat map of labeled metabolites in crops grown on 13C2-acetate.
a, Representative images showing crop plants grown with 2 mM 13C-acetate 1/2 MS media and control 1/2 MS media. These plants were later used for metabolomic analysis seen in Fig. 4a. b, Heat map of the log2 enrichment of 13C-labeling in all crop replicates compared to the average value of non-treated controls used to create Fig. 4a. The number after each plant name at the top of the figure corresponds to the replicate number. Samples grown without acetate with only one replicate are not included. There is enrichment in all treated crop samples. There are lower levels of 13C-enrichment in green pea relative to other crops. This may be due to a larger reliance for carbon and energy from the endosperm of the seed resulting in less absorption of nutrients and 13C2-acetate from the growth media.
Extended Data Fig. 6 Effect of increasing levels of acetate exposure on lettuce germination and growth (0.0, 0.1, 0.3, 0.6, 1.0, 2.0, 3.0, 6.0, 10 mM).
a, b, Seed germination rate for lettuce after 28 days on (a) agar + 1/2 MS + sucrose + acetate (b) agar + 1/2 MS + acetate. 1/2 MS is a typical plant nutrient mix for growth on agar. Seed germination percent is not significantly different from controls across all treatments (Tukey’s HSD all p-values >0.15). All error bars represent the standard deviation between the germination percentage of the replicates (3 biological replicates with 10 seeds each). Images of lettuce seed germination and growth. c, d, The media was made of (c) 1/2 MS + acetate, and (d) 1/2 MS + sucrose + acetate. (c) 1/2 MS + acetate visually shows the effect of acetate on plant growth in a concentration dependent manner. (d) Agar + 1/2 MS + sucrose + acetate shows that supplementing lettuce with an additional carbon source, sucrose, does not affect the concentration dependent inhibition. All images are representative of all biological replicates for each treatment (3 biological replicates with 10 seeds each). e-l Quantify plant growth measurements: weight, root length, stem height, and leaf count for lettuce plants (~30 individuals shown, and 3 biological replicates visualized in red, grey, and blue for each treatment). e, g, i and k, show agar + 1/2 MS + acetate. There is an acetate concentration dependent inhibition of plant growth that occurs at different concentrations of acetate for different traits, height being the most sensitive and leaf count being the least sensitive. f, h, j and l, show agar + 1/2 MS + sucrose + acetate. Again, there is inhibition of growth at higher acetate concentrations of acetate (2-10 mM). It did not appear that the addition of sucrose had a strong effect on growth, positive or negative, in combination with acetate. The box plots encompasses the quartiles of the dataset and the whiskers capture the rest of the data distribution, except for points that are determined to be “outliers” based on a function of the interquartile range60.
Extended Data Fig. 7 Lettuce plants grown on electrolysis effluent and liquid media with acetate feeding.
a, Representative images of lettuce seeds that were germinated and grown on control 1/2 MS no acetate media, 1/2 MS media supplemented with 1 mM acetate KOH effluent (0.691 M acetate: 1 M KOH), and 1/2 MS media supplemented with 1 mM acetate Bicarbonate effluent (0.648 M acetate: 1 M KHCO3) (3 biological replicates with 10 seeds each with at least 18 total seeds germinating in each treatment). White scale bar is 2 cm. All treatments had 3 biological replicates with 10 seeds each that were grown for 28 days and then growth parameters were measured and photographed. The white scale bar represents 2 cm. b, c, d, e and f, show the leaf number, height, fresh weight, root length, and germination rate, respectively, of lettuce plants germinated and grown on no acetate, 1 mM acetate KOH effluent (0.691 M acetate: 1 M KOH), and 1 mM acetate bicarbonate effluent (0.648 M acetate: 1 M KHCO3). There was no statistical difference in weight between lettuce grown with (n = 21) and without (n = 18) effluent (p-value >0.05, Tukey’s HSD). However there was a significant difference between the 0.691 M acetate: 1 M KOH for root length and height, which could suggest that the potassium salt may cause some level of inhibition. g, Representative images of the acetate feeding experiment performed on lettuce seedling. Lettuce seedlings were germinated for 11 days and then cut at the base of the stem and transferred to water and acetate solutions at acetate concentrations of 0, 0.1, 0.3, 0.6, 1, 3, 6 and 10 mM (n = 9 biological replicates for each treatment). h, i, j and k, show the height, leaf count, length of the roots, and the weight of the plants respectively. Root length was significantly increased in the presence of acetate (p-value <0.001, Tukey’s HSD) (n = 9 for each treatment). Height for 6 and 10 mM is not included due to incompatibility with the method of measuring the height, which was the distance from the top of the stem to the first lateral root, at these higher levels of acetate concentration there was poor root development, which led to artificially inflated height. There is a significant increase in root length in lower levels of acetate concentration (0.1, 1, and 3 mM) based on Tukey’s HSD statistical test, before higher acetate levels start to inhibit root growth. Significant p-values from Tukey’s HSD comparing controls to treatment are denoted by an asterisk, p-value <0.05, double asterisk is a p-value <0.001, N.S. denotes no significant difference from controls (p-value > 0.05). All box plots encompass the quartiles of the dataset and the whiskers capture the rest of the data distribution, except for points that are determined to be “outliers” based on a function of the interquartile range60.
Extended Data Fig. 8 Energy efficiency of food production from artificial photosynthesis compared to biological photosynthesis.
The energy efficiency of sunlight to food production through artificial photosynthesis and biological photosynthesis are compared. Major steps in energy conversion from sunlight (100% solar energy) to food are represented by arrows, with the percentage of remaining energy after each step indicated. The energy efficiency of each step is noted in parentheses. a, Same data as in Fig. 5. The current efficiency numbers for algae production through artificial photosynthesis were determined in this work through experimentation (electricity to acetate and acetate to algae efficiencies). The value for solar to electricity efficiency is based on a commercially available silicon solar cell49,50. The value for biological photosynthetic efficiency was obtained from the literature1. b, Theoretical maximum efficiencies for both biological photosynthesis and artificial photosynthesis are given to show the potential of these approaches. The theoretical maximum efficiency for biological photosynthesis (6%) is for C4 plants (including maize, sugarcane, and sorghum). The theoretical maximum efficiency of biological photosynthesis for C3 plants (including rice, tomato, pepper, and cowpea) is 4.6% (not shown)57. The value for solar to electricity efficiency (47.1%) is from a lab demonstration of a multi-junction solar cell under concentrated illumination49. Continued improvements to electrolysis technology and acetate utilization in plants and algae would increase the overall efficiency of this artificial photosynthetic approach. c, The current efficiency numbers for nutritional yeast production through artificial photosynthesis were determined in this work through experimentation (electricity to acetate and acetate to yeast efficiencies). The value for solar to electricity efficiency is based on a commercially available silicon solar cell49,50. Nutritional yeast is typically grown using glucose derived from plants, such as corn. The efficiency was calculated using values for solar irradiance and corn productivity in Illinois, USA51,52.
Supplementary information
Supplementary Information
Supplementary Tables 1–6, Figs. 1 and 2, and Note.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hann, E.C., Overa, S., Harland-Dunaway, M. et al. A hybrid inorganic–biological artificial photosynthesis system for energy-efficient food production. Nat Food 3, 461–471 (2022). https://doi.org/10.1038/s43016-022-00530-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s43016-022-00530-x
This article is cited by
-
Scanning the active center of formolase to identify key residues for enhanced C1 to C3 bioconversion
Bioresources and Bioprocessing (2024)
-
Solar-driven sugar production directly from CO2 via a customizable electrocatalytic–biocatalytic flow system
Nature Communications (2024)
-
Kilowatt-scale tandem CO2 electrolysis for enhanced acetate and ethylene production
Nature Chemical Engineering (2024)
-
Tandem reactors and reactions for CO2 conversion
Nature Chemical Engineering (2024)
-
Integrated germination related traits and transcriptomic analysis elucidate the potential mechanism of rapeseed under drought stress
Plant Growth Regulation (2024)