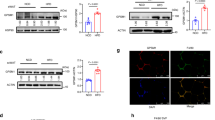
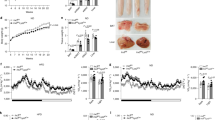
Abstract
Liver macrophages (LMs) have been proposed to contribute to metabolic disease through secretion of inflammatory cytokines. However, anti-inflammatory drugs lead to only modest improvements in systemic metabolism. Here we show that LMs do not undergo a proinflammatory phenotypic switch in obesity-induced insulin resistance in flies, mice and humans. Instead, we find that LMs produce non-inflammatory factors, such as insulin-like growth factor–binding protein 7 (IGFBP7), that directly regulate liver metabolism. IGFBP7 binds to the insulin receptor and induces lipogenesis and gluconeogenesis via activation of extracellular-signal-regulated kinase (ERK) signalling. We further show that IGFBP7 is subject to RNA editing at a higher frequency in insulin-resistant than in insulin-sensitive obese patients (90% versus 30%, respectively), resulting in an IGFBP7 isoform with potentially higher capacity to bind to the insulin receptor. Our study demonstrates that LMs can contribute to insulin resistance independently of their inflammatory status and indicates that non-inflammatory factors produced by macrophages might represent new drug targets for the treatment of metabolic diseases.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding author upon reasonable request. Custom codes used for GSEA are available upon request. All sequence data for this study have been deposited in the Sequence Read Archive (SRA) under accession numbers PRJNA483744 (mouse data) and PRJNA491664 (human data).
Change history
21 December 2022
Editor’s Note: Readers are alerted that the reliability of data presented in this article is currently in question. Appropriate editorial action will be taken once this matter is resolved.
04 April 2019
In the version of this article initially published, author Volker M. Lauschke had affiliation number 13; the correct affiliation number is 12. The error has been corrected in the HTML and PDF versions of the article.
19 January 2021
A Correction to this paper has been published: https://doi.org/10.1038/s42255-021-00343-5
References
Moore, M. C., Coate, K. C., Winnick, J. J., An, Z. & Cherrington, A. D. Regulation of hepatic glucose uptake and storage in vivo. Adv. Nutr.3, 286–294 (2012).
Petersen, M. C., Vatner, D. F. & Shulman, G. I. Regulation of hepatic glucose metabolism in health and disease. Nat. Rev. Endocrinol.13, 572–587 (2017).
Lackey, D. E. & Olefsky, J. M. Regulation of metabolism by the innate immune system. Nat. Rev. Endocrinol.12, 15–28 (2016).
Tilg, H. & Moschen, A. R. Evolution of inflammation in nonalcoholic fatty liver disease: the multiple parallel hits hypothesis. Hepatology52, 1836–1846 (2010).
Jager, J., Aparicio-Vergara, M. & Aouadi, M. Liver innate immune cells and insulin resistance: the multiple facets of Kupffer cells. J. Intern. Med.280, 209–220 (2016).
Esser, N., Paquot, N. & Scheen, A. J. Anti-inflammatory agents to treat or prevent type 2 diabetes, metabolic syndrome and cardiovascular disease. Expert Opin. Investig. Drugs24, 283–307 (2015).
Pollack, R. M., Donath, M. Y., LeRoith, D. & Leibowitz, G. Anti-inflammatory agents in the treatment of diabetes and Its vascular complications. Diabetes Care39(Suppl. 2), S244–S252 (2016).
Everett, B. M. et al. Anti-Inflammatory therapy with canakinumab for the prevention and management of diabetes. J. Am. Coll. Cardiol.71, 2392–2401 (2018).
Chen, L. et al. Selective depletion of hepatic Kupffer cells significantly alleviated hepatosteatosis and intrahepatic inflammation induced by high fat diet. Hepatogastroenterology59, 1208–1212 (2012).
Clementi, A. H., Gaudy, A. M., van Rooijen, N., Pierce, R. H. & Mooney, R. A. Loss of Kupffer cells in diet-induced obesity is associated with increased hepatic steatosis, STAT3 signaling, and further decreases in insulin signaling. Biochim. Biophys. Acta1792, 1062–1072 (2009).
Lanthier, N. et al. Kupffer cell activation is a causal factor for hepatic insulin resistance. Am. J. Physiol. Gastrointest. Liver Physiol.298, G107–G116 (2010).
Papackova, Z. et al. Kupffer cells ameliorate hepatic insulin resistance induced by high-fat diet rich in monounsaturated fatty acids: the evidence for the involvement of alternatively activated macrophages. Nutr. Metab.9, 22 (2012).
Stienstra, R. et al. Kupffer cells promote hepatic steatosis via interleukin-1β-dependent suppression of peroxisome proliferator-activated receptor α activity. Hepatology51, 511–522 (2010).
Figueroa-Clarevega, A. & Bilder, D. Malignant Drosophila tumors interrupt insulin signaling to induce cachexia-like wasting. Dev. Cell33, 47–55 (2015).
Kwon, Y. et al. Systemic organ wasting induced by localized expression of the secreted insulin/IGF antagonist ImpL2. Dev. Cell33, 36–46 (2015).
Evdokimova, V. et al. IGFBP7 binds to the IGF-1 receptor and blocks its activation by insulin-like growth factors. Sci. Signal.5, ra92 (2012).
Kratz, M. et al. Metabolic dysfunction drives a mechanistically distinct proinflammatory phenotype in adipose tissue macrophages. Cell Metab.20, 614–625 (2014).
Hardy, O. T. et al. Body mass index–independent inflammation in omental adipose tissue associated with insulin resistance in morbid obesity. Surg. Obes. Relat. Dis.7, 60–67 (2011).
Haukeland, J. W. et al. Systemic inflammation in nonalcoholic fatty liver disease is characterized by elevated levels of CCL2. J. Hepatol.44, 1167–1174 (2006).
Ahrens, M. et al. DNA methylation analysis in nonalcoholic fatty liver disease suggests distinct disease-specific and remodeling signatures after bariatric surgery. Cell Metab.18, 296–302 (2013).
Horvath, S. et al. Obesity accelerates epigenetic aging of human liver. Proc. Natl Acad. Sci. USA111, 15538–15543 (2014).
Morinaga, H. et al. Characterization of distinct subpopulations of hepatic macrophages in HFD/obese mice. Diabetes64, 1120–1130 (2015).
Aouadi, M. et al. Orally delivered siRNA targeting macrophage Map4k4 suppresses systemic inflammation. Nature458, 1180–1184 (2009).
Tesz, G. J. et al. Glucan particles for selective delivery of siRNA to phagocytic cells in mice. Biochem. J.436, 351–362 (2011).
Tencerova, M. et al. Activated Kupffer cells inhibit insulin sensitivity in obese mice. FASEB J.29, 2959–2969 (2015).
Wan, F. & Lenardo, M. J. The nuclear signaling of NF-κB: current knowledge, new insights, and future perspectives. Cell Res.20, 24–33 (2010).
Fang, B. et al. Circadian enhancers coordinate multiple phases of rhythmic gene transcription in vivo. Cell159, 1140–1152 (2014).
Aparicio-Vergara, M., Tencerova, M., Morgantini, C., Barreby, E. & Aouadi, M. Isolation of Kupffer cells and hepatocytes from a single mouse liver. Methods Mol. Biol.1639, 161–171 (2017).
Huang, W. et al. Depletion of liver Kupffer cells prevents the development of diet-induced hepatic steatosis and insulin resistance. Diabetes59, 347–357 (2010).
Halpern, K. B. et al. Single-cell spatial reconstruction reveals global division of labour in the mammalian liver. Nature542, 352–356 (2017).
Scott, C. L. et al. Bone marrow–derived monocytes give rise to self-renewing and fully differentiated Kupffer cells. Nat. Commun.7, 10321 (2016).
Levanon, E. Y. et al. Evolutionarily conserved human targets of adenosine to inosine RNA editing. Nucleic Acids Res.33, 1162–1168 (2005).
Godfried Sie, C., Hesler, S., Maas, S. & Kuchka, M. IGFBP7’s susceptibility to proteolysis is altered by A-to-I RNA editing of its transcript. FEBS Lett.586, 2313–2317 (2012).
Ahmed, S. et al. Proteolytic processing of IGFBP-related protein-1 (TAF/angiomodulin/mac25) modulates its biological activity. Biochem. Biophys. Res. Commun.310, 612–618 (2003).
Bader, R. et al. The IGFBP7 homolog Imp-L2 promotes insulin signaling in distinct neurons of the Drosophila brain. J. Cell Sci.126, 2571–2576 (2013).
Jiao, P., Feng, B., Li, Y., He, Q. & Xu, H. Hepatic ERK activity plays a role in energy metabolism. Mol. Cell Endocrinol.375, 157–166 (2013).
Han, M. S. et al. JNK expression by macrophages promotes obesity-induced insulin resistance and inflammation. Science339, 218–222 (2013).
Cai, D. et al. Local and systemic insulin resistance resulting from hepatic activation of IKK-β and NF-κB. Nat. Med.11, 183–190 (2005).
Obstfeld, A. E. et al. C-C chemokine receptor 2 (CCR2) regulates the hepatic recruitment of myeloid cells that promote obesity-induced hepatic steatosis. Diabetes59, 916–925 (2010).
Arkan, M. C. et al. IKK-β links inflammation to obesity-induced insulin resistance. Nat. Med.11, 191–198 (2005).
Odegaard, J. I. et al. Alternative M2 activation of Kupffer cells by PPARδ ameliorates obesity-induced insulin resistance. Cell Metab.7, 496–507 (2008).
Lanthier, N. et al. Kupffer cell depletion prevents but has no therapeutic effect on metabolic and inflammatory changes induced by a high-fat diet. FASEB J.25, 4301–4311 (2011).
Honegger, B. et al. Imp-L2, a putative homolog of vertebrate IGF-binding protein 7, counteracts insulin signaling in Drosophila and is essential for starvation resistance. J. Biol.7, 10 (2008).
Ussar, S., Bezy, O., Bluher, M. & Kahn, C. R. Glypican-4 enhances insulin signaling via interaction with the insulin receptor and serves as a novel adipokine. Diabetes61, 2289–2298 (2012).
Boothe, T. et al. Inter-domain tagging implicates caveolin-1 in insulin receptor trafficking and Erk signaling bias in pancreatic beta-cells. Mol. Metab.5, 366–378 (2016).
Belfiore, A., Frasca, F., Pandini, G., Sciacca, L. & Vigneri, R. Insulin receptor isoforms and insulin receptor/insulin-like growth factor receptor hybrids in physiology and disease. Endocr. Rev.30, 586–623 (2009).
Leibiger, B. et al. PI3K-C2α knockdown results in rerouting of insulin signaling and pancreatic beta cell proliferation. Cell Rep.13, 15–22 (2015).
Kumar, S., Bradley, C. L., Mukashyaka, P. & Doerrler, W. T. Identification of essential arginine residues of Escherichiacoli DedA/Tvp38 family membrane proteins YqjA and YghB. FEMS Microbiol. Lett.363, fnw133 (2016).
Dahms, S. O., Hardes, K., Steinmetzer, T. & Than, M. E. X-ray structures of the proprotein convertase furin bound with substrate analogue inhibitors reveal substrate specificity determinants beyond the S4 pocket. Biochemistry57, 925–934 (2018).
Eisenberg, E. & Levanon, E. Y. A-to-I RNA editing—immune protector and transcriptome diversifier. Nat. Rev. Genet.19, 473–490 (2018).
Donath, M. Y. Targeting inflammation in the treatment of type 2 diabetes: time to start. Nat. Rev. Drug Discov.13, 465–476 (2014).
Lee, J. H. et al. Hepatic steatosis index: a simple screening tool reflecting nonalcoholic fatty liver disease. Dig. Liver Dis.42, 503–508 (2010).
Bell, C. C. et al. Transcriptional, functional, and mechanistic comparisons of stem cell–derived hepatocytes, HepaRG cells, and three-dimensional human hepatocyte spheroids as predictive in vitro systems for drug-induced liver injury. Drug Metab. Dispos.45, 419–429 (2017).
Wijkstrom, J. et al. Renal morphology, clinical findings, and progression rate in Mesoamerican nephropathy. Am. J. Kidney Dis.69, 626–636 (2017).
Weibel, E. R. Stereological methods in cell biology: where are we—where are we going? J. Histochem. Cytochem.29, 1043–1052 (1981).
Parini, P., Johansson, L., Broijersen, A., Angelin, B. & Rudling, M. Lipoprotein profiles in plasma and interstitial fluid analyzed with an automated gel-filtration system. Eur. J. Clin. Invest.36, 98–104 (2006).
Tennessen, J. M., Barry, W. E., Cox, J. & Thummel, C. S. Methods for studying metabolism in Drosophila. Methods68, 105–115 (2014).
Schneedorferova, I., Tomcala, A. & Valterova, I. Effect of heat treatment on the n-3/n-6 ratio and content of polyunsaturated fatty acids in fish tissues. Food Chem.176, 205–211 (2015).
Kutsukake, M. et al. Circulating IGF-binding protein 7 (IGFBP7) levels are elevated in patients with endometriosis or undergoing diabetic hemodialysis. Reprod. Biol. Endocrinol.6, 54 (2008).
Kim, D. et al. TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol.14, R36 (2013).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics30, 923–930 (2014).
Quinlan, A. R. & Hall, I. M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics26, 841–842 (2010).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol.15, 550 (2014).
Robinson, M. D. & Oshlack, A. A scaling normalization method for differential expression analysis of RNA-seq data. Genome Biol.11, R25 (2010).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc.57, 289–300 (1995).
Mootha, V. K. et al. PGC-1α-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat. Genet.34, 267 (2003).
Falcon, S. & Gentleman, R. Using GOstats to test gene lists for GO term association. Bioinformatics23, 257–258 (2007).
Barrett, T. et al. NCBI GEO: archive for functional genomics data sets—update. Nucleic Acids Res.41, D991–D995 (2013).
Ritchie, M. E. et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res.43, e47 (2015).
Rosner, B. Fundamental of Biostatistics 7th edn (Brooks/Cole CENGAGE Learning, 2010).
Acknowledgements
We are grateful to R. Harris, S. Craige and B. Craige for their considerable input on the manuscript. We thank T. Dolezal and M. Jindra for helpful suggestions on the Drosophila work. We also thank T. Schröder and R. Kuiper for the tissue histology, M. Taipale and J. Liu for performing sequencing, and A. Krstic for providing flow cytometry support. We thank M. Nordstrand for organising the collection of human obese liver biopsies. This work was supported by research grants from AstraZeneca through the ICMC (M.A.), the Swedish Research Council (M.A.; 2015-03582), the Stockholm County Council (E.N.), the Novo Nordisk Foundation, including the Tripartite Immuno-metabolism Consortium (M.A. and TrIC; NNF15CC0018486), the Strategic Research Program in Diabetes at Karolinska Institutet (M.A. and E.N.), the Diabetes Wellness Foundation Sweden (J.J.) and the Ruth and Richard Julin Foundation (C.M.).
Author information
Authors and Affiliations
Contributions
C.M. carried out most experiments. G.K. and A.B. performed and analysed the data from the Drosophila experiments. J.J., L.L., V.A., E.B., A.S., C.X., M.T., C.K. and F.V. helped with experiments and interpretation of the data. S.S. measured circulating lipid levels in mice. A.T. measured lipids in Drosophila. K.H. generated all electron microscopy images and measurements. N.K.B. and E.E. provided biopsies and liver cells from lean individuals. E.N. provided liver biopsies from obese human subjects. M.R. provided serum from obese and lean volunteers. T.H. and V.M.L. performed experiments in human liver spheroids. X.L. and C.K. performed the bioinformatics analysis and interpretation. J.B. contributed to the design of experiments related to IGFBP7 signalling. C.M. and M.A. conceived the project, analysed data and wrote the manuscript with input from all co-authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–8 and Supplementary Tables 1–8
Supplementary Data 1
Significantly regulated pathways in human LMs isolated from obese as compared to lean patients. GSEA depicting significantly up- or downregulated pathways (FDR < 0.05) in human LMs according to MES.
Supplementary Data 2
Significantly regulated pathways in LMs isolated from HFD-fed mice as compared to ND-fed mice. GSEA depicting significantly up- or downregulated pathways (FDR < 0.05) in mouse LMs according to MES.
Supplementary Data 3
Obesity-regulated genes in mouse LMs and in mouse whole liver. List of regulated genes in LMs and in whole liver from mice fed a HFD as compared to mice fed a ND. Data include gene name, log2 fold change (log2FC), P value and FDR.
Supplementary Data 4
Highly expressed and upregulated genes in LMs isolated from HFD-fed mice as compared to ND-fed mice. List of upregulated genes in LMs isolated from HFD-fed as compared to ND-fed mice with median RPKM > 50. Data include gene name, log2 fold change (log2FC), P value and FDR.
Supplementary Data 5
Gene expression regulated by obesity and insulin resistance in human LMs. List of regulated genes in LMs isolated from humans: obese as compared to lean patients and obese insulin-resistant as compared to obese insulin-sensitive patients. Data include gene name, log2 fold change (log2FC), P value and FDR.
Supplementary Data 6
Gene expression regulated in mouse LMs following Igfbp7 silencing. List of regulated genes in LMs isolated from mice treated with GeRP-Scr versus GeRP-Igfbp7. Data includes gene name, log2 fold change (log2FC), P value and FDR.
Supplementary Data 7
Gene ontology enrichment analysis of inversely regulated genes in LMs from HFD- versus ND-fed mice and mice treated with GeRP-Igfbp7 versus GeRP-Scr. Complete list of inversely regulated pathways in mice fed a HFD versus ND as compared to mice treated with GeRP-Igfbp7 versus GeRP-Scr.
Supplementary Data 8
Genes regulated in mouse hepatocytes following Igfbp7 silencing in LMs. List of regulated genes in hepatocytes isolated from mice treated with GeRP-Scr versus GeRP-Igfbp7. Data include gene name, log2 fold change (log2FC), P value and FDR.
Supplementary Data 9
Significantly regulated pathways in mouse hepatocytes following Igfbp7 silencing in LMs. GSEA depicting significantly up- or downregulated pathways (FDR < 0.05) in human and mouse LMs according to MES.
Rights and permissions
About this article
Cite this article
Morgantini, C., Jager, J., Li, X. et al. Liver macrophages regulate systemic metabolism through non-inflammatory factors. Nat Metab 1, 445–459 (2019). https://doi.org/10.1038/s42255-019-0044-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-019-0044-9
This article is cited by
-
Tissue-specific macrophages: how they develop and choreograph tissue biology
Nature Reviews Immunology (2023)
-
Human resident liver myeloid cells protect against metabolic stress in obesity
Nature Metabolism (2023)
-
Insulin-like growth factor-binding protein-7 (IGFBP7) links senescence to heart failure
Nature Cardiovascular Research (2022)
-
Macrophage functional diversity in NAFLD — more than inflammation
Nature Reviews Endocrinology (2022)
-
Obesity — no role for pro-inflammatory liver macrophages
Nature Reviews Endocrinology (2019)