Abstract
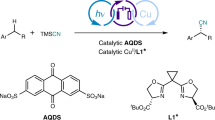
The efficient generation of high structural complexity, which correlates with the number of stereocentres, is an important objective in organic synthesis. Ideally, from a perspective of economy and sustainability, the conversion should include the direct functionalization of unactivated C(sp3)–H bonds. Here we introduce a methodology that enables the generation of complex cyclobutanes with up to four consecutive stereocentres, including all-carbon quaternary stereocentres, from a direct reaction of C–C single bonds with C=C double bonds. The asymmetric photoelectrocatalysis combines photocatalysis, electrochemical redox catalysis and asymmetric catalysis. It avoids the use of chemical oxidants, exhibits excellent enantioselectivity and diastereoselectivity, reveals high functional group compatibility, and also succeeds in the simultaneous conversion of two C(sp3)–H bonds into consecutive carbon stereocentres. This work demonstrates the power of combining electrochemistry with photochemistry and asymmetric catalysis to generate complex structures in an economic and sustainable fashion.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the paper and its Supplementary Information, or from the authors on reasonable request. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre under deposition number CCDC 2258577 (11) and CCDC 2258578 (28). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/.
References
Kobayashi, S. & Jørgensen, K. A. Cycloaddition Reactions in Organic Synthesis (Wiley‐VCH, 2002).
Nishiwaki, N. Methods and Applications of Cycloaddition Reactions in Organic Syntheses (Wiley‐VCH, 2014).
Schreiber, S. L. [2 + 2] photocycloadditions in the synthesis of chiral molecules. Science 227, 857–863 (1985).
Crimmins, M. T. Synthetic applications of intramolecular enone–olefin photocycloadditions. Chem. Rev. 88, 1453–1473 (1988).
Schuster, D. I., Lem, G. & Kaprinidis, N. A. New insights into an old mechanism: [2 + 2] photocycloaddition of enones to alkenes. Chem. Rev. 93, 3–22 (1993).
Xu, Y., Conner, M. L. & Brown, M. K. Cyclobutane and cyclobutene synthesis: catalytic enantioselective [2 + 2] cycloadditions. Angew. Chem. Int. Ed. 54, 11918–11928 (2015).
Poplata, S., Tröster, A., Zou, Y.-Q. & Bach, T. Recent advances in the synthesis of cyclobutanes by olefin [2 + 2] photocycloaddition reactions. Chem. Rev. 116, 9748–9815 (2016).
Sarkar, D., Bera, N. & Ghosh, S. [2 + 2] photochemical cycloaddition in organic synthesis. Eur. J. Org. Chem. 2020, 1310–1326 (2020).
Sicignano, M., Rodríguez, R. I. & Alemán, J. Recent visible light and metal free strategies in [2 + 2] and [4 + 2] photocycloadditions. Eur. J. Org. Chem. 2021, 3303–3321 (2021).
Yang, P., Jia, Q., Song, S. & Huang, X. [2+2]-Cycloaddition-derived cyclobutane natural products: structural diversity, sources, bioactivities, and biomimetic syntheses. Nat. Prod. Rep. 40, 1094–1129 (2023).
Cox, B., Booker-Milburn, K. I., Elliott, L. D., Robertson-Ralph, M. & Zdorichenko, V. Escaping from flatland: [2 + 2] photocycloaddition; conformationally constrained sp3-rich scaffolds for lead generation. ACS Med. Chem. Lett. 10, 1512–1517 (2019).
van der Kolk, M. R., Janssen, M., Rutjes, F. & Blanco-Ania, D. Cyclobutanes in small-molecule drug candidates. ChemMedChem 17, e202200020 (2022).
Winkler, J. D., Bowen, C. M. & Liotta, F. [2 + 2] photocycloaddition/fragmentation strategies for the synthesis of natural and unnatural products. Chem. Rev. 95, 2003–2020 (1995).
Murakami, M. & Ishida, N. Cleavage of carbon–carbon σ-bonds of four-membered rings. Chem. Rev. 121, 264–299 (2021).
Kleinmans, R. et al. Intermolecular [2π + 2σ]-photocycloaddition enabled by triplet energy transfer. Nature 605, 477–482 (2022).
Guo, R. et al. Strain-release [2π + 2σ] cycloadditions for the synthesis of bicyclo[2.1.1]hexanes initiated by energy transfer. J. Am. Chem. Soc. 144, 7988–7994 (2022).
Huang, H. et al. Electrophotocatalysis with a trisaminocyclopropenium radical dication. Angew. Chem. Int. Ed. 58, 13318–13322 (2019).
Wang, F. & Stahl, S. S. Merging photochemistry with electrochemistry: functional-group tolerant electrochemical amination of C(sp3)−H bonds. Angew. Chem. Int. Ed. 58, 6385–6390 (2019).
Yan, H., Hou, Z.-W. & Xu, H.-C. Photoelectrochemical C−H alkylation of heteroarenes with organotrifluoroborates. Angew. Chem. Int. Ed. 58, 4592–4595 (2019).
Zhang, L. et al. Photoelectrocatalytic arene C–H amination. Nat. Catal. 2, 366–373 (2019).
Moutet, J.-C. & Reverdy, G. Photochemistry of cation radicals in solution: photoinduced oxidation by the phenothiazine cation radical. Tetrahedron Lett. 20, 2389–2392 (1979).
Moutet, J.-C. & Reverdy, G. Phototochemistry of cation radicals in solution: photoinduced electron-transfer reactions between alcholos and the N,N,N’,N’-tetraphenyl-p-phenylenediamine cation radical. Chem. Commun. 654–655 (1982).
Barham, J. P. & König, B. Synthetic photoelectrochemistry. Angew. Chem. Int. Ed. 59, 11732–11747 (2020).
Liu, J., Lu, L., Wood, D. & Lin, S. New redox strategies in organic synthesis by means of electrochemistry and photochemistry. ACS Cent. Sci. 6, 1317–1340 (2020).
Huang, H., Steiniger, K. A. & Lambert, T. H. Electrophotocatalysis: combining light and electricity to catalyze reactions. J. Am. Chem. Soc. 144, 12567–12583 (2022).
Wu, S., Kaur, J., Karl, T. A., Tian, X. & Barham, J. P. Synthetic molecular photoelectrochemistry: new frontiers in synthetic applications, mechanistic insights and scalability. Angew. Chem. Int. Ed. 61, e202107811 (2022).
Yan, M., Kawamata, Y. & Baran, P. S. Synthetic organic electrochemical methods since 2000: on the verge of a renaissance. Chem. Rev. 117, 13230–13319 (2017).
Meyer, T. H., Choi, I., Tian, C. & Ackermann, L. Powering the future: how can electrochemistry make a difference in organic synthesis? Chem 6, 2484–2496 (2020).
Prier, C. K., Rankic, D. A. & MacMillan, D. W. C. Visible light photoredox catalysis with transition metal complexes: applications in organic synthesis. Chem. Rev. 113, 5322–5363 (2013).
Stephenson, C., Yoon, T. & MacMillan, D. W. C. Visible Light Photocatalysis in Organic Chemistry (Wiley‐VCH, 2018).
Cai, C.-Y. et al. Photoelectrochemical asymmetric catalysis enables site- and enantioselective cyanation of benzylic C–H bonds. Nat. Catal. 5, 943–951 (2022).
Fan, W. et al. Electrophotocatalytic decoupled radical relay enables highly efficient and enantioselective benzylic C–H functionalization. J. Am. Chem. Soc. 144, 21674–21682 (2022).
Huang, X. et al. Direct visible-light-excited asymmetric Lewis acid catalysis of intermolecular [2 + 2] photocycloadditions. J. Am. Chem. Soc. 139, 9120–9123 (2017).
Huang, X. & Meggers, E. Asymmetric photocatalysis with bis-cyclometalated rhodium complexes. Acc. Chem. Res. 52, 833–847 (2019).
Huang, X., Zhang, Q., Lin, J., Harms, K. & Meggers, E. Electricity-driven asymmetric Lewis acid catalysis. Nat. Catal. 2, 34–40 (2019).
Xiong, P., Hemming, M., Ivlev, S. I. & Meggers, E. Electrochemical enantioselective nucleophilic α-C(sp3)–H alkenylation of 2-acyl imidazoles. J. Am. Chem. Soc. 144, 6964–6971 (2022).
Liang, K., Zhang, Q. & Guo, C. Nickel-catalyzed switchable asymmetric electrochemical functionalization of alkenes. Sci. Adv. 8, eadd7134 (2022).
Jie, X., Shang, Y., Zhang, X. & Su, W. Cu-catalyzed sequential dehydrogenation–conjugate addition for β-functionalization of saturated ketones: scope and mechanism. J. Am. Chem. Soc. 138, 5623–5633 (2016).
Li, J., He, L., Liu, X., Cheng, X. & Li, G. Electrochemical hydrogenation with gaseous ammonia. Angew. Chem. Int. Ed. 58, 1759–1763 (2018).
Huang, B., Li, Y., Yang, C. & Xia, W. Electrochemical 1,4-reduction of α,β-unsaturated ketones with methanol and ammonium chloride as hydrogen sources. Chem. Commun. 55, 6731–6734 (2019).
Nguyen, T. H. et al. Three quinolinone alkaloid–phenylpropanoid adducts from Melicope pteleifolia. Nat. Prod. Res. 36, 3858–3864 (2022).
Schmittel, M. & Burghart, A. Understanding reactivity patterns of radical cations. Angew. Chem. Int. Ed. 36, 2550–2589 (1997).
Ohmatsu, K., Nakashima, T., Sato, M. & Ooi, T. Direct allylic C–H alkylation of enol silyl ethers enabled by photoredox–Brønsted base hybrid catalysis. Nat. Commun. 10, 2706 (2019).
Nakashima, T., Fujimori, H., Ohmatsu, K. & Ooi, T. Exploiting transient radical cations as Brønsted acids for allylic C–H heteroarylation of enol silyl ethers. Chem. Eur. J. 27, 9253–9256 (2021).
Shono, T., Matsumura, Y. & Nakagawa, Y. Electroorganic chemistry. XII. Anodic oxidation of enol esters. J. Am. Chem. Soc. 96, 3532–3536 (1974).
Shono, T., Okawa, M. & Nishiguchi, I. Electroorganic chemistry. XXI. Selective formation of α-acetoxy ketones and general synthesis of 2,3-disubstituted 2-cyclopentenones through the anodic oxidation of enol acetates. J. Am. Chem. Soc. 97, 6144–6147 (1975).
Wilsey, S., González, L., Robb, M. A. & Houk, K. N. Ground- and excited-state surfaces for the [2 + 2]-photocycloaddition of α,β-enones to alkenes. J. Am. Chem. Soc. 122, 5866–5876 (2000).
Chiba, K., Miura, T., Kim, S., Kitano, Y. & Tada, M. Electrocatalytic intermolecular olefin cross-coupling by anodically induced formal [2 + 2] cycloaddition between enol ethers and alkenes. J. Am. Chem. Soc. 123, 11314–11315 (2001).
Miura, T., Kim, S., Kitano, Y., Tada, M. & Chiba, K. Electrochemical enol ether/olefin cross-metathesis in a lithium perchlorate/nitromethane electrolyte solution. Angew. Chem. Ed. 45, 1461–1463 (2006).
Elgrishi, N. et al. A practical beginner’s guide to cyclic voltammetry. J. Chem. Educ. 95, 197–206 (2018).
McKenzie, E. C. R. et al. Versatile tools for understanding electrosynthetic mechanisms. Chem. Rev. 122, 3229–3335 (2022).
Gnaim, S. et al. Electrochemically driven desaturation of carbonyl compounds. Nat. Chem. 13, 367–372 (2021).
Jung, H. et al. Understanding the mechanism of direct visible-light-activated [2+2] cycloadditions mediated by Rh and Ir photocatalysts: combined computational and spectroscopic studies. Chem. Sci. 12, 9673–9681 (2021).
Acknowledgements
We are grateful for funding from the Deutsche Forschungsgemeinschaft (ME 1805/17-1 for E.M.) and to X. Huang for his early related explorations. P.X. is grateful for a postdoctoral fellowship from the Alexander von Humboldt Foundation.
Author information
Authors and Affiliations
Contributions
E.M. and P.X. wrote the manuscript. E.M. and P.X. conceived of the project and devised the synthetic experiments. P.X. performed the synthetic experiments and analysed the data. S.I. collected the crystallographic data and solved and refined the X-ray crystal structures.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Qingquan Lu, Maurizio Benaglia and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–68, Tables 1–5, experimental procedures and characterization data, HPLC and NMR spectra.
Supplementary Data 1
CIF file of the crystal structure of compound 11.
Supplementary Data 2
CIF file of the crystal structure of compound 28.
Supplementary Data 3
Checkcif file of the crystal structure of compound 11.
Supplementary Data 4
Checkcif file of the crystal structure of compound 28.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xiong, P., Ivlev, S.I. & Meggers, E. Photoelectrochemical asymmetric dehydrogenative [2 + 2] cycloaddition between C–C single and double bonds via the activation of two C(sp3)–H bonds. Nat Catal 6, 1186–1193 (2023). https://doi.org/10.1038/s41929-023-01050-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-023-01050-y
This article is cited by
-
Cycloaddition with asymmetric photoelectrocatalysis
Nature Catalysis (2023)