Abstract
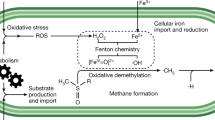
The enzymatic oxidation of methane to methanol was discovered in methanotrophs over 110 years ago. Nevertheless, the mechanism of action of particulate methane monooxygenase (pMMO) remains elusive, especially regarding O2 activation and the nature of the active species of the enzyme. Here we decipher the catalytic cycle of pMMO in the presence of the physiological reductant duroquinol (DQH2). We demonstrate that O2 activation is in fact initiated by a CuC(ii)–DQH− species generated by deprotonation of DQH2. Our simulations capture the exclusive pathway for the sequential formation of the intermediates, CuC(ii)−O2•−, CuC(ii)−OOH− and H2O2, along the O2 reduction pathway. Furthermore, H2O2 activation by CuC(ii)−DQH− is initiated by dissociation of DQH• to yield CuC(i), followed by CuC(i)-catalysed O−O homolysis, en route to the formation of the CuC(ii)−O•− species, which is responsible for C−H oxidations. These findings uncover the important roles of the phenol co-substrate for O2 activation and help resolve the enigmatic mechanism of pMMO.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The authors declare that all the data supporting the findings of this work are available within the article and its Supplementary Information, Supplementary Data or from the corresponding authors upon request.
References
Ross, M. O. & Rosenzweig, A. C. A tale of two methane monooxygenases. J. Biol. Inorg. Chem. 22, 307–319 (2017).
Jin, Z. et al. Hydrophobic zeolite modification for in situ peroxide formation in methane oxidation to methanol. Science 367, 193–197 (2020).
Newton, M., Knorpp, A. J., Sushkevich, V., Palagin, D. & van Bokhoven, J. A. Active sites and mechanisms in the direct conversion of methane to methanol using Cu in zeolitic hosts: a critical examination. Chem. Soc. Rev. 49, 1449–1486 (2020).
Schwarz, H., Shaik, S. & Li, J. Electronic effect on room-temperature, gas-phase C-H activation by cluster oxides and metal carbides: the methane challenge. J. Am. Chem. Soc. 139, 17201–17212 (2017).
Hakemian, A. S. & Rosenzweig, A. C. The biochemistry of methane oxidation. Annu. Rev. Biochem. 76, 223–241 (2007).
Sirajuddin, S. & Rosenzweig, A. C. Enzymatic oxidation of methane. Biochemistry 54, 2283–2294 (2015).
Sazinsky, M. H. & Lippard, S. J. Methane monooxygenase: functionalizing methane at iron and copper. Met. Ions Life Sci. 15, 205–256 (2015).
Tol, R. S., Heintz, R. J. & Lammers, P. E. M. Methane emission reduction: an application of FUND. Clim. Change 57, 71–98 (2003).
Chan, S. I. & Yu, S. S. F. Copper protein constructs for methane oxidation. Nat. Catal. 2, 286–287 (2019).
Kim, H. J. et al. Biological conversion of methane to methanol through genetic reassembly of native catalytic domains. Nat. Catal. 2, 342–353 (2019).
Murrell, J. C., Gilbert, B. & McDonald, I. R. Molecular biology and regulation of methane monooxygenase. Arch. Microbiol. 173, 325–332 (2000).
Banerjee, R., Jones, J. C. & Lipscomb, J. D. Soluble methane monooxygenase. Annu. Rev. Biochem. 88, 409–431 (2019).
Baik, M. H., Newcomb, M., Friesner, R. A. & Lippard, S. J. Mechanistic studies on the hydroxylation of methane by methane monooxygenase. Chem. Rev. 103, 2385–2419 (2003).
Banerjee, R., Proshlyakov, Y., Lipscomb, J. D. & Proshlyakov, D. A. Structure of the key species in the enzymatic oxidation of methane to methanol. Nature 518, 431–434 (2015).
Christine, E. & Tinberg, S. J. L. Dioxygen activation in soluble methane monooxygenase. Acc. Chem. Res. 44, 280–288 (2011).
Jasniewski, A. J. & Que, L. Dioxygen activation by nonheme diiron enzymes: diverse dioxygen adducts, high-valent intermediates, and related model complexes. Chem. Rev. 118, 2554–2592 (2018).
Sirajuddin, S. et al. Effects of zinc on particulate methane monooxygenase activity and structure. J. Biol. Chem. 289, 21782–21794 (2014).
Lieberman, R. L. & Rosenzweig, A. C. Crystal structure of a membrane-bound metalloenzyme that catalyses the biological oxidation of methane. Nature 434, 177–182 (2005).
Smith, S. M. et al. Crystal structure and characterization of particulate methane monooxygenase from Methylocystis species strain M. Biochemistry 50, 10231–10240 (2011).
Ro, S. Y. et al. From micelles to bicelles: effect of the membrane on particulate methane monooxygenase activity. J. Biol. Chem. 293, 10457–10465 (2018).
Chan, S. I., Lee, S. J. & Lee, E. Y. in Lecture Notes in Microbiology Monographs Vol. 32, 71−120 (Springer, 2019).
Chan, S. I. et al. Efficient oxidation of methane to methanol by dioxygen mediated by tricopper clusters. Angew. Chem. Int. Ed. 52, 3731–3735 (2013).
Wang, V. C. C. et al. Alkane oxidation: methane monooxygenases, related enzymes, and their biomimetics. Chem. Rev. 117, 8574–8621 (2017).
Hakemian, A. S. et al. The metal centers of particulate methane monooxygenase from Methylosinus trichosporium OB3b. Biochemistry 47, 6793–6801 (2008).
Culpepper, M. A. & Rosenzweig, A. C. Architecture and active site of particulate methane monooxygenase. Crit. Rev. Biochem. Mol. Biol. 47, 483–492 (2012).
Ro, S. Y. et al. Native top-down mass spectrometry provides insights into the copper centers of membrane bound methane monooxygenase. Nat. Commun. 10, 2675 (2019).
Ross, M. O. et al. Particulate methane monooxygenase contains only mononuclear copper centers. Science 364, 566–570 (2019).
Lieberman, R. L. et al. Purified particulate methane monooxygenase from Methylococcus capsulatus (Bath) is a dimer with both mononuclear copper and a copper-containing cluster. Proc. Natl Acad. Sci. USA 100, 3820–3825 (2003).
Balasubramanian, R. et al. Oxidation of methane by a biological dicopper centre. Nature 465, 115–119 (2010).
Cao, L., Caldararu, O., Rosenzweig, A. C. & Ryde, U. Quantum refinement does not support dinuclear copper sites in crystal structures of particulate methane monooxygenase. Agnew. Chem. Int. Ed. 57, 162–166 (2018).
Ciano, L., Davies, G. J., Tolman, W. B. & Walton, P. H. Bracing copper for the catalytic oxidation of C−H bonds. Nat. Catal. 1, 571–577 (2018).
Balasubramanian, R. & Rosenzweig, A. C. Structural and mechanistic insights into methane oxidation by particulate methane monooxygenase. Acc. Chem. Res. 40, 573–580 (2007).
Rosenzweig, A. C. & Sazinsky, M. H. Structural insights into dioxygen-activating copper enzymes. Curr. Opin. Struct. Biol. 16, 729–735 (2006).
Culpepper, M. A., Cutsail, G. E.III, Gunderson, W. A., Hoffman, B. M. & Rosenzweig, A. C. Identification of the valence and coordination environment of the particulate methane monooxygenase copper centers by advanced EPR characterization. J. Am. Chem. Soc. 136, 11767–11775 (2014).
Liew, E. F., Tong, D., Coleman, N. V. & Holmes, A. J. Mutagenesis of the hydrocarbon monooxygenase indicates a metal centre in subunit-C, and not subunit-B, is essential for copper-containing membrane monooxygenase activity. Microbiology 160, 1267–1277 (2014).
Shiota, Y. & Yoshizawa, K. Comparison of the reactivity of Bis(μ-oxo)CuIICuIII and CuIIICuIII species to methane. Inorg. Chem. 48, 838–845 (2009).
Miyanishi, M., Abe, T., Hori, Y., Shiota, Y. & Yoshizawa, K. Role of amino acid residues for dioxygen activation in the second coordination sphere of the dicopper site of pMMO. Inorg. Chem. 58, 12280–12288 (2019).
Shiota, Y., Juhász, G. & Yoshizawa, K. Role of tyrosine residue in methane activation at the dicopper site of particulate methane monooxygenase: a density functional theory study. Inorg. Chem. 52, 7907–7917 (2013).
Mahyuddin, M. H., Shiota, Y., Staykov, A. & Yoshizawa, K. Theoretical overview of methane hydroxylation by copper-oxygen species in enzymatic and zeolitic catalysts. Acc. Chem. Res. 51, 2382–2390 (2018).
Shiota, K. Y. Y. Conversion of methane to methanol at the mononuclear and dinuclear copper sites of particulate methane monooxygenase (pMMO): a DFT and QM/MM study. J. Am. Chem. Soc. 128, 9873–9881 (2006).
Liu, Y.F. & Du, L. Theoretical study of the oxidation of methane to methanol by the [CuIICuII(μ-O)2CuIII(7‑N‑Etppz)]1+ complex. Inorg. Chem. 57, 3261–3271 (2018).
Morris, R. H. Methane activation by a single copper center in particulate methane monooxygenase: a computational study. Inorg. Chim. Acta 503, 119441 (2020).
Shiemke, A. K., Cook, S. A., Miley, T. & Singleton, P. Detergent solubilization of membrane bound methane monooxygenase requires plastoquinol analogs as electron donors. Arch. Biochem. Biophys. 321, 421–428 (1995).
Cook, S. A. & Shiemke, A. K. Evidence that a type-2 NADH: quinone oxidoreductase mediates electron transfer to particulate methane monooxygenase in methylococcus capsulatus. Arch. Biochem. Biophys. 398, 32–40 (2002).
Solomon, E. I. et al. Copper active sites in biology. Chem. Rev. 114, 3659–3853 (2014).
Labourel, A. et al. A fungal family of lytic polysaccharide monooxygenase-like copper proteins. Nat. Chem. Biol. 16, 345–350 (2020).
Garcia-Santamarina, S. et al. A lytic polysaccharide monooxygenase-like protein functions in fungal copper import and meningitis. Nat. Chem. Biol. 16, 337–344 (2020).
Adelson, C. N. et al. Characterization of the preprocessed copper site equilibrium in amine oxidase and assignment of the reactive copper site in topaquinone biogenesis. J. Am. Chem. Soc. 141, 8877–8890 (2019).
Wu, P. et al. Theory demonstrated a “coupled” mechanism for O2 activation and substrate hydroxylation by binuclear copper monooxygenases. J. Am. Chem. Soc. 141, 19776–19789 (2019).
Bailey, W. D., Dhar, D., Cramblitt, A. C. & Tolman, W. B. Mechanistic dichotomy in proton-coupled electron-transfer reactions of phenols with a copper superoxide complex. J. Am. Chem. Soc. 141, 5470–5480 (2019).
Lawton, T. J., Ham, J., Sun, T. & Rosenzweig, A. C. Structural conservation of the B subunit in the ammonia monooxygenase/particulate methane monooxygenase superfamily. Proteins 82, 2263–2267 (2014).
Wang, B., Walton, P. H. & Rovira, C. Molecular mechanisms of oxygen activation and hydrogen peroxide formation in lytic polysaccharide monooxygenases. ACS Catal. 9, 4958–4969 (2019).
Jones, S. M., Transue, W. J., Meier, K. K., Kelemen, B. & Solomon, E. I. Kinetic analysis of amino acid radicals formed in H2O2-driven CuI LPMO reoxidation implicates dominant homolytic reactivity. Proc. Natl Acad. Sci. USA 45, 8372–8381 (2020).
Wang, B. et al. QM/MM studies into the H2O2‑dependent activity of lytic polysaccharide monooxygenases: evidence for the formation of a caged hydroxyl radical intermediate. ACS Catal. 8, 1346–1351 (2018).
Miyaji, A., Suzukib, M., Baba, T., Kamachi, T. & Okura, I. Hydrogen peroxide as an effecter on the inactivation of particulate methane monooxygenase under aerobic conditions. J. Mol. Catal. B 57, 211–215 (2009).
Bissaro, B. et al. Oxidative cleavage of polysaccharides by monocopper enzymes depends on H2O2. Nat. Chem. Biol. 13, 1123–1128 (2017).
Paradisi, A. et al. Formation of a copper(ii)–tyrosyl complex at the active site of lytic polysaccharide monooxygenases following oxidation by H2O2. J. Am. Chem. Soc. 141, 18585–18599 (2019).
Op den Camp, H. J. M. et al. Environmental, genomic and taxonomic perspectives on methanotrophic Verrucomicrobia. Env. Microbiol. Rep. 1, 293–306 (2009).
CP2K v. 4.1 (CP2K Developers Group, 2016); https://www.cp2k.org/
VandeVondele, J. et al. QUICKSTEP: fast and accurate density functional calculations using a mixed Gaussian and plane waves approach. Comput. Phys. Commun. 167, 103–128 (2005).
Laino, T., Mohamed, F., Laio, A. & Parrinello, M. An efficient real space multigrid QM/MM electrostatic coupling. J. Chem. Theory Comput. 1, 1176–1184 (2005).
Laio, A., VandeVondele, J. & Rothlisberger, U. A Hamiltonian electrostatic coupling scheme for hybrid Car–Parrinello molecular dynamics simulations. J. Chem. Phys. 116, 6941–6947 (2002).
VandeVondele, J. & Hutter, J. Gaussian basis sets for accurate calculations on molecular systems in gas and condensed phases. J. Chem. Phys. 127, 114105 (2007).
Goedecker, S., Teter, M. & Hutter, J. Separable dual-space Gaussian pseudopotentials. Phys. Rev. B 54, 1703–1710 (1996).
Hartwigsen, C., Goedecker, S. & Hutter, J. Relativistic separable dual-space Gaussian pseudopotentials from H to Rn. Phys. Rev. B 58, 3641–3662 (1998).
Guidon, M., Hutter, J. & VandeVondele, J. Auxiliary density matrix methods for Hartree−Fock exchange calculations. J. Chem. Theory Comput. 6, 2348–2364 (2010).
Laio, A. & Parrinello, M. Escaping free-energy minima. Proc. Natl Acad. Sci. USA 99, 12562–12566 (2002).
Barducci, A., Bussi, G. & Parrinello, M. Well-tempered metadynamics: a smoothly converging and tunable free-energy method. Phys. Rev. Lett. 100, 020603 (2006).
Acknowledgements
We thank B. M. Hoffman for helpful discussions. B. W. is grateful for financial support from National Natural Science Foundation of China (nos. 21933009 and 22073077). S.S. thanks the Israel Science Foundation for support (grant ISF 520/18).
Author information
Authors and Affiliations
Contributions
B.W. and W.P. conceived and designed the project. W.P. conducted the computational studies and made all figures, with X.Q. providing assistance. All authors participated in the discussion, and W.P., B.W. and S.S. co-wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Catalysis thanks Jeewon Lee, Lou Noodleman and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Table 1, Figs. 1–28, Notes 1−4 and References.
Supplementary Data 1
The Cartesian coordinates of the truncated PDB (solvation waters 3 Å away from the protein are removed) of all species involved in the catalytic cycle (Fig. 5) from QM/MM metadynamics.
Supplementary Data 2
The Cartesian coordinates of all computed species in QM calculations.
Rights and permissions
About this article
Cite this article
Peng, W., Qu, X., Shaik, S. et al. Deciphering the oxygen activation mechanism at the CuC site of particulate methane monooxygenase. Nat Catal 4, 266–273 (2021). https://doi.org/10.1038/s41929-021-00591-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-021-00591-4
This article is cited by
-
σ–σ Stacked supramolecular junctions
Nature Chemistry (2022)
-
Microbial oxidation of atmospheric trace gases
Nature Reviews Microbiology (2022)
-
Determination of the iron(IV) local spin states of the Q intermediate of soluble methane monooxygenase by Kβ X-ray emission spectroscopy
JBIC Journal of Biological Inorganic Chemistry (2022)
-
Recent Computational Insights into the Oxygen Activation by Copper-Dependent Metalloenzymes
Topics in Catalysis (2022)