Abstract
We demonstrate the dry release transfer of single-layer and bi-layer graphene and few-layer h-BN by utilizing poly(propylene) carbonate (PPC) films. Because of the strong adhesion between PPC and 2D materials around room temperature, we demonstrate that single-layer to few-layer graphene, as well as few-layer h-BN can be fabricated on a spin-coated PPC film/SiO2/Si substrate via the mechanical exfoliation method. In addition, we show that these single-layer to few-layer crystals are clearly distinguishable using an optical microscope with the help of optical interference. Because of the thermoplastic properties of PPC film, the adhesion between the 2D materials and PPC significantly decreases at about 70 °C. Therefore, by tuning the temperature, the graphene and h-BN flakes on PPC can be easily dry-transferred onto another h-BN substrate. We demonstrate the fabrication of encapsulated h-BN/graphene/h-BN devices and graphene/few-layer h-BN/graphene vertical-tunnel-junction devices using this method. Our finding provides a simple method for constructing high-quality graphene and h-BN-based van der Waals heterostructures with a minimum amount of polymer contamination.
Similar content being viewed by others
Introduction
Up until now, various kinds of van der Waals (vdW) heterostructures composed of graphene, hexagonal boron nitridee (h-BN), and other two-dimensional (2D) materials have been fabricated for the purposes of fundamental research and applications.1,2 To construct these heterostructures, various transfer methods for 2D materials have been explored,3 including the (1) wet release transfer,4 (2) dry release transfer,5,6,7 and (3) stamping methods.8,9,10,11 Among them, the dry release transfer method has been recognized as the most versatile and flexible technique to explore various kinds of heterostructures. In this method, flakes of the 2D material are first fabricated on a polymer sheet and are subsequently transferred onto another material (typically, exfoliated h-BN on a Si substrate) without dissolving the polymer sheets by organic solvents. The most commonly used polymer for the dry transfer method is polydimethylpolysiloxane (PDMS).5 Various vdW heterostructures have been fabricated based on this method for utilizing transition metal dichalcogenides such as MoS2, WSe2, and NbSe2, as well as black phosphorus. Examples of the applications of these heterostructures are high-quality quantum Hall effect devices,12,13 superconducting devices,14,15,16,17 optoelectronic devices,18,19,20,21 and spintronics devices.22,23,24 However, the PDMS-based dry transfer method has a limited capability for use in single-layer, bi-layer, and few-layer-thick graphene and few-layer-thick h-BN because of the poor adhesion between PDMS and these materials which makes it difficult to fabricate these thin crystals on PDMS sheets, and owing to the poor visibility of the thin flakes prepared on PDMS sheet.5,25,26,27 Alternative polymers that enable reliable exfoliation and dry release transfer of thin graphene and h-BN have been in high demand. In this study, we demonstrate the dry release transfer method using poly(propylene) carbonate (PPC) for fabricating graphene-based and h-BN-based vdW heterostructures. The main advantages of this method are summarized in the following three points. (1) Because of the strong adhesion of PPC to graphene and h-BN at room temperature (RT), appropriately large flakes of thin graphene and h-BN can be obtained with the mechanical exfoliation method. (2) Single-layer graphene, as well as thin h-BN, fabricated on a PPC/SiO2/Si structure is visible under an optical microscope. (3) As the adhesion between PPC and the flakes significantly decreases at high temperatures, we demonstrate the dry release transfer of graphene and h-BN on a h-BN/SiO2/Si substrate at about 70 °C. The proposed dry release transfer method is suitable for fabricating graphene-based and h-BN-based vdW heterostructures.
Results and discussion
First, we show the numerical calculation of optical contrast for graphene and h-BN on different substrates, and the results are presented in Fig. 1. The optical contrast for each substrate was calculated based on the multilayer Fresnel reflection method.28,29,30 We consider the case of visible light incidence from air onto the multi-layer structure including graphene, h-BN, PPC, SiO2, and Si. The refractive index values of graphene, PPC, SiO2, and Si are taken from the literature.31,32,33 We consider the results for graphene/PPC/SiO2/Si as depicted in Fig. 1a. The single-layer graphene is assumed to have a thickness d1 = 0.34 nm and complex wavelength-independent refractive index n1(λ) = 2.6–1.3i.28 For multilayer graphene, we use the thickness of Nd1, where N represents the number of layers. We changed the thickness of the PPC layer d2 from 0 to 1000 nm with a refractive index of n2(λ) = 1.463. The thickness of the SiO2 layer d3 is fixed at d3 = 290 nm with a wavelength-dependent refractive index n3(λ) consisting of only the real part only (typically, n3(λ) ~1.458 for λ ~600 nm). The thickness of the Si layer is assumed to be semi-infinite with a wavelength-dependent complex refractive index n4(λ) (the refractive index data for Si and SiO2 are provided in the supplementary information). Using the geometry depicted in Fig. 1a, the reflected light intensity can be written as
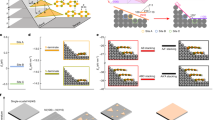
Schematics, calculated results, and optical micrographs of a–e graphene (Gr)/ poly(propylene) carbonate (PPC)/SiO2/Si, f–j Gr/SiO2/Si, k–o h-BN/PPC/SiO2/Si, and p–r h-BN/SiO2/Si structures. a, f, k, p Schematics of the device structure with refractive indexes n1, n2, n3, n4, and n5 for Gr, PPC, SiO2, Si, and h-BN, respectively. The thicknesses d1, d2, d3, d4, and d5 are also depicted for Gr, PPC, SiO2, Si, h-BN, respectively. b, g, l, q Calculated optical contrast as a function of wavelength and thickness for b, l PPC and g, q SiO2. c, h, m, r Representative calculated optical contrast for single-layer and bi-layer c, h Gr and m, r h-BN. d, e, i, j, n, o Optical micrographs of d, i single-layer Gr, e, j bi-layer Gr, and n, o three-layer and four-layer h-BN. All the scale bars are 10 μm
where
are the reflection coefficients for different interfaces, and δ1 = 2πn1d1/λ, δ2 = 2πn2d2/λ, and δ3 = 2πn3d3/λ characterize the phase shifts when light passes through the nth layer (detail information of calculation procedure is provided in the supplementary information). Then, the optical contrast C is defined as the relative intensity of reflected light in the presence (n1 ≠ 1) and absence (n1 = n0 = 1) of graphene:
The results are plotted as a function of the incident light wavelength and PPC thickness, as shown in Fig. 1b. The contrast curve for single and bi-layer graphene on PPC with a thickness of 900 nm is presented in Fig. 1c. Contrast exhibits zero to positive values and it depends on wavelength. The graphene-covered region exhibits significant absorption for green, blue, and red light, thus rendering a region on the image that is darker than the surrounding area. This demonstrates that graphene on the PPC/SiO2/Si substrate can be distinguishable with an optical microscope. For comparison, the optical contrast for graphene/SiO2/Si, as depicted in Fig. 1f, is calculated. The variation in optical contrast for different thicknesses of SiO2 and wavelengths is plotted in Fig. 1g, and the results for single-layer and bi-layer graphene on 290-nm-thick SiO2/Si are presented in Fig. 1h. The calculated optical contrast in Fig. 1h is fully consistent with the literature and clear layer-dependent contrast is exhibited.28,29 The contrast curves in Fig. 1b, g are quite similar. This is due to the similarity of the refractive index of PPC to that of SiO2; thus, the spin-coated PPC on SiO2/Si gives rise to a similar effect with the increase in SiO2 thickness in the SiO2/Si structure. Therefore, it is a natural consequence that few-layer graphene is visible on a PPC/SiO2/Si substrate with a moderately thin PPC, as studied here. Optical micrographs for single- and bi-layer graphene on 900-nm-thick PPC/290-nm-thick SiO2/Si are shown in Fig. 1d, e, respectively. We find that single- and bi-layer graphene on PPC/SiO2/Si can be visible under an optical microscope. For comparison, optical micrographs of single-layer and bi-layer graphene on 290-nm-thick SiO2/Si taken with the same optical microscope are also shown in Fig. 1i, j, respectively. Here, images were taken in high-dynamic-range mode to enhance the visibility of the micrograph.
We also calculated the contrast for the h-BN/PPC/290-nm-thick SiO2/Si structure, as illustrated in Fig. 1k. Here, the thickness for h-BN is assumed to be d5 = 0.335 nm with a wavelength-independent refractive index of n5(λ) = 2.2.34 The results are plotted as a function of the incident light wavelength and PPC thickness, as shown in Fig. 1l. The contrast curves for single and bi-layer h-BN on PPC with a thickness of 900 nm are presented in Fig. 1m. For comparison, the optical contrast for h-BN/SiO2/Si, as depicted in Fig. 1p, is calculated. The variation in optical contrast for different thicknesses of SiO2 and wavelengths is plotted in Fig. 1q, and the results for single-layer and bi-layer h-BN on 290-nm-thick SiO2/Si are presented in Fig. 1r. For both the PPC/SiO2/Si and SiO2/Si substrate, h-BN exhibits a positive or negative optical contrast depending on the wavelength. These positive and negative contrasts could easily cancel out each other under optical microscope observation with white light. Thus, compared to graphene, few-layer h-BN is more difficult to distinguish with a microscope. Previous experiments on few-layer h-BN on a SiO2/Si substrate used optical filters and optimization of the SiO2 thickness to enhance visibility.34 Because of the similarity between Fig. 1l, q, we believe that few-layer h-BN can be easily distinguishable using filters and by optimizing the thickness of both PPC and SiO2. Nevertheless, we show that even without optimization, the technologically important three-layer and four-layer h-BN can be distinguishable on a 900-nm-thick PPC/SiO2/Si substrate, and their optical micrographs are presented in Fig. 1n, o. These results indicate that the PPC/SiO2/Si structure enables atomically thin 2D crystals to be exfoliated, and their thickness can be identified with an optical microscope.
Here, we show a method to demonstrate the dry transfer of single-layer graphene on a h-BN substrate. As the thin graphene and h-BN are visible on the PPC film, we used this 2D material/PPC stack for the dry transfer of 2D materials onto another substrate. The detailed transfer procedure for fabricating the graphene/h-BN structure is illustrated in the schematic in Fig. 2. First, the PPC solution was spin-coated on the SiO2/Si substrate at a spin-coating speed of 4000 rpm; this created a PPC film with a thickness of about 900 nm. Prior to spin coating, the substrate was cleaned by acetone and IPA with ultrasonic agitation. Then, the PPC-coated substrate was baked on a hot plate at 70 °C for 5 min. The PPC/SiO2/Si substrate was taken away from the hot plate and graphene or h-BN was then deposited on the substrate by the mechanical exfoliation method at RT, as shown in Fig. 2a. As PPC is known to exhibit a strong adhesion to 2D materials at around RT,8,10,35,36 both few-layer graphene and thick graphite or h-BN with a reasonably large size can be easily fabricated. Separately, a piece of PDMS sheet with a size of approximately 3 mm (width) × 3 mm (length) × 0.4 mm (depth) was placed on a glass slide. To ensure a strong adhesion between the PDMS and the glass slide, the backside of the PDMS was treated with air plasma for a few minutes. On the PDMS sheet, graphene flakes on the PPC film were transferred from the SiO2/Si substrate by the following procedure, as depicted in Fig. 2b, c: (1) prepare the tape window with a size of about 4 mm × 4 mm on the PPC that surrounds the graphene region; (2) clean the front side of the PDMS sheet with air plasma for a few minutes to ensure a strong adhesion of the PDMS surface; (3) remove the graphene/PPC membrane structures together with the tape by gently removing the tape from the substrate; (4) attach the graphene/PPC membrane on the PDMS sheet by aligning the tape window to the PDMS sheet (Fig. 2c). At the baking temperature of 70 °C, PPC can be easily removed from the substrate without using a sacrificed polymer layer between PPC and the substrate.6,7 We note that baking with temperature higher than 70 °C, smooth detaching of the PPC sheet from the substrate with this method becomes more difficult. Separately, h-BN flakes were prepared on another SiO2/Si substrate by mechanical exfoliation (Fig. 2d). The substrate was pre-cleaned with piranha solution prior to the deposition of h-BN. For transfer, the relative positions of graphene/PPC and h-BN were aligned under optical microscope observation, as shown in Fig. 2e, and the graphene and h-BN were made to gently come into contact each other without heating up the stage. Once they made contact, the substrate stage was heated up to 70 °C (Fig. 2f). Here, we changed only the setpoint of the substrate heater from RT to 70 °C; then, the substrate heated up with the maximum speed of the heater. When the substrate stage temperature was stable at 70 °C, the substrate and PPC film were slowly separated by moving either the substrate stage or the glass slide stage (Fig. 2g). We found that at temperatures equal to or higher than about 70 °C, the adhesion between graphene and PPC significantly weakens such that graphene can be released from the PPC film and transferred onto h-BN. Thus, the graphene/h-BN structure can be fabricated. Similarly, thick or thin h-BN can be transferred onto the h-BN or graphene. The dry transfer operates within the temperature range of 70–100 °C, depending on the adhesion between PPC and PDMS. The PPC film tends to detach from PDMS during transfer when the substrate temperature is higher than 100 °C. Here, we would note that increasing temperature have dual roles for successively transferring the graphene onto thick h-BN; one is for easier detaching graphene or thin h-BN from PPC sheet and second is for preventing pick up targeted h-BN from SiO2/Si substrate with PPC; both of these are crucial to success our method. The process does not require graphene (or h-BN) to be exposed to organic solvents; thus, this can be regarded as a dry release transfer. To completely remove the PPC residue from the graphene surface, the sample was annealed at 350 °C in an Ar/3% H2 atmosphere for 1 h; this temperature is above the decomposition temperature of PPC of about 280 °C.37 The air-plasma treatment process during transfer can be replaced with an O2 plasma or an equivalent process to improve the adhesion at the PPC/PDMS and PDMS/glass interfaces. We also note that these plasma treatment processes are not crucial steps to achieve flake transfer but they reduce the risk of PDMS or PPC sheet falling off from the glass slide or PDMS during transfer.
a–g Schematics of dry transfer fabrication process. a Preparation of graphene (Gr) on PPC/SiO2/Si substrate. An optical micrograph of Gr/PPC/290-nm-thick SiO2/Si is also shown. Separately, a piece of polydimethylpolysiloxane (PDMS) is prepared on the glass slide. b Preparation of tape window surrounding graphene area, with its optical micrograph shown in the top panel. Subsequently, this tape window, as well as the Gr/PPC sheet, is transferred onto the PDMS. An optical micrograph of PDMS on the glass slide is shown in the bottom panel. c Prepared Gr/PPC/PDMS structure on glass slide; the optical micrograph is also shown. d h-BN flake with thickness of about 30 nm prepared on SiO2/Si substrate by mechanical exfoliation technique; the optical micrograph is also shown. e Adjusting the relative positions of Gr and h-BN flakes under an optical micrograph and making gentle contact at room temperature. f Heating the stage to 70 °C while graphene and h-BN are in contact. g Gentle separation of glass slide from SiO2/Si substrate; the optical micrograph of the fabricated Gr/h-BN structure on the SiO2/Si substrate is also shown. All the scale bars are 10 μm
Here, we discuss the mechanism of the PPC-based dry transfer. PPC is known as a thermoplastic material such that it becomes very soft at higher temperatures, whereas it hardens at RT. To compare this to PDMS, we plot the storage modulus of these materials taken from the literature in Fig. 3a.31,38 The storage modulus represents the mechanical hardness of the material. The glass transition temperature (Tg) of PPC is about 40 °C. Below this temperature, the storage modulus rapidly increases and saturates at a lower temperature. Here, PPC behaves as a solid and is difficult to deform. The adhesion between the 2D crystal and PPC is known to be strong in this region. In fact, the pick-up of thick h-BN from the SiO2/Si substrate with a PPC sheet has been performed around this temperature regime.8,10,35,36 We demonstrate that reasonably large graphene and h-BN flakes can be easily fabricated with mechanical exfoliation. An optical micrograph of graphene and graphite on PPC fabricated under this condition is presented in Fig. 3b, c, respectively. Heating the PPC above about 40 °C drastically reduces its storage modulus. Here, PPC is glass and becomes soft. The adhesion between the 2D crystal and PPC gradually decreases with the increase in temperature above this region. At high temperatures (about 70 °C) where the adhesion between PPC and graphene or PPC and h-BN is sufficiently small, these crystals tend to detach from the PPC and transfer to another substrate. Previous works done by other groups also demonstrated that a h-BN or a h-BN/graphene/h-BN stack can be dry transferred from PPC sheet to SiO2/Si substrate in the temperature range of 70–110 °C.10,36,39 Here, graphene and the graphite flakes respectively shown in Fig. 3b, c are transferred onto the 290-nm-thick SiO2/Si substrate using the method explained in Fig. 2, and their optical micrographs are presented in Fig. 3d, e, respectively. Nearly all the graphene and graphite flakes can be transferred from PPC to the SiO2 substrate. This indicates that our dry transfer method relies on the strong reduction of adhesion between the 2D crystal and PPC membrane at high temperatures. Thus, the flakes on the PPC can be easily transferred even on the SiO2 surface. For comparison, the storage modulus curve for PDMS is also presented in Fig. 3a. As the Tg of PDMS is –28 °C, PDMS does not show a dramatic change in storage modulus with the increase in temperature and is always in the glass state and soft above RT. Because of this softness, PDMS is very effective for the dry transfer of the flake once the flake has been exfoliated onto the PDMS surface. The drawback of this method is the difficulty of the exfoliation step of the thin graphene and h-BN flakes onto PDMS due to the poor adhesion. The advantage of using the PPC presented here is the strong adhesion at RT, which becomes very weak at higher temperatures.
a Storage modulus as a function of temperature for PPC and PDMS polymers. The data have been obtained from refs.,31,38 b, c Optical micrographs of Gr and graphite flakes on 900-nm-thick PPC/290-nm-thick SiO2/Si, respectively. Scale bars in b and c are 50 and 10 μm, respectively. d, e Optical micrographs of transferred Gr and graphite flakes on 290-nm-thick SiO2/Si, respectively. Scale bars in d and e are 50 and 10 μm, respectively
In Fig. 4a–d, optical micrographs of the fabricated single-layer and bi-layer graphene and three-layer and four-layer h-BN on a thick h-BN/SiO2/Si heterostructure are presented, respectively. Topographic image samples were measured by atomic force microscopy (AFM) and are respectively presented in Fig. 4e–h. From both the optical micrograph and AFM image, it appears that in each case, the surface of the transferred graphene or h-BN is clean, without noticeable polymer residue. There are some bubbles presented in the transferred flakes. We infer that the number of bubbles can be reduced by further optimizing transfer conditions such as substrate temperature.10 The thicknesses of the single-layer and bi-layer graphene and three-layer and four-layer h-BN on the thick h-BN in Fig. 4e–h were measured and the results are presented in Fig. 4i–l, respectively. The thicknesses are determined to be about 0.5, 0.9, 1.2, and 1.5 nm, respectively. The thickness difference between the flakes is 0.3–0.4 nm and they are close to the single-layer thickness of both graphene and h-BN; thus, we think these prove the successful dry transfer of single-layer to few-layer-thick graphene and h-BN with a reasonable control of thickness.
a–d Optical micrograph, e–h atomic force microscopy (AFM) topographic image, and i–l AFM height profiles of a, e, i single-layer Gr on thick h-BN, b, f, j bi-layer Gr on thick h-BN, c, g, k three-layer h-BN on thick h-BN, and d, h, l four-layer h-BN on thick h-BN, respectively. The locations of the displayed AFM height profiles (i, j, k, l) are indicated by white lines in panels (e, f, g, h), respectively. Scale bars in a–d and e–h are 10 μm and 500 nm, respectively
Finally, we show the transport properties of graphene and h-BN devices fabricated with our proposed dry transfer method. By repeating the PPC-based dry transfer, encapsulated h-BN/graphene/h-BN and h-BN/graphene/few-layer h-BN/graphene/h-BN structures were fabricated. By opting to use a top h-BN layer with a size smaller than the graphene layer, we allow the edge of the graphene to be not fully covered by the top h-BN layer. This enables us to construct a vdW heterostructure without using a one-dimensional edge-contact scheme.8 This greatly simplifies the device fabrication as well as eliminates the requirement of fabricating of a global graphite gate with edge contact, which is difficult. We fabricated both top-contact (Fig. 5a) and edge-contact (Fig. 5b) h-BN/graphene/h-BN devices. In addition to this, a top-contact vertical-tunnel-junction device (Fig. 5c) was fabricated. Optical micrographs of the devices are presented in Fig. 5d–f, respectively. Note that top-contact device in Fig. 5a contains two graphene channels that are prepared by successive transfers: one uses a Si back-gate and the other uses a graphite back-gate. Magnetotransport measurements were performed at ~2.0 K. The resistances of the graphene channels were measured under the sweep of the carrier density n by applying a gate voltage to the doped Si back-gate or graphite back-gate, and the results are presented in Fig. 5g, h, respectively. The capacitance of the h-BN/SiO2 dielectric was determined from the quantum Hall effect. The mobilities and charge inhomogeneity (extracted from the width of a charge neutrality point resistance peak) of the two top-contact h-BN/graphene/h-BN devices were calculated as ~260,000 cm2/Vs and ~3 × 1010 cm−2 for back-gated device, ~140,000 cm2/Vs and ~5 × 1010 cm−2 for graphite-gated device, respectively. These values are comparable to the mobility and the charge inhomogeneity of the edge contact device (Fig. 5h) of ~610,000 cm2/Vs and ~5× 1010 cm−2; thus, we think that quality of the device is not limited by the contact scheme. The extracted mobilities of the h-BN/graphene/h-BN devices approach that of modern h-BN/graphene/h-BN devices;7,8,10,40 this indicates the cleanness of our transfer process. This is noticeably high quality considering the fact that the top surface of the transferred graphene flake has been contacted to the PPC polymer during our dry transfer process. This is in contrast to the stamping method8,9,10,11 where graphene has never contacted to polymer during fabrication. For the top-contacted vertical-tunnel-junction device shown in Fig. 5f, we selected an h-BN tunnel barrier with a thickness of about 1.5 nm, as measured by AFM. Current–voltage (I–V) characteristics at about 2.0 K are presented in Fig. 5i, showing a non-linear change in I with respect to V. There are kinks in I–V at around ±0.2 V; we attribute these are due to the tunneling through the localized state within h-BN layer as similar results are published in recent literature based on similar graphene/h-BN/graphene device.41 The extracted junction resistance–area product RA is about 1 × 1012 Ω μm2; this RA is comparable to that of the h-BN barrier with a similar thickness.42,43,44 These results further prove that the PPC-based dry transfer method can be used to fabricate a multi-stack graphene/h-BN vdW heterostructure with clean interfaces.
a–c Schematics of the device structure of a top-contacted h-BN/Gr/h-BN sample, b edge-contacted h-BN/Gr/h-BN sample, and c h-BN/Gr/few-layer h-BN/Gr/h-BN vertical-tunnel-junction sample. d, e Photographs of d top-contact and e edge-contact h-BN/Gr/h-BN devices. Dashed lines outline the location of graphene. Scale bars are 10 μm. g, h Resistance data as a function of carrier density of Gr for the devices shown in d and e, respectively; conductivity data is shown in the insets. In g, data shown in red and blue indicates the resistance value of left and right graphene in d, respectively. f Photograph of h-BN/Gr/few-layer h-BN/Gr/h-BN vertical-tunnel-junction device. Solid and dashed red lines indicate the bottom and top graphene, respectively. The white line indicates the thin h-BN tunnel barrier. The thickness of few-layer h-BN is determined via AFM to be about 1.5 nm. Scale bar is 10 μm. i Current–voltage characteristics of the device shown in c at 3 K
In summary, we demonstrate the dry transfer fabrication of graphene as well as h-BN using the PPC-based method. The main advantages of this method are summarized in the following three points: (1) Because of the strong adhesion of PPC to graphene and h-BN at RT, appropriately large flakes of thin graphene and h-BN can be obtained via the mechanical exfoliation method. (2) Single-layer graphene, as well as thin h-BN, fabricated on a PPC/SiO2/Si structure is visible under an optical microscope. (3) As the adhesion between the PPC and flakes significantly decreases at high temperatures, we are able to demonstrate the dry release transfer of graphene and h-BN on a h-BN/SiO2/Si substrate at about 70 °C. Furthermore, we show that the fabricated graphene/h-BN devices exhibit excellent properties. So far, the PDMS-based dry transfer method has been widely used for fabricating TMD-based and black phosphorus-based vdW heterostructures.5 With the straightforward addition of a PPC sheet onto the PDMS, we demonstrate that graphene as well as few-layer h-BN can be also dry transferred. Our PPC-based dry transfer method is sufficiently simple and enables us to construct the heterostructure using various type of contact scheme. Thus, our results further extend the capability of the dry release transfer method for constructing multilevel high-quality vdW heterostructures.
Methods
PPC (Sigma-Aldrich, CAS 25511-85-7) was dissolved into an anisole with a ratio of 15% PPC: 85% anisole by weight. The PPC film was prepared by spin-coating the PPC solution onto a 290-nm-thick SiO2/highly-doped Si substrate. The same substrate was used for the mechanical exfoliation of graphene and h-BN. A commercially available PDMS sheet (Gel-Pak PF-X4-17 mil.) was used. Graphene was mechanically exfoliated from a graphite crystal (NGS Naturgraphit GmbH) using adhesive tape (Nitto Denko, 32B) and was deposited on a PPC-coated SiO2/Si substrate or SiO2/Si substrate. Using the same method, thin h-BN flakes were exfoliated from its bulk crystal, which was synthesized by the high-pressure and high-temperature method and deposited onto the substrate. We chose thick h-BN with thicknesses in the range of 30–50 nm for the substrate and encapsulation for the graphene and thin h-BN. A commercial optical microscope (Keyence, VHX-5000) was used to observe fabricated flakes with a halogen lamp as a light source. To improve the adhesion of the PDMS, a plasma cleaner was operated with air as the plasma source (Harrick Plasma, PDC-32G). The thickness of the flakes was measured with an atomic force microscope (Hitachi High Technologies, SPM400). The device was spin-coated with a PMMA resist, and using electron beam (EB) lithography, electrode patterns were prepared. Au (~100 nm)/Cr (5 nm) contact electrodes were deposited by EB evaporation. For the edge-contact device, graphene and h-BN were etched using reactive ion etching with a CF4-based gas (Samco, RIE-200iP). Immediately after plasma etching, the sample was loaded into the EB evaporation system and the electrodes were deposited. Transport properties were measured in a variable-temperature cryostat equipped with a superconducting magnet that can apply a magnetic field in the out-of-plane direction. After each dry transfer process, the vdW stacks were annealed in a tube furnace under the 0.3 L/min. of flow of Ar/3% H2 gas. During the annealing process, temperature setting of the furnace was controlled such that 30 min for heating up from room temperature to 350 °C, kept at 60 min at 350 °C, and 30 min for cooling down from 350 °C to room temperature.
Data availability
The data generated during this study are available from the corresponding author on reasonable request.
References
Geim, A. K. & Grigorieva, I. V. Van der Waals heterostructures. Nature 499, 419–425 (2013).
Novoselov, K. S., Mishchenko, A., Carvalho, A. & Castro Neto, A. H. 2D materials and van der Waals heterostructures. Science 353, aac9439 (2016).
Frisenda, R. et al. Recent progress in the assembly of nanodevices and van der Waals heterostructures by deterministic placement of 2D materials. Chem. Soc. Rev. 47, 53–68 (2018).
Dean, C. R. et al. Boron nitride substrates for high-quality graphene electronics. Nat. Nanotech. 5, 722–726 (2010).
Castellanos-Gomez, A. et al. Deterministic transfer of two-dimensional materials by all-dry viscoelastic stamping. 2D Mater. 1, 011002 (2014).
Taychatanapat, T., Watanabe, K., Taniguchi, T. & Jarillo-Herrero, P. Quantum Hall effect and Landau-level crossing of Dirac fermions in trilayer graphene. Nat. Phys. 7, 621–625 (2011).
Kretinin, A. V. et al. Electronic properties of graphene encapsulated with different two-dimensional atomic crystals. Nano Lett. 14, 3270–3276 (2014).
Wang, L. et al. One-dimensional electrical contact to a two-dimensional material. Science 342, 614–617 (2013).
Zomer, P. J., Guimarães, M. H. D., Brant, J. C., Tombros, N. & Van Wees, B. J. Fast pick up technique for high quality heterostructures of bilayer graphene and hexagonal boron nitride. Appl. Phys. Lett. 105, 013101 (2014).
Pizzocchero, F. et al. The hot pick-up technique for batch assembly of van der Waals heterostructures. Nat. Commun. 7, 11894 (2016).
Masubuchi, S. et al. Autonomous robotic searching and assembly of two-dimensional crystals to build van der Waals superlattices. Nat. Commun. 9, 1413 (2018).
Cui, X. et al. Multi-terminal transport measurements of MoS2 using a van der Waals heterostructure device platform. Nat. Nanotechnol. 10, 534–540 (2015).
Li, L. et al. Quantum oscillations in a two-dimensional electron gas in black phosphorus thin films. Nat. Nanotechnol. 10, 608 (2015).
Xi, X. et al. Ising pairing in superconducting NbSe2 atomic layers. Nat. Phys. 12, 139–143 (2016).
Yabuki, N. et al. Supercurrent in van der Waals Josephson junction. Nat. Commun. 7, 10616 (2016).
Kim, M. et al. Strong proximity Josephson coupling in vertically stacked NbSe2–graphene–NbSe2 van der Waals Junctions. Nano Lett. 17, 6125–6130 (2017).
Singh, V. et al. Optomechanical coupling between a multilayer graphene mechanical resonator and a superconducting microwave cavity. Nat. Nanotechnol. 9, 820–824 (2014).
Ota, Y. et al. Optical coupling between atomically thin black phosphorus and a two dimensional photonic crystal nanocavity. Appl. Phys. Lett. 110, 223105 (2017).
Palacios-Berraquero, C. et al. Large-scale quantum-emitter arrays in atomically thin semiconductors. Nat. Commun. 8, 15093 (2017).
Li, D. et al. Two-dimensional non-volatile programmable p–n junctions. Nat. Nanotechnol. 12, 901–906 (2017).
Xie, H., Jiang, S., Shan, J. & Mak, K. F. Valley-selective exciton bistability in a suspended monolayer semiconductor. Nano Lett. 18, 3213–3220 (2018).
Lee, J., Wang, Z., Xie, H., Mak, K. F. & Shan, J. Valley magnetoelectricity in single-layer MoS2. Nat. Mater. 16, 887–891 (2017).
Yamasaki, Y. et al. Exfoliation and van der Waals heterostructure assembly of intercalated ferromagnet Cr1/3TaS2. 2D Mater. 4, 041007 (2017).
Arai, M. et al. Construction of van der Waals magnetic tunnel junction using ferromagnetic layered dichalcogenide. Appl. Phys. Lett. 107, 103107 (2015).
Uwanno, T., Hattori, Y., Taniguchi, T., Watanabe, K. & Nagashio, K. Fully dry PMMA transfer of graphene on h -BN using a heating/cooling system. 2D Mater. 2, 041002 (2015).
Goler, S. et al. Self-assembly and electron-beam-induced direct etching of suspended graphene nanostructures. J. Appl. Phys. 110, 064308 (2011).
Tien, D. H. et al. Study of graphene-based 2D-heterostructure device fabricated by all-dry transfer process. ACS Appl. Mater. Interfaces 8, 3072–3078 (2016).
Blake, P. et al. Making graphene visible. Appl. Phys. Lett. 91, 063124 (2007).
Ni, Z. H. et al. Graphene thickness determination using reflection and contrast spectroscopy. Nano Lett. 7, 2758–2763 (2007).
Byrnes, S. J. Multilayer optical calculations. arXiv: 1603.02720 (2016).
Luinstra, G. A. & Borchardt, E. in Synthetic Biodegradable Polymers 29–48 (Springer, Berlin, Heidelberg, 2012).
Philipp, H. R. in Handbook of Optical Constants of Solids (ed. Palik, E. D.) 749–763 (Academic Press, Cambridge, 1997).
Edwards, D. F. in Handbook of Optical Constants of Solids (ed. Palik, E. D.) 547–569 (Academic Press, Cambridge, 1997).
Gorbachev, R. V. et al. Hunting for monolayer boron nitride: optical and Raman signatures. Small 7, 465–468 (2011).
Wang, J. I.-J. et al. Electronic transport of encapsulated graphene and WSe2 devices fabricated by pick-up of prepatterned hBN. Nano Lett. 15, 1898–1903 (2015).
Jessen, B. S. et al. Lithographic band structure engineering of graphene. Nat. Nanotechnol. 14, 340–346 (2019).
Wang, S. et al. Structure and properties of poly(Propylene Carbonate). Int. J. Polym. Anal. Charact. 3, 131–143 (1997).
Barreau, V., Yu, D., Hensel, R. & Arzt, E. Elevated temperature adhesion of bioinspired polymeric micropatterns to glass. J. Mech. Behav. Biomed. Mater. 76, 110–118 (2017).
Larentis, S. et al. Reconfigurable complementary monolayer MoTe2 field-effect transistors for integrated circuits. ACS Nano 11, 4832–4839 (2017).
Mayorov, A. S. et al. Micrometer-scale ballistic transport in encapsulated graphene at room temperature. Nano Lett. 11, 2396–2399 (2011).
Greenaway, M. T. et al. Tunnel spectroscopy of localised electronic states in hexagonal boron nitride. Commun. Phys. 1, 94 (2018).
Britnell, L. et al. Electron tunneling through ultrathin boron nitride crystalline barriers. Nano Lett. 12, 1707–1710 (2012).
Kamalakar, M. V., Dankert, A., Bergsten, J., Ive, T. & Dash, S. P. Enhanced tunnel spin injection into graphene using chemical vapor deposited hexagonal boron nitride. Sci. Rep. 4, 6146 (2014).
Yamaguchi, T. et al. Electrical spin injection into graphene through monolayer hexagonal boron nitride. Appl. Phys. Express 6, 073001 (2013).
Acknowledgements
This work was supported by CREST, Japan Science and Technology Agency (JST) under Grant Number JPMJCR15F3, and by JSPS KAKENHI Grant Numbers JP25107001, JP25107003, JP25107004, JP26248061, JP15K21722, JP15K17433, and JP16H00982.
Author information
Authors and Affiliations
Contributions
K.K., R.M. and T.M. conceived and designed the experiments; K.K. fabricated the devices with the help of R.M., M.O., Y.W. and S.M.; K.W. and T.T. grew the h-BN crystals; K.K performed the device characterization with the help of R.M; R.M., K.K. and T.M. wrote the manuscript using contributions from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kinoshita, K., Moriya, R., Onodera, M. et al. Dry release transfer of graphene and few-layer h-BN by utilizing thermoplasticity of polypropylene carbonate. npj 2D Mater Appl 3, 22 (2019). https://doi.org/10.1038/s41699-019-0104-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41699-019-0104-8
This article is cited by
-
Low Ohmic contact resistance and high on/off ratio in transition metal dichalcogenides field-effect transistors via residue-free transfer
Nature Nanotechnology (2024)
-
The practice of reaction window in an electrocatalytic on-chip microcell
Nature Communications (2023)
-
Coaxial boron nitride nanotubes as interfacial dielectric layers to lower interface trap density in carbon nanotube transistors
Nano Research (2023)
-
All-dry flip-over stacking of van der Waals junctions of 2D materials using polyvinyl chloride
Scientific Reports (2022)
-
Evaluation of polyvinyl chloride adhesion to 2D crystal flakes
npj 2D Materials and Applications (2022)