Abstract
The utilization of biochar’s as soil amendments for enhancing nutrient retention in subsoils present potential limitations. To address this issue, we conducted a greenhouse experiment to assess the effects of various biochar’s derived from animal manures (swine manure, poultry litter, cattle manure) and plant residues (rice straw, soybean straw, corn straw) when applied to surface of an acidic soil. Our study focused on wheat crops under a no-tillage system, with a subsequent evaluation of the residual impacts on soybeans. The experimental design involved the application of biochar’s at different rates i.e. 10 and 20 Mg ha−1, followed by the assessment of their influence on NPK levels, pH, and exchangeable Al in stratified soil layers (0–5, 5–10, 10–15, and 15–25 cm). Furthermore, we investigated the interplay between biochar doses and the application of nitrogen (N) in the top 5 cm of soil, specifically examining \({\text{NO}}_{{3}}^{ - }\), \({\text{NH}}_{{4}}^{ + }\), P and K levels. Our findings revealed that in the top 5 cm of soil, biochar doses and N application significantly affected \({\text{NO}}_{{3}}^{ - }\), \({\text{NH}}_{{4}}^{ + }\), P and K concentrations. However, in deeper soil layers, no significant differences were observed among biochar doses with or without N application. Interestingly, K levels were impacted throughout all soil depths, regardless of the presence or absence of N application. Moreover, biochar application up to a 5 cm depth induced favorable changes in soil pH and reduced exchangeable Al. In contrast, deeper layers experienced a decrease in soil pH and an increase in exchangeable Al following biochar treatment. In conclusion, our study demonstrates that biochar’s can effectively retain NPK nutrients, enhance soil pH, and decrease exchangeable Al, independent of the type and dosage of application under a no-tillage system. Nonetheless, the efficacy of biochar amendments may vary with soil depth and type of nutrient, warranting careful consideration for maximizing their benefits in sustainable agricultural practices.
Similar content being viewed by others
Introduction
Biochar is a solid product derived from the carbonization or pyrolysis of biomass, such as agricultural residues, wood waste, or organic matter1,2,3,4. This process involves heating the biomass in a low-oxygen environment, preventing complete combustion, and resulting in the formation of a stable, carbon-rich material5,6,7,8. The production and application of biochar’s to soil instigate fundamental changes in soil nutrient cycling, leading to enhanced soil fertility and increased crop productivity9,10,11,12,13,14,15. Particularly in acidic, infertile soils with low organic matter content, biochar application yields positive responses. In acidic soils, the functional groups on biochar reduce aluminum phytotoxicity by forming surface complexes with aluminum cations16. As biochar application rate increased, significant increases were observed in exchangeable base cations and decreases in exchangeable acidity and Al saturation, but no additional changes in soil pH. The elimination of exchangeable acidity and the strong buffering capacity of biochar may be partially responsible for the lack of change in soil pH at the higher biochar application rate, inhibiting a further liming effect17. However, the outcomes of this application in such soils can exhibit variability18,19. The nitrification process is regulated by several factors, including soil pH, temperature, soil moisture, nitrogen (N) supplying substrate, soil microbes, and soil types. These elements play crucial roles in shaping the rate and efficiency of nitrification in the soil ecosystem20, as the nitrification process is major factor in N cycle in soil as well as nutrient use efficiency21,22,23. Understanding the nitrification process and its environmental implications for various soil types is crucial for enhancing soil fertility and promoting environmental protection. It is essential to comprehend soil processes affected by factors such as Low pH, high Al, and low CEC, which significantly limit crop growth. The common practice of using liming on acid soils to elevate pH and boost crop yields requires careful consideration24. Prolonged and excessive liming can lead to soil compaction, disrupt the balance of Ca, K, and Mg in the soil, ultimately resulting in reduced crop productivity25. A number of studies have been reporting to understand the nitrification and acidification in forests and temperate soil26. The attention towards the potential advantages of no-tillage has increased, particularly concerning carbon sequestration, CO2 emissions mitigation, and improvement of soil quality27. The understanding of \({\text{NH}}_{{4}}^{ + }\) application effects in tropical and subtropical regions under a no-tillage system is currently limited, with scant information available28,29,30. Considering the significance of managing acidic soils and improving soil fertility to enhance agricultural production, we chose to utilize biochar’s derived from animal manures and plant residues.
The biochars, mostly negatively charged material31 and being high surface charge can enhance the nutrient retention and use in soil32 having great potential for improving soil fertility33. Biochar application to soils can magnificently hold onto the nutrients that are required plants. However, the relationship between nutrient retention and loss pathways is still not obvious. The present study, we designed to find out the retention of nutrients C, N (\({\text{NO}}_{{3}}^{ - }\), \({\text{NH}}_{{4}}^{ + }\)), P, K and micronutrients through the application of animal manures and plant residues derived biochars under greenhouse conditions. Our objective was to raise soil pH, reduce soil acidity, and boost nutrients retention in soil by using biochars derived from animal manures and plant residues. This study focused on evaluating nutrient retention in the soil after two consecutive crops of wheat and soybean, comparing plots with and without \({\text{NH}}_{{4}}^{ + }\) fertilizer application. We hypothesized that the biochar’s from various animal manures and plant residues would enhance soil nutrient retention capacity and increase soil pH in different soil layers. Additionally, we expected that the biochar’s would slow down the nitrification process, thereby promoting the retention of nitrogen in the \({\text{NH}}_{{4}}^{ + }\) form.
Material and methods
Soil collection
The soil collection site was selected on basis of no-till areas, according to the data available from the Department of Soil Science of the Federal University of Santa Maria (29° 43′ 14.2″ S 53° 42′ 15.0″ W). The vegetative cover and grasses were removed manually prior to collecting the soil. The un-disturbed soil was collected in polyvinyl pipes (PVC) (0.29 m height × 0.20 m diameter) up to 25 cm were collected for experiments under no-tillage system for pre-sowing analysis and for experimental use. Prior to installing experiments, the soil was analyzed for pH (4.8 (1:2.5 w/v)), total C (1.2%), N (0.8%), P (4.8 mg kg−1), K (28 mg kg−1), Ca (15.5 cmolc dm−3), Mg (9.3 cmolc dm−3) and Al (16.89 cmolc dm−3). The collected soil having sandy loam texture of the soil (61.71% sand, 25.72% silt, 12.56% clay) was classified as typic hapludults (USDA Soil Taxonomy).
Biochar preparation and analysis
To prepare the biochars, data was collected to find out the quantity and type of feedstock available and decided to collect the materials available easily and are even having any kind of difficulty in their dispose-off. For biochar’s preparation, all feedstocks were collected from the experimental areas of the Federal University of Santa Maria—RS (29° 43′ 14.4″ S 53° 43′ 31.2″W) while corn straw was collected from a nearby city Paraíso do Sul—RS (29° 35′ 10.3″ S 53° 07′ 26.3″ W). Biochar’s, swine manure biochar (SMB), poultry litter biochar (PLB), cattle manure biochar (CMB), rice straw biochar (RSB), soybean straw biochar (SSB) and corn straw biochar (CSB) were prepared at 450 °C for 1 h in muffle furnace with an increase in temperature 10 °C min−1. All the biochar’s were analyzed for pH, electrical conductivity (EC) total carbon (C: Thermo Scientific, Flash EA 1112, Milan, Italy), total nitrogen (N: Thermo Scientific, Flash EA 1112, Milan, Italy), phosphorus (P: Murphy & Riley, 1962), potassium (K: Tedesco et al. 1995), calcium (Ca: Tedesco et al. 1995), magnesium and (Mg: Tedesco et al. 1995).
Experimental setup and treatment plan
A greenhouse experiment was conducted to evaluate the influence of different biochar types on wheat under no-tillage system with biochar application rate at 0 (0 g column−1), 10 (33.5 g column−1) and 20 Mg ha−1 (67 g column−1) with three replicates and their subsequent effect on soybean under complete randomized design (CRD) with three factors i.e. biochar types, biochar dose, nitrogen levels (6 × 2 × 2) with two controls (control 1: No biochar, no nitrogen and control 2: no biochar, recommended nitrogen). Recommended doses of nitrogen (110 kg ha−1 ~ 1.6 g ammonium sulfate column−1), P2O5 (170 kg ha−1 ~ 1.3 g triple superphosphate column−1) and 120 kg K2O ha−1 (~ 0.65 g potassium chloride column−1) were also recommended along with biochar treatments.
As the biochars derived from plant residues had a huge volume, all the biochars were mixed up to 3 cm to have a good contact between soil and biochar to ensure seed placement in good contact with soil biochar mixture. Eight wheat seeds (Sinuelo variety) were sown into each PVC column, after germination thinning was done and four healthy seedlings were left for growth up to 93 days. After wheat harvest, three out of six soybean (5958 RSF IPRO variety) were left for 66 days with basal dose of 90 kg P2O5 ha−1 (0.69 g triple superphosphate column−1) and 120 kg K2O ha−1 (0.65 g potassium chloride column−1), but no nitrogen was added to PVC columns and after 66 days soybean aerial part was of soybean was collected for further analysis. The PVC columns were irrigated on daily basis depending upon the visual soil conditions due to the sandy loam texture of soil, to fulfill the water requirements of both crops respectively.
Soil stratification
After the soybean harvest soil columns were cut into two halves vertically and stratified soil samples were as 0–5, 5–10, 10–15 and 15–25 cm to evaluate the influence of different biochar’s on nutrient retention in topsoil as well as subsoil. The stratified soil samples were then air-dried, ground and passed through 2 mm sieve, then were analyzed for \({\text{NH}}_{{4}}^{ + }\), \({\text{NO}}_{{3}}^{ - }\), P, K, Ca, Mg and Al in different soil layers through recommended procedures used in Soil Science Lab at Federal University of Santa Maria.
Statistical analysis
Standard statistical analysis was performed on collected data34. Analysis of variance was conducted to check the significance of treatments and to compare means of the treatment with controls and with other treatment using software R using linear model (version 3.5) with compatible services by R-studio (version 1.1461). The mean comparison was done using Tukey Multiple comparison test at p < 0.05 using the “emmeans” package. The Figs. 1 and 2 were drawn through SigmaPlot 12.3 version.
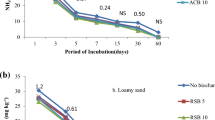
pH change in different soil layers with surface application of swine manure biochar (SMB), poultry litter biochar (PLB), Cattle manure biochar (CMB), rice straw biochar (RSB), soybean straw biochar (SSB) and corn straw biochar (CSB), (A) 10 Mg ha−1, (B) 20 Mg ha−1, (C) 10 Mg ha−1 with N and (D) 20 Mg ha−1 with N.
Soil exchangeable Al in different soil layers with surface application of swine manure biochar (SMB), poultry litter biochar (PLB), Cattle manure biochar (CMB), rice straw biochar (RSB), soybean straw biochar (SSB) and corn straw biochar (CSB), (A) 10 Mg ha−1, (B) 20 Mg ha−1, (C) 10 Mg ha−1 with N and (D) 20 Mg ha−1 with N.
Ethics approval and consent to participate
We all declare that manuscript reporting studies do not involve any human participants, human data, or human tissue. So, it is not applicable. Our experiment follows the with relevant institutional, national, and international guidelines and legislation.
Results and discussions
Primary nutrients concentration
It has been recognized that biochars can adsorb both \({\text{NO}}_{{3}}^{ - }\) and \({\text{NH}}_{{4}}^{ + }\) nitrogen because of their large surface areas and presence of a range of different functional groups, consequently increasing the soil fertility and crop production. On biochar’s surface both acidic and basic sites can be found which can affect the adsorption of cations as well anions35. From the results (Table 1) in layer 0–5 cm, it can be seen that retention of \({\text{NO}}_{{3}}^{ - }\) is influenced by the different biochar types in both levels of application. Maximum \({\text{NO}}_{{3}}^{ - }\) (25.0 mg kg−1) was adsorbed in treatment with CSB. The increase in dose of biochars increases \({\text{NO}}_{{3}}^{ - }\) retention in soil. Minimum \({\text{NO}}_{{3}}^{ - }\) was found in PLB (3.0 mg kg−1) and control (5.5 mg kg−1) treatment respectively. The addition of N fertilizer had no significant effect on \({\text{NO}}_{{3}}^{ - }\) retention in top 5 cm soil while an increase in \({\text{NO}}_{{3}}^{ - }\) was observed when N was applied as \({\text{NH}}_{{4}}^{ + }\) form. The N application to soil decreased the \({\text{NO}}_{{3}}^{ - }\) in columns with CMB, SSB and CSB both in 10 as well as 20 Mg ha−1. Data on soil layer 5–10 cm (Table 2) shows that increase in depth of soil decreased the \({\text{NO}}_{{3}}^{ - }\) retention in soil both in different biochar types as well as doses of biochar’s. There was no effect was noted among different biochar types, even with different doses of biochar’s i.e., 10 and 20 Mg ha−1. The N application to crops also didn’t affect the \({\text{NO}}_{{3}}^{ - }\) in soil after harvest. Maximum \({\text{NO}}_{{3}}^{ - }\) (8.0 mg kg−1) was observed in soil column amended with SMB at 20 Mg ha−1 while Minimum (1.1 mg kg−1) was observed in soil column treated with SSB at 20 Mg ha−1. No \({\text{NO}}_{{3}}^{ - }\) was found in deeper layers i.e., 10–15 and 15–10 cm (Tables 3 and 4) even an application of N fertilizer to an acidic soil had no effect on soil \({\text{NO}}_{{3}}^{ - }\) contents under no tillage conditions. Presence of \({\text{NO}}_{{3}}^{ - }\) in soil layer 0–5 cm confirms the nitrification process occurs in topsoil which was mixed with biochar (2.5–3 cm) based on the great volumes of the plant residues derived biochars. The application of alkaline biochar with high adsorptive capability adsorbs \({\text{NO}}_{{3}}^{ - }\) and \({\text{NH}}_{{4}}^{ + }\) and hence reduce N loss from soil36. Biochars being porous and high exchange nature material can adsorb more nutrients, enhancing the soil nitrogen contents37,38. A high amount of nitrogen is attached by biochars when they are applied in high rates in soil39. The \({\text{NH}}_{{4}}^{ + }\) contents in soil were not affected by different biochar types after crop harvest (Table 1), even increase in dose of biochar’s had no significant effect in \({\text{NH}}_{{4}}^{ + }\) retention in soil in topsoil layer (0–5 cm). A slight increase in \({\text{NH}}_{{4}}^{ + }\) was observed with increase in dose of each biochar. Ammonium content in soil was also influenced directly with application of \({\text{NH}}_{{4}}^{ + }\) fertilizer in soil in wheat crop under no tillage system. The N application increased the \({\text{NH}}_{{4}}^{ + }\) retention in soil while the dose of biochar had not a significant effect on \({\text{NH}}_{{4}}^{ + }\) retention in top 5 cm soil layer. Maximum \({\text{NH}}_{{4}}^{ + }\) (67.6 mg kg−1) was observed with application of CSB at 20 Mg ha−1 whereas minimum was observed in control treatment (control with N application). The most important biochar physical property to retain \({\text{NH}}_{{4}}^{ + }\) and NH3 is the surface area and pore structure. The NH3 also act as Lewis’s acid that could react with carboxyl groups pf biochar and produce \({\text{NH}}_{{4}}^{ + }\) or amide group40. However, NH3 being an alkaline gas, the acidic surface groups on biochar with low pH can protonate NH3 gas to \({\text{NH}}_{{4}}^{ + }\) ions thereby promoting their adsorption onto the cation exchange sites of biochar41 hence reducing the \({\text{NH}}_{{4}}^{ + }\) loss through NH3. In soil layer 5–10 cm decreased \({\text{NH}}_{{4}}^{ + }\) content as compared to top 5 cm soil, the decrease in \({\text{NH}}_{{4}}^{ + }\) concentration shows the weak influence of surface application of biochar’s derived from animal manures and plant residues. Resaee et al.42 noted that biochars with higher O/C ratio can have more \({\text{NH}}_{{4}}^{ + }\) adsorption as compared to biochars with less O/C, likewise Wang et al.43 found a direct relationship between functional groups and \({\text{NH}}_{{4}}^{ + }\) adsorption. An increase can be seen with increase in dose of biochar’s but there was not statistically (p < 0.05) significant difference found between the two doses of biochars. The application of N to soil also didn’t affect the \({\text{NH}}_{{4}}^{ + }\) in soil up to 10 cm depth. A decrease and slight increase can be observed in both doses of biochar’s for example, in soil column SMB had \({\text{NH}}_{{4}}^{ + }\) contents 15.7 mg kg−1 at 10 Mg ha−1 that decreased with 20 Mg ha−1 to 6.6 mg kg−1 while in case of CSB increased from 9.3 mg kg−1 to 12.2 mg kg−1 with increase in dose of biochar. In both control treatments (with N and without N) \({\text{NH}}_{{4}}^{ + }\) was almost same 15.5 mg kg−1 without N and 14.6 m kg−1 with N application. As compared to \({\text{NO}}_{{3}}^{ - }\), the \({\text{NH}}_{{4}}^{ + }\) was found continuously up to 25 cm layers collections (Tables 3 and 4), but with the increase in soil depth the concentration also remained gradually decreasing. There are number of studies showing that the addition of biochars lower the loss of \({\text{NO}}_{{3}}^{ - }\) through leaching and increase its concentrations in soil were for short period of time while long term experiments were still overlooked. According to Coa et al.44, the inclusion of biochar improved \({\text{NO}}_{{3}}^{ - }\) retention in the early phases of the experiment, while \({\text{NO}}_{{3}}^{ - }\) N loss by leaching increased in the later stages. Kameyama et al.45 reported adsorption of \({\text{NO}}_{{3}}^{ - }\) primarily caused by base functional groups rather than physical sorption thus biochar and NO3−adsorption relationship is weak. On the other hand, the \({\text{NO}}_{{3}}^{ - }\) adsorption may be attributed to electrostatic interactions and ion exchange phenomena46. The increase in dose of biochar’s had also a little influence in \({\text{NH}}_{{4}}^{ + }\) contents whereas there was not a significant between doses of biochar’s, even in case of SSB and CSB the \({\text{NH}}_{{4}}^{ + }\) content decreased 78 and 38% respectively with increase in dose of biochar (Table 3). With increase in soil depth the \({\text{NO}}_{{3}}^{ - }\) contents decreased and in final 2 layers (10–15 and 15–25 cm) no \({\text{NO}}_{{3}}^{ - }\) was noted that can be directly attributed to the no tillage soil conditions that we couldn’t mix the soil and biochar at grater depths. The available P remained changing with increase in depth, in top 0–5 cm layer P was influenced with biochar types as well as the increase in dose of biochar’s under no tillage system. Highest P (177.9 mg kg−1) was found in soil column treated with CMB at 10 Mg ha−1 while minimum (33.7 mg kg−1) was observed in control (no biochar, no N). The addition of N fertilizer enhanced the P retention in soil in all treatments with 20 Mg ha−1 except the soil treated with SMB where the addition of N fertilizer decreased the P content in soil i.e., 247 mg kg−1 without N and 206.6 mg kg−1 with the addition of N, while the P contents remained non-significant with biochar’s dose at 10 Mg ha−1 with N application together. In control treatments addition of N also increased the P retention in soil. With increase in depth, decrease in available P (Table 2) was observed but among different biochar types, no difference was observed when applied at 10 and 20 Mg ha−1 without N fertilizer while a huge increase was noted in column treated with SMB at 20 Mg ha−1 as compared to 10 Mg ha−1. The addition of N fertilizer also had not a significant impact on available P contents between 5 and 10 cm depth. In control treatments, no difference was found with and without application of mineral N fertilizer. As compared to topsoil layers 0–5 and 5–10 cm, the available P in the subsoil layers (10–15 and 15–25 cm) was remained uninfluenced with different biochar types, doses of biochar’s as well as in combination with mineral N fertilizer (Tables 3 and 4), while a minute different among treatments and doses can be noted. In the acidic soils, the P sorption is higher than the neutral or alkaline soil because of its low pH and Fe, Al and Mn oxides are dominant at low pH and fix P and reduce its availability47. Addition of biochar’s in low pH soil can decrease the soil pH and increase available P in soil solution by the increase in negative charged surfaces and pH may be increased by proton consumption reaction and hence forming hydro-oxides of Al and Fe. The biochar types, doses of biochar’s as well as combination of N strongly affected the available K in soil after the crop harvest. The available K ranged from 15 to 248 mg kg−1 affected by control (no biochar, no N) and RSB respectively without an application of N fertilizer (Table 1). The application of N decreased the k retention significantly in both 10 and 20 Mg ha−1. With increase in soil depth the available K concentration decreased while the influence of different biochar’s on K remained significant among different biochar types, doses, and combination of N. in sublayer 5–10 cm highest K (267.7 mg kg−1) was observed in soil column treated with RSM at 20 Mg ha−1 while minimum (14.3 mg kg−1) was observed in control treatment (no biochar, no N) (Table 2). From the data (Tables 3 and 4) similar behavior has been observed that with increase in biochar dose the K content increases while the addition of mineral N fertilizer decreases the K contents in soil.
Soil pH and Al alteration
Biochar pH ranges from 5.5 to 10.5, that depends on content and composition of the mineral fractions that may be different depending upon the feedstock and pyrolysis48,49,50,51,52 That’s why biochar can alter the \({\text{NH}}_{{4}}^{ + }\) and \({\text{NO}}_{{3}}^{ - }\) dynamic in soil system through their adsorptive properties and pH. Soil pH was greatly influenced by the addition of biochar’s alone, and along with \({\text{NH}}_{{4}}^{ + }\) fertilizer (Fig. 1A, B, C and D). The addition of biochar’s at 10 Mg ha−1 increased the soil pH sufficiently, highest soil pH was observed from the soil column treated with CSB, SSB, PLB and RSB as well in layer 0–5 cm. With increase in soil depth pH also decreased gradually even CSB, SSB and PLB decreased in layer 5–10 cm and remained decreased up to 15–25 cm (Fig. 1A). Increased dose of biochar’s also increased the soil pH drastically. Soil treated with RSB at 20 Mg ha−1 showed maximum soil pH, whereas SSB didn’t increase the soil pH with an increase in its dose (Fig. 1B). On the other hand, RSB decreased the soil pH in sublayer (15–25 cm) again up to an acidic level. Application of ammonium fertilizer also had an influence on soil pH in layer 0–5 cm, because in addition to NH4 fertilizer pH was increased up to a certain level (PLB, SMB), after that level then decreased quickly in 5–10 cm layer at 10 Mg ha−1 while PLB remained in slow decrease as compared to SMB at 20 Mg ha−1. In deeper soil layers there are not significant differences can be noted but the pH remained decreasing with increase in soil depth. The pH increase in surface layer can be related to the presence of biochar’s negatively charged phenolic, carboxyl and hydroxyl groups on surface of biochar which tend to bind H+ from soil solution by reducing soil H+ and hence increase in pH53,54. The pH increment increases the CEC by reducing the base cations leaching in competition H+ ions55. In our studies the biochar affected the only surface layer while underneath layers were not affected directly with addition of biochar even at 20 Mg ha−1. The addition of \({\text{NH}}_{{4}}^{ + }\) as fertilizer in soil decreases the soil pH whereas an increase occurs with application of biochar to an acid soil56 A huge gradient can be seen by addition of different biochar’s in soil under undisturbed soil (no tillage system). Minimum exchangeable Al was observed in soil layer 0–5 cm (Fig. 2A, B, C, D), that kept it increasing with increase in soil depth. Lowest exchangeable Al was observed in SSB at both 10 and 20 Mg ha−1. The addition of ammonium fertilizer didn’t influence the exchangeable Al content in an acidic soil under no tillage system while the influence of amendment was limited to a very shallow depth (5 cm), after that remained increasing and reached near to its original Al content in both 10 and 20 Mg ha−1. The addition of biochar’s increases the alkaline metals (Ca2+, Mg2+ and K+) oxides in acidic soil and hence soluble Al3+ reduces by an increase in pH3,57,58,59,60,61.
Conclusion
Surface application of different biochar can have a limited impact on soil nutrients especially to an acidic soil. The biochar’s had a significant effect up to 5 cm soil depth by retaining \({\text{NO}}_{{3}}^{ - }\) while can hold higher quantities of \({\text{NH}}_{{4}}^{ + }\) up to more depths under no tillage system. Phosphorus can be adsorbed by biochar’s when applied at surface while in deeper layers biochar’s don’t influence the P retention in soil. Potassium is greatly influenced with surface application of biochar’s but decrease the K retention in soil with application of \({\text{NH}}_{{4}}^{ + }\) fertilizer together. Soil pH and exchangeable Al also can have a prodigious positive impact up to a certain depth with superficial application of biochar’s that may not have an impact in depth.
Data availability
All data generated or analyzed during this study are included in this published article.
References
Cui, C. et al. Bioremediation of phenanthrene in saline-alkali soil by biochar-immobilized moderately halophilic bacteria combined with Suaeda salsa L. Sci. Total Environ. 880, 163279 (2023).
Liu, Z. et al. Phosphate-solubilizing microorganisms regulate the release and transformation of phosphorus in biochar-based slow-release fertilizer. Sci. Total Environ. 869, 161622 (2023).
Tammeorg, P., Kalu, S., Karhu, K. & Simojoki, A. Long-term effects of softwood biochars on boreal soils: results from two experiments through 13 years on soils, nutrient cycling and crops (2023) https://doi.org/10.5194/egusphere-egu23-9707.
Javeed, H. M. R. et al. Biochar and arbuscular mycorrhizae fungi to improve soil organic matter and fertility. In Sustainable Agriculture Reviews 61: Biochar to Improve Crop Production and Decrease Plant Stress under a Changing Climate 331–354 (Springer, 2023).
Gunamantha, I. M. & Widana, G. A. B. Characterization the potential of biochar from cow and pig manure for geoecology application. In IOP Conference Series: Earth and Environmental Science vol. 131 (Institute of Physics Publishing, 2018).
Patel, M. R. & Panwar, N. L. Biochar from agricultural crop residues: Environmental, production, and life cycle assessment overview. Resour. Conserv. Recycl. Adv. 200173 (2023).
Liang, S. et al. Combination of biochar and functional bacteria drives the ecological improvement of saline-alkali soil. Plants 12, 284 (2023).
Brtnicky, M. et al. Pre-activated biochar by fertilizers mitigates nutrient leaching and stimulates soil microbial activity. Chem. Biol. Technol. Agric. 10, 57 (2023).
Jahan, S., Iqbal, S., Rasul, F. & Jabeen, K. Evaluating the effects of biochar amendments on drought tolerance of soybean (Glycine Max L.) using relative growth indicators. Pak. J. Bot. 54, 1629–1641 (2022).
Kakar, H., Memon, M., Rajpar, I. & Chachar, Q. Comparative role of Dwarf Cavendish banana leaf formulated biochar and compost in corn—nutrient uptake and its relationship with yield. Pak. J. Bot. 54, 2303–2309 (2022).
Aslam, M. A. et al. Effects of biochar and zeolite integrated with nitrogen on soil characteristics, yield and quality of maize (Zea mays L.). Pak. J. Bot. 53, 2047–2057 (2021).
Ibrahim, M. E. H. et al. Mitigation effect of biochar on sorghum seedling growth under salinity stress. Pak. J. Bot. 53, 387–392 (2021).
Jabborova, D. et al. Growth of spinach as influenced by biochar and Bacillus endophyticus IGPEB 33 in drought condition. Pak. J. Bot. 55, 53–59 (2023).
Ashraf, F. & Chen, Y. Synergistic effects of biochar and arbuscular mycorrhizal fungi on enhancing Elymus elymoides growth in saline coastal soil. Pak. J. Bot. 55, 119–126 (2023).
Sheikh, L. et al. Evaluating the effects of cadmium under saline conditions on leafy vegetables by using acidified biochar. Pak. J. Bot. 55, 33–39 (2023).
Qian, L., Chen, B. & Hu, D. Effective alleviation of aluminum phytotoxicity by manure-derived biochar. Environ. Sci. Technol. 47, 2737–2745 (2013).
Wang, L. et al. Effect of crop residue biochar on soil acidity amelioration in strongly acidic tea garden soils. Soil Use Manag. 27, 119–128 (2014).
Prommer, J. et al. Biochar decelerates soil organic nitrogen cycling but stimulates soil nitrification in a temperate arable field trial. PLoS One 9, e86388 (2014).
Pandit, N. R. et al. Biochar improves maize growth by alleviation of nutrient stress in a moderately acidic low-input Nepalese soil. Sci. Total Environ. 625, 1380–1389 (2018).
Che, J., Zhao, X. Q., Zhou, X., Jia, Z. J. & Shen, R. F. High pH-enhanced soil nitrification was associated with ammonia-oxidizing bacteria rather than archaea in acidic soils. Appl. Soil Ecol. 85, 21–29 (2015).
Yi, X. et al. Silicon improves cotton growth and photosynthesis under low nitrogen by regulating photosynthetic nitrogen use efficiency. Plant Growth Regul. 89, 249–257 (2019).
Cardenas, L. M. et al. Nitrogen use efficiency and nitrous oxide emissions from five UK fertilised grasslands. Sci. Total Environ. 661, 696–710 (2019).
Yi, X. et al. Silicon enhances the nitrogen use efficiency of rice by regulating the activities of nitrogen metabolism enzymes. J. Plant Nutr. Soil Sci. 183, 649–657 (2020).
Karan, S. K., Osslund, F., Azzi, E. S., Karltun, E. & Sundberg, C. A spatial framework for prioritizing biochar application to arable land: A case study for Sweden. Resour. Conserv. Recycl. 189, 106769 (2023).
Wang, J. Soil Chemistry of Plant Nutrition (University Press, 1995).
De Boer, W., Tietema, A., Gunnewiek, P. J. A. K. & Laanbroek, H. J. The chemolithotrophic ammonium-oxidizing community in a nitrogen-saturated acid forest soil in relation to ph-dependent nitrifying activity. Soil Biol. Biochem. 24, 229–234 (1992).
Cole, C. V. et al. Global estimates of potential mitigation of greenhouse gas emissions by agriculture. Nutr. Cycl. Agroecosyst. 49, 221–228 (1997).
Kumar, K. S., Tiwari, K. N. & Jha, M. K. Design and technology for greenhouse cooling in tropical and subtropical regions: A review. Energy Build. 41, 1269–1275 (2009).
Mankasingh, U., Choi, P. C. & Ragnarsdottir, V. Biochar application in a tropical, agricultural region: A plot scale study in Tamil Nadu, India. Appl. Geochem. 26, S218–S221 (2011).
Cardoso, I. M. & Kuyper, T. W. Mycorrhizas and tropical soil fertility. Agric. Ecosyst. Environ. 116, 72–84 (2006).
Lou, K., Rajapaksha, A. U., Ok, Y. S. & Chang, S. X. Pyrolysis temperature and steam activation effects on sorption of phosphate on pine sawdust biochars in aqueous solutions. Chem. Speciat. Bioavailab. 28(1–4), 42–50 (2016).
Kongthod, T., Thanachit, S., Anusontpornperm, S. & Wiriyakitnateekul, W. Effects of biochars and other organic soil amendments on plant nutrient availability in an ustoxic quartzipsamment. Pedosphere 25(5), 790–798 (2015).
Ahmad, M. et al. Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 99, 19–33 (2014).
Calinski, T., Steel, R. G. D. & Torrie, J. H. Principles and procedures of statistics: A biometrical approach. Biometrics 37, 859 (1981).
Joseph, S. D. et al. An investigation into the reactions of biochar in soil. Soil Res. 48, 501–515 (2010).
Chen, C. R. et al. Impacts of greenwaste biochar on ammonia volatilisation from bauxite processing residue sand. Plant Soil 367, 301–312 (2013).
Sarfaraz, Q. et al. Characterization and carbon mineralization of biochars produced from different animal manures and plant residues. Sci. Rep. 10, 955. https://doi.org/10.1038/s41598-020-57987-8 (2020).
Ahmed, R. et al. Biochar effects on mineral nitrogen leaching, moisture content, and evapotranspiration after 15N urea fertilization for vegetable crop. Agronomy https://doi.org/10.3390/agronomy9060331 (2019).
Zhang, M., Liu, Y., Wei, Q. & Gou, J. Biochar enhances the retention capacity of nitrogen fertilizer and affects the diversity of nitrifying functional microbial communities in karst soil of Southwest China. Ecotoxicol. Environ. Saf. https://doi.org/10.1016/j.ecoenv.2021.112819 (2021).
Spokas, K. A., Novak, J. M. & Venterea, R. T. Biochar’s role as an alternative N-fertilizer: Ammonia capture. Plant Soil 350, 35–42 (2012).
Bandosz, T. J. Chapter 5 Desulfurization on activated carbons. Interface Sci. Technol. 7, 231–292 (2006).
Rezaee, M., Saeed, G. & Sarrafzadeh, M.-H. Evaluation of phosphate and ammonium adsorption desorption of slow pyrolyzed wood biochar. Environ. Eng. Manag. J. 20(2) (2021).
Wang, Z. et al. Biochar produced from oak sawdust by Lanthanum (La)-involved pyrolysis for adsorption of ammonium (NH4+), nitrate (NO3−), and phosphate (PO43−). Chemosphere 119, 646–653 (2015).
Cao, T., Meng, J., Liang, H., Yang, X. & Chen, W. Can biochar provide ammonium and nitrate to poor soils?: Soil column incubation. Journal of soil science and plant nutrition 17(2), 253–265 (2017).
Kameyama, K., Miyamoto, T., Shiono, T. & Shinogi, Y. Influence of sugarcane bagasse-derived biochar application on nitrate leaching in calcaric dark red soil. J. Environ. Qual. 41(4), 1131–1137 (2012).
Jassal, R. S. et al. Nitrogen enrichment potential of biochar in relation to pyrolysis temperature and feedstock quality. J Environ. Manag. 152, 140–144 (2015).
Geelhoed, J. S., Hiemstra, T. & Van Riemsdijk, W. H. Phosphate and sulfate adsorption on goethite: Single anion and competitive adsorption. Geochim. Cosmochim. Acta 61, 2389–2396 (1997).
Zhu, L. et al. A review of biochar derived from pyrolysis and its application in biofuel production. SF J. Mater. Chem. Eng. 1 (2018).
Jassal, R. S. et al. Nitrogen enrichment potential of biochar in relation to pyrolysis temperature and feedstock quality. J. Environ. Manag. 152, 140–144 (2015).
Bruun, E. W., Ambus, P., Egsgaard, H. & Hauggaard-Nielsen, H. Effects of slow and fast pyrolysis biochar on soil C and N turnover dynamics. Soil Biol. Biochem. 46, 73–79 (2012).
Zwieten, L. V. et al. Effects of biochar from slow pyrolysis of papermill waste on agronomic performance and soil fertility. Plant Soil 327, 235–246 (2009).
Ye, Z., Liu, L., Tan, Z., Zhang, L. & Huang, Q. Effects of pyrolysis conditions on migration and distribution of biochar nitrogen in the soil-plant-atmosphere system. Sci. Total Environ. 723, 138006 (2020).
Chintala, R., Mollinedo, J., Schumacher, T. E., Malo, D. D. & Julson, J. L. Effect of biochar on chemical properties of acidic soil. Arch. Agron. Soil Sci. 60, 393–404. https://doi.org/10.1080/03650340.2013.789870 (2013).
Wang, Z., Yang, Z., Liu, L., Ye, Y. & Xie, X. Electron transfer mechanism of chitosan-modified natural manganese ore-cornstalk biochar composites with activated peroxymonosulfate: The role of functional groups on the surface of biochar-based composites. Sep. Purif. Technol. 302, 122107 (2022).
Gul, S., Whalen, J. K., Thomas, B. W., Sachdeva, V. & Deng, H. Physico-chemical properties and microbial responses in biochar-amended soils: Mechanisms and future directions. Agric. Ecosyst. Environ. 206, 46–59 (2015).
Li, W., Feng, X., Song, W. & Guo, M. Transformation of phosphorus in speciation and bioavailability during converting poultry litter to biochar. Front. Sustain. Food Syst. 2, 374321 (2018).
Estefan, G., Sommer, R. & Ryan, J. Methods of Soil, Plant, and Water Analysis: A Manual for the West Asia and North Africa Region (International Center for Agricultural Research in the Dry Areas, 2013).
Schmidt, H.-P. et al. Biochar and biochar-compost as soil amendments to a vineyard soil: Influences on plant growth, nutrient uptake, plant health and grape quality. Agric. Ecosyst. Environ. 191, 117–123 (2014).
Agbede, T. M. et al. Assessing the synergistic impacts of poultry manure and biochar on nutrient-depleted sand and sandy loam soil properties and sweet potato growth and yield. Exp. Agric. 58, e54 (2022).
Major, J., Steiner, C., Downie, A. & Lehmann, J. Biochar effects on nutrient leaching. In Biochar for Environmental Management 303–320 (Routledge, 2012).
Gelardi, D. L. et al. Biochar alters hydraulic conductivity and impacts nutrient leaching in two agricultural soils. Soil 7, 811–825 (2021).
Acknowledgements
Authors are thankful to the Brazilian Council for Scientific and Technological Development (CNPq) and the Brazilian Federal Agency for Support and Evaluation of Graduate Education (CAPES) for providing scholarships and the laboratory equipment for analysis (Financing Code 001) through The World Academy of Sciences (TWAS). The authors extend their appreciation to the Researchers Supporting Project number (RSPD2024R1091), King Saud University, Riyadh, Saudi Arabia.
Funding
Authors are thankful to the Brazilian Council for Scientific and Technological Development (CNPq) and the Brazilian Federal Agency for Support and Evaluation of Graduate Education (CAPES) for providing scholarships and the laboratory equipment for analysis (Financing Code 001) through The World Academy of Sciences (TWAS). The authors extend their appreciation to the Researchers Supporting Project number (RSPD2024R1091), King Saud University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Q.S.; G.L.D.; M.Z.; contributed to the conceptualization and design of the study, as well as data collection, analysis, and interpretation. F.Z.; L.S.d.D.; S.D.; contributed to the statistical analysis and interpretation of the data. M.N.S.; A.E.-Z.M.A.M.; M.S.E.; contributed to the writing, statistical analysis and editing of the manuscript. All authors have reviewed and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sarfaraz, Q., Drescher, G.L., Zafar, M. et al. Nutrient retention after crop harvest in a typic hapludults amended with biochar types under no-tillage system. Sci Rep 14, 5126 (2024). https://doi.org/10.1038/s41598-024-55430-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-55430-w
Keywords
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.