Abstract
The inspired sinewave technique (IST) is a non-invasive method to measure lung heterogeneity indices (including both uneven ventilation and perfusion or heterogeneity), which reveal multiple conditions of the lung and lung injury. To evaluate the reproducibility and predicted clinical outcomes of IST heterogeneity values, a comparison with a quantitative lung computed tomography (CT) scan is performed. Six anaesthetised pigs were studied after surfactant depletion by saline-lavage. Paired measurements of lung heterogeneity were then taken with both the IST and CT. Lung heterogeneity measured by the IST was calculated by (a) the ratio of tracer gas outputs measured at oscillation periods of 180 s and 60 s, and (b) by the standard deviation of the modelled log-normal distribution of ventilations and perfusions in the simulation lung. In the CT images, lungs were manually segmented and divided into different regions according to voxel density. A quantitative CT method to calculate the heterogeneity (the Cressoni method) was applied. The IST and CT show good Pearson correlation coefficients in lung heterogeneity measurements (ventilation: 0.71, and perfusion, 0.60, p < 0.001). Within individual animals, the coefficients of determination average ventilation (R2 = 0.53) and perfusion (R2 = 0.68) heterogeneity. Strong concordance rates of 98% in ventilation and 89% when the heterogeneity changes were reported in pairs measured by CT scanning and IST methods. This quantitative method to identify heterogeneity has the potential to replicate CT lung heterogeneity, and to aid individualised care in ARDS.
Similar content being viewed by others
Introduction
The recent coronavirus pandemic caused > 6.5 million recognized deaths worldwide and highlighted the impact that severe acute respiratory distress syndrome (ARDS) has on healthcare systems1. Mechanical ventilation is a life-saving therapy required by nearly half of ARDS patients. However, with the exception of some basic ground rules, there is no clear guidance for nuanced ventilatory management in ARDS2,3. Ventilator induced lung injury is thought to occur mainly through two mechanisms: atelectotrauma, the opening/collapsing of lung units, and volutrauma, the repetitive over-inflation of the lung4,5,6. To prevent ventilator-induced lung injury, endpoints by which Positive End-Expiratory Pressure (PEEP) might be titrated for individual patients remains elusive7,8. Lung heterogeneity may provide an individualised ventilatory target.
Ventilatory heterogeneity is the “uneven ventilation” or the unequal distribution of the airflow (ventilation) relative to the size of the lung regions to which it is distributed9,10. Ventilation heterogeneity and perfusion heterogeneity of the lung have been considered as key research points of lung diseases11,12,13. Computed tomography (CT) measured lung heterogeneity increases with severity of disease in ARDS14,15. Quantification of ventilatory heterogeneity has the potential to benefit patients with lung disease16,17,18. CT scanning is widely considered the gold-standard to determine lung recruitability, effective lung volume, and ventilatory heterogeneity to help determine preliminary PEEP selection19. Work by Cressoni, et al. in 148 ARDS patients showed the potential of quantitative CT image analysis to determine lung inhomogeneity. However, CT scanning is labour intensive and not without risks.
The Inspired Sinewave Technique (IST) was developed to measure deadspace volume, effective lung volume, pulmonary blood flow, and lung heterogeneity continuously and non-invasively at the bedside, without the need for patient co-operation, with an operator-independent approach, and not requiring ionising radiation20. The IST adds a forced oscillation of tracer gas into inspiratory airflow to measure cardiopulmonary parameters20,21. Additionally, the IST in conjunction with advanced lung simulation can determine ventilation and perfusion heterogeneity within the lung22. Given the limitations and risks of CT scanning, a simple and non-invasive alternative technology, like the IST, has a clear clinical need. Preliminary results of this work have been published previously23 wherein simple CT quantitative analysis, using the ratio between atelectatic and over-extended lung, was used to identify heterogeneity of the lung, and compared to IST measurements. However, this is the first study comparing Cressoni’s CT analysis with IST heterogeneity, and comparing IST with simulation-based calculations of the lung heterogeneity24.
We hypothesised that lung heterogeneity values measured by the CT imaging could be similar to the bedside IST values in the ARDS porcine model. We then further investigated the changes in lung heterogeneity with the changes in different PEEP levels to suggest that measuring lung heterogeneity at the bedside has the potential to optimise the ventilator setting to provide better support for ARDS subjects.
Methods
Ethical approval
This study was approved by Animal Research Ethics Committee at Uppsala University (Ref: C98/16). All the animal experiments conformed to the National Institutes of Health Guidelines for the Use of Laboratory Animals. We abide by the Animal Research: Reporting of In Vivo Experiments guidelines (ARRIVE)25. Previous ethical documents were included here26. All methods were performed following the relevant guidelines and regulations by ARRIVE, APPROVAL, and ACCORDANCE.
Protocol
Six anaesthetised pigs underwent surfactant depletion by saline lavage. The summary baseline data are shown in Table 1. The animals were premedicated with i.m. xylazine 2 mg/kg, ketamine 20 mg/kg, and midazolam 0.5 mg/kg27. Then, animals underwent induction of anaesthesia with i.v. propofol titrated to effect (1–3 mg/kg). The trachea was intubated. Volume-control ventilation was implemented with Servo-I, Maquet ventilator (Rastatt, Germany) at 20–25 breaths per minute, and with a tidal volume of 10 mL/kg. The inspiration:expiration ratio was 1:2. Surfactant depletion was undertaken according to the Lachmann method28. This process was repeated until the PaO2:FIO2 ratio (PFR) was < 300 mmHg at a PEEP level of 5 cmH2O and FIO2 0.7.
During the experiments, the PEEP level was incrementally increased from 5 through to 10, 15 and 20 cmH2O and decreased back to 15, 10 and 5 cmH2O. At each PEEP level, two paired measurements of lung volume by the IST and CT were performed. Supplementary Figure A1 shows a summary timeline of each measurement.
Lung heterogeneity measured by the IST
The IST uses a forced oscillation of a low-dose tracer gas (N2O) in the inspired air with a set sinusoidal period20,22,26,29,30. Measurements using IST were made using sinusoid periods of both 60 s and 180 s. By measuring the system output (in the expired gas), compared to the forced input signal, and applying mass conservation principles, the IST analysis allows the calculation of the lung parameters. Recoverable variables of IST are deadspace volume (VD), (volume not involved in gas exchange); effective lung volume (ELV), (the apparent volume of the alveolar compartment, assuming it to be heterogeneously ventilated); pulmonary blood flow (Qp), and lung heterogeneity index (H), which quantifies the heterogeneity of the lung, as both ventilation and perfusion per unit lung compartment volume.
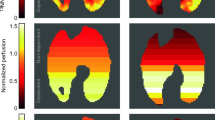
In this study, we focus upon two methods to measure heterogeneity by the IST. A schematic diagram of both calculations is presented in Fig. 1A and B. The conventional IST method exploits the fact that errors in the ELV estimated by the IST are increasingly frequency-dependent as heterogeneity increases. The ratio of the ELV outputs at 60 s and 180 s oscillation periods will therefore deviate from 1.0 as heterogeneity increases, in Fig. 1A. Further details of this method are discussed here30. In this method the IST heterogeneity ratios (\({H}_{IST} ratios\)) are calculated by the ratios of the ELV180/ELV60 for ventilation, and Qp60/Qp180 for perfusion.
Methods to measure lung heterogeneity by IST and CT images. Panel (A) shows the working principle of the IST, using N2O tracer-gas and applied an inversed model to calculate pulmonary-vascular parameters. Panel (B) shows a schematic diagram of a method to optimise the heterogeneity simulation results versus measured IST results to calculate the ventilation/perfusion heterogeneity. Panels (C) and (D) illustrate two quantitative methods to calculate the heterogeneity index from CT imaging scans. Panel (C) shows compare surrounding voxel method (Cressoni’s method) and panel (D) shows the classification method based on Hounsfield unit ranges.
In the second method, the IST heterogeneity indices are calculated using a multi-compartment simulation model, in Fig. 1B. The same oscillatory signals that are fed into the animal are input into the simulation which is a multi-compartment model with log-normally distributed specific ventilations and perfusions, to represent the real lung. The optimisation process adjusts this simulation such that its output best matches the output from the experimental animal. The simulation-based optimisation therefore recovers the log-normal distribution of specific ventilation and perfusion of the lung. Then, the width of these distributions within the simulation (the log standard deviations) will now represent lung heterogeneity. LogSDv presents the ventilatory heterogeneity and LogSDp is the perfusion heterogeneity (called the HIST LogSD indices). Further details and mathematical equations of the optimisation process are presented in24, which could be summarised in the optimisation of the loss function:
where \(y_{180s}\) is the IST results measurement at 180 s, similar to the \(y_{60s}\), \(\hat{y}\left( {LogSD} \right)\) is the lung simulation controlled by the \(LogSD \) in both perfusion and ventilation.
Measurement of the lung heterogeneity using CT scan
A Somatom Definition Flash (Siemens, Munich, Germany) was used to acquire images as a series of transverse sections with a reconstituted voxel size of 0.5 × 0.5 × 5 mm. We conducted a 20 s end-expiratory pause without ventilator disconnection to scan the whole-lung volume. The scans were segmented by 3D Slicer version 4.10.2 (https://www.slicer.org). Segmentation does not include the mediastinum, diaphragm, inferior vena cava, and hilar vessels. Lung CT images were divided into four different functional compartments31:
-
Over-distended volume: − 1000 to − 901 HU
-
Normally aerated volume: − 900 to − 501 HU
-
Poorly aerated lung: − 500 to − 101 HU
-
Atelectasis or Collapse: − 100 to + 100 HU
An example is presented in Fig. 1D. The mass of each lung fraction (e. g., collapse) was then calculated using the mean density and volume of each fraction assuming that the lung is composed solely of air and water. In previous work23, we compared a simple lung heterogeneity index using the ratio between atelectasis and over-distended volume to IST ratio indices. However, since there is a lack of clinical validation for this latter CT metric, we applied Cressoni’s method to quantify the inhomogeneity of the lung14.
According to Cressoni’s method, validated in 148 ARDS human subjects, the heterogeneity of a single voxel was measured by calculating based on one voxel’s HU density compared to the density of the voxels surrounding it, from above R1 = 2.41 mm and R2 = 3.675 mm, Fig. 1C14. According to Cressoni, et al., CT-derived lung inhomogeneity (termed HCT in this paper) could be calculated by a ratio of pathologic regions (index showing a value greater than the 95th percentile of the control group—1.61) versus normal regions (index less than 1.61) 14.
Statistical analysis
Statistical analyses were performed in GraphPad Prism 9 (GraphPad Software, La Jolla, CA, USA; https://www.graphpad.com/). Pearson’s correlation highlights the primary results by identifying the correlation between paired repeated measurements of HIST, and HCT. After that, linear regression analysis was applied to assess the agreement between lung heterogeneity measured with IST (HIST) and that measured with CT (HCT). For repeated measurements at the same PEEP level, average values were calculated. The changes in lung heterogeneity (ΔH), throughout the different levels of applied PEEP, were reported by the four-quadrant plot32. ΔH was calculated by subtracting one absolute HIST(or HCT) from its value at the preceding PEEP level. The distribution of the data was tested for the normal distribution, as well as, and the mean and standard deviation that were reported.
Results
We obtained a total of n = 84 paired measurements, including both IST and CT scans at four incremental and decremental PEEP levels. The changes in absolute values and changes in lung heterogeneity were captured and analysed. The changes in VD/ELV and VD/VT ratios are also reported. The IST heterogeneity measurements had good repeatability, with variations in the same condition being less than 10%.
Figure 2A shows the Pearson’s correlation and p values of heterogeneity results in both CT scanning and IST methods. There was a strong correlation between HCT and both ventilation and perfusion heterogeneity measured by the IST. Ventilation heterogeneity (either measured by the ratio, r = 0.60, p < 0.001, or the LogSDv, r = 0.71, p < 0.001) had stronger correlations to the HCT than perfusion heterogeneity (measured by the ratio, r = 0.10, p > 0.05, or LogSDp, r = 0.49, p < 0.001). Interestingly, in the method using LogSD, both ventilation and perfusion heterogeneity correlated with HCT (p < 0.001). However, in the ratio method, only the ventilation ratio correlated with the CT (p < 0.001).
The correlation matrix of the lung heterogeneity was measured by the CT scan versus IST values (panel A) and the heterogeneity values vs PEEP changes (panels B, C and D). In panel (A), Pearson correlation values are in the lower triangular and the p-values are in the upper triangular. A/N is Atelectasis over Normally aerated volume; O/N is Over-distended over Normally aerated volume; HIST is the IST Heterogeneity including v—ventilation and p—perfusion; HCT is the Cressoni’s Heterogeneity. Panels (B) and (C) show mean and standard deviation values of the ratio between atelectasis and normally aerated lung against PEEP, and the overextended volume over the normally aeriated ratio against PEEP. Panel D shows the HCT versus PEEP.
Furthermore, to validate Cressoni’s score (HCT), we also compared its correlation to the aerated and over-extended volumes/normal aerated volumes. HCT correated well with both (A/N, r = 0.98 and O/N, r = 0.80, p < 0.001) in Fig. 2A. Figure 2B–D show the overall influence of PEEP on heterogeneity measured by CT images. The difference between incremental and decremental PEEP levels was negligible (continuous lines vs. dash lines in Fig. 2B–D, differences were < 10% of the mean values). When PEEP increased, the lung became more homogeneous. Also, both the ratios of atelectasis: normal aerated lung (2C) and over-extended: normal aerated lung (2D) decreased when PEEP increased. A similar result was seen in HCT.
Figure 3 shows the impacts of PEEP levels on IST results. Increasing PEEP led to a more homogeneous lung (HIST LogSDv was from 1.0 to 0.4 and HIST LogSDp was from 1.5 to 1.2), in panel b. However, for the HISTratio (in panel a) where the ventilation heterogeneity decreased with increasing PEEP, perfusion heterogeneity worsened. The ventilation and perfusion heterogeneity vs. PEEP plots are seen to cross over at about PEEP 12 cmH2O. Furthermore, in Fig. 3C and D, when PEEP levels rose, VD/ELV ratios decreased and VD/VT ratios increased with negligible variation between incremental and decremental step changes in PEEP.
The effect of PEEP on various IST-derived parameters. Panels (A) and (B) show lung heterogeneity measured by the IST using both ratio and LogSD methods versus PEEP. Solid lines are incremental and dashed lines are decremental PEEP levels. Panels (C) and (D) show the changes in VD/ELV and VD/VT ratios versus PEEP. Mean and standard deviation are plotted; black solid lines are incremental and dashed lines are decremental PEEP levels.
Figure 4 shows the colour-coded scatterplots and linear regression comparison of the lung heterogeneity between IST and CT scanning methods in each animal (numbered 1–6). The full report of the linear regression in individual pigs is presented in Table 2. Due to the contribution of the PEEP level and characteristics of different animals, the correlation of mixed regression results was generally lower than the regression results in individual animals. In individual animals, there was a strong correlation between HIST and HCT. For example, the mixed regression of HIST LogSDv versus HCT had R2 = 0.50, but in individual animals, the highest recorded, R2 = 0.87 was found in animal number 6. Additionally, the HIST LogSD had a stronger correlation with HCT values than the HIST ratios. In the perfusion heterogeneity comparison (Fig. 4B,D), the results of HIST ratios perfusion did not correlate with the HCT, while there was only a weak correlation between HIST logSDp and HCT.
Figure 5 shows the four quadrant plots demonstrating the relationship of the changes in heterogeneity in response to a change in PEEP, measured by CT and IST. These comparisons show strong concordances. The \(\Delta {\text{H}}_{{{\text{IST}}}} {\text{ LogSD}}\) (panels c and d) showed better concordance with the \(\Delta {\text{H}}_{{{\text{CT}}}}\) than \(\Delta {\text{H}}_{{{\text{IST}}}}\) ratios (panels a and b). In panels c and d, the concordance of \(\Delta {\text{H}}_{{{\text{IST}}}} {\text{ logSD}}\) to the \(\Delta {\text{H}}_{{{\text{CT}}}}\) was 98% in ventilation and 89% in perfusion. Additionally, in the perfusion, the concordance of the \(\Delta {\text{H}}_{{{\text{IST}}}}\) ratio was 54% (R2 = 0.30), which was much lower than 89% (R2 = 0.60) of the \(\Delta {\text{H}}_{{{\text{IST}}}} {\text{ LogSD}}\).
Four quadrant plots show the changes in lung heterogeneity measured by the IST versus CT image with the changes in PEEP level. Panels (A) and (B) illustrated the comparisons of the HIST ratios method versus HCT. Panels (C) and (D) illustrated the comparisons of the HIST LogSD method versus HCT. The exclusion zone (black rectangle) is 15% of the maximum values. R2 and slope values are from linear regressions. The concordance rate is the percentage of data points, which are in the \(I\left( { + ; + } \right)\) and \(III\left( { - ; - } \right)\) quadrants, not including the exclusion zone.
Discussion
In this study, we compared lung heterogeneity measured by a bedside-deployable method (Inspired Sinewave Technique) versus CT scan images. Agreements in both absolute heterogeneity values and induced changes in heterogeneity were observed. The negative correlation of PEEP on lung heterogeneity is shown at four different PEEP levels. There was good repeatability of the IST heterogeneity measurement (less than 10% differences in the same condition). Furthermore, when comparing PEEP-induced changes in ventilation heterogeneity, IST indices were > 95% concordant with CT.
There is currently no ‘gold standard’ bedside method to quantify lung heterogeneity. However, researchers point out that the degree of ventilation heterogeneity increases with the severity of lung injury, and it is an important marker of it24,33. Normally, the airway wall expanded by the airflow is proportional to the pressure change. However, in the mismatched ventilation region, this caused a high driving pressure significantly to the airway wall, leading to the increasing shear stress between alveolus34. In this study, we quantified and compared the absolute changes in heterogeneity using both the IST and CT scan analysis. The CT scan images were processed by Cressoni’s method and used as the ‘gold standard’ for comparison. However, each of these techniques has limitations. Both the acquisition and the analysis of the CT image is labour intensive, and not without clinical risks in transporting critically ill patients to and from the scanning facility. Electrical Impedance Tomography (EIT) shows promise in imaging ventilatory heterogeneity, but it requires a robust quantitative framework to reconstruct data and obtain helpful data35. Furthermore, it is possible that the fidelity of EIT in detecting heterogeneity could be improved when combined with IST data. This approach may be a particularly useful fusion because IST alone provides quantification of ventilation distributions only in the statistical sense, whereas EIT provides spatial detail.
The measurement of lung heterogeneity is a newly developed feature of IST. The validation of pulmonary cardiovascular parameters measured by the IST was validated elsewhere29,30. In this paper, we validated two IST methods to quantify lung heterogeneity in comparison with CT scans. The ‘IST ratio’ is the ratio of ELV values obtained at tracer periods 180 and 60 s (ELV180/ELV60) and is a simple representation of the ventilation heterogeneity distribution within the lung. Similarly, Qp60/Qp180 presents the perfusion heterogeneity distribution. A further method, using lung simulation to characterise the log-normal distribution of ventilation and perfusion, shows advantages compared to the first method. Overall, compared to Cressoni’s score, the logSDv method (r = 0.71, p < 0.001) had a stronger correlation than the IST ratio of ventilation (r = 0.60, p < 0.001). Both IST heterogeneity measurements have their strengths and weaknesses. The IST ratio is quick to implement in near real-time (less than 10 min per test). However, the IST logSD method is more robust and reliable in comparison to the CT scan but requires more computing resources for the optimisation process (about 10 min in a standard office desktop—core i7, GPU NVIDIA RTX 3050 Ti). Therefore, depending on the purpose of use, both IST methods could be used for bedside monitoring applications or clinical research.
Cressoni’s method considers regional mechanical inhomogeneity on a voxel-by-voxel basis, at the level of the acinus. We have shown that Cressoni’s score correlates well with heterogeneity on a more global scale, as represented by the ratios of atelectatic: normal aeration and over-extended: normal aerated volumes on CT, Fig. 2. We performed the linear regression analysis to compare IST and CT heterogeneity, however, due to the complexity of the PEEP levels and characteristics of individual animals, the results for individual animals had a higher correlation to the mixed regression model (Fig. 4 and summary in Table 2). Although absolute values comparison between IST and CT were not well correlated, changes in heterogeneity (\(\Delta H\)) in response to a change in PEEP, measured by CT and IST have high concordance, 95% and 98% for ventilation and 54% and 89% for perfusion (Fig. 5). Since this global heterogeneity is detected by the IST, this result suggests that the IST heterogeneity is also a marker of mechanical heterogeneity through the lung parenchyma.
There is no agreed universal means of determining the best PEEP, and indeed; perhaps the concept of best PEEP itself is questionable6,7. In this paper, we present a means of determining several key lung parameters including lung volume, pulmonary blood flow and the heterogeneity of ventilation and perfusion. Consideration might be given to the concept that PEEP should be applied according to its ability to reduce heterogeneity in ventilation without compromising heterogeneity in perfusion. The study of this trade-off could be the subject of future research.
This research contains several limitations. There is no comparison between healthy versus injured lungs in this research because the experiments were performed in parallel with other experiments, which imposed time and protocol restrictions. The degree of heterogeneity between healthy and injured lungs could potentially be an interesting comparison study. The data collected in this experiment were performed in parallel with another experiment, therefore, the comparison might contain further limitations, such as FiO2 levels were not similar. FiO2 levels might be an important factor in the distribution of ventilation and pulmonary blood flow inside the lung. However, in this study, there was no significant correlation between FiO2 and the reported heterogeneity indices (Supplementary Table A1). We experimented with guidelines to obtain objective outcomes. Furthermore, we used the CT images processed by Cressoni’s method as “gold standard”. However, there are several quantitative methods to interrogate the heterogeneous lung, such as EIT36 and hyperpolarized gas MRI37. Nevertheless, this work may support useful and more practicable monitoring of ARDS patients under mechanical ventilation. Further research in ventilated patients is needed.
The IST could measure lung heterogeneity noninvasively at the bedside. In this animal study, the heterogeneity of the lung measured by the IST had a strong correlation with the heterogeneity measured with CT scan image analysis. This IST technology and approach open the possibility that PEEP may be optimised for individual patients, aiming at reducing lung heterogeneity. This study supports the implementation of the IST in ventilated patients in future clinical research.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
WHO. WHO Coronavirus (COVID-19) Dashboard.
NHLBI ARDS Network. ABOUT THE NHLBI ARDS NETWORK.
cute Respiratory Distress Syndrome Network. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N. Engl. J. Med. 342, 1301–1308 (2000).
Slutsky, A. S. & Ranieri, V. M. Ventilator-induced lung injury. N. Engl. J. Med. 369, 2126–2136 (2013).
de Prost, N., Ricard, J.-D., Saumon, G. & Dreyfuss, D. Ventilator-induced lung injury: Historical perspectives and clinical implications. Ann. Intensive Care 1, 28 (2011).
Pelosi, P. et al. Different methods to improve the monitoring of noninvasive respiratory support of patients with severe pneumonia/ARDS Due to COVID-19: An update. JCM 11, 1704 (2022).
Gattinoni, L. & Marini, J. J. In search of the holy grail: Identifying the best PEEP in ventilated patients. Intensive Care Med. 48, 728–731 (2022).
Cavalcanti, A. B. et al. Effect of lung recruitment and titrated Positive End-Expiratory Pressure (PEEP) vs low PEEP on mortality in patients with acute respiratory distress syndrome: A randomized clinical trial. JAMA 318, 1335–1345 (2017).
West. Respiratory physiology. J. Physiol. 487, 105–115 (1995).
West, J. B. Respiratory Physiology: The Essentials (Williams & Wilkins, 2012).
Rodríguez-Roisin, R. et al. Ventilation-perfusion imbalance and chronic obstructive pulmonary disease staging severity. J. Appl. Physiol. 106, 1902–1908 (2009).
Verbanck, S. A. B. et al. Ventilation heterogeneity in smokers: Role of unequal lung expansion and peripheral lung structure. J. Appl. Physiol. 129, 583–590 (2020).
Downie, S. R. et al. Ventilation heterogeneity is a major determinant of airway hyperresponsiveness in asthma, independent of airway inflammation. Thorax 62, 684–689 (2007).
Cressoni, M. et al. Lung inhomogeneity in patients with acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 189, 149–158 (2014).
Cressoni, M. et al. Opening pressures and atelectrauma in acute respiratory distress syndrome. Intensive Care Med. 43, 603–611 (2017).
Mountain, J. E. et al. Potential for noninvasive assessment of lung inhomogeneity using highly precise, highly time-resolved measurements of gas exchange. J. Appl. Physiol. 124, 615–631 (2018).
Cressoni, M. et al. Lung inhomogeneities, inflation and [18F]2-fluoro-2-deoxy-D-glucose uptake rate in acute respiratory distress syndrome. Eur. Respir. J. 47, 233–242 (2016).
Stahr, C. S. et al. Quantification of heterogeneity in lung disease with image-based pulmonary function testing. Sci. Rep. 6, 1–10 (2016).
Gattinoni, L. et al. Positive end-expiratory pressure: How to set it at the individual level. Ann. Transl. Med. 5, 1–10 (2017).
Phan, P. A., Farmery, A. & Hahn, C. Method and apparatus for measurement of cardiopulmonary function (2015).
Phan, P. A., Hahn, C. E. W. W. & Farmery, A. D. A modification of the Bohr method to determine airways deadspace for non-uniform inspired gas tensions. Physiol. Meas. 38, 107–117 (2017).
Tran, M. C., Crockett, D. C., Phan, P. A., Payne, S. J. & Farmery, A. D. A tidal lung simulation to quantify lung heterogeneity with the Inspired Sinewave Test. In 2020 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC) 2438–2441 (2020). https://doi.org/10.1109/EMBC44109.2020.9176375.
Tran, M. C. et al. Validating the inspired sinewave technique to measure lung heterogeneity compared to atelectasis & over-distended volume in computed tomography images. In ICASSP 2021–2021 IEEE International Conference on Acoustics, Speech and Signal Processing (ICASSP) I, 1010–1014 (2021).
Tran, M. C. et al. Simulation-based optimisation to quantify heterogeneity of specific ventilation and perfusion in the lung by the Inspired Sinewave Test. Sci. Rep. 11, 1–10 (2021).
Kilkenny, C., Browne, W. J., Cuthill, I. C., Emerson, M. & Altman, D. G. Improving bioscience research reporting: The arrive guidelines for reporting animal research. Animals 4, 35–44 (2013).
Crockett, D. C. et al. Validating the inspired sinewave technique to measure the volume of the ‘baby lung’ in a porcine lung-injury model. Br. J. Anaesth. 124, 345–353 (2020).
Crockett, D. C. et al. Tidal changes in PaO2 and their relationship to cyclical lung recruitment/derecruitment in a porcine lung injury model. Br. J. Anaesth. 122, 277–285 (2019).
Lachmann, B., Robertson, B. & Vogel, J. In vivo lung lavage as an experimental model of the respiratory distress syndrome. Acta Anaesthesiol. Scand. 24, 231–236 (1980).
Bruce, R. M. et al. Noninvasive cardiac output monitoring in a porcine model using the inspired sinewave technique: A proof-of-concept study. Br. J. Anaesth. 123, 126–134 (2019).
Tran, M. C. et al. Lung heterogeneity and deadspace volume in acute respiratory distress syndrome animals using the inspired sinewave test. J. Physiol. Meas. 41, 115009 (2020).
Gattinoni, L. et al. Relationships between lung computed tomographic density, gas exchange, and PEEP in acute respiratory failure. Anesthesiology 69, 824–832 (1988).
Perrino, A. C., Harris, S. N. & Luther, M. A. Intraoperative determination of cardiac output using multiplane transesophageal echocardiography: A comparison to thermodilution. Anesthesiology 89, 350–357 (1998).
Rutting, S., Chapman, D. G., Farah, C. S. & Thamrin, C. Lung heterogeneity as a predictor for disease severity and response to therapy. Curr. Opin. Physiol. 22, 100446 (2021).
Jamaati, H., Nazari, M., Darooei, R., Ghafari, T. & Raoufy, M. R. Role of shear stress in ventilator-induced lung injury. Lancet Respir. Med. 4, e41–e42 (2016).
Brazey, B., Haddab, Y. & Zemiti, N. Robust imaging using electrical impedance tomography: Review of current tools. Proc. R. Soc. A. 478, 20210713 (2022).
Frerichs, I. et al. Chest electrical impedance tomography examination, data analysis, terminology, clinical use and recommendations: Consensus statement of the TRanslational EIT developmeNt stuDy group. Thorax 72, 83–93 (2017).
Zhang, X. et al. Quantification of lung ventilation defects on hyperpolarized MRI: The Multi-Ethnic Study of Atherosclerosis (MESA) COPD study. Magn. Reson. Imaging 92, 140–149 (2022).
Acknowledgements
The authors are grateful for the important work from veterinary anaesthetists and for the technical assistance from the Hedenstierna Laboratory of Uppsala University, Sweden.
Funding
The Swedish Research Council grant to GP (2018-02438); the Swedish Heart and Lung Foundation to GP (20200877, 20200825 and 20220681) and the Alvar Gullstrand research Grant to GP (ALF-938050). National Institute for Health Research (II-LA-0214-20005 and NIHR200029) to PAP and ADF; Royal Academy of Engineering Enterprise Fellowship to PAP. Medical Research Council (MC_PC_17164) to FF; The Physiological Society (Formenti 2018) to FF.
Author information
Authors and Affiliations
Contributions
M.T., D.C., and P.P. carried out the experiments. A.F., D.C., F.F. and P.P. designed the experiments. M.T., D.C. analysed the data. D.C., M.T., T.T., and A.F. interpreted the data. F.F., R.B., D.C., T.T., S.P., G.P. and A.F. contributed to financial support. M.T. and A.F. wrote the manuscript. M.T., D.C., R.B., F.F., G.P., S.P. and A.F. critically revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tran, M.C., Crockett, D.C., Tran, T.K. et al. Quantifying heterogeneity in an animal model of acute respiratory distress syndrome, a comparison of inspired sinewave technique to computed tomography. Sci Rep 14, 4897 (2024). https://doi.org/10.1038/s41598-024-55144-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-55144-z
Keywords
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.







