Abstract
As one of the main sources of natural antioxidants, flowering plants play a role in the prevention and treatment of many diseases directly and indirectly. Honey is considered as an important nutrient in the supply of natural antioxidants, the amount of which is directly dependent on the plant origin and geographical location of the bee feeding place. The existence of valuable communities of native and endemic plant species has turned Alborz, Zagros and Azerbaijan into the most important hubs of honey production in Iran. In this study, we collected samples of honey from more than 90 regions in Alborz, Zagros and Azerbaijan during the years 2020 to 2021. We evaluated the samples using melissopalynology method and measuring the amount of antioxidant activity. The rise of antioxidant activity in honey is dependent on the abundance of some plant families as well botanical origins. The abundance of plant families Rosaceae, Amaranthaceae, Fabaceae and Asteraceae showed a higher influence on the amount of antioxidants in honey than other plant families. Also, the abundance of plant families Rosaceae and Fabaceae increased with increasing altitude. In general, the amount of antioxidant activity of honey samples shows a different percentage under the influence of ecological and geographical changes.
Similar content being viewed by others
Introduction
Today, all kinds of diseases, especially cancers, are a serious concern for modern humans in terms of their abundance, existence of different types and the response to treatment1. Cancer is a disease that can be caused by the imbalance between antioxidants and reactive oxygen species2. Therefore, a preventive action for diseases in the first place can greatly reduce the possibility of contracting this disease and also provide the possibility of treating it with less side effects and longer survival3. The key role of natural antioxidants in biological systems and their relationship with the treatment and prevention of various diseases has been the focus of nutrition and health researchers4,5. Natural antioxidants in food have shown positive effects in the prevention and treatment of many types of cancer6,7. It also prevents metabolic disorders by eliminating free radicals8,9. Antioxidants in food are generally secondary metabolites of plants, whose activity level is influenced by the chemical structure of their constituents10,11. Therefore, green plants are considered as important food sources containing natural antioxidants in the human diet12.
Honey is considered as a valuable food that is rich in natural antioxidants13. The amount of antioxidants in honey depends on several factors such as harvest season, humidity, the type of bee and most importantly, the type of plants that the bee fed on14,15,16. The vegetable origin of honey is considered the most determining parameter for the amount of antioxidants in honey17,18. Therefore, the place of honey production can determine the amount of antioxidants due to the type of plants in that area19,20. Plants have been used as food and medicine since the past, and today they are the basis of many commercial medicines21,22. Molecular studies have shown that plants have various medicinal properties due to their different chemical compounds23. Meanwhile, some plants specifically contain more antioxidants24. Plants have pharmacological effects on humans due to the production of some chemical substances, among these compounds are phenolic compounds, alkaloids and flavonoids, which are also known as secondary metabolites25,26. Medicinal plants can be used in the prevention or treatment of some diseases due to their antioxidant and phenolic compounds27.
Asteraceae is a large family of flowering plants with more than 2500 species and 1600 genera. The plants of this family have the ability to grow in most habitats due to their high adaptability and are scattered in different parts of the world except Antarctica. Among the important plants of this family, we can mention chicory, daisies and dandelion. Many plants of the Asteraceae family contain amounts of polyphenol compounds, antioxidants, and flavonoids that increase biological activity and protect the consuming organisms against oxidative stress conditions. Therefore, the plants of this family are considered as a useful source of vitamins, antioxidants, polyphenol compounds and proteins27,28. Amaranthaceae family, known as Amaranthus, which is found mainly in the form of leafy vegetables in the world, is considered as a good source of antioxidants29. The plants of this large family are not only flexible against adverse environmental conditions such as lack of water, salinity, dryness and poor soil, but also increase the amount of flavonoids, phenolic acids, vitamins and natural antioxidants30,31. This family contains more than 2000 plant species and 175 genera that are found in cold, temperate and tropical regions. A series of plants of the Amaranthaceae family can be used as herbal medicine in the world due to the presence of phytochemical properties such as phenolic compounds, terpenoids and betalains32. Fabaceae as one of the largest plant families has about 19,500 species and 770 genera33,34. The plants of this family with chemical compounds such as flavonoids, alkaloids, lectins and phenolic acids can have antioxidant, anti-inflammatory, anti-ulcer, anti-rheumatic and anti-cancer properties and activities35. Therefore, in today's world, plants of this family can be used in paint, insecticide, fuel, etc. industries36. The Rosaceae family, known as roses, is one of the largest plant families, including about 120 genera and 4000 species37.
Approximately 7800 plant taxa comprise the flora of Iran38, about 2700 of which are confined to the geographical boundaries of Iran39. The climatological diversity, history of vegetation, geographical isolation40, complex tectonic history, special soils41, and intersection of diverse phytogeographical units42 lead to the high potential of this region for diversification and make it a global center of diversity for plants43 and one of the most important distribution centers of crop wild relatives in the world44. Accordingly, a wide range of nectar and pollen producing plants were distributed in Iran which has led to the production of about 55 different types of mono-floral and polyfloral honey in Iran45, a wide range of which are classified as medical food and medical grades46. Melissopalynology is considered as an effective method of identifying and determining the abundance of plants selected by honey bees, which, along with measuring the antioxidant activity of honey, can determine the effect of plants on the antioxidant activity of honey47,48. In this study, to determine the frequency of the presence of plant families of the antioxidant activity of honey, the method of melissopalynology and the laboratory measurement of the antioxidant activity of honeys produced in the Zagros and Alborz Mountains were used. Samples (unrefined and non-clay honeys) were collected directly from beekeepers that were present in natural areas and their bees depended on the natural plants of the region. Identifying and determining the frequency of plant families that are effective in the antioxidant activity of honey showed that their distribution and frequency depend on their coordinates.
Materials and methods
Introduction of the study area
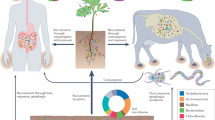
Due to its geographical and topographical conditions, Iran is home to about 8200 plant species, of which more than 2300 species are known as medicinal and aromatic plant species49. Zagros, Alborz, Kope Dagh and several other internal mountain ranges are considered as the main geomorphological regions of Iran. The Zagros mountain range extends from the northwest to the southeast, and the Alborz mountain range is located at 36 degrees north of the Caspian Sea with a height of 5671 m50. The Zagros and Alborz mountain ranges and the highlands of Azerbaijan in Iran have formed valuable plant communities with natural plant species, some of which are endemic. Alborz and Zagros are the habitats of many 2597 native vascular plant species in Iran51,52. The diverse plant communities of Zagros and Alborz and the highlands of Azerbaijan have turned them into hubs of honey production in Iran48. Therefore, these areas were chosen to collect honey samples (Fig. 1).
Sample collection
Samples were collected in 2020–2021 directly from beekeepers that were present in natural areas and their bees depended on the natural plants of the region. The geographic coordinates of each sample were recorded separately by a Garmin Map64s GPS device. Each collected sample contained 300 g of unrefined and non-clay honey from each colony. Each sample was coded separately and placed in a sterile container and transported to the laboratory at a temperature of < 20 °C. In this study, honey samples were collected from 94 different regions in Iran.
Melissopalynological analyses
Louveaux method was used in this study to analyze Melissopalynology53. The International Commission on Bee Botany (ICBB) has proposed the melissopalynological method53,54. Pollens were extracted from honey samples according to the Louveaux method. First, 10 g of honey in about 20 mL of distilled water was dissolved on a hot plate at a temperature below 40 °C. Then, the honey samples were filtrate to remove large honey wax particles, the liquid was poured into 30 mL conical centrifuge tubes. After several centrifugation times (2500–3000 rpm) at room temperature (21–25 °C), pollen grains were extracted from the honey. In the first round, the samples were centrifuged for 10 min (approximately 2500 rpm) and the supernatant was carefully removed to avoid pollen loss. The remaining precipitate was mixed again with about 10 mL of distilled water, but this time the samples were centrifuged for 5 min (2500–3000 rpm). After the last centrifugation, we poured the last remaining liquid into the watch glass and placed it on the hot plate so that the water evaporated completely and the pollen grains remained at the bottom of the container. We placed the prepared pollen grains on special bases and, after coating the samples with gold metal, we placed them inside the scanning electron microscope (SEM) and then the pollen grains obtained from honey sediments were counted using a SEM. In order to determine the relative abundance, the number of 500–1000 pollen grains should be identified in each honey sample55. Pollen grains were identified based on their morphology using atlases and international palynology websites.
Chemical analyses
Antioxidant activity was measured based on 2,2-Diphenyl-1-picrylhydrazyl (DPPH). All honey samples were analyzed using DPPH free radical scavenging activity by antioxidant activity analysis56. Honeys were diluted 1/1 (w/v) with distilled water. DPPH solution was prepared by dissolving 0.1 mM of DPPH in methanol. In its radical form, DPPH solution with purple color shows an absorbance maximum at 515 nm which disappears upon reduction by an antiradical (anti-oxidant) compounds which turn its purple to yellow color. Ascorbic acid (20 mg/mL) was used as a positive control. Ascorbic acid was used as a strong antioxidant compound, which in this concentration can completely reduce DP and cause the maximum decrease in DP absorption at 530 nm. This mode can help in reducing absorption in DPPH when comparing honey samples. To measure the antioxidant activity of the honey samples, 10 μL of diluted honey was mixed with 100 μL of DPPH and incubated for 30 min in the dark. Absorbance was recorded using a spectrophotometer at a wavelength of 520 nm. The control sample consists of 10 μL of water and 100 μL of DPPH.
Antioxidant activity was calculated using the following formula
Instrumentations and reagents
Hitachi SU3500 scanning electron microscope (SEM) made in Japan.
Spectrophotometer JENUS V-1100 made by DLAB China.
Ascorbic acid and 2,2-Diphenyl-1-picrylhydrazyl (DPPH) were obtained from Sigma and Merck Chemical Company respectively.
Statistical analysis
To determine the degree of dependence of antioxidant activity on plant families, between the abundance of each family and their antioxidant levels, regression was taken by Fitted line plot, Minitab19 with a confidence level of 95%. ArcGIS 10.8 software was also used to describe the abundance of effective plant families and the amount of antioxidant activity of honey samples.
Statement
In the above study, the beekeepers provided us with honey samples voluntarily and with full consent, and we did not need to obtain a permit due to not entering protected areas or national parks.
Results
Of the 94 collected samples, it was not possible to identify pollen in 37 samples, and for this reason, it was excluded from the melissopalynology part of the study. From the studied honey samples, 42 plant families from 55 plant genera were identified and their frequency was determined (Fig. 2).
Among the number of identified plant families, 4 families Rosaceae (p 0.000, R2 > 62%), Amaranthaceae (p 0.040, R2 > 47%), Fabaceae (p 0.000, R2 > 45%) and Asteraceae (p 0.000, R2 > 31%), were respectively the most effective families in the amount of antioxidant activity of honey. The result showed that the increase in abundance of plant families Rosaceae, Asteraceae, Amaranthaceae and Fabaceae in honey has increased the amount of antioxidant activity of the studied honey samples (Fig. 3).
The four identified families effective on the amount of honey antioxidants have different influence power. As Fig. 4 shows, for 1% of the presence of Rosaceae family in honey, it causes an rise of about 3% of antioxidants, which shows more effective power compared to other samples (Fig. 4).
The abundance of Rosaceae (p 0.003, R2 > 41%) and Fabaceae (p 0.001, R2 > 31.7%) families showed a significant increase with the rise in the altitude of the beekeepers' settlement. Also, the results showed that the abundance of the Rosaceae and Fabaceae family in the collected honey samples had a direct relationship with the increase in altitude (Fig. 5).
The abundance of Rosaceae and Fabaceae families in honey shows an increasing trend with rise altitude up to 3000 m. The four families influencing the amount of antioxidant activity of honey were sampled throughout the region and showed different frequencies. Figure 6 describes the abundance of four families in the collected honey samples.
Antioxidants of the studied samples showed that geographical coordinates can act as an important determining factor in the amount of honey antioxidants. Figure 7 describes the amount of antioxidant activity of honey geographically in the sampled area.
Discussion
This study was conducted in order to identify effective plant families in determining the antioxidant activity of honey. Also, the results of this study compared the influence of effective families on the amount of antioxidant activity of honey. So that for 1% abundance of Rosaceae family, the antioxidant of honey can increase by about 3%. The results of this study showed that the height and abundance of Rosaceae and Fabaceae families have a direct relationship, so that the abundance of these two families increased with the rise in attitude. The geographical part of the results of this study showed that the abundance of four plant families in honey is not uniform and in some areas, each of them shows a high relative abundance. Also, the amount of antioxidant activity of honey varied from 98 to 19% in the sampled areas.
The results of this study showed that the Rosaceae family has the greatest effect in increasing the antioxidant activity of honey because the plant species of the Rosaceae family are considered an important natural source of antioxidants57. Also, increasing the abundance of plant species of the Fabaceae family in honey compared to other families has a greater impact on increasing the antioxidant activity of honey18. The results of studies have shown that almost all components of the Rosaceae family are considered as an important source of natural antioxidants58,59.
The results showed that the Amaranthaceae family is an effective family on the amount of antioxidants in honey. With a 1% rise in the abundance of Amaranthaceae family in honey, its antioxidant activity increased by about 2.3%. The Amaranthaceae family, having a significant amount of natural antioxidants, has turned this family into an important plant family for medicinal purposes29,60,61.
The Fabaceae family was identified as an important family in determining the antioxidant content of honey in this study, which combined melissopalynology studies, confirms our results62. An increase in the abundance of the Fabaceae family by 1% rise the antioxidant activity of honey by about 1.3%, because molecular studies have shown that the species of the Fabaceae family have an effective and significant amount of antioxidants63,64,65. The amount of antioxidants of some species of the Fabaceae family is so impressive that some studies have suggested for the prevention and treatment of various cancers66,67. Also, the Fabaceae family plays an effective role in increasing the amount of antioxidants in honey.
The Asteraceae family is the fourth identified family in this study, the increase in its abundance is directly related to the rise in the antioxidant activity of honey. The significant amount of antioxidant activity in the plant species of the Asteraceae family has made this plant family an important medicinal plant family in the studies of prevention and treatment of various diseases68,69. The results of this study showed that a 1% rise in its frequency causes an increase of about 1.3% in the antioxidant activity of honey. Some studies confirm the effect of the presence of the Asteraceae family on the amount of antioxidants in honey70,71.
The ecological results of this study showed that the abundance of Rosaceae and Fabaceae families, which play an effective role in the antioxidant activity of honey, rise with increasing altitude. Therefore, it seems that the increase in antioxidant activity due to the rise in altitude up to 3000 m above the surface of open water48 is largely influenced by the abundance of these two families. For example, the Rosaceae family grows in high areas72. Therefore, there is a direct relationship between the plant-geographical origin of honey production and its biological properties73,74. Also, the contents of medicinal and aromatic plants show significant changes depending on the region and climatic conditions27.
Conclusion
The antioxidant activity of honey has a direct and significant relationship with the plant and geographical origin of honey, and the presence of some plant families significantly increases the antioxidant activity in honey. The four plant families Rosaceae, Amaranthaceae, Fabaceae, and Asteraceae, respectively, in this study had the greatest influence on the amount of antioxidant activity of honey compared to other plant families. The abundance of two plant families, Rosaceae and Fabaceae, which play an effective role in the antioxidant activity of honey, also increases compared to altitude. Many species of the four plant families effective in the amount of antioxidants in honey are native and endemic species of the Iranian plateau. Therefore, these areas have the potential to produce honeys with high antioxidant activity, which has made the protection of plant communities in these areas important.
Data availability
Datasets analyzed during the current study are available on Figshare as https://figshare.com/s/cc9d170a8530fb798e26.
References
Hanahan, D. Hallmarks of cancer: New dimensions. Cancer Discov. 12(1), 31–46 (2022).
Luo, M. et al. Antioxidant therapy in cancer: Rationale and progress. Antioxidants 11(6), 1128 (2022).
Fitzgerald, R. C., Antoniou, A. C., Fruk, L. & Rosenfeld, N. The future of early cancer detection. Nat. Med. 28(4), 666–677 (2022).
Caponio, G. R. et al. Nutraceuticals: Focus on anti-inflammatory, anti-cancer, antioxidant properties in gastrointestinal tract. Antioxidants 11(7), 1274 (2022).
Mendonça, J. D. S. et al. Natural antioxidant evaluation: A review of detection methods. Molecules 27(11), 3563 (2022).
Paesa, M. et al. Valorization of onion waste by obtaining extracts rich in phenolic compounds and feasibility of its therapeutic use on colon cancer. Antioxidants 11(4), 733 (2022).
Mišković Špoljarić, K. et al. Antioxidant and antiproliferative potentials of phenolic-rich extracts from biotransformed grape pomace in colorectal Cancer. BMC Complement. Med. Ther. 23(1), 29 (2023).
Gulcin, İ. Antioxidants and antioxidant methods: An updated overview. Arch. Toxicol. 94(3), 651–715 (2020).
Azlan, A., Sultana, S., Huei, C. S. & Razman, M. R. Antioxidant, anti-obesity, nutritional and other beneficial effects of different chili pepper: A review. Molecules 27(3), 898 (2022).
Lawal, B., Shittu, O. K., Oibiokpa, F. I., Berinyuy, E. B. & Mohammed, H. African natural products with potential antioxidants and hepatoprotectives properties: A review. Clin. Phytosci. 2(1), 1–66 (2017).
Cianciosi, D. et al. The reciprocal interaction between polyphenols and other dietary compounds: Impact on bioavailability, antioxidant capacity and other physico-chemical and nutritional parameters. Food Chem. 375, 131904 (2022).
Meenigea, N. Evaluation of antioxidant potential and antimicrobial activity. Trans. Latest Trends Health Sector 14(14) (2022).
Pena Júnior, D. S. et al. Antioxidant activities of some monofloral honey types produced across Minas Gerais (Brazil). PLoS One 17(1), e0262038 (2022).
Fernández-Estellé, M., Hernández-González, V., Saurina, J., Núñez, O. & Sentellas, S. Characterization and classification of Spanish honeydew and blossom honeys based on their antioxidant capacity. Antioxidants 12(2), 495 (2023).
Mduda, C. A., Hussein, J. M. & Muruke, M. H. The effects of bee species and vegetation on the antioxidant properties of honeys produced by Afrotropical stingless bees (Hymenoptera, Apidae, Meliponini). J. Agric. Food Res. 14, 100736 (2023).
Zapata-Vahos, I. C. et al. Physicochemical parameters, antioxidant capacity, and antimicrobial activity of honeys from tropical forests of Colombia: Apis mellifera and Melipona eburnea. Foods 12(5), 1001 (2023).
Shamsudin, S., Selamat, J., Abdul Shomad, M., Ab Aziz, M. F. & Haque Akanda, M. J. Antioxidant properties and characterization of Heterotrigona itama honey from various botanical origins according to their polyphenol compounds. J. Food Qual. 2022, 2893401 (2022).
Tedesco, R. et al. Characterization of botanical origin of Italian honey by carbohydrate composition and volatile organic compounds (VOCs). Foods 11(16), 2441 (2022).
Bayram, N. E. et al. Characterization of physicochemical and antioxidant properties of Bayburt honey from the North-east part of Turkey. J. Apic. Res. 60(1), 46–56 (2021).
Tesfaye, O. Evaluating the antioxidant properties of unifloral honey (Apis mellifera L.) from Ethiopia. Int. J. Food Sci. 2023, 7664957 (2023).
Gurib-Fakim, A. Medicinal plants: Traditions of yesterday and drugs of tomorrow. Mol. Aspects Med. 27(1), 1–93 (2006).
Petrovska, B. B. Historical review of medicinal plants’ usage. Pharmacogn. Rev. 6(11), 1 (2012).
Süntar, I. Importance of ethnopharmacological studies in drug discovery: Role of medicinal plants. Phytochem. Rev. 19(5), 1199–1209 (2020).
Krishnaiah, D., Sarbatly, R. & Nithyanandam, R. A review of the antioxidant potential of medicinal plant species. Food Bioprod. Process. 89(3), 217–233 (2011).
Duraipandiyan, V., Ayyanar, M. & Ignacimuthu, S. Antimicrobial activity of some ethnomedicinal plants used by Paliyar tribe from Tamil Nadu, India. BMC Complement. Altern. Med. 6, 1–7 (2006).
Ashrafi, S. et al. Chemico-biological profiling of Blumea lacera (Burm. f.) DC. (Family: Asteraceae) provides new insights as a potential source of antioxidant, cytotoxic, antimicrobial, and antidiarrheal agents. Evid. Based Complement. Altern. Med. 2022, 2293415 (2022).
Güneş, A., Kordali, Ş, Turan, M. & Bozhüyük, A. U. Determination of antioxidant enzyme activity and phenolic contents of some species of the Asteraceae family from medicinal plants. Ind. Crops Prod. 137, 208–213 (2019).
Lemus-Mondaca, R., Vega-Gálvez, A., Zura-Bravo, L. & Ah-Hen, K. Stevia rebaudiana Bertoni, source of a high-potency natural sweetener: A comprehensive review on the biochemical, nutritional and functional aspects. Food Chem. 132(3), 1121–1132 (2012).
Adegbola, P. I., Adetutu, A. & Olaniyi, T. D. Antioxidant activity of Amaranthus species from the Amaranthaceae family—A review. S. Afr. J. Bot. 133, 111–117 (2020).
Terletskaya, N. V. et al. Genetic polymorphism in the Amaranthaceae species in the context of stress tolerance. Plants 12(19), 3470 (2023).
Bhargava, A. & Srivastava, S. Response of Amaranthus sp. to salinity stress: A review. In Emerging Research in Alternative Crops 245–263 (Springer, 2020).
Mroczek, A. Phytochemistry and bioactivity of triterpene saponins from Amaranthaceae family. Phytochem. Rev. 14, 577–605 (2015).
Maroyi, A. Medicinal uses of the Fabaceae family in Zimbabwe: A review. Plants 12(6), 1255 (2023).
Christenhusz, M. J. & Byng, J. W. The number of known plants species in the world and its annual increase. Phytotaxa 261(3), 201–217 (2016).
Usman, M. et al. Exploring the phytochemicals and anti-cancer potential of the members of Fabaceae family: A comprehensive review. Molecules 27(12), 3863 (2022).
Semenya, S. S. & Maroyi, A. Ethnomedicinal uses of Fabaceae species for respiratory infections and related symptoms in the Limpopo province, South Africa. J. Pharm. Nutr. Sci. 8, 219–229 (2018).
Ramdhanawati, V. U., Octaviani, W. T. & Supriyatna, A. Inventory and identification of Rosaceae family in the surroundings of farm house Lembang. IJESPG (Int. J. Eng. Econ. Soc. Politic Gov.) 1(1), 29–35 (2023).
Assadi, M. et al. (eds) Flora of Iran, vol. 31 (Iridaceae) (Research Institute of Forests and Rangelands Publication, 1999) (in Persian).
Mehrabian, A. R., Sayadi, S., Kuhbanani, M. M., Hashemi, V. & Abdoljabari, M. Priorities for conservation of endemic trees and shrubs of Iran: Important Plant Areas (IPAs) and Alliance for Zero Extinction (AZE) in SW Asia. J. Asia-Pac. Biodivers. 13, 295–305 (2020).
Frey, W. & Probst, W. A synopsis of the vegetation of Iran. In Contributions to the Vegetation of Southwest Asia (ed. Kürschner, H.) 61–73 (Ludwig Reichert, 1986).
Hedge, I. C. & Wendelbo, P. Patterns of distribution and endemism in Iran. Notes R. B. G. Edinb. 36, 441–464 (1978).
Takhtajan, A. Floristic Regions of the World (University of California Press, 1986).
Kier, G. et al. Global patterns of plant diversity and floristic knowledge. J. Biogeogr. 32(7), 1107–1116 (2005).
Vavilov, N. I. Studies on the origin of cultivated plants. Bull. Appl. Bot. Plant Breed. 14, 1–245 (1926) (Russian).
Refahi, M. A. & Alipour, M. J. Bee Plants of Iran (Zarih Aftab, 2019).
Mehrabian. A. R. Medical grade of honey: Authenticity, criteria and characteristics. The First Apitherapy Symposium of Iran and Turkey, 25–26 Oct, Tehran (2023).
Iosageanu, A. et al. Comparative palynological, physicochemical, antioxidant and antibacterial properties of Romanian honey varieties for biomedical applications. Chem. Biodivers. 19(8), e202200406 (2022).
Shakoori, Z., Mehrabian, A., Minai, D., Salmanpour, F. & Khajoei Nasab, F. Assessing the quality of bee honey on the basis of melissopalynology as well as chemical analysis. PLoS One 18(8), e0289702 (2023).
Hassanpouraghdam, M. B. et al. Diversity and distribution patterns of endemic medicinal and aromatic plants of Iran: Implications for conservation and habitat management. Int. J. Environ. Res. Public Health 19(3), 1552 (2022).
Noroozi, J. & Körner, C. A bioclimatic characterization of high elevation habitats in the Alborz mountains of Iran. Alpine Bot. 128, 1–1 (2018).
Noroozi, J., Moser, D. & Essl, F. Diversity, distribution, ecology and description rates of alpine endemic plant species from Iranian mountains. Alpine Bot. 126, 1–9 (2016).
Noroozi, J. et al. Endemic diversity and distribution of the Iranian vascular flora across phytogeographical regions, biodiversity hotspots and areas of endemism. Sci. Rep. 9(1), 12991 (2019).
Louveaux, J., Maurizio, A. & Vorwohl, G. Methods of melissopa-lynology. Bee World. 51, 25–138 (1970).
Von Der Ohe, W., Oddo, L. P., Piana, M. L., Morlot, M. & Martin, P. Harmonized methods of melissopalynology. Apidologie 35(Suppl. 1), S18–S25. https://doi.org/10.1051/apido:2004050 (2004).
Behm, F., Von Der Ohe, K. & Henrich, W. Zuverlässigkeit der pollenanalyse von honig: bestimmung der pollenhäufigkeit. Deutsche Lebensmittel-Rundschau. 92(6), 183–188 (1996).
Chua, L. S., Rahaman, N. L. A., Adnan, N. A. & Eddie Tan, T. T. Antioxidant activity of three honey samples in relation with their biochemical components. J. Anal. Methods Chem. 2013, 13798 (2013).
Zhang, X. K. et al. Comprehensive analysis of 34 edible flowers by the determination of nutritional composition and antioxidant capacity planted in Yunnan Province China. Molecules 28(13), 5260 (2023).
Ekin, H. N., Gokbulut, A., Aydin, Z. U., Donmez, A. A. & Orhan, I. E. Insight into anticholinesterase and antioxidant potential of thirty-four Rosaceae samples and phenolic characterization of the active extracts by HPLC. Ind. Crops Prod. 91, 104–113 (2016).
Sytar, O., Hemmerich, I., Zivcak, M., Rauh, C. & Brestic, M. Comparative analysis of bioactive phenolic compounds composition from 26 medicinal plants. Saudi J. Biol. Sci. 25(4), 631–641 (2018).
Miguel, M. G. Betalains in some species of the Amaranthaceae family: A review. Antioxidants 7(4), 53 (2018).
Shegebayev, Z. et al. Pharmacological properties of four plant species of the genus Anabasis, Amaranthaceae. Molecules 28(11), 4454 (2023).
Majid, M., Ellulu, M. S. & Abu Bakar, M. F. Melissopalynological study, phenolic compounds, and antioxidant properties of Heterotrigona itama honey from Johor, Malaysia. Scientifica 2020, 2529592 (2020).
Phuong, D. L. et al. Extraction process, identification of fatty acids, tocopherols, sterols and phenolic constituents and antioxidant evaluation of seed oils from five Fabaceae species. Processes 7, 456 (2019).
Tungmunnithum, D., Drouet, S., Lorenzo, J. M. & Hano, C. Characterization of bioactive phenolics and antioxidant capacity of edible bean extracts of 50 Fabaceae populations grown in Thailand. Foods 10(12), 3118 (2021).
Janarny, G., Ranaweera, K. K. D. S. & Gunathilake, K. D. P. P. Digestive recovery of polyphenols, antioxidant activity, and anti-inflammatory activity of selected edible flowers from the family Fabaceae. J. Food Biochem. 46(2), e14052 (2022).
Myrtsi, E. D., Evergetis, E., Koulocheri, S. D. & Haroutounian, S. A. Bioactivity of wild and cultivated legumes: Phytochemical content and antioxidant properties. Antioxidants 12(4), 852 (2023).
Youssef, A. M., Maaty, D. A. & Al-Saraireh, Y. M. Phytochemical analysis and profiling of antioxidants and anticancer compounds from Tephrosia purpurea (L.) subsp. apollinea family Fabaceae. Molecules 28(9), 3939 (2023).
Sowa, P. et al. Analysis of cytotoxicity of selected Asteraceae plant extracts in real time, their antioxidant properties and polyphenolic profile. Molecules 25(23), 5517 (2020).
Piątkowska, E., Biel, W., Witkowicz, R. & Kępińska-Pacelik, J. Chemical composition and antioxidant activity of Asteraceae family plants. Appl. Sci. 12(23), 12293 (2022).
Ávila, S. et al. Bioactive compounds and biological properties of Brazilian stingless bee honey have a strong relationship with the pollen floral origin. Food Res. Int. 123, 1–10 (2019).
Ghorab, A. et al. Sensorial, melissopalynological and physico-chemical characteristics of honey from Babors Kabylia’s region (Algeria). Foods 10(2), 225 (2021).
Peyravi, M. Prioritizing areas for conservation of Rosaceae in Iran based on the geographic distribution analysis. Iran. J. Bot. 21(1), 47–58 (2015).
Becerril-Sánchez, A. L., Quintero-Salazar, B., Dublán-García, O. & Escalona-Buendía, H. B. Phenolic compounds in honey and their relationship with antioxidant activity, botanical origin, and color. Antioxidants 10(11), 1700 (2021).
Moreni, A., Cabrera, M. C., Cracco, P., Cadenazzi, M., Pirotti, F. & Santos, E. Trace elements content and antioxidant capacity in honey from protected phytogeographical regions in Uruguay. Agrociencia Uruguay, 27 (2023).
Acknowledgements
We are grateful to Mr. Saeed Javadi Anaghizi is thanked for his assistance in SEM work.
Author information
Authors and Affiliations
Contributions
Z.S., E.S., F.S., A.M., D.T., N.F.: Writing—review and editing. Z.S., E.S., F.S., A.M., D.T.: Conceptualization, Methodology. Z.S., E.S., F.S., N.F.: Data curation. Z.S., F.S.: Formal analysis, Investigation, Project administration, Software, Supervision. Z.S.: Writing—original draft.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shakoori, Z., Salaseh, E., Mehrabian, A.R. et al. The amount of antioxidants in honey has a strong relationship with the plants selected by honey bees. Sci Rep 14, 351 (2024). https://doi.org/10.1038/s41598-023-51099-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-51099-9
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.