Abstract
Fifty-five children aged 2 years from a birth cohort in the largest dioxin-contaminated area in Bien Hoa city, Vietnam participated in this survey to examine gaze behavior. Exposure levels were indicated by 2,3,7,8-tetrachlorodibenzo-p-dibenzodioxin (TCDD) and toxic equivalent of polychlorinated dibenzo-p-dioxin and polychlorinated dibenzofuran (TEQ-PCDD/Fs) levels in maternal breast milk. The percentage of the total fixation duration on the face (% Face), mouth (% Mouth), and eye areas (% Eyes) when viewing silent and conversation scenes was used as gaze behavior indices. When they reached 3-year-old, autistic behavior was assessed using the Autism Spectrum Rating Scale (ASRS). A general linear model adjusted for confounding factors was used to compare gaze indices and ASRS scores between high and low dioxin exposure groups. Effects of perinatal dioxin exposure on gaze behavior were found only when viewing conversation scenes indicated by lower % Face for boys in high TCDD exposure group and lower % Eyes for girls in high TEQ-PCDD/Fs group. Increased autistic traits showed by higher ASRS scores at 3-year-old were found in both gender in the high TCDD exposure group. These findings indicate that perinatal TCDD and TEQ-PCDD/Fs exposure may reduce gaze behavior in 2-year-old children, predicting increased autistic traits at 3-year-old.
Similar content being viewed by others
Introduction
Bien Hoa airbase is the largest dioxin-contaminated area in Vietnam because of the use of herbicide, particularly Agent Orange. Agent Orange contains 2,3,7,8-tetrachlorinated-p-dibenzodioxin (TCDD), and was used by the US army from 1961 to 1970 in Vietnam. In 2012, we recruited 210 mother–newborn pairs living in 10 communes close to Bien Hoa airbase (Bien Hoa birth cohort 2012) and reported dioxin levels in maternal breast milk1. The mean concentrations of TCDD and toxic equivalent (TEQ) of polychlorinated dibenzodioxins and polychlorinated dibenzofurans (PCDD/Fs) in breast milk were 2.6 pg/g lipid and 10.5 pg-TEQ/g lipid for primiparous mothers, and 2.2 pg/g lipid and 9.0 pg-TEQ/g lipid for multiparous mothers, respectively. TCDD levels were nearly four to five times higher in primiparous mothers and seven times higher in multiparous mothers compared with those in unsprayed areas. In 2015, we additionally collected 78 mother–child pairs as the second birth cohort in the Bien Hoa region (Bien Hos cohort 2015). We combined the two groups and reported the effects of perinatal dioxin exposure on expressive language in boys at 2 years old2.
In newborn infants from the Bien Hoa birth cohort 2015, we recorded neonatal electroencephalography (EEG) during sleep time on the second day after birth and reported that dioxin exposure, particularly TCDD exposure, altered relative EEG power and coherence in the active sleep stage3. We also reported decreased relative EEG power in the quiet sleep stage associated with increasing TCDD exposure, which was leading to poor gaze behavior at 2 years old as shown by a shorter fixation duration on the face of a child talking in videos4. However, we didn’t investigate direct effects of dioxin exposure on gaze behavior in these 2-year-old children from Bien Hoa cohort.
We followed up the children at 3 years old in Bien Hoa cohort 2012 and found that increased TCDD exposure decreased the percentage of face fixation duration when viewing face static pictures in girls, which associated with poorer social communication ability examined by the Autism Spectrum Rating Scale5. In addition, previous reports have shown a shorter fixation duration on facial areas including eye and mouth areas during dynamic stimuli of viewing a video in children with autism compared with typical children6,7. These results suggest that poor gaze behavior at 2 years of age may be a good predictor for increased autistic traits which can be diagnosed at 3 years of age in perinatally dioxin-exposed children in a dioxin hot spot in Vietnam.
Therefore, we firstly investigated direct associations between perinatal dioxin exposure and gaze behavior indicated by fixation duration of eye, mouth, and whole face areas at 2 years old. And then, we investigated the neurodevelopmental status at 3 years old, including general neurodevelopment and autistic traits, in highly exposed children who showed poor gaze behavior at 2 years old compared with lower exposed children.
Methods
Subjects and location
Bien Hoa airbase is considered as the most severely dioxin contaminated areas in Vietnam. The Office of the Vietnam National Steering Committee 33 and Hatfield Consultants (2011) reported TCDD soil levels as high as 61,400 pg/g dry weight and TEQ-PCDD/Fs in sediment as high as 5970 pg/g dry weight in samples collected from the Bien Hoa airbase. The contribution of TCDD to these TEQ-PCDD/Fs was more than 80% in most samples8.
Seventy-eight mother–newborn pairs living in 10 communes close to Bien Hoa airbase were enrolled in the present study from August to December in 2015. The mother–newborn pairs were included if they met the following criteria: (i) mothers had resided in the target area for at least 1 year before giving birth; (ii) newborns were born at full-term, and (iii) there were no complications during giving birth. At that time, breast milk sample was collected from 67 mothers for dioxin measurement. There were no breast milk samples or not enough breast milk samples for dioxin measurement were found in eleven mothers. In December 2017 and January 2018, we carried out a 2-year follow-up survey in which 66 mother–child pairs (84.6%) participated in an examination of gaze behavior and neurodevelopment4. Twelve pairs did not participate in the survey for the following reasons. One mother stayed in hospital, three refused to participate in the survey, five moved to other locations, and three were parents who were too busy with work. Additionally, six children who were not able to achieve successful calibration in the gaze examination or their total fixation duration for all stimuli was < 2.7 s (< 5th percentile), and five children who had missing data of exposure markers or gestational weeks at birth were excluded. Finally, data of 55 children (24 boys and 31 girls) were available for analysis of associations between perinatal dioxin exposure and gaze behavior. There were no significant differences in dioxin concentrations between participants and non-participants (Supple. Data 1). The characteristics of the mothers and families of children, gestational weeks and body size of children at birth, and age and body size of children at the survey are shown in Table 1. Only maternal education was significantly higher in girls compared with boys.
A follow-up study was performed when children reached 3 years old. At this time, 50 (21 boys and 29 girls) of 55 (90.9%) children who participated in the 2-year-old survey had their neurodevelopment examined. Five cases who did not participate the survey because of two cases who stayed in the hospital and three cases with their parents who were too busy on the examination day. The number of participants, age, and mean (SD) body size indices of children who had their neurodevelopment examined in the 3-year-old survey are shown in Table 1. The time line of the present study is shown in Supple. data 2.
Informed consent was obtained from all the subjects and their legal guardian (mother’s) involved in the study. All methods were performed in accordance with the relevant guidelines and regulations by the Dong Nai Health Department and the Vietnam Military Medical University. The institutional ethics board for epidemiological studies at Kanazawa Medical University reviewed and approved the study design (No. 187).
Exposure assessment
Dioxins in maternal breast milk were used as exposure markers because we previously showed a significant association of dioxins between cord blood and breast milk samples9.
One month after birth, nurses from community health centers visited the mother’s house and collected approximately 20 ml of breast milk from them. Breast milk samples were frozen, transported to Japan, and levels of 7 congeners of PCDDs and 10 congeners PCDFs were quantified in the High Technology Center at Kanazawa Medical University. After using an EYELA freeze-dryer (FDU-1200; Tokyo-rika Inc., Tokyo, Japan) to dehydrate 10 ml of breast milk, the fat content was extracted using an ASE-200 accelerated solvent extractor (Dionex Co., CA, USA). Next, 13C-labeled 2,3,7,8-substituted PCDDs/Fs (DF-LCS-A40; Wellington Inc., Ontario, Canada) were added as an internal standard. After a series of purification and separation processes, levels of 7 PCDD and 10 PCDF congeners were quantified using a gas chromatograph (HP-6980; Hewlett-Packard, Palo Alto, CA, USA) equipped with a high-resolution mass spectrometer (MStation-JMS700; JEOL, Tokyo, Japan). The concentration of each congener below the detection limit was set by half of the detection limit. TEQ-PCDDs/Fs were calculated as the sum of all values obtained by multiplying each congener concentration by its toxic equivalent factor from WHO 2005-TEF10. The established method for analysis was described in detail previously1,2.
Gaze behavior examination
Examination method
At 2 years old, the gaze behavior test was performed in community health centers using a Tobii X2-60 Compact eye tracker running at 60 Hz (Tobii Technology, Stockholm, Sweden) to measure the position of both eyes with an infrared camera below the screen. To minimize noise or other factors from outside, which might disturb the attention of the children, the device was set up in a private room with a closed door. Only the examiner and the child with their caregiver stayed inside the room. Children were comfortably seated on the lap of their caregiver in front of the screen of a computer with a distance from their eyes to the screen of approximately 60 cm and free viewing.
Gaze data were extracted and analyzed using Tobii Studio software (Tobii Technology, Stockholm, Sweden). Before extracting data, a default I-VT filter (Tobii Studio software, User manual version 3.3.1, 2015) was applied with a velocity threshold of 30 degrees/s and the minimum fixation duration was set at 100 m.sec. to determine fixation. Two video clips contained a Vietnamese child (a boy for one clip and a girl for the other clip) who was similar in age to the participants. The Vietnamese child engaged in social interaction, such as talking to the camera operator or having a conversation with someone who did not appear on the screen. The video clips were shown for 43 s. to children by the Tobii system. In the interval between the two video clips, one dynamic video of a bear cartoon figure with sound appeared in the center of screen for 2 s. to attract the child’s attention to the screen. Before data recording, children had to achieve successful calibration with a dynamic bear cartoon figure that appeared at the four corners in the top and bottom of the screen and in the middle of the screen, accompanied by sound. Details of this method were described in our previous study4.
Full frame analysis
The whole frame and facial area were manually set to define areas of interest (AOIs) in each frame for 1336 frames. The total fixation duration in the facial area and in the whole frame were extracted in each frame using Tobii Studio software. By adding up the values in the 1336 frames, the total fixation duration on the screen and facial areas were calculated. The percentage of the total fixation duration on facial areas (% Face) was defined as the ratio of the total fixation duration on facial areas in all frames divided by the total fixation duration on the whole frame in all frames. These ratios were then multiplied by 100 to calculate the percentage of the total fixation duration on facial areas for all stimuli.
Analysis for silent and talking scenes
Gaze attention allocation patterns in the eye and mouth regions are different between viewing silent and talking facial scenes. This finding can be explained by the influence of the context of social scenes, particularly auditory speech modulation of gaze attention when viewing a dynamic face11,12. We chose five epochs from the videos that lasted for 18.6 s showing talking facial scenes to set up AOIs for gaze analysis. Additionally, we chose five epochs that lasted for 12.4 s. showing a child with a silent face (non-talking), in which both eyes and the mouth of the child were clearly identifiable, to set up AOIs for gaze analysis.
Similarly, the whole frame, face, mouth, and eye areas were manually set to define AOIs in each frame. Each AOI contained the entire feature for talking and non-taking facial scenes such as eye areas is defined by a single rectangle that included both eyes and eyebrows and mouth areas is defined by a single rectangle that framed the mouth (Fig. 1). There were 557 frames of 5 epochs for talking facial scenes and 370 frames of 5 epochs for silent facial scenes. We extracted the total fixation duration in the whole frame, face, mouth, and eye areas in each frame. We added up the values of the 557 and 370 frames as the total fixation duration on the whole frame, facial areas, mouth areas, and eye areas for viewing talking and silent facial scenes, respectively. The percentages of the total fixation duration on faces (% Face), the mouth (% Mouth), and eye areas (% Eyes) when viewing talking and silent facial scenes were calculated and used as gaze behavior indices for data analysis. Details of this method were described in our previous study5,13.
Neurodevelopmental assessment
At 3 years old, face to face interview mothers or caregivers of children was conducted using full-length parent rating forms (2–5 years) of the Autism Spectrum Rating Scale ([ASRS] MSH, North Tonawanda, NY, USA) to assess child behavior associated with autism spectrum disorder. Before using the ASRS scale for this study, a trial examination was conducted with 15 Vietnamese children to ensure the feasibility and appropriateness of the ASRS for the Vietnamese population. Four main scores, which were the total (TOT) score, the DSM-IV-TR (DSM) score, the Social Communication (SC) score, and the Unusual Behavior (UB) score, were calculated. An increase in TOT and DSM scores is associated with increased autistic behavioral traits. Details of this method were described in our previous study14.
The Bayley-III scale (NCS Pearson, Inc., Bloomington, MN, USA) was used to examine general neurodevelopment across five domains of cognition, expressive and receptive language, and fine and gross motor skills. Similarly, we had previously conducted several surveys using The Bayley Scales of Infant and Toddler Development, Third Edition (Bayley-III) scale in groups of Vietnamese infants and children in our previous studies2,14,15,16. Only one examiner who was trained in the previous surveys and blinded to the dioxin exposure levels of the children performed the test to provide reliable scoring. In the present examination, results for ASRS scores were obtained from 50 children and those for general neurodevelopmental scores were obtained from 48 children because 2 children refused to perform some tests.
Statistical analysis
IBM SPSS version 22.0 (IBM Corp., Armonk, NY, USA) was used for statistical analysis. Children were divided into high and low exposure groups in relation to concentrations of TCDD and TEQ-PCDD/Fs in breast milk. The cut-off value for TCDD (3.5 pg/g lipid) was calculated from the geometric mean × the geometric standard deviation3 of TCDD concentrations in breast milk samples from unsprayed areas as we previously reported15. Because of a limited number of subjects with TEQ-PCDD/Fs levels higher than the geometric mean × the geometric standard deviation3, the cut-off value for TEQ-PCDD/Fs was set at the 75th percentile of TEQ-PCDD/F levels (9.2 pg-TEQ/g lipid) of the subjects.
Gaze behavior indices were converted into z-scores to improve the normal distribution. The Mann–Whitney U test was used to compare unadjusted gaze behavior indices between the high and low exposure groups. A general linear model was used to compare adjusted gaze behavior indices after adjusting for maternal years of education. Since smoking have been reported that associated with increase autistic trait and neurodevelopmental disorder in previous studies17,18. Therefore, adjusted ASRS and Bayley-III scores were compared between high and low exposure group using a general linear model after adjusting for maternal years of education and family member smoking (yes/no). The p-value ≤ 0.05 was considered statistically significant.
Ethics approval
All methods were performed in accordance with the relevant guidelines and regulations by the Dong Nai Health Department and the Vietnam Military Medical University. The institutional ethics board for epidemiological studies at Kanazawa Medical University reviewed and approved the study design (No. 187).
Consent to participate
Informed consent was obtained from all the subjects and their legal guardian(mother's) involved in the study.
Results
Comparison of gaze behavior indices between boys and girls
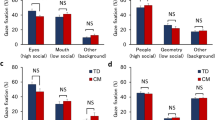
The total fixation duration on the whole screen, % Face, % Mouth, and % Eyes for viewing all, silent, and talking scenes were compared between sexes after adjusting for covariates (Table 2). The adjusted means of the total fixation duration on the screen for viewing all scenes, silent scenes, and talking scenes were higher in girls than in boys. However, only the total fixation duration on the whole screen for talking scenes was significant (p = 0.037). Adjusted means of % Mouth were lower and adjusted means of % Eyes were higher in boys for viewing silent and talking scenes compared with girls, but these differences were not significant.
Associations between perinatal TCDD exposure and gaze behavior
The adjusted means of standardized % Face for all scenes and % Face, % Mouth, and % Eyes for silent and talking scenes were compared between the high and low TCDD exposure groups with a cut-off value of 3.5 pg/g lipid (Table 3). For all participants, there was no significant difference in any gaze indices between high and low TCDD exposure groups. In boys, the means of standardized % Face for all scenes, silent scenes, and talking scenes were lower in the high TCDD group compared with the low TCDD group, but this was only significant for talking scenes (p < 0.05). However, in girls, there were no significant differences in the standardized means of any gaze markers for any scenes.
Associations between perinatal TEQ-PCDD/Fs exposure and gaze behavior
The adjusted means of standardized % Face for all scenes and % Face, % Mouth, and % Eyes for silent and talking scenes were compared between the high and low TEQ-PCDD/Fs exposure groups with a cut-off value of 9.2 pg-TEQ/g lipid (Table 4). No significant difference was found in the standardized means of any gaze marker for all participants. Similarly, in boys, there were no significant differences in the standardized means of any gaze markers for any scenes. However, in girls, the adjusted mean of standardized % Eyes for talking scenes was significantly lower in the high TEQ-PCDD/Fs exposure group than in the low TEQ-PCDD/Fs exposure group (p < 0.05). These results suggest a shorter fixation duration of gaze on the eye area of a talking person in girls who are exposed to high TEQ-PCDD/Fs.
Comparisons of autistic behavior indicated by the ASRS scores between high and low dioxin exposure groups
Autistic behavior in the children was examined when they reached 3 years old by the ASRS. Adjusted mean scores of TOT and the three subscales DSM, SC, and UB were compared between the high and low TCDD exposure groups after adjusting for maternal years of education and family member smoking status (Table 5). High TCDD group showed significantly decrease in the adjusted mean TOT, DSMT, and SCT scores when compared with the low TCDD exposure group for all participants (p < 0.05). Similarly, the adjusted mean DSM score was significantly higher in the high TCDD group compared with the low TCDD group in boys (p < 0.05). In girls, the adjusted mean TOT and DSM scores were significantly higher in the high TCDD group compared with the low TCDD group. There were no significant differences in ASRS scale scores between the high and low TEQ-PCDD/Fs exposure groups in either sex (data not shown). These findings suggest that perinatal TCDD exposure increases autistic traits in both sexes.
Comparisons of general neurodevelopment indicated by the Bayley-III scores between high and low dioxin exposure groups
The general neurodevelopmental status of children at 3 years old was also examined using the Bayley-III. We compared these neurodevelopmental scale scores between the high and low TCDD groups after adjusting for maternal years of education and family member smoking status (Table 6). The adjusted mean score was significantly lower in the high TCDD group than in the low TCDD group only for the gross motor scale in girls. There were no significant differences in Bayley III scores between the high and low TEQ-PCDD/Fs groups in either sex (data not shown).
Discussion
In the present study, we found significant effects of perinatal dioxin exposure on gaze behavior only when viewing conversation scenes in 2-year-old Vietnamese children with sex differences in affected gaze areas and exposure markers, lower % Face in the high TCDD group for boys and lower % Eyes in the high TEQ-PCDD/Fs group for girls. A few studies have reported effects of dioxin on gaze behavior in previous reports. In Japan, Doi et al.19 investigated prenatal exposure to 17 polychlorinated biphenyl (PCB) congeners on gaze behavior of Japanese infants when viewing biological motion stimuli displayed by point-light. They reported that no significant difference in the fixation duration on inverted and upright biological motions between the high and low PCB#118 exposure groups. However, infants exposed to high PCB#118 preferred to fix their gaze on inverted biological stimuli rather than reducing attention to upright biological motion19. These results from a Japanese cohort are consistent with our results, albeit different stimuli were used between our study and the previous study from Japan. Taken together, these findings suggest that there are limited effects of perinatal dioxin and PCB exposure on gaze behavior of infants when viewing only biological motion, including upright facial motion.
In children aged 3 years from our larger Bien Hoa birth cohort 2012, we found decreased % Face associated TCDD exposure in girls, although we used static facial pictures of children for stimuli5,13. However, dynamic facial stimuli are likely to have a more natural form, and motion and higher social complexity than point-light stimuli or static pictures7,20. In addition, Schilbach et al. suggested that the use of more complex and ecologically valid social stimuli was useful to test social abilities21. To the best of our knowledge, the present study is the first to report effects of perinatal dioxin exposure on gaze behavior when viewing dynamic facial stimuli, particularly talking facial scenes.
Prenatal PCB exposure has been reported to affect gaze behavior in infants19. We previously reported concentrations of dioxin-like PCBs in breast milk samples that were collected from a birth cohort in Bien Hoa City in 20121. We found that dioxin-like PCB concentrations were low and they were even lower compared with concentrations in samples that were collected from unsprayed areas. These findings suggest that the contribution of TEQ-dl-PCBs to the TEQ is not high and that TEQ-PCDDs/Fs may be an indicator of dioxin toxicity in this contaminated area.
In our previous study of the present subjects, we found that perinatal TCDD and TEQ-PCDD/F exposure affected neonatal neuronal activity and functional connectivity as indicated by relative EEG power and coherence between various brain areas during active and quiet sleep3,4. This exposure might also be associated with poorer language development3 and less fixation on facial areas when they reach 2 years old4. However, we did not stratify by sex in these previous studies. At that time, we used only the fixation duration on facial areas, including the fixation duration on eye areas when viewing all stimuli as a gaze behavior index. Our findings suggest that altered brain development before birth induced by TCDD and TEQ-PCDD/F exposure affects gaze behavior when viewing dynamic facial stimuli.
In this study, we also found that the adjusted mean DSM score for boys and the adjusted mean TOT and DSM scores for girls were significantly higher in the high TCDD exposure group than in the low TCDD exposure group at 3 years of age. Using the same ASRS questionnaires in another cohort of dioxin contamination in Da Nang, Vietnam, we reported that increased perinatal exposure to TCDD levels was associated with increased TOT scores at 3 years old in boys and girls14. This association were more pronounced in boys than in girls. These results indicate that perinatal TCDD exposure increases autistic trait behavior in early childhood in Bien Hoa and Da Nang Cities in Vietnam. Reduced facial attention as shown by a shorter fixation duration on facial areas when viewing talking facial scenes has also been reported in autistic children6,7. Taken together, these findings suggest that perinatal TCDD exposure increases autistic trait behavior in boys, which might be associated with less attention on facial areas when viewing the face while talking.
According our previous report that there was no significant effect of TEQ-PCDD/Fs on autistic behavioral traits in 3-year-old children in Da Nang14, which is consistent with findings in the present study. We did not find any significant association between perinatal TEQ-PCDD/Fs exposure and ASRS scores or neurodevelopmental scores as indicated by Bayley-III scores examined at 3 years old in boys and girls (unpublished data). In our previous studies where we recorded EEG signals during active and quiet sleep in the present subjects, we reported that TEQ-PCDD/Fs levels were significantly associated with relative EEG power or coherence in fewer brain regions than with TCDD levels3,4. In a neurodevelopmental study in 2-year-old children in Bien Hoa City including the present subjects, increased perinatal TCDD exposure levels were associated with a decreased expressive language score. However, there were no significant effects of perinatal TEQ-PCDD/Fs exposure on Bayley-III scores in boys or girls2. These results indicate that adverse effects of perinatal dioxin exposure originating from Agent Orange on neurodevelopment of children are mainly characterized by effects of TCDD exposure.
In the present study, we only found a decreased percentage of the total fixation duration on eye areas when viewing talking in girls who were exposed to high TEQ-PCDD/Fs levels. This finding suggested that eye contact was reduced when viewing talking in girls who were exposed to high TEQ-PCDD/Fs levels. In previous studies in Da Nang city, Pham et al. (2015) reported that increased perinatal TEQ-PCDD/Fs exposure levels were associated with decreased social emotional scores in infants at 1 year old22, expressive language in 3 years-old-girls14, increased hyperactivity score in girls at 8 years of age23. In the Netherlands, alterations in visual processing and cognition induced by perinatal dioxin exposure as indicated by TEQ-PCCD/Fs levels in maternal breast milk were found in school-aged children24. These results suggest that perinatal TEQ-PCDD/Fs exposure might affect specific domains of neurodevelopment, such as communication and social cognition in girls. Additionally, it was also reported that children with hyperactivity often show social interaction impairments related to processing of other’s eye gaze such as lack of attention to the eye region of faces appears25. Therefore, follow-up of these children to carefully examine higher cognitive function and symptoms of attention deficit hyperactivity disorder in childhood is necessary.
Sex-specific effects of perinatal dioxin exposure on neurodevelopment or physical growth have been reported in epidemiological studies conducted not only in Vietnam, but also in Japan26,27 and The Netherlands28. A previous study investigated dioxin and PCB exposure on neurodevelopment of infants in a Hokkaido cohort in Japan and reported that adverse effects of perinatal dioxin exposure were more pronounced in boys than in girls27. In Rotterdam, The Netherlands, adverse effects of perinatal exposure to TEQ-PCDD/Fs on playing behavior were found only in boys and not in girls28. In Da Nang City, Vietnam, effects of perinatal exposure to TCDD and TEQ-PCDD/Fs on child neurodevelopment were found only in boys at 3, 5, and 8 years14,16,29,30. In the Bien Hoa region, we reported that perinatal exposure to high TCDD levels decreased expressive language and gross motor scores only in 2-year-old-boys2. Similarly, in the current study, a change in the percentage of the total fixation duration on face areas when viewing talking scenes was found only in boys and not in girls with perinatal exposure to TCDD. Our findings in Vietnam also suggest that boys are more susceptible to toxicity of perinatal TCDD exposure on neurodevelopment than girls.
However, in a 3-year-follow-up study for children from Bien Hoa birth cohort 2012, we found a decreased percentage of the total fixation duration on facial areas when viewing static facial images was also found in 3-year-old-girls who were exposed to high TCDD levels (3.5 pg/g lipid)5,13. Additionally, in the Da Nang cohort, we found effects of perinatal TCDD exposure on neurodevelopment in girls at 3 and 8 years old, which resulted in increased autistic behavior at 3 years old14 and feminine gaze behavior at 8 years old31. However, no effect on learning ability was found in these girls at 8 years old30. These findings suggest that neurodevelopmental effects induced by perinatal TCDD exposure in girls may be detectable in different domains of neurodevelopment and at a different age from boys.
In the present study, we found that perinatal TEQ-PCDD/Fs exposure decreased attention to the eyes when viewing talking scenes in girls. Seeing the face, including eye contact, can help children to improve their learning ability and social skills, leading to good neurodevelopment in the future32,33. A developmental milestone from 2 years can predict development in children and adulthood34,35 or is associated with psychiatric disorders, including neurodevelopmental disorders, such as autism diagnosed later in life36. Therefore, a follow-up study of these children is necessary until school age to clarify the association between dioxin exposure and neurodevelopment, especially language ability and social communication skills.
Limitations
In this study, we used eye tracking, which is an objective tool for assessment of gaze behavior. We found an adverse effect of perinatal dioxin exposure on the gaze pattern when viewing dynamic facial stimuli, particularly viewing talking facial scenes in 2-year-old Vietnamese children. However, we should interpret the present data within the context of several limitations. We used two videos of a Vietnamese boy in one video and a Vietnamese girl in the other who were interacting with the camera operator as stimuli to attract children to engage in the gaze test. However, these stimuli appeared to attract visual attention more effectively in girls compared with boys. Furthermore, this study had a relatively small sample size which made dose–response analysis difficult and did not have a control group of participants in non-exposed areas. Therefore, further epidemiological studies with a sufficient number of subjects and un-exposed controls with gaze behavior measurement when viewing a dynamic face are necessary to clarify this issue.
Conclusion
Perinatal dioxin exposure indicated by TCDD and TEQ-PCDD/Fs levels in breast milk reduced gaze behavior in 2-year-old children when viewing dynamic facial stimuli, particularly talking scenes, reflects their social communication ability. However, the effects of TCDD exposure on gaze behavior was more specific in boys than in girls, and TEQ-PCDD/Fs exposure was associated with decreased gaze behavior in girls. Furthermore, children with high TCDD levels in both sexes show increased autistic traits after 1 year of follow-up. These results suggest that poor gaze behavior, particularly in the face of children while talking, may predict increased autistic traits in early childhood.
Data availability
The datasets used and analysed during the current study available from the corresponding author on reasonable request.
Abbreviations
- TCDD:
-
2,3,7,8-Tetrachlorodibenzo-p-dioxin
- TEQ:
-
Toxic equivalency
- PCDDs:
-
Polychlorinated dibenzo-p-dioxins
- PCDFs:
-
Polychlorinated dibenzo-p-dioxins
- PCDD/Fs:
-
Polychlorinated dibenzo-p-dioxins/dibenzofurans
- FD:
-
Fixation duration
- AOI:
-
Areas of interest
- Bayley-III:
-
The Bayley scales of infant and toddler development, third edition
- ASRS:
-
The Autism spectrum rating scale
- FD:
-
Fixation duration
References
Nghi, T. N. et al. Dioxins and nonortho PCBs in breast milk of vietnamese mothers living in the largest hot spot of dioxin contamination. Environ. Sci. Technol. 49, 5732–5742. https://doi.org/10.1021/es506211p (2015).
Pham, N. T. et al. Perinatal dioxin exposure and neurodevelopment of 2-year-old Vietnamese children in the most contaminated area from Agent Orange in Vietnam. Sci. Total Environ. 678, 217–226. https://doi.org/10.1016/j.scitotenv.2019.04.425 (2019).
Nghiem, G. T. et al. Adverse effects of maternal dioxin exposure on fetal brain development before birth assessed by neonatal electroencephalography (EEG) leading to poor neurodevelopment; a 2-year follow-up study. Sci. Total Environ. 667, 718–729. https://doi.org/10.1016/j.scitotenv.2019.02.395 (2019).
Pham, N. T. et al. Effects of perinatal dioxin exposure on neonatal electroencephalography (EEG) activity of the quiet sleep stage in the most contaminated area from Agent Orange in Vietnam. Int. J. Hyg. Environ. Health 232, 113661. https://doi.org/10.1016/j.ijheh.2020.113661 (2021).
Pham, T. N. et al. Effect of perinatal dioxin exposure originating from agent orange on gaze behavior in 3-year-old children living in the most dioxin-contaminated areas in Vietnam. Toxics. https://doi.org/10.3390/toxics10040150 (2022).
Hosozawa, M., Tanaka, K., Shimizu, T., Nakano, T. & Kitazawa, S. How children with specific language impairment view social situations: An eye tracking study. Pediatrics 129, e1453-1460. https://doi.org/10.1542/peds.2011-2278 (2012).
Irwin, J. R. & Brancazio, L. Seeing to hear? Patterns of gaze to speaking faces in children with autism spectrum disorders. Front. Psychol. 5, 397. https://doi.org/10.3389/fpsyg.2014.00397 (2014).
The Office of the Vietnam National Steering Committee 33, Hatfield Consultants. Environmental and Human Health Assessment of Dioxin Contamination at Bien Hoa Airbase, Viet Nam; Final Report (Hatfield Consultants, 2011). https://www.hatfieldgroup.com/wp-content/uploads/2020/05/bien-hoa-final-full-report-august-2011.pdf. Assessed 30 Oct 2023.
Boda, H. et al. Prenatal dioxin exposure estimated from dioxins in breast milk and sex hormone levels in umbilical cord blood in Vietnamese newborn infants. Sci. Total Environ. 615, 1312–1318. https://doi.org/10.1016/j.scitotenv.2017.09.214 (2018).
Van den Berg, M. et al. The 2005 World Health Organization reevaluation of human and Mammalian toxic equivalency factors for dioxins and dioxin-like compounds. Toxicol. Sci. 93, 223–241. https://doi.org/10.1093/toxsci/kfl055 (2006).
Chawarska, K., Macari, S. & Shic, F. Context modulates attention to social scenes in toddlers with autism. J. Child. Psychol. Psychiatry 53, 903–913. https://doi.org/10.1111/j.1469-7610.2012.02538.x (2012).
Vo, M. L., Smith, T. J., Mital, P. K. & Henderson, J. M. Do the eyes really have it? Dynamic allocation of attention when viewing moving faces. J. Vis. 12, 3. https://doi.org/10.1167/12.13.3 (2012).
Thao, P. N. et al. Effects of dioxin exposure on gaze behaviour in 3-year-old children in Vietnam. Organohalogen Compd. 80, 125–128 (2018).
Nishijo, M. et al. 2,3,7,8-Tetrachlorodibenzo-p-dioxin in breast milk increases autistic traits of 3-year-old children in Vietnam. Mol. Psychiatry 19, 1220–1226. https://doi.org/10.1038/mp.2014.18 (2014).
Tai, P. T. et al. Dioxin exposure in breast milk and infant neurodevelopment in Vietnam. Occup. Environ. Med. 70, 656–662. https://doi.org/10.1136/oemed-2012-101021 (2013).
Tai, P. T. et al. Effects of perinatal dioxin exposure on development of children during the first 3 years of life. J. Pediatr. 175, 159–166. https://doi.org/10.1016/j.jpeds.2016.04.064 (2016).
Jung, Y., Lee, A. M., McKee, S. A. & Picciotto, M. R. Maternal smoking and autism spectrum disorder: Meta-analysis with population smoking metrics as moderators. Sci. Rep. 7, 4315. https://doi.org/10.1038/s41598-017-04413-1 (2017).
Herrmann, M., King, K. & Weitzman, M. Prenatal tobacco smoke and postnatal secondhand smoke exposure and child neurodevelopment. Curr. Opin. Pediatr. 20, 184–190. https://doi.org/10.1097/MOP.0b013e3282f56165 (2008).
Doi, H. et al. Prenatal exposure to a polychlorinated biphenyl (PCB) congener influences fixation duration on biological motion at 4-months-old: A preliminary study. PLoS ONE 8, e59196. https://doi.org/10.1371/journal.pone.0059196 (2013).
Dobs, K., Bulthoff, I. & Schultz, J. Use and usefulness of dynamic face stimuli for face perception studies: A review of behavioral findings and methodology. Front. Psychol. 9, 1355. https://doi.org/10.3389/fpsyg.2018.01355 (2018).
Schilbach, L. Towards a second-person neuropsychiatry. Philos. Trans. R. Soc. Lond. B 371, 20150081. https://doi.org/10.1098/rstb.2015.0081 (2016).
Pham, T. T. et al. Perinatal dioxin exposure and the neurodevelopment of Vietnamese toddlers at 1 year of age. Sci. Total Environ. 536, 575–581. https://doi.org/10.1016/j.scitotenv.2015.07.055 (2015).
Pham-The, T. et al. Perinatal dioxin exposure and attention deficit hyperactivity disorder (ADHD) symptoms in children living in a dioxin contamination hotspot in Vietnam. Toxics https://doi.org/10.3390/toxics10050212 (2022).
Schellart, N. A. & Reits, D. Influences of perinatal dioxin load to visual motion and oddball stimuli examined with an EEG and MEG analysis. Clin. Neurophysiol. 119, 1486–1495. https://doi.org/10.1016/j.clinph.2008.03.002 (2008).
Airdrie, J. N., Langley, K., Thapar, A. & van Goozen, S. H. M. Facial emotion recognition and eye gaze in attention-deficit/hyperactivity disorder with and without comorbid conduct disorder. J. Am. Acad. Child. Adolesc. Psychiatry 57, 561–570. https://doi.org/10.1016/j.jaac.2018.04.016 (2018).
Tsukimori, K. et al. Maternal exposure to high levels of dioxins in relation to birth weight in women affected by Yusho disease. Environ. Int. 38, 79–86. https://doi.org/10.1016/j.envint.2011.08.010 (2012).
Nakajima, S. et al. Sex-specific differences in effect of prenatal exposure to dioxin-like compounds on neurodevelopment in Japanese children: Sapporo cohort study. Environ. Res. 159, 222–231. https://doi.org/10.1016/j.envres.2017.08.006 (2017).
Vreugdenhil, H. J., Slijper, F. M., Mulder, P. G. & Weisglas-Kuperus, N. Effects of perinatal exposure to PCBs and dioxins on play behavior in Dutch children at school age. Environ. Health Perspect. 110, A593-598. https://doi.org/10.1289/ehp.021100593 (2002).
Tran, N. N. et al. Impacts of perinatal dioxin exposure on motor coordination and higher cognitive development in vietnamese preschool children: A five-year follow-up. PLoS ONE 11, e0147655. https://doi.org/10.1371/journal.pone.0147655 (2016).
Pham The, T. et al. Effects of perinatal dioxin exposure on learning abilities of 8-year-old children in Vietnam. Int. J. Hyg. Environ. Health 223, 132–141. https://doi.org/10.1016/j.ijheh.2019.09.010 (2020).
Pham, T. N. et al. Dioxin exposure and sexual dimorphism of gaze behavior in prepubertal Vietnamese children living in Da Nang, a hot spot for dioxin contamination. Sci. Total Environ. 749, 141083. https://doi.org/10.1016/j.scitotenv.2020.141083 (2020).
Heron-Delaney, M. et al. Perceptual training prevents the emergence of the other race effect during infancy. PLoS ONE 6, e19858. https://doi.org/10.1371/journal.pone.0019858 (2011).
Altvater-Mackensen, N. & Grossmann, T. Learning to match auditory and visual speech cues: Social influences on acquisition of phonological categories. Child Dev. 86, 362–378. https://doi.org/10.1111/cdev.12320 (2015).
Murray, G. K., Jones, P. B., Kuh, D. & Richards, M. Infant developmental milestones and subsequent cognitive function. Ann. Neurol. 62, 128–136. https://doi.org/10.1002/ana.21120 (2007).
Peyre, H. et al. Do developmental milestones at 4, 8, 12 and 24 months predict IQ at 5–6 years old? Results of the EDEN mother-child cohort. Eur. J. Paediatr. Neurol. 21, 272–279. https://doi.org/10.1016/j.ejpn.2016.11.001 (2017).
Falck-Ytter, T. et al. Reduced orienting to audiovisual synchrony in infancy predicts autism diagnosis at 3 years of age. J. Child. Psychol. Psychiatry 59, 872–880. https://doi.org/10.1111/jcpp.12863 (2018).
Acknowledgements
We would like to thank all mother-infant participating in this study. We are grateful to all mother-infant participating in the present study. We would like to thank staffs in Dong Nai Health Department, Dr Nguyen Xuan Hung, Dr Nguyen Thi Phuong Thao in Bien Hoa Health Center and staffs in ten communes around Bien Hoa airbase, and Dr. Le Thi Phuong Tram and staffs in Department of Obstetrics in Dong Nai prefectural hospital for their collaboration. We thank Ellen Knapp, PhD, from Edanz (https://edanz.com/ac) for editing a draft of this manuscript.
Funding
This work was supported partly by the Ministry of Education, Science, Sports and Culture, Grant-in-Aid for Scientific Research (B) (25305024) and (17H04665). The founders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript. The authors declare they have no actual or potential competing financial interests.
Author information
Authors and Affiliations
Contributions
P.N.T.: Conceptualization, formal analysis, writing—original draft, Writing—review & editing; M.N.: Conceptualization, Investigation, Formal analysis, Writing—review & editing; P.T.T.: Investigation; T.N.N.: Investigation; T.H.A.: Investigation; V.T.H.: Investigation; T.V.T.: Conceptualization, Investigation; Y.N.: Formal analysis, Writing—review; H.N.: Conceptualization, Writing—review & editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Thao, P.N., Nishijo, M., Tai, P.T. et al. Impacts of perinatal dioxin exposure on gaze behavior in 2-year-old children in the largest dioxin-contaminated area in Vietnam. Sci Rep 13, 20679 (2023). https://doi.org/10.1038/s41598-023-47893-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-47893-0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.