Abstract
To compare fecal level of short-chain fatty acid (SCFA) and some serum inflammatory markers between the low-carbohydrate (LCD) and the habitual (HD) diet, subjects were enrolled from our previous study on the effect of LCD vs. HD on gut microbiota in obese women following an energy-restricted diet. Serum interleukin-6 (IL-6) significantly increased in the HD group (p < 0.001). Adjusted for the baseline parameters, fecal level of butyric, propionic, and acetic acid were significantly different between the LCD and HD groups (p < 0.001, p = 0.02, and p < 0.001, respectively). Increase in serum insulin level correlated with decrease in fecal propionic acid by 5.3-folds (95% CI = − 2.7, − 0.15, p = 0.04). Increase in serum high sensitive C-reactive protein (hs-CRP) correlated with decrease in the percentage of fecal butyric acid by 25% (p = 0.04). Serum fasting blood sugar (FBS) and insulin showed a significant effect on fecal acetic acid (p = 0.009 and p = 0.01, respectively). Elevated serum FBS and insulin correlated with increase in fecal acetic acid by 2.8 and 8.9-folds (95%CI = 0.34, 1.9 and 1.2, 9.2), respectively. The LCD increased fecal SCFAs and a significant correlation was seen between serum IL-6 and fecal propionic acid level. More studies are needed to reach a concise correlation.
Trial registration number: The trial was registered in Iranian ClinicalTrials.gov IRCT20200929048876N3.
Similar content being viewed by others
Due to the high prevalence and incidence of obesity and its strong relationship with all chronic complications, successful and harmless anti-obesity strategies are the primary proceeding in the health system1. The effect of diet on insulin sensitivity is definite2; however, the optimal diet is not clear. People consume macronutrients in different percentages3. Low carbohydrate diet can be varied in terms of carbohydrate content and quality4. Therefore there is no consensus on precise definitions and comparisons among studies5. Recently, prevalence of obesity has increased due to high carbohydrate and fat consumption across the world6. Low-carb diet is more popular for inducing rapid weight loss7. However, the side effects due to high intake of fat reduce the adherence to this pattern8,9. Changes in dietary fatty acids are suggested to prevent metabolic complications induced by a high fat diet10. It is reported that saturated fatty acids (SFAs) are more obesogenic than mono- and polyunsaturated fatty acids (MUFAs and PUFAs), because diet-induced thermogenesis are higher in a diet rich in MUFAs and PUFAs than SFAs11. Low-carb diet has been shown an improve effect on blood glucose, serum insulin, homeostasis model assessment of insulin resistance (HOMA-IR), and blood pressure in obese patients12.
Short-chain fatty acids (SCFAs) are the end product of undigested/unabsorbed dietary components by the gut microbiota that has been received more attention, recently due to their role in the gut barrier and metabolism13,14. Previously, some randomized controlled trials showed that a western-style diet promotes inflammation, changes the profile of gut microbiota to the obese pattern, and decreases the amount of beneficial gut microbiota, especially Lactobacillus sp. and Bifidobacterium sp15,16. However, the plant-based and Mediterranean diets increased the abundance of protective microbiota and the protectors of intestinal barrier including Bifidobacteria and Lactobacillus17. Moreover, butyrate-producing bacteria including increased and inflammation-inducing lipopolysaccharides decreased17,18,19. In our previous study, the gut Actinobacteria population significantly increased in women who received a low-carb diet, however the Proteobacteria population significantly decreased in this group compared to the habitual diet. Moreover, changes in the gut microbiota population affected on the serum atherogenic and antioxidant status20. Following this, we hypothesized that the metabolites of the gut microbiota, especially SCFAs, will change after alterations in the microbiota composition. Acetate, propionate, and butyrate are the main SCFAs with 60:20:20 molar ratios in the colon and stool, depending on the dietary components and the diversity of gut microbiota21,22. In a recent cell culture study, a potential correlation was founded between fecal SCFAs and inflammation23; however this association has not been studied in a human population, up to date. Herein, we compared the effects of the low-carb (LCD) and habitual (HD) diets on fecal level of SCFAs, and some inflammatory markers in women who participated in our previous study20. In addition, the correlation between serum inflammatory markers and fecal SCFAs was assessed.
Results
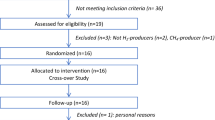
The study protocol is illustrated in Fig. 1. Total calorie, protein, fat, carbohydrate, and fiber had no significant difference between the two studied groups at the baseline20.
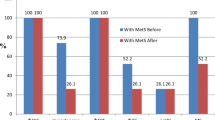
As shown in Table 1, a significant difference was seen in weight and waist circumference (WC) at the end of intervention in both dietary groups (p < 0.001 in all). The mean changes in weight and WC were not statistically significant between the two groups. Waist-to-hip ratio (WHR) significantly decreased from baseline up to the end in the LCD (p = 0.001) and HD (p = 0.01) groups. Serum insulin and HOMA-IR significantly decreased (p = 0.001 and p = 0.003, respectively) in the HD compared to the LCD. Serum interleukin-6 (IL-6) significantly increased in the HD group from baseline up to the end (p < 0.001). Serum high sensitive C-reactive protein (hs-CRP) level significantly decreased in both dietary groups at the end (p = 0.01 in the LCD and p = 0.04 in the HD). There was no significant difference between the two dietary groups in Bacteroidetes and Firmicutes population, before and after the intervention. Positive-Actinobacteria and Proteobacteria participants significantly increased and decreased after six weeks of the LCD intake (p = 0.002 and p = 0.004, respectively). Positive-Actinobacteria participants were significantly higher in the LCD than the HD group at the end (p = 0.03). Fecal acetic acid significantly increased after six weeks of intervention in both dietary groups (p < 0.001), however the fecal acetic acid level was significantly higher in the LCD compared to the HD group at the end (p < 0.001). Fecal propionic acid significantly increased in both dietary groups from baseline up to the end (p < 0.001). After six weeks of intervention, propionic acid level was significantly higher in the stool of participants received the LCD than the HD (p = 0.003). Butyric acid significantly increased in both dietary groups at the end, however, this elevation was significantly higher in the stool of women received the LCD than the HD (p < 0.001 in all comparisons). As shown in Table 2, the mean change of WHR was significantly higher in the LCD than in the HD group (p = 0.01). The decrease in IL-6 was significantly higher in the serum of women received the LCD than the HD (p = 0.009). The mean changes of serum insulin and HOMA-IR were significantly higher in women on a HD than the LCD (p = 0.008 and p = 0.01, respectively). The mean changes of fecal acetic, propionic, and butyric acid were significantly higher in women on the LCD than the HD (p < 0.001, p = 0.001 and p < 0.001, respectively).
Baseline comparisons between the LCD to HD and HD to LCD groups showed a significant difference in serum IL-6 and hs-CRP level (p = 0.02, and p < 0.001, respectively). Weight (p < 0.001), WC (p < 0.001), WHR (p = 0.04), serum fasting blood sugar (FBS) (p = 0.03), insulin (p = 0.001) and HOMA-IR (p = 0.003) significantly decreased in the HD to LCD group, after six weeks of intervention. In the LCD to HD group, weight (p < 0.001), waist circumference (p = 0.001), WHR (p = 0.01), serum IL-6 (p = 0.001) and hs-CRP (p = 0.009) significantly decreased after six weeks. Positive-Actinobacteria participants significantly increased in women on a HD to LCD at the end (p = 0.02) (Table 3). The mean changes of serum insulin and HOMA-IR were significantly higher in the HD to LCD than the LCD to HD group (p = 0.04, and p = 0.04, respectively). The mean changes of serum hs-CRP were significantly higher in the LCD to HD than in the HD to LCD group (p = 0.04). Other parameters had no significant difference between the two groups. (Table 4).
The mean changes of fecal SCFAs were adjusted for the baseline parameters and no significant effect was observed. Adjusted for the baseline parameters, fecal level of butyric, propionic, and acetic acid were significantly different between women on the LCD and HD (p < 0.001, p = 0.02, and p < 0.001, respectively). Only serum insulin level showed a significant effect on the fecal level of propionic acid (p = 0.04). Increase in serum insulin level decreased fecal level of propionic acid by 5.3-folds (95% CI = − 2.7, − 0.15). (Table 5).
Moreover, fecal butyric, propionic, and acetic acid had no significant difference between the LCD to HD and the HD to LCD group, adjusting for the parameters. Serum hs-CRP showed a significant effect on fecal level of butyric acid (p = 0.04). Increase in serum hs-CRP decreased the percentage of fecal butyric acid by 25%. Fecal propionic acid showed a significant effect on butyric acid level (p = 0.03). Serum FBS and insulin showed a significant effect on fecal level of acetic acid (p = 0.009 and p = 0.01, respectively). Elevated serum FBS and insulin increased fecal level of acetic acid by 2.8 and 8.9-folds (95% CI = 0.34, 1.9 and 1.2, 9.2), respectively. Fecal propionic and butyric acid showed a significant effect on acetic acid (p = 0.01 and p = 0.02, respectively). (Table 6).
In the LCD group, fecal level of propionic acid showed a significant correlation with the positive- Actinobacteria population (r = 0.45, p = 0.04). Moreover, a significant correlation was shown between changes in fecal level of propionic acid with serum IL-6 (r = 0.46, p = 0.04). In the HD to LCD group, a significant correlation was founded between the positive- Proteobacteria population and fecal level of propionic acid (r = 0.7, p = 0.005). (Table 7).
Discussion
As a novel finding, serum hs-CRP level showed a significant effect on fecal level of butyric acid. Moreover, a mutual relationship was observed between the Actinobacteria population and fecal level of propionic acid. Moreover, a mutual relationship was observed between fecal level of propionic acid and serum IL-6, as an initiating factor of inflammatory pathways. The SCFAs, carboxylic acids with aliphatic tails of 1–6 carbons, are volatile bacterial metabolites of unabsorbed/ undigested food components, especially carbohydrates in the large intestine. Non-digestible dietary fibers are the main substrates for bacterial fermentation to produce acetic (C2), propionic (C3), and butyric (C4) acids, as the most abundant SCFAs in the colon which have various impacts on human metabolism and health24. Different microorganisms in the gut produce SCFAs through various pathways25,26,27,28. The main butyrate producing-bacteria in the human gut belong to the Firmicutes phyla. Moreover, sugar and lactate-utilizing bacteria, such as Eubacterium hallii and Anaerostipes spp. produce butyrate from lactate and acetate29. The Actinobacteria, Bacteroidetes, Fusobacteria, Proteobacteria, Spirochaetes, and Thermotogae can produce butyrate through an increase in gene expression of butyryl-CoA dehydrogenase, butyryl-CoA transferase and/or butyrate kinase30. The Actinobacteria phyla regulate the production of acetate and propionate in the gut31. These data are consistent with our study that the positive-Actinobacteria women increased, but positive-Proteobacteria population decreased in the LCD group20,32. In the present study, the Actinobacteria showed a significant correlation with fecal level of propionic acid. Alteration in fecal SCFAs occurred due to changes in phyla population in the gut33. Obesity has been associated with increase in the Firmicutes and decrease in the Bacteroidetes population in previous studies34,35. In the present study, no change in the Firmicutes and Bacteroidetes population was observed before and after intervention. Difference in foods, region, culture, climate, and ethnicity make variations in these results that create novelty in this field.
The SCFAs induce epigenetic modifications such as changes in DNA methylation and micro-RNA expression36. They regulate appetite, lipogenesis, gluconeogenesis, and inflammation which have potential effects on health status, susceptibility to obesity, and related complications37. The SCFAs affect inflammatory pathways via several mechanisms including regulating the cytokine production, activating the acetylation of G-protein-coupled receptors (GPCRs), and tight junction proteins that finally strengthen the intestinal integrity, which is one of the important factors for inflammatory pathways38. There is a relationship between oxidative stress, inflammation, and the gut barrier status. Oxidative stress degrades the intestinal integrity by activating the signaling pathways of nuclear factor kappa- B (NF-κB), insulin receptor kinase, and mitogen-activated protein kinase (MAPK). Inflammation and damage to the intestinal barrier interact by regulating the expression of tumor necrosis factor (TNF), claudin-2, occludin, and zonula occludens-1 (ZO1). Oxidative stress directly promotes inflammation by inhibiting the NF-κB activity and the expression of TNF-α and interleukin-1 beta (IL-1β)39. Our previous study showed that the LCD increases the Actinobacteria population in the gut and improves serum total antioxidant capacity which is associated with higher capability of the body for reactive oxygen scavenges. Moreover, decrease in the Proteobacteria population lead to lower oxidant status in the body19. Association between the SCFAs and serum inflammatory markers has been studied in some previous animal models40,41. For example, dietary sodium butyrate supplementation reduced serum IL-6 and TNF-α level in pigs. The number of Clostridium and Escherichia coli decreased, but the number of Lactobacillus spp increased in the gut of pigs40. Lactobacillus is facultative anaerobic bacteria belong to the Firmicutes phyla that metabolize carbohydrates to produce lactic acid41. We did not assess the species of bacteria in each phylum in the present study. Participants only studied for positive or negative phyla and no difference was observed in the Firmicutes population between the groups before and after six weeks.
Dietary composition changes the gut microbiota and the produced SCFAs, as the final metabolites of undigested food in the large intestine42,43. Previous human study reported that a western style diet with a high intake of refined carbohydrates and saturated fats promotes inflammation by a change in the Actinobacteria population16. But, plant-based diet increased butyrate-producing bacteria belonging to the Actinobacteria phyla, however decreased inflammation-inducing bacteria, as the members of Proteobacteria phyla17,18,19. Our results are in accordance with the mentioned studies. Dietary fat was provided from PUFAs in the present LCD that leads to increase in Actinobacteria population in the gut. Higher Actinobacteria population correlated with higher fecal level of propionate and lower serum IL-6. Animal models feeding propionate and butyrate-enriched high-fat diet were resistant to obesity and improved blood glucose levels44,45,46,47. In human studies, propionate supplementation increased the satiety hormones including peptide YY (PYY) and glucagon-like peptide (GLP-1), which have been related to lower serum FBS and higher insulin secrection in the body48,49. Herein, fecal level of butyric acid significantly increased in the LCD compared to the HD group. As we previously reported, positive- Actinobacteria and Proteobacteria participants significantly increased and decreased after the LCD, respectively20. An inverse association has been reported between the intestinal propionate and butyrate level with inflammation50 which is consistent with our results. Propionate inhibits histone deacetylases (HDACs) and activates histone acetyltransferases (HATs), which are associated with inflammatory- and immune-regulatory pathways51. In addition, it regulates cytokine expression in T-cells and generates the regulatory T-cells (Tregs) through HDAC inhibition52. Recently, a population-based study in China showed a positive correlation between the butyrate and BMI status. No statistical significant difference was observed between the SCFAs-producers of bacteria and BMI. Plasma levels of SCFAs positively associated with BMI. They concluded that the colonic fermentation of undigested/unabsorbed foods differs in adults with and without overweight/obesity53. Our results showed no correlation between the fecal levels of butyrate with anthropometric measures, and serum inflammatory markers. Differences in the ethnicity make variety in the gut microbiota population and their species that change the final produced metabolites, especially SCFAs. A recent study on morbid obese patients referred for one anastomosis gastric bypass- mini gastric bypass showed the beneficial effect of probiotic on serum IL-6, TNF-α and hs-CRP after 16 weeks of supplementation, however the mean changes of serum TNF-α was only statistically significant between the supplemented [-6.18 (-12.69, 0.32)] and placebo [4.04 (− 1.18, 9.26)] groups. Moreover, serum FBS, insulin and HOMA-IR improved at the end of study in the supplemented group, but the mean changes were not statistically significant between the two groups. In addition anthropometric measures including the percentage excess weight loss, WC, BMI and weight significantly decreased after sixteen weeks of supplementation in the probiotic group, however the mean changes of WC was not statistically significant between the supplemented and placebo group54. Our results are in accordance with the mentioned study about inflammatory markers; however we did not measure serum TNF-α. This study was a randomized controlled trial that compared the effect of probiotic supplementation containing seven species of bacteria belonging to the Actinobacteria and Firmicutes phyla with placebo on anthropometric measures, glycemic indices and serum inflammatory markers in patients under the bypass surgery that is different in the study design and intervention with our study. But, the beneficial effect of the Actinobacteria phyla on serum inflammatory markers has been observed in both studies. Recent reviews have been discussed about the role of SCFAs in the redox signaling pathways, protection against bone loss, and inflammation13,14, however no human randomized controlled trial with a cross-over design was founded in this field. Therefore, similar to all novel studies, the present study has some limitations. The sample size was very small, and only women were enrolled. More clinical trials with larger sample sizes are needed. The levels of SCFAs were only measured in the stool and there is no data about their levels in serum. In addition, we did not study the absorption of SCFAs. It is still an open question whether the elevation of fecal SCFAs is because of a decrease in the gut absorption or not. The bacterial species did not assay in the present study. Determination of actual values of phyla and species make the changes more debatable. This is a new field of study that needs more future attempts to clearly describe underlying mechanisms and impacts of these changes in human health. A complex interaction between the genetic background, the gut microbiota, and diet has been opened a new target and tool for the personalized medical nutrition therapy.
Materials and methods
Participants and interventions
Block randomization was used as two groups with 5-number blocks, including four participants in each block. The randomization unit was the person, and we used random allocation software for this purpose. Random coded boxes were used for concealment. In this method, cans with similar weight, shape, and color, which were numbered according to the random sequence, was used. Our previous study on effects of the HD and LCD on the gut microbiota in women with obesity (BMI ≥ 30 kg/m2)20, is followed here by measuring the diet’s impact on fecal levels of SCFAs, as the main metabolites of the gut phyla. The hypocaloric HD and LCD were prescribed for six weeks with two weeks of washout period. According to the previous study, two weeks is sufficient for removing the effect of diets on the gut microbiota55. Hypocalorie diets were prescribed with 500 kcal reductions from the total daily calorie requirements for 0.5 kg weight loss in each week. From total energy requirements, 55%, 25%, and 20% were provided from fat, protein, and carbohydrate, respectively. The HD was a 500 kcal- reduced calorie diet that provided 20%, 15%, and 65% of total daily calories from fat, protein, and carbohydrate. The PUFAs were advised as the main source of dietary fat and fiber was prescribed in similar amounts (20 g/day) in both diets. In the washout period, the weight maintenance HD was prescribed based on 1.4–1.5 × resting energy expenditure for all participants. Compliance was assessed by the food diary and participants who followed < 80% of the dietary plan were excluded. The present study was ethically approved by the ethical committee of Zanjan University of Medical Sciences (IR.ZUMS.REC.1400.094). The informed consent form was obtained from all subjects. The present trial has been registered at the IRCT on 08-01-2021 under the registration number: IRCT20200929048876N3. All of the procedure was performed according to the Declaration of Helsinki.
Anthropometric and biochemical measurements
Anthropometric measurements were recorded, and fasting blood samples were collected in our previous study20. Fasting serum insulin, IL-6, and hs-CRP were measured according to the ELISA method based on the manufacturer’s instruction (Pars Azmoon Co., Iran). The HOMA-IR was computed according to the below formula;
Extraction of SCFAs
The stool sample was gathered at the baseline and end of each dietary intervention and maintained in a refrigerator ( − 80 °C) for final analysis. The fecal SCFA analysis was carried out using gas chromatography-mass spectrometry (GC–MS). Before GC analysis, the fecal samples were subjected to an acid–base treatment followed by ether extraction, and derivatization.
The concentrations of volatile fatty acids were determined using a gas chromatography system (Agilent Chromatography System, model 7890B), equipped with a capillary column according to the method described previously56.
Briefly, 1 mL of 25% metaphosphoric acid was mixed with 1 g of sample in a centrifuge tube and the mixture was frozen overnight. The samples were then thawed, mixed with 0.4 mL of 25% NaOH, and vortexed, followed by the addition of 0.64 mL of 0.3 mol L−1 oxalic acid. The samples were centrifuged for 20 min at 3000g at 4 °C. Then, 2 mL of the supernatant was transferred into a gas chromatography vial. Helium as the carrier gas was used at a constant flow rate of 1 ml min−1. The initial column oven temperature was 50 °C for 2 min and increased to 70 °C at a rate of 10 °C min-1. Then, the temperature was increased to 85 °C at a rate of 3 °C min−1, then increased to 110 °C at a rate of 5 °C min−1, and then increased at a rate of 30 °C min−1 to a final temperature of 290 °C, where it was held for 5 min. The temperatures of the front inlet, transfer line, and mass source were set at 260 °C, 290 °C, and 230 °C, respectively.
Statistical analyses
The number of participants was calculated according to the previous study with the effects of dietary intervention on the gut microbiota to change in the production of SCFAs, as the post-hoc endpoint57. Considering a power of 80% in a two-sided test, and α = 0.05 (type I error), eight people were sufficient to show this effect. Therefore, eight participants were randomly selected from our previous study20. Correlation analysis was performed by the Kendall’s and Spearman tests. The effects of the dietary interventions on all outcomes were analyzed using SPSS 18v through parallel repeated measures. A linear regression model was used to adjust the effect of baseline variables on outcomes. Analysis was performed in two models of grouping; (1) the LCD vs. HD, and (2) the LCD to HD vs. HD to LCD.
Ethical statement
All of the procedure was performed according to the Declaration of Helsinki. The protocol was approved by the ethical committee of Zanjan University of Medical Sciences, Zanjan, Iran (IR.ZUMS.REC.1400.094).
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Lean, M. E. et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): An open-label, cluster-randomized trial. Lancet 391, 541–551 (2018).
Te Morenga, L., Docherty, P., Williams, S. & Mann, J. The effect of a diet moderately high in protein and fiber on insulin sensitivity measured using the dynamic insulin sensitivity and secretion test (DISST). Nutrients 9(12), 1291 (2017).
Venn, B. J. Macronutrients and human health for the 21st century. Nutrients 8, 2363 (2020).
Trumbo, P., Schlicker, S., Yates, A. A. & Poos, M. Food and nutrition board of the institute of medicine, the national academies. Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein and amino acids. J Am Diet Assoc. 102, 1621–30 (2002).
Dowis, K. & Banga, S. The potential health benefits of the ketogenic diet: A narrative review. Nutrients 13, 1654 (2021).
Boutari, C. & Mantzoros, C. S. A 2022 update on the epidemiology of obesity and a call to action: As its twin COVID-19 pandemic appears to be receding, the obesity and dysmetabolism pandemic continues to rage on. Metabolism 133, 155217 (2022).
Goss, A. et al. Effects of weight loss during a very low carbohydrate diet on specific adipose tissue depots and insulin sensitivity in older adults with obesity: A randomized clinical trial. Nutr. Metab. 17, 64 (2020).
Chawla, S., Tessarolo Silva, F., Amaral Medeiros, S., Mekary, R. A. & Radenkovic, D. The effect of low-fat and low-carbohydrate diets on weight loss and lipid levels: A systematic review and meta-analysis. Nutrients. 12, 3774 (2020).
Mousavi, S. N. et al. Extra virgin olive oil in maternal diet increases osteogenic genes expression, but high amounts have deleterious effects on bones in mice offspring at adolescence. Iran. J. Basic. Med. Sci. 19, 1299–1307 (2016).
Tutunchi, H., Ostadrahimi, A. & Saghafi-Asl, M. The effects of diets enriched in monounsaturated oleic acid on the management and prevention of obesity: A systematic review of human intervention studies. Adv. Nutr. 11, 864–877 (2020).
Krishnan, S. & Cooper, J. A. Effect of dietary fatty acid composition on substrate utilization and body weight maintenance in humans. Eur. J. Nutr. 53, 691–710 (2014).
Van Baak, M. A. et al. Dietary intake of protein from different sources and weight regain, changes in body composition and cardiometabolic risk factors after weight loss: The DIOGenes study. Nutrients. 9, E1326 (2017).
Lucas, S. et al. Short-chain fatty acids regulate systemic bone mass and protect from pathological bone loss. Nat Commun. 9, 55 (2018).
González-Bosch, C., Boorman, E., Zunszain, P. A. & Mann, G. E. Short-chain fatty acids as modulators of redox signaling in health and disease. Redox Biol. 47, 102165 (2021).
Bell, D. S. Changes seen in gut bacteria content and distribution with obesity: Causation or association?. Postgrad. Med. 127, 863–868 (2015).
Statovci, D., Aguilera, M., MacSharry, J. & Melgar, S. The impact of Western diet and nutrients on the microbiota and immune response at mucosal interfaces. Front. Immunol. 8, 838 (2017).
Tomova, A. et al. The effects of vegetarian and vegan diets on gut microbiota. Front. Nutr. 6, 47 (2019).
Garcia-Mantrana, I., Selma-Royo, M., Alcantara, C. & Collado, M. C. Shifts on gut microbiota associated to Mediterranean diet adherence and specific dietary intakes on general adult population. Front. Microbiol. 9, 890 (2018).
Coelho, O. G., Cândido, F. G. & Alfenas, R. C. Dietary fat and gut microbiota: Mechanisms involved in obesity control. Crit. Rev. Food Sci. Nutr. 59, 3045–3053 (2019).
Tehrani, L. H. G., Mousavi, S. N., Chiti, H. & Afshar, D. Effect of Atkins versus a low-fat diet on gut microbiota, and cardiometabolic markers in obese women following an energy-restricted diet: Randomized, crossover trial. Nutr. Metab. Cardiovasc. Dis. 32, 1734–1741 (2022).
Binder, H. J. Role of colonic short-chain fatty acid transport in diarrhea. Annu. Rev. Physiol. 72, 297–313 (2010).
Hijova, E. & Chmelarova, A. Short chain fatty acids and colonic health. Bratisl. Lek. Listy. 108, 354–358 (2007).
Rutting, S. et al. Short-chain fatty acids increase TNFα-induced inflammation in primary human lung mesenchymal cells through the activation of p38 MAPK. Am. J. Physiol. Lung Cell Mol. Physiol. 316, L157–L174 (2019).
Ríos-Covián, D. et al. Intestinal short chain fatty acids and their link with diet and human health. Front. Microbiol. 7, 185 (2016).
Louis, P., Hold, G. L. & Flint, H. J. The gut microbiota, bacterial metabolites and colorectal cancer. Nat. Rev. Microbiol. 12, 661–672 (2014).
Rey, F. E. et al. Dissecting the in vivo metabolic potential of two human gut acetogens. J. Biol. Chem. 285, 22082–22090 (2010).
Scott, K. P., Martin, J. C., Campbell, G., Mayer, C. D. & Flint, H. J. Whole-genome transcription profiling reveals genes up-regulated by growth on fucose in the human gut bacterium “Roseburia inulinivorans”. J. Bacteriol. 188(12), 4340–4349 (2006).
Duncan, S. H., Barcenilla, A., Stewart, C. S., Pryde, S. E. & Flint, H. J. Acetate utilization and butyryl coenzyme A (CoA): Acetate-CoA transferase in butyrate-producing bacteria from the human large intestine. Appl. Environ. Microbiol. 68, 5186–5190 (2002).
Louis, P. & Flint, H. J. Diversity, metabolism and microbial ecology of butyrate-producing bacteria from the human large intestine. FEMS Microbiol. Lett. 294, 1–8 (2009).
Vital, M., Howe, A. C. & Tiedje, J. M. Revealing the bacterial butyrate synthesis pathways by analyzing (meta) genomic data. MBio 5, 1–11 (2014).
Rivière, A., Selak, M., Lantin, D., Leroy, F. & De Vuyst, L. Bifidobacteria and butyrate-producing colon bacteria: Importance and strategies for their stimulation in the human gut. Front. Microbiol. 7, 979 (2016).
Karbalaiee, M., Chiti, H., Mousavi, S. N. & Afshar, D. Low-carbohydrate hypo calorie diet has a beneficial effect on gut phyla and metabolic markers in healthy women with obesity: A randomized crossover study. Obes. Med. 35, 100461 (2022).
Sun, D., Chen, Y. & Fang, J. Influence of the microbiota on epigenetics in colorectal cancer. Natl. Sci. Rev. 6, 1138–1148 (2019).
Turnbaugh, P. et al. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 444, 1027–1031 (2006).
Ley, R. E., Turnbaugh, P. J., Klein, S. & Gordon, J. I. Human gut microbes associated with obesity. Nature 444, 1022–1023 (2006).
Sun, Q., Jia, Q., Song, L. & Duan, L. Alterations in fecal short-chain fatty acids in patients with irritable bowel syndrome: A systematic review and meta-analysis. Medicine. 98, e14513 (2019).
Cuevas-Sierra, A., Ramos-Lopez, O., Riezu-Boj, J. I., Milagro, F. I. & Martinez, J. A. Diet, gut microbiota, and obesity: links with host genetics and epigenetics and potential applications. Adv. Nutr. 10, S17–S30 (2019).
Venegas, P. et al. Short chain fatty acids (SCFAs)-mediated gut epithelial and immune regulation and its relevance for inflammatory bowel diseases. Front. Immunol. 10, 277 (2019).
Liu, P. et al. The role of short-chain fatty acids in intestinal barrier function, inflammation, oxidative stress, and colonic carcinogenesis. Pharmacol. Res. 165, 105420 (2021).
Wen, Z. S., Lu, J. J. & Zou, X. T. Effects of sodium butyrate on the intestinal morphology and DNA-binding activity of intestinal nuclear factor-κB in weanling pigs. J. Anim. Vet. Adv. 11, 814–21 (2012).
Hammes, W. P. & Hertel, C. Lactobacillus. In: Whitman WB, Rainey F, Kämpfer P, Trujillo M, Chun J, DeVos P, et al, editors. Bergey's Manual of Systematics of Archaea and Bacteria. (John Wiley & Sons, Inc.) In Association With Bergey’s Manual Trust. 1–76 (2015).
Schnorr, S. L. et al. Gut microbiome of the Hadza hunter-gatherers. Nat. Commun. 5, 3654 (2014).
Sonnenburg, E. D. et al. Diet-induced extinctions in the gut microbiota compound over generations. Nature 1529, 212–215 (2016).
De Vadder, F. et al. Microbiota-generated metabolites promote metabolic benefits via gut-brain neural circuits. Cell 156, 84–96 (2014).
Lin, H. V. et al. Butyrate and propionate protect against diet-induced obesity and regulate gut hormones via free fatty acid receptor 3-independent mechanisms. PLoS ONE. 7, e35240 (2012).
Yamashita, H. et al. Improvement of obesity and glucose tolerance by acetate in Type 2 diabetic Otsuka Long-Evans Tokushima Fatty (OLETF) rats. Biosci. Biotechnol. Biochem. 71, 1236–1243 (2007).
Gao, Z. et al. Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes 58, 1509–1517 (2009).
Chambers, E. S. et al. Effects of targeted delivery of propionate to the human colon on appetite regulation, body weight maintenance and adiposity in overweight adults. Gut 64, 1744–1754 (2015).
Freeland, K. R. & Wolever, T. M. S. Acute effects of intravenous and rectal acetate on glucagon-like peptide-1, peptide YY, ghrelin, adiponectin and tumour necrosis factor-alpha. Br. J. Nutr. 103, 460–466 (2010).
Machiels, K. et al. A decrease of the butyrate-producing species Roseburia hominis and Faecalibacterium prausnitzii defines dysbiosis in patients with ulcerative colitis. Gut. 63, 1275–1283 (2014).
Flint, H. J., Scott, K. P., Louis, P. & Duncan, S. H. The role of the gut microbiota in nutrition and health. Nat. Rev. Gastroenterol. Hepatol. 9, 577–589 (2012).
Furusawa, Y. et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature 504, 446–450 (2013).
Wang, Y. et al. Circulating short-chain fatty acids are positively associated with adiposity measures in Chinese adults. Nutrients. 12, 2127 (2020).
Karbaschian, Z. et al. Probiotic supplementation in morbid obese patients undergoing one anastomosis gastric bypass-mini gastric bypass (OAGB-MGB) surgery: A randomized, double-blind, placebo-controlled, clinical trial. Obes. Surg. 28, 2874–2885 (2018).
David, L. et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature. 505, 559e63 (2014).
Erwin, E. S., Marco, G. J. & Emery, E. M. Volatile fatty acid analyses of blood and rumen fluid by gas chromatography. J. Dairy Sci. 44, 1768–1771 (1961).
Yamamura, R. et al. Associations of gut microbiota, dietary intake, and serum short-chain fatty acids with fecal short-chain fatty acids. Biosci. Microbiota Food Health. 39, 11–17 (2020).
Acknowledgements
All authors are very thanks from women who participated in the present study.
Funding
The present study is financially supported by Zanjan Metabolic Diseases Research Center, Zanjan University of Medical Sciences, Zanjan, Iran.
Author information
Authors and Affiliations
Contributions
S.N.M. and H.C. conceived and designed research. S.N.M. and Z.A. conducted experiments. S.N.M. and Z.A. analyzed data. S.N.M. wrote the manuscript. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rad, Z.A., Mousavi, S.N. & Chiti, H. A low-carb diet increases fecal short-chain fatty acids in feces of obese women following a weight-loss program: randomized feeding trial. Sci Rep 13, 18146 (2023). https://doi.org/10.1038/s41598-023-45054-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-45054-x
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.