Abstract
Several Eurydema species (Hemiptera: Pentatomidae) are considered as pests, however, reports on their chemical ecology are scarce. In the current study we focused on Eurydema ornata (Linnaeus) a pentatomid pest of several brassicaceous crops. Since the species is known to feed preferably on generative parts of plants, a series of floral and green leaf volatiles were tested by electroantennography and compounds eliciting remarkable responses were also tested in the field. Three compounds elicited the most outstanding responses from antennae of E. ornata: allyl isothiocyanate, phenylacetaldehyde and ± linalool. Field experiments were conducted in Hungary between 2017 and 2021 to test the potential attractive effects of the compounds. Three Eurydema species were caught in the experiments E. ornata, E. oleracea (Linnaeus) and E. ventralis Kolenati. In the experiments combinations containing allyl isothiocyanate attracted both males and females of E. ornata. The compound was also attractive on its own, in a positive, dose-dependent manner. When presented alone, neither phenylacetaldehyde nor ± linalool was attractive to the species, furthermore, addition of these compounds to allyl isothiocyanate did not affect attraction considerably. To our knowledge this is the first demonstration of field attration of an Eurydema species to a semiochemical and one of the few reports on trapping of a pentatomid species with a synthetic plant volatile in the field. Perspectives regarding research and potential practical applications are discussed in the paper.
Similar content being viewed by others
Introduction
Stink bugs (Pentatomidae) is a species rich family of true bugs (Heteroptera). Several pentatomids are considered as pests, and some may cause severe damages to crops1. Knowledge of the chemical ecology of pests is important, as it may provide tools for agricultural practice (e.g.2).
Recently, Weber et al.3 published a comprehensive review on the chemical ecology of Pentatomoidea. Studies on chemical ecology of Pentatomidae primarily focused on pheromone composition, however, for a lot of presumed pheromones information on their field activity is missing3,4. Furthermore, for several pentatomid pheromones it was found that bugs are attracted only to the vicinity of the traps, but they do not enter4. Beside pheromones plant volatiles may provide possibilities as attractants of insect pests (e.g.5). However, reports on attraction of pentatomids to plant volatiles are scarce3.
The genus Eurydema (Hemiptera: Pentatomidae) includes several, in general oligophagous pentatomids, which primarily feed on cruciferous plants, several species are considered as pests6,7,8,9,10,11.
Despite their importance, very few studies have been published concerning the chemical ecology of Eurydema spp. Aldrich et al.12 identified compounds from metathoracic scent glands of males of E. ventralis Kolenati and E. oleracea (Linnaeus), but behavioral responses were not recorded. Other studies focused on host plant choice of E. ornata (Linnaeus) and E. pulchrum Westwood13,14. Although the authors found differences in preferences for different host plants, no semiochemicals were identified in these studies. To our knowledge to date no reports on behavioral response of Eurydema spp. to semiochemicals have been published.
E. ornata is an important pest of cruciferous crops and caper6,8,11,15. The species was found to feed preferably on generative parts of plants11,16.
In our study we conducted electroantennographic screenings and field experiments on E. ornata in search for plant volatiles as potential attractants.
Materials and methods
Collected insects
Insects for electrophysiological studies were collected in the field with a sweep net at Halásztelek (Hungary). The collected individuals were taken to the laboratory and determined to species with a stereomicroscope, based on the works of Benedek17 and Derjanschi and Péricart18.
In the laboratory E. ornata adults collected were maintained in cylindric glass jars (diameter: 12.5 cm, height: 18 cm) on cauliflower sprouts on room temperature. Before the electrophysiological tests individuals were sexed and kept individually in glass vials.
Electrophysiology
All chemicals for electrophysiological tests and field experiments were obtained from Sigma Aldrich Kft. (Budapest, Hungary). For each compound 1 mg/ml hexane solutions were prepared. Stimuli for electroantennographic (EAG) screenings were prepared as follows: 5 µL of the test solutions were loaded on 10 mm diameter Rotilabo filter discs (RKTech Kft., Budapest, Hungary) inside a Pasteur pipette.
In accordance with the observation that E. ornata prefers to feed on generative parts of plants11,16, the series of tested stimuli included several floral volatiles along with green leaf volatiles and allyl isothiocyanate, a characteristic compound of brassicaceous plants, which was found attractive to several pestiferous insects with cruciferous host plants (e.g.19). During the screening myrcene was used as a standard and was applied before and after the tested stimuli. Tested stimuli also included solvent control, in which only hexane was loaded on the filter disc and blank control, containing only an unloaded filter disc.
For electrophysiological tests, the antennae of live E. ornata adults were cut near the base and were mounted between glass capillaries containing Ringer solution. One of the electrodes was grounded while the other was connected to a high impedance DC amplifier (IDAC-2, Ockenfels Syntech GmbH, Buchenbach, Germany).
For presenting the stimuli to the antennae, a stainless steel tube (teflon coated inside) with a constant humidified airflow was set up. Stimuli were presented by a stimulus controller (CS-55, Ockenfels Syntech GmbH, Buchenbach, Germany), with a 0.5 s pulse duration. Measurements were made with the GC-EAD sofware (Ockenfels Syntech GmbH, Buchenbach, Germany). Stimuli were administered at ca. 20–30 s intervals. For evaluation of results, response amplitudes were normalized against the response means of the standard.
Preparation of baits
Baits were prepared as follows: individual compounds, or blends of compounds were loaded on cotton wicks in polyethylene vial dispensers with lid (No. 730, Kartell Co., Italy). In Experiment 1 and 2, load of individual compounds was kept at 100 µL, both if loaded individually or in combination. In the dose–response experiment (Exp. 3) 1, 10 or 100 µL of allyl isothiocyanate was loaded in the dispensers. The lids of loaded dispensers were closed and for easier handling dispensers were attached to 8 × 1 cm plastic handles. Baits were wrapped singly in pieces of aluminium foil and stored at −18 °C until used. In the field baits were replaced after 4–5 weeks, as previous experience showed that the baits do not lose their activity during this period20.
Field experiments
All field experiments were conducted at Halásztelek (Central Hungary) at an adandoned field with wild cruciferous plants. For field testing of synthetic compounds, CSALOMON® VARb3 funnel traps were used (produced by the Plant Protection Institute, Centre for Agricultural Research, Budapest, Hungary). A small piece (1 × 1 cm) of household anti-moth strip (Chemotox®, Sara Lee; Temana Intl. Ltd, Slough, UK; active ingredient 15% dichlorvos) was placed in the containers to kill captured insects.
In the experiments treatments were set out in a randomized complete block design, with 5–8 m distance between traps. To avoid positional effects, trap positions were rotated regularly, as a rule on a fortnightly basis.
Based on electroantennographic results, allyl isothiocyanate, ± linalool and phenylacetaldehyde were chosen to be tested in field experiments. Treatments of field experiments are listed in Table 1.
In Experiment 1 binary and ternary combinations of ± linalool, phenylacetaldehyde and allyl isothiocyanate were tested along with unbaited, control traps. The experiment was run at Halásztelek, from 17th May to 11th September, 2017, with four blocks.
In Experiment 2 ± linalool, phenylacetaldehyde and allyl isothiocyanate were tested alone and in ternary combination along with unbaited, control traps. The experiment was run at Halásztelek, from 5th August to 11th October, 2018, with 5 blocks.
In Experiment 3, 1, 10 and 100 mg doses of allyl isothiocyanate and unbaited traps were tested. The experiment was run at Halásztelek, from 5th July to 25th September, 2019, with 5 blocks.
In Experiment 4, traps baited with allyl isothiocyanate, allyl isothiocyanate + phenylacetaldehyde and unbaited traps were tested. The experiment was run at Halásztelek, from 30th June to 5th October, 2021, with five blocks.
Trapped insects were collected on a weekly basis and were brought to the laboratory, where the individuals were sexed and determined to species based on the works of Benedek17 and Derjanschi and Péricart18 with a stereomicroscope.
Statistics
Data were tested for normality by Shapiro–Wilk test. Since none of the experimental data were normally distributed, nonparametric tests were used. In field experiments catch data of individual traps were summed for trap rotation periods. Periods with low catches, accounting for less than 5% of total catches of the respective experiment were excluded from the analysis. Data from electroantennographic screenings and from field experiments were analyzed by Kruskal–Wallis test, and differences between treatments were evaluated by pairwise Wilcoxon test with Benjamini–Hochberg correction. For the dose–response test, Spearman’s rank correlation was calculated. Statistical procedures were conducted using the software R21.
Results
Electroantennographic screenings
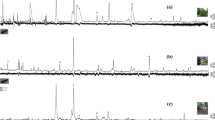
Several plant volatiles elicited electroantennographic responses, different from solvent control (Fig. 1). Three compounds elicited significantly higher responses than the standard, these were the following: allyl isothiocyanate, phenylacetaldehyde and ± linalool. These compounds were tested in consecutive field experiments.
Electroantennographic responses of antennae of Eurydema ornata adults (N = 13) to plant volatiles relative to a standard (myrcene). Treatments marked with the same letter are not significantly different at p = 5% by Kruskal–Wallis test, pairwise comparison by Wilcoxon test with Benjamini–Hochberg correction.
Field experiments
In the course of the field experiments three Eurydema species were caught: E. oleracea, E. ornata and E. ventralis.
Field catches of Eurydema ornata
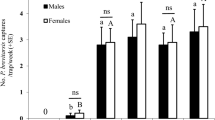
In Experiment 1, only combinations containing allyl isothiocyanate caught more E. ornata than unbaited traps. The treatments containing allyl isothiocyanate did not differ significantly (Fig. 2A). Both males and females were caught (ratio of females 51.04%), with no apparent sex ratio differences between treatments.
Field catches of Eurydema ornata (A) and E. oleracea (B) adults with binary and ternary combinations of ± linalool, phenylacetaldehyde and allyl isothiocyanate and in unbaited traps (Exp. 1). Treatments marked with the same letter are not significantly different within one diagram at p = 5% by Kruskal–Wallis test, pairwise comparison by Wilcoxon test with Benjamini–Hochberg correction.
In Experiment 2, allyl isothiocyanate and the ternary combination of allyl isothiocyanate, phenylacetaldehyde and ± linalool attracted more E. ornata than unbaited traps. Unbaited traps and traps baited with either phenylacetaldehyde alone or ± linalool alone did not catch any individuals (Fig. 3A). Both males and females were caught (ratio of females 39.62%), with no apparent sex ratio differences between treatments.
Field catches of Eurydema ornata (A) and E. oleracea (B) adults with ± linalool, phenylacetaldehyde, allyl isothiocyanate, their ternary combination and in unbaited traps (Exp. 2). Treatments marked with the same letter are not significantly different within one diagram at p = 5% by Kruskal–Wallis test, pairwise comparison by Wilcoxon test with Benjamini–Hochberg correction.
In Experiment 3, traps baited with 10 mg of allyl isothiocyanate caught more E. ornata than unbaited traps, however, catches did not differ from those of traps baited with 1 mg dose of the compound. On the other hand, traps baited with 100 mg of allyl isothiocyanate caught more than all other treatments. Unbaited traps did not catch any individuals (Fig. 4A). Again both males and females were caught (ratio of females 52.63%) with no apparent sex ratio differences between treatments. Catches showed a significant positive correlation with increasing dose, both for males (Spearman's rank correlation rho = 0.393, p < 0.001) and females (Spearman's rank correlation rho = 0.494, p < 0.001).
Field catches of Eurydema ornata (A) and E. oleracea (B) adults in traps baited with different doses of allyl isothiocyanate and in unbaited traps (Exp. 3). Treatments marked with the same letter are not significantly different at p = 5% by Kruskal–Wallis test, pairwise comparison by Wilcoxon test with Benjamini–Hochberg correction.
In Experiment 4, both allyl isothiocyanate and allyl isothiocyanate + phenylacetaldehyde caught more E. ornata than unbaited traps. Catches of the baited treatments did not differ significantly. Males and females responded similarly (Table 2).
Field catches of Eurydema oleracea
In Experiment 1, catches of E. oleracea in different treatments did not differ significantly (Fig. 2B). Both males and females were caught (ratio of females 55.88%), with no apparent sex ratio differences between treatments.
In Experiment 2, traps baited with the ternary combination attracted more E. oleracea than unbaited traps and than traps baited with ± linalool, neither of which caught any individuals of this species. On the other hand, catches with the ternary combination did not differ from those baited with either allyl isothiocyanate or phenylacetaldehyde (Fig. 3B). Both males and females were caught (ratio of females 72.22%), with no apparent sex ratio differences between treatments.
In Experiment 3, only a few E. oleracea individuals were caught. Only traps baited with either 10 or 100 mg of allyl isothiocyanate caught E. oleracea, however, treatments did not differ significantly (Fig. 4B). Both males and females were caught (ratio of females 80%), with no apparent sex ratio differences between treatments. Due to the low catches correlations were not calculated.
In Experiment 4, again only few E. oleracea were caught, treatments did not differ significantly. Both males and females were caught (Table 2).
Field catches of Eurydema ventralis
In the experiments E. ventralis was only caught in Experiment 4, nevertheless, the species was represented in low numbers, treatments did not differ significantly. Both males and females were caught (Table 2).
Discussion
In the current study E. ornata was found to be attracted to baits containing allyl isothiocyanate. To our knowledge this is the first report on field attraction of an Eurydema species to a synthetic semiochemical. As for other pentatomids, Thrift et al.22 found higher abundance of Murgantia histrionica (Hahn) on plants baited with either allyl isothiocyanate or benzyl isothiocyanate as compared to control plants with benzyl isothiocyanate showing more pronounced effect, and although the authors reported weak attraction to these plant volatiles, the compounds further increased the attractive effect of the pheromone.
Reports on field attraction of pentatomids to plant volatiles are scarce3. For instance, for Halyomorpha halys (Stal) plant volatiles were found to be only slightly attractive or even inhibitory23. Furthermore, it is rather commonly reported in pentatomids that although the pheromone blends show activity, several species were found to rarely enter traps4. This is possibly due to the importance of vibratory signals in their intraspecific communication24. For instance, Thrift et al.22 applied baited plants in their studies. In the current study bugs were attracted to the baits and were caught in the traps, which is especially promising in respect of development of tools for potential practical applications in the future. Optimisation of bait composition and trap design may result in further enhanced effectiveness of the traps.
According to the literature E. ornata primarily feeds on generative parts of plants11,16. However, in our research, common floral volatiles, phenylacetaldehyde and ± linalool, present also in cruciferous plants25, despite their electroantennographic activity, did not elicit behavioral response in field experiments, when presented on their own. Furthermore, these compounds did not influence activity of allyl isothiocyanate considerably.
Breakdown products of glucosinolates are characteristic for cruciferous plants26. Thiocyanates and isothiocyanates were found to attract pests of brassicaceous crops, for instance, allyl isothiocyanate is a known attractant for Phyllotreta spp. (e.g.19,27).
In the current research three Eurydema species were caught, however, E. oleracea and E. ventralis were usually represented by few individuals in trap catches, mostly in insufficient numbers for statistical evaluation. However, research targeting these pests may bring fruitful results, as it was found in previous studies that even closely related species with overlapping host plant ranges may respond differently to plant volatiles (e.g.19,28). Therefore, other compounds characteristic for cruciferous plants may also be of interest, for instance for Phyllotreta spp. it was found that for some species, other isothyocyanates were more attractive than allyl isothyocyanate (e.g.27).
Furthermore, research on combinations of semiochemicals may also bring novel results as combinations of multiple stimuli may elicit more pronounced behavioural response than individual compounds29.
Conclusions
Pentatomidae is a highly species rich group of true bugs, with considerable diversity in their chemical ecology3,4. As several species are important pests of various crops, tools for monitoring or possibly for direct control are of high potential value for agricultural practice.4
As research on chemical ecology of Pentatomidae primarily focuses on identification of pheromones, studies on attraction of pentatomids to plant volatiles are scarce3. To our knowledge, this is the first report of attraction of an Eurydema species to a semiochemical and one of the few examples of attraction of a pentatomid bug to a synthetic plant volatile in the field. The results may provide a first step in the development of practically applicable lures for trapping E. ornata, which may open perspectives for developments aiming applications in agricultural practice in the future. Furthermore, the results suggest that plant volatiles may provide prospects for practical applications for other pentatomid pests as well.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Panizzi, A. R. Wild hosts of pentatomids: Ecological significance and role in their pest status on crops. Annu. Rev. Entomol. 42, 99–122 (1997).
Witzgall, P., Kirsch, P. & Cork, A. Sex pheromones and their impact on pest management. J. Chem. Ecol. 36, 80–100 (2010).
Weber, D. C., Khrimian, A., Blassioli-Moraes, M. C. & Millar, J. G. Semiochemistry of Pentatomoidea. in Invasive Stink Bugs and Related Species (Pentatomoidea) (ed. McPherson, J.E.). 677–726 (CRC Press, 2018)
Millar, J. G. (2005) Pheromones of true bugs. in The Chemistry of Pheromones and Other Semiochemicals II. Topics in Current Chemistry. (ed. Schulz, S.). Vol 240. (Springer, 2005)
Vuts, J. et al. Performance and selectivity of floral attractant-baited traps targeted for cetoniin scarabs (Coleoptera: Scarabaeidae) in Central and Southern Europe. Crop. Prot. 29, 1177–1183 (2010).
Atalay, R. & Çağlayan, L. Investigations on the population-injury relationships of Eurydema ornatum L. (Heteroptera, Pentatomidae) which harmful on cabbage and cauliflower seedlings. Türk. Entomol. Derg. 14, 109–114 (1990).
Trdan, S., Žnidarčič, D. & Valič, N. Field efficacy of three insecticides against cabbage stink bugs (Heteroptera: Pentatomidae) on two cultivars of white cabbage. Int. J. Pest. Manag. 52, 79–87 (2006).
Infantino, A., Tomassoli, L., Peri, E. & Colazza, S. Viruses, fungi and insect pests affecting caper. Eur. J. Plant Sci. Biotechnol. 1, 170–179 (2007).
Bohinc, T. & Trdan, S. Trap crops for reducing damage caused by cabbage stink bugs (Eurydema spp.) and flea beetles (Phyllotreta spp.) on white cabbage: Fact or fantasy?. J. Food Agric. Environ. 10, 1365–1370 (2012).
Simoglou, K. B. & Dioli, P. First report of caper (Capparis spinosa) serious infestation by Eurydema eckerleini in Cyclades Islands, Greece (Hemiptera: Heteroptera, Pentatomidae). Fragm. Entomol. 49, 65–69 (2017).
Stankevych, S. et al. Eurydema bugs: Review of distribution, ecology, harmfulness, and control. Ukr. J. Ecol. 11, 131–149 (2021).
Aldrich, J. R. et al. Semiochemistry of cabbage bugs (Heteroptera: Pentatomidae: Eurydema and Murgantia). J. Entomol. Sci. 31, 172–182 (1996).
Eltez, S. & Karsavuran, Y. Food preference in the cabbage bug Eurydema ornatum (L.) (Heteroptera: Pentatomidae). Pak. J. Zool. 42, 407–412 (2010).
Rather, A. H., Azim, M. N. & Maqsood, S. Host plant selection in a pentatomid bug Eurydema pulchrum Westwood. J. Plant. Prot. Res. 50, 229–232 (2010).
Badenes-Pérez, F. R., Márquez, B. P. & Petitpierre, E. Simultaneous use of winter cress (Brassicaceae) in trap cropping, conservation biological control, and pollinator conservation. IOBC/WPRS Bull. 122, 11–15 (2017).
Benedek, P. Poloskák—Heteroptera. in The Handbook of Plant Protection Zoology 1. (ed. Jermy, T. & Balázs, K.). 306–431 (Akadémiai Kiadó, 1988) (in Hungarian).
Benedek, P. About Eurydemae in Hungary I. Problems of taxonomy. Növényvédelem 1, 17–26 (1965) ((in Hungarian with English abstract)).
Derjanschi, V. &, Pericart, J. Hémiptères Pentatomoidea Euro-Méditerranéens. Volume I. Faune de France. Vol. 90. 2nd Ed. 1–500. (Fédération Française des Sociétés de Sciences Naturelles, 2016) (in French).
Benedek, P., Bakcsa, F., Tóth, M. & Csonka, É. Allyl isothiocyanate baited traps to monitor cabbage flea beetles (Phyllotreta spp. Coleoptera: Chrysomelidae). Int. J. Hortic. Sci. 11, 95–99 (2005).
Koczor, S. et al. Sex pheromone of the alfalfa plant bug, Adelphocoris lineolatus: Pheromone composition and antagonistic effect of 1-hexanol (Hemiptera: Miridae). J. Chem. Ecol. 47, 525–533 (2021).
R Core Team. R: A Language and Environment for Statistical Computing. https://www.R-project.org/ (R Foundation for Statistical Computing, 2016).
Thrift, E. M., Herlihy, M. V., Wallingford, A. K. & Weber, D. C. Fooling the harlequin bug (Hemiptera: Pentatomidae) using synthetic volatiles to alter host plant choice. Environ. Entomol. 47, 432–439 (2018).
Weber, D. C. et al. Chemical ecology of Halyomorpha halys: Discoveries and applications. J. Pest. Sci. 90, 989–1008 (2017).
Gogala, M. Vibratory signals produced by Heteroptera—Pentatomorpha and Cimicomorpha. in Insect Sounds and Communication: Physiology, Behaviour, Ecology, and Evolution. (ed. Drosopoulos, S. & Claridge, M.F.). 275–295 (CRC Press/Taylor & Francis Group, 2006)
Knudsen, J. T., Eriksson, R., Gershenzon, J. & Stahl, B. Diversity and distribution of floral scent. Bot. Rev. 72, 1–120 (2006).
Cole, R. A. Isothiocyanates, nitriles and thiocyanates as products of autolysis of glucosinolates in Cruciferae. Phytochemistry 15, 759–762 (1976).
Csonka, É., Tóth, M. & Ujváry, I. Differences in host–plant related chemical communication of the flea beetles Phyllotreta cruciferae Goeze and Ph. vittula Redtenbacher (Coleoptera, Chrysomelidae). Acta Phytopathol. Entomol. Hung. 42, 343–352 (2007).
Liblikas, I. et al. Flea beetle (Coleoptera: Chrysomelidae) response to alkyl thiocyanates and alkyl isothiocyanates. Agron. Res. 1, 175–184 (2003).
Bruce, T. J. A. & Pickett, J. A. Perception of plant volatile blends by herbivorous insects—Finding the right mix. Phytochemistry 72, 1605–1611 (2011).
Acknowledgements
The current research was partially financed by the National Research Development and Innovation Office (NKFIH, grant FK134744). We would like to express our gratitude to Anita Koczor-Bagarus, Viktória Jurkó, Gábor Balogh and Eszter Matula (Department of Chemical Ecology, Plant Protection Institute, Centre for Agricultural Research) for their help in preparation of baits for field experiments and to Júlia Katalin Szilasné Dr. Jósvai (Department of Chemical Ecology, Plant Protection Institute, Centre for Agricultural Research) for her kind help in field experiments.
Funding
Open access funding provided by ELKH Centre for Agricultural Research. The current research was partially financed by the National Research Development and Innovation Office (NKFIH, grant FK134744).
Author information
Authors and Affiliations
Contributions
S.K. and M.T. conceived and designed the experiment. S.K. performed electroantennographic screenings and field experiments. S.K. determined the trapped material and performed data analysis. Both authors contributed to, read, reviewed and approved the manusript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Koczor, S., Tóth, M. Field attraction of Eurydema ornata (Hemiptera: Pentatomidae) to allyl isothiocyanate. Sci Rep 13, 11063 (2023). https://doi.org/10.1038/s41598-023-37705-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-37705-w
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.