Abstract
The brain is a complex system whose understanding enables potentially deeper approaches to mental phenomena. Dynamics of wide classes of complex systems have been satisfactorily described within q-statistics, a current generalization of Boltzmann-Gibbs (BG) statistics. Here, we study human electroencephalograms of typical human adults (EEG), very specifically their inter-occurrence times across an arbitrarily chosen threshold of the signal (observed, for instance, at the midparietal location in scalp). The distributions of these inter-occurrence times differ from those usually emerging within BG statistical mechanics. They are instead well approached within the q-statistical theory, based on non-additive entropies characterized by the index q. The present method points towards a suitable tool for quantitatively accessing brain complexity, thus potentially opening useful studies of the properties of both typical and altered brain physiology.
Similar content being viewed by others
Introduction
The brain is widely recognized as a complex system since it is composed by billions of cells (neurons) which express individual behaviors and, at same time, they build a fully interconnected network with emergent, self-organized collective behaviors1. Thus, traditional reductionist scientific methodology from mechanistic rationality appears to fail for deeply understanding the brain and its associated mind inside a multidimensional environment2. On one hand, a humanity’s great unresolved problem is to establish a suitable mental medicine, from epistemology3 to the biomedical perspective. The problem begins in differentiating normality from typicality, illness from neurodiversity. And, upon this basis, to establish a taxonomy about mental typology for a more realistic nosography. On the other hand, several studies have explored brain complexity through entropic measures within the electroencephalogram (EEG), and found relationships between brain complexity and different mind conditions4. However, this issue yet is incipient. One way of accessing brain complexity is through the electroencephalogram (EEG) signal5, which is the electrical result of millions of neurons under each of the leads (electrodes) over time. The EEG is the simplest, least invasive and universally used form of functional recording of the human brain dynamics.
The pioneering works of Boltzmann6 and Gibbs7 (BG) established a magnificent theory which is structurally associated with the BG entropic functional
and consistent expressions for continuous or quantum variables; k is a conventional positive constant (in physics, k is chosen to be the Boltzmann constant \(k_B\); in information theory and computational sciences, \(k=1\) is frequently adopted).
In the simple case of equal probabilities, this functional becomes \(S_{BG}=k \ln W\). Equation (1) is generically additive8. Indeed, if A and B are two probabilistically independent systems (i.e., \(p_{ij}^{A+B}= p_i^A p_j^B\)), we straightforwardly verify that \(S_{BG}(A+B)=S_{BG}(A)+S_{BG}(B)\). The celebrated entropic functional (1) is consistent with thermodynamics for all systems whose N elements are either independent or weakly interacting in the sense that only basically local (in space/time) correlations are involved. For example, if we have equal probabilities and the system is such that the number of accessible microscopic configurations is given by \(W(N) \propto \mu ^N\; (\mu >1; \,N\rightarrow \infty )\), then \(S_{BG}(N)\) is extensive (i.e., proportional to the number of elements) as required by thermodynamics. Indeed \(S_{BG}(N)=k\ln W(N) \sim k(\ln \mu )N\).
However, complex systems are typically composed of many elements which essentially are non-locally correlated, building an intricate network of interdependencies from where collective states can emerge9. BG statistical mechanics appears to be generically inadequate for such systems because this theory assumes (quasi) independent components with short-range (stochastic or deterministic) interactions.
Indeed, if the correlations are nonlocal in space/time, \(S_{BG}\) may become thermodynamically inadmissible. Such is the case of equal probabilities with say \(W(N) \propto N^\nu \;(\nu >0; \,N\rightarrow \infty )\): it immediately follows \(S_{BG}(N) \propto \ln N\), which violates thermodynamical extensivity9. To satisfactorily approach cases such as this one, it was proposed in 198810 to build a more general statistical mechanics based on the nonadditive entropic functional
with the q-logarithmic function \(\ln _q z \equiv \frac{z^{1-q}-1}{1-q} \; (\ln _1 z=\ln z)\), its inverse being the q-exponential \(e_q^z \equiv [1+(1-q)z]_{+}^{1/(1-q)}\); \((e_1^z=e^z\); \([z]_{+}=z\) if \(z>0\) and vanishes otherwise); for \(q<0\), it is necessary to exclude from the sum the terms with vanishing \(p_i\). We easily verify that equal probabilities yield \(S_q=k\ln _q W\). Also, we generically have the following functional nonadditivity
Consequently, in the \((1-q)/k \rightarrow 0\) limit, we recover the \(S_{BG}\) additivity. For the anomalous class of systems mentioned above, namely if \(W(N) \propto N^\nu\), we obtain, \(\forall \nu\), the extensive entropy \(S_{1-1/\nu }(N)=k\ln _{1-1/\nu }W(N) \propto N\), as required by the Legendre structure of thermodynamics11,12. Finally, the optimization of \(S_q\) under simple constraints yields q-exponential distributions for the (quasi)stationary states, instead of the usual BG exponentials.
Sequence of inter-event time intervals from EEG signal, as detected in Fig. 1.
Since EEG is a massive electrical phenomenon, its amplitude is correlated with the cell synchronization. The regularity of time intervals between amplitude peaks that overcomes a typical threshold (in this case, one standard deviation), would reflect the system’s complexity. If synchronization would be a stochastic and uncorrelated phenomenon, the distribution of inter-peak distances could possibly be estimated within the BG frame. But EEG is a highly non-equilibrium phenomenon, and it requires more general approaches. Independently of the nature of regularities, this phenomenon exhibits the complex nature of the system. It cannot be excluded that, in the realm of q-statistics where q is a scalar measure of complexity, a possibly satisfactory description could be attained.
Motivation, methodology and results
The above nonadditive entropies, as well as the nonextensive statistical mechanics grounded on them, have been already used to characterize various aspects of complexity. Various data obtained from EEG, magnetoencephalograms (MEG), electrocardiograms (ECG), and others, have been analyzed in connection to q-statistics13,14,15,16. However, the discussion frequently focuses on qualitative ingredients. Our aim here is to demonstrate that nonextensive statistical mechanics is applicable to the brain as a complex system, thus providing specific values for the relevant parameters. Thus, we are analyzing human EEG’s in a specific manner herein described which eventually provides a small number of real numbers (such as q) having the potential of satisfactorily characterizing different regions of the brain, different functional neuro-states, nosologically different classes of human phenomenologies.
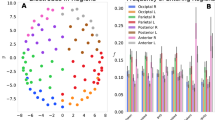
Probability distributions of EEG inter-occurrence times (500 equal logarithmic bins) and fittings with statistical models. Superimposed signal recorded on the \(P_z\) location of ten subjects performing a work memory task. Amplitude threshold = 1.0 standard deviation. Fitting within Boltzmann-Gibbs statistical mechanics for non-complex systems (i.e., \(q=1\), dashed red curve). Fitting within nonextensive statistical mechanics for complex systems (i.e., \(q\ne 1\), black continuous curve). See Methodology for details.
We analized the EEG signal of ten typical adult humans from a match-to-sample task experiment with neutral affective interference for access working memory and attention, such in Yang and Zhen’s study17. This work was approved by our ethical board for human research, under CAAE 50137721.4.0000.5269. Each EEG signal has 5–10 min length recorded with open eyes at 1000Hz sampling rate, through 20 channels disposed at 10–20 montage with eyes open. The high, low and band-pass filters were respectively 0.5, 150 Hz and 60 Hz. We did not apply any other filter to minimize signal manipulation.
We accessed signal recorded at the midparietal (\(P_z\)) site (see Fig. 3), where classical cognitive event-related potentials, as P30018, manifest during attention tasks. A threshold was set at − 1.0 standard deviation from \(P_z\) signal average (Fig. 1, from subject B006). Taking negative voltages we are minimizing the effect of blink artifacts, which are positive waves, amplier in frontal places.
Each event is the numerical position i of signal vector (1 s = 1000 positions) where the amplitude crossed the threshold downwards. The inter-event distances \(i_n - i_{n-1}\) (where n = 1,...,N) were calculated (Fig. 2, from B006). The logarithm distribution of inter-event distances (with 500 distance classes) of all ten EEG signals at \(P_z\) were superimposed, and the fitting was performed to the following q-statistical function (Fig. 3):
where \((a_{q},\beta _{q},c_{q},\eta _{q},q)=(2.1 \times 10^{-5},2.0 \times 10^{-5},2.12,2.96,1.89)\) for the best fitting. And, for comparison, we also included the classical statistical BG function (where \(q=1\)), as follows:
where \((a_{BG},\beta _{BG},c_{BG},\eta _{BG},q_{BG})=(4.3 \times 10^{-4},0.023,0.94,0.93,1)\) for the best fitting.
The fitting was performed using three different methods: dog leg trust region19, trust region reflective20 and crow search21 algorithms, all available in Scipy library.
The constant a is determined by imposing normalization, i.e., \(\int _0^\infty dx\,y(x)=1\). Consequently,
for \(q>1\) and \(\frac{1}{q-1}-\frac{1+c_q}{\eta _q}>0\). In the \(q\rightarrow 1\) limit, we obtain
It is observed that EEGs at \(P_z\) position from all subjects express very similar distributions of distances. The EEG regularity was modelled by the q-statistics function instead BG one (Fig. 3).
Discussion
Consistently with the use of \(S_q\) entropy in numerous articles as a measure of complexity in neural systems, we believe that we bring here the demonstration of the applicability of non-extensive statistical mechanics on the collective behavior of a neural system through the regularities of EEG. This preliminary study exhibits as a proof of concept that q-statistics easily can quantitatively reveal some aspects of brain complexity through the q parameter. Future research needs to be carried out to determine whether this measure will be sensitive enough to discriminate the complexity of different regions or different states of the brain, as well as aspects of inter-individual diversity (among them, brain diseases or even mental disorders). Consistently, we have verified here that brain phenomenology is not properly described within BG statistics (i.e., q = 1). This is by no means surprising since BG statistics generically disregards inter-component long-range correlations and their collective behavior, which is well known in neural systems1. In contrast, q-statistics has been empirically shown to be a useful generalization of BGSM12,22,23,24,25,26. In addition to other quite informative complexity measures and related methodologies applied to neurosciences27,28,29,30,31, q-statistics hopefully also is useful in the present case. Here, it was applied through a quite simple methodology, using a functional model involving stretched q-exponentials which satisfactorily fit the empirical distributions of scalar inter-event intervals (see Fig. 3). Many of these complex systems present \(c_q \ne 0\), from basic chemical reactions through quantum tunneling32 to financial market behavior33, COVID-19 spreading34, commercial air traffic networks35 We are led to believe that we are dealing with universality classes of complexity, thus revealing, in what concerns information processing and energy dynamics, far more integrative networks than one might a priori expect from neural structures36.
By generalizing the BG theory, q-statistics shows that it could be a suitable and promising path to explore brain complexity. Our expectancy is that the q parameter can be sensitive to different brain/mental states, to brain/mind development, and to neural diversity, perhaps clarifying the boundaries between the normal and the ill brain, including extreme cases such as Alzheimer, Pick, and Parkinson diseases. Consistently, a key outcome of emergence of self-organized new states in complex systems is an adaptive behavior facing environmental constraints1. Indeed, the concept of disease has also been related to reduced adaptive capabilities, and to the alteration of complexity4,37,38. Along the lines of the seminal philosophical work of G. Canguilhem3, normality should be related to the ability to create new rules (i.e., adaptation) instead of living by the same old norms. We intend to further explore, in the future, the neural diversity through the most remarkable paradigm of complexity.
Data availability
The raw EEG wavesfrom all subjects are provided at data.mendeley.com under https://doi.org/10.17632/dm3922zmpj.1.
References
Bassett, D. S. & Gazzaniga, M. S. Understanding complexity in the human brain. Trends Cogn. Sci. 15(5), 200–9 (2011).
Braslow, J. T., Brekke, J. S. & Levenson, J. Psychiatry’s myopia-reclaiming the social, cultural, and psychological in the psychiatric gaze. JAMA Psychiatry 78(4), 349–350 (2021).
Canguilhem, G. The Normal and the Pathological trans. Carolyn R. Fawcett and Robert S. Cohen (Zone Books, 1991).
Lau, Z. J., Pham, T., Chen, S. H. A. & Makowski, D. Brain entropy, fractal dimensions and predictability: A review of complexity measures for EEG in healthy and neuropsychiatric populations. Eur. J. Neurosci. 56(7), 5047–5069 (2022).
Speckmann, E.-J., Elger, C. E. & Gorji, A. Neurophysiologic basis of EEG and DC potentials. In Niedermeyer’s Electroencephalography: Basic Principles, Clinical Applications, and Related Fields (eds Schomer, D. L. & da Silva, F. H. L.) 17–31 (Lippincott Williams & Wilkins, 2011).
Boltzmann, L. Weitere Studien u̇ber das Wrmegleichgewicht unter Gas moleku̇len [Further Studies on Thermal Equilibrium Between Gas Molecules], Wien. Ber. 66, 275 (1872).
Gibbs, J. W. Elementary Principles in Statistical Mechanics—Developed with Especial Reference to the Rational Foundation of Thermodynamics (C. Scribner’s Sons, 1902; Yale University Press, 1948; OX Bow Press, 1981).
Penrose, O. Foundations of Statistical Mechanics: A Deductive Treatment 167 (Pergamon, Oxford, 1970).
Gell-Mann, M. & Tsallis, C. (eds) Nonextensive Entropy—Interdisciplinary Applications (Oxford University Press, 2004).
Tsallis, C. Possible generalization of Boltzmann-Gibbs statistics. J. Stat. Phys. 52, 479–487 (1988).
Tsallis, C. Entropy. Encyclopedia 2, 264–300 (2022).
Tsallis, C. Introduction to Nonextensive Statistical Mechanics–Approaching a Complex World, (Springer, 2009); Second Edition, (Springer 2023).
Montangie, L. & Montani, F. Quantifying higher-order correlations in a neuronal pool. Phys. A Stat. Mech. Appl. 421, 388–400 (2015).
Montangie, L. & Montani, F. Higher-order correlations in common input shapes the output spiking activity of a neural population. Phys. A Stat. Mech. Appl. 471, 845–861 (2017).
Montangie, L. & Montani, F. Common inputs in subthreshold membrane potential: The role of quiescent states in neuronal activity. Phys. Rev. E 97(6), 060302(R) (2018).
Guisante, N., Pallares di Nunzio, M., Martinez, N., Rosso, O. A. & Montani, F. Chaotic dynamics of the henon map and neuronal inputoutput: A comparison with neurological data. Chaos 33, 043111 (2023).
Yang, H., Li, J. & Zheng, X. Different influences of negative and neutral emotional interference on working memory in trait anxiety. Front. Psychol. 12, 570552 (2021).
Polich, J. & Criado, J. R. Neuropsychology and neuropharmacology of P3a and P3b. Int. J. Psychophysiol. 60(2), 172–85 (2006).
Powell, M. J. D. A New Algorithm for Unconstrained Optimization, Nonlinear Programming 31–66 (Academic Press, 1970).
Coleman, T. F. & Li, Y. An interior trust region approach for nonlinear minimization subject to bounds. SIAM J. Optim. 6, 418–445 (1996).
Askarzadeh, A. A novel metaheuristic method for solving constrained engineering optimization problems: Crow search algorithm. Comput. Struct. 169, 1–12 (2016).
Ludescher, J., Tsallis, C. & Bunde, A. Universal behaviour of interoccurrence times between losses in financial markets: An analytical description. Europhys. Lett. 95, 68002 (2011).
Lutz, E. & Renzoni, F. Beyond Boltzmann-Gibbs statistical mechanics in optical lattices. Nat. Phys. 9, 615–619 (2013).
Bogachev, M. I., Kayumov, A. R. & Bunde, A. Universal internucleotide statistics in full genomes: A footprint of the DNA structure and packaging?. PLoS ONE 9(12), e112534 (2014).
Combe, G., Richefeu, V., Stasiak, M. & Atman, A. P. F. Experimental validation of nonextensive scaling law in confined granular media. Phys. Rev. Lett. 115, 238301 (2015).
Greco, A. et al. Acoustic emissions in compression of building materials: q-statistics enables the anticipation of the breakdown point. Eur. Phys. J. Special Top. 229(5), 841–849 (2020).
Tononi, G., Sporns, O. & Edelman, G. M. A measure for brain complexity: Relating functional segregation and integration in the nervous system. PNAS 91(11), 5033–5037 (1994).
Rosas, F., Mediano, P. A. M., Gastpar, M. & Jensen, H. J. Quantifying high-order interdependencies via multivariate extensions of the mutual information. Phys. Rev. E 100(3), 032305 (2019).
Varley, T. F., Pope, M., Faskowitz, J. & Sporns, O. Multivariate information theory uncovers synergistic subsystems of the human cerebral cortex. Commun. Biol. 6(1), 451 (2023).
Meisel, C., Olbrich, E., Shriki, O. & Achermann, P. Fading signatures of critical brain dynamics during sustained wakefulness in humans. J. Neurosci. 33(44), 17363–17372 (2013).
Shriki, O. et al. Neuronal avalanches in the resting MEG of the human brain. J. Neurosci. 33(16), 7079–7090 (2013).
Wild, R., Nötzold, M., Simpson, M., Tran, T. D. & Wester, R. Tunnelling measured in a very slow ion-molecule reaction. Nature https://doi.org/10.1038/s41586-023-05727-z (2023).
Tsallis, C., Anteneodo, C., Borland, L. & Osorio, R. Nonextensive statistical mechanics and economics. Phys. A Stat. Mech. Appl. 324(1–2), 89–100 (2003).
Tsallis, C. & Tirnakli, U. Predicting COVID-19 peaks around the world. Front. Phys. 8, 217 (2020).
Mitsokapas, E., Schäfer, B., Harris, R. J. & Beck, C. Statistical characterization of airplane delays. Sci. Rep. 11, 7855 (2021).
Sauseng, P., Griesmayr, B., Freunberger, R. & Klimesch, W. Control mechanisms in working memory: A possible function of EEG theta oscillations. Neurosci. Biobehav. Rev. 34, 1015–1022 (2010).
Goldberger, A. L. Fractal variability versus pathologic periodicity: Complexity loss and stereotypy in disease. Perspect. Biol. Med. 40(4), 543–561 (1997).
Yang, A. C. & Tsai, S. J. Is mental illness complex? From behavior to brain. Prog. Neuropsychopharmacol. Biol. Psychiatry. 45, 4253–4257. https://doi.org/10.1016/j.pnpbp.2012.09.015 (2013).
Acknowledgements
We acknowledge fruitful conversations with E.M.F. Curado, A.R. Plastino and R. Wedemann, as well as partial financial support by CNPq and Faperj.
Author information
Authors and Affiliations
Contributions
D.M.A. research design, data analysis, figure design, text writing, and revision. C.T. research design, mathematical formulation, text writing, and revision. H.S.L. mathematical formulation, data analysis, figure design, and text revision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abramov, D.M., Tsallis, C. & Lima, H.S. Neural complexity through a nonextensive statistical–mechanical approach of human electroencephalograms. Sci Rep 13, 10318 (2023). https://doi.org/10.1038/s41598-023-37219-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-37219-5
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.