Abstract
This prospective study assessed the exposure to phthalates of preterm neonates who received total parenteral nutrition (TPN) during their stay in the neonatal intensive care unit (NICU) and the risk of neurodevelopment delays at the age of 2 months. Our study recruited 33 preterm neonates who required TPN upon NICU admission. Urine samples for analyzing phthalate metabolites were obtained at admission and then daily until the last day of receiving TPN. Phthalates in the daily TPN received by the preterm neonates were analyzed. The neurodevelopment of the neonates was assessed using the Ages and Stages Questionnaire Edition 3 (ASQ-3). Diethyl phthalate and butyl benzyl phthalate were found in all TPN samples, while 27% and 83% contained dibutyl phthalate and di-(2-ethylhexyl) phthalate (DEHP), respectively. Yet, the daily dose of each phthalate that our preterm neonates received from TPN was much lower than the recommended tolerable limit. Urinary levels of monobenzyl phthalate and four metabolites of DEHP [i.e., mono-(2-ethylhexyl) phthalate (MEHP), mono-(2-ethyl-5-hydroxyhexyl) phthalate, mono-(2-ethyl-5-oxohexyl) phthalate (MEOHP), and mono-(2-ethyl-5-carboxypentyl) phthalate (MECPP)] and the sum of four DEHP metabolites (∑4DEHP) increased significantly in preterm neonates before discharge. However, these levels were not correlated with their phthalate parent compounds in TPN, suggesting other sources of exposure in the NICU. At 2 months, we found that urinary levels of mono-iso-butyl phthalate (MiBP), MECPP, MEHP, and ∑4DEHP were inversely related to fine motor skills. After adjusting for head circumference, the inverse relationships remained significant, suggesting direct effects from phthalates. Given the extreme vulnerability of our population, it is critical to minimize exposure to phthalates during their NICU stay.
Similar content being viewed by others
Introduction
Premature neonates are those born before 37 weeks of pregnancy due to low birth weight < 5.5 pounds (2500 g) or a medical condition requiring specialized medical care. According to the World Health Organization, preterm birth complications are the leading cause of death among children under 5 years and were responsible for approximately 1 million deaths in 20151. Additionally, premature neonates are prone to health complications later in life, such as respiratory diseases2, cardiovascular problems3, renal dysfunction4, and neurodevelopment delays5, and have a potentially higher risk of specific cancers6.
Total parenteral nutrition (TPN) has been an integral part of the clinical management of low birth weight premature neonates, as feeding via the enteral route is not attainable7,8. Studies have reported the risks to premature neonates from TPN solutions contaminated with chemicals and/or microbes9,10. Preterm neonates in a neonatal intensive care unit (NICU) setting are highly vulnerable to harmful chemicals, which can impact their growth and neurodevelopment in the long term11, and the authors suggest improving the NICU environment.
Phthalates can be another significant source of contamination of TPN solutions because they are used to soften plastics and thus are widely utilized in medical devices, primarily in bags and infusion tubing for intravenous solutions and feeding12,13,14,15. As phthalates are not chemically bound to the plastic matrix, they leach out and migrate into the air, food, or other materials13. Among phthalates, di (2-ehylhexyl) phthalate (DEHP) is the most widely used plasticizer in medical devices16, and may contribute to chronic complications, particularly in preterm infants17. Although alternative DEHP-free devices have been introduced, studies have found that a wide range of plasticizers, in addition to DEHP, are still detected in medical devices used in pediatric units18, including the NICU19. Following exposure, phthalates are rapidly metabolized to their corresponding hydrolytic monesters, which can be further transformed to more hydrophobic oxidative products, conjugated to glucuronic acid via enzymatic hydrolysis, and eliminated in the urine20,21. The glucuronidation process facilitates the urinary excretion of phthalates, reducing their potential toxicity21. Newborns and preterm infants have immature glucuronidation pathways, leading to slower urinary excretion of phthalates compared with older children and adults22. High phthalate levels in urine were reported in the first weeks of intensive care, following invasive procedures, and in preterm infants with a birth weight < 1000 g23. Infants with low birth weight and those diagnosed with septicemia or bronchopulmonary dysplasia had higher urinary phthalate levels because of prolonged exposure to medical equipment containing phthalates24. A limited number of studies have explored the relationship between exposure to some phthalates and neurobehavioral outcomes25,26,27,28. However, the results are still controversial. The potential toxicity of phthalates to infants in the NICU is a concern because of their small body size and health status29.
There is limited evidence on the exposure of preterm neonates in the NICU to contaminants through TPN solutions and their potential impact on neurologic development. The present study aimed to examine: (a) the contribution of TPN solutions contaminated with phthalate to the overall phthalate exposure of preterm neonates during their NICU stay and (b) the risk of neurodevelopmental delays at 2 months.
Patients and methods
Study design
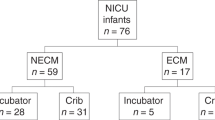
A prospective cohort study that included 33 premature neonates admitted to the NICU at King Faisal Specialist Hospital & Research Centre (KFSH&RC) was conducted between January 2020 and March 2021. The inclusion criteria were preterm neonates born at < 35 weeks of gestation or of birth weight < 1500 g and prescribed TPN within the first hour of their life for a minimum of 2 consecutive days. Preterm neonates who received a vaccination or were prenatally diagnosed with genetic or metabolic disorders or gastrointestinal genetic anomalies were excluded. This study was conducted after obtaining the approval of the Research Ethics Committee of the KFSH&RC (RAC#2191092). The study details were explained to the mothers, and their written consent was obtained on behalf of the children. Participants were encouraged to ask questions about the study to ensure their understanding. The demographics (gender, gestational age), baseline measurements (body length and weight), and general health information of the studied infants at delivery and during their stay at NICU were gathered from the patient chart. The TPN solution administered during the study period was Olimel N9E®; SmofKabiven® (Olimel; Baxter, Lessines, Belgium), the components of which were adjusted and individualized according to the patient's clinical condition. It contains fixed concentrations of trace elements (copper, zinc, manganese, selenium, sodium fluoride, and potassium chloride) at 0.1 mL/kg body weight. Figure 1 shows the data for the study population. Study quality was assessed using the STROBE template for observational studies30 (Table S1).
All experiments and methods were performed in accordance with relevant guidelines and regulations.
Throughout the manuscript, "admission" refers to samples collected from preterm neonates at admission before receiving TPN solutions. "Before discharge" refers to samples (daily for urine) collected from preterm neonates after receiving TPN solutions.
Biological materials
Urine samples (1–2 mL) were collected at admission and then daily until TPN was discontinued, by applying a sterile adhesive plastic bag to their perineum. Urine was transferred using 10 mL plastic syringes into 10 mL glass screw-capped tubes and stored at − 30 °C until the analysis of phthalates. Aliquots of 100 µL were collected into 0.5 mL microcentrifuge Eppendorf tubes and stored at − 30 °C until the analysis of creatinine.
TPN solutions
Leftover TPN solution was collected daily from each discarded bag received by a preterm neonate and aliquoted immediately into glass tubes. Both were stored at − 20 °C until analysis. For logistic reasons, phthalate compounds were measured in 195 samples instead of 230.
Analytical techniques
Phthalate assessment of urine (metabolites) and TPN (parent compounds)
The levels of eight phthalate metabolites (µg/L), monoethyl phthalate (MEP), mono-n-butyl phthalate (MnBP), mono-iso-butyl phthalate (MiBP), mono-benzyl phthalate (MBzP), mono-(2-ethylhexyl) phthalate (MEHP), mono-(2-ethyl-5-hydroxyhexyl) phthalate (MEHHP), mono-(2-ethyl-5-oxohexyl) phthalate (MEOHP), and mono-(2-ethyl-5-carboxypentyl) phthalate (MECPP) were measured in urine samples using ultra-performance liquid chromatography and tandem mass spectrometry (Waters, Milford, USA), based on a modified method from previously published studies31,32,33. These metabolites were associated with five parental phthalates. MEP is a metabolite of diethyl phthalate (DEP), MnBP and MiBP are metabolites of dibutyl phthalate (DBP), MBzP is a metabolite of butyl benzyl phthalate (BBzP), and MECPP, MEHP, MEOHP, and MEHHP are metabolites of DEHP. The method detection limits (MDLs) for MEP, MiBP, MnBP, MBzP, MECPP, MEHHP, MEOHP, and MEHP were 1.7, 1.51, 1.76, 0.99, 1.57, 1.62, 0.61, and 2.07 µg/L, respectively. The accuracy of the metabolite measurements (except MEP) was assessed using the German External Quality Assessment Scheme. We also used internal quality control by spiking urine with known concentrations of the metabolites. The recovery of each metabolite in urine spiked with three concentrations (15, 80, and 150 µg/L) was 94–103%. Each run included blanks, quality controls, matrix-spiked samples, and unknown samples. The analytical procedures and method validation are presented in the Supplemental Material (Tables S2–S5).
DEP, DBP, BBzP, and DEHP in TPN solutions were analyzed using solid-phase microextraction (SPME) followed by gas chromatography/mass spectrometry (GC–MS). The calibration curves showed good linearity within the given concentration ranges of 0.5–16 µg/L for DEP, DBP, BBzP, and DEHP, with r2 > 0. 997. The MDLs for DEP, DBP, BBzP, and DEHP in µg/L were 0.05, 0.047, 0.169, and 0.05, respectively. The recoveries for both ranges were good. The relative standard deviations (RSDs) for within- and between-run precision were < 10%. Therefore, the SPME–GC–MS method exhibited good precision and accuracy for quantifying the six selected phthalate compounds in the TPN samples. The validation parameters for the analytical methods are presented in Tables S6 and S7 (Supplemental Material).
Molar sum of DEHP metabolites (∑4DEHP) was calculated by summing the ratio of each metabolite to its molecular weight: [MEHP/278.34] + [MEHHP/294.34] + [MECPP/308.33] + [MEOHP/292.33]. ∑3DEHP was calculated similarly but without MEHP. The molar concentrations of two DBP metabolites was calculated as [MiBP/222.24 + MnBP/222.24] and expressed as ∑DBP.
Other measurements
The urine samples for neonates were analyzed for creatinine (creat) to adjust for the effect of urinary flow rate on the urinary excretion of tested analytes34, presenting the results as micrograms of phthalate monoester per gram of urinary creatinine (µg/g creat).
Neurodevelopment screening tools
After NICU discharge, 33 preterm neonates were scheduled for a 2-month follow-up visit at the Center for Autism Research at KFSH&RC during which the Ages and Stages Questionnaire 3 (ASQ-3) was conducted to identify whether the infant was at risk of delays in neurological development. Age was corrected for prematurity using the following link: Calculator: Add to or subtract from a date (timeanddate.com). The median corrected age at assessment was 4.2 months. The test is a comprehensive parent checklist, standardized for children between the ages of 1 and 66 months, that consists of 30 questions covering five domains of development: communication, gross motor, fine motor, problem-solving, and personal-social35. Each domain has a set of six items. The response of the parent to each item is yes (score = 10), sometimes (score = 5), or not yet (score = 0). The questionnaire takes 10–15 min for parents to complete. A total of 30 mothers agreed to answer the AQS-3 questionnaire.
Statistical analysis
The characteristics of the preterm neonates were described using means with standard deviations (SDs) or medians (minimum–maximum) for continuous variables, or numbers (%) for categorical variables. Geometric means were calculated to describe the distributions of tested analytes that were logarithmically transformed to approximate a normal distribution. Values below the MDL were replaced with ½ MDL to provide unbiased estimates for the mean and SD, particularly with a small proportion of readings below MDL36. However, this approach might be of concern when more than 50% of the data are below MDL37. This method has been adopted by many researchers38,39. As shown in the Results section, a small proportion of phthalate metabolites measurements in the current study were < MDL, suggesting that an unbiased estimate of the relationship between exposure and the neurodevelopment outcome was provided40. Spearman rank correlation analysis (rs) was performed to test for associations between pairs of continuous variables. Mann–Whitney test was used for categorical variables. Using mixed-effects models, we calculated the variability in the levels of the phthalate metabolites measured in 13 urine samples of preterm neonates by estimating interclass correlation coefficients (ICCs) and their 95% confidence intervals (95%CIs). Reproducibility based on the ICCs was categorized as excellent (> 0.75), moderate (0.4 to 0.75), or weak (< 0.4)41. Generalized estimating equations (GEE) with Bonferroni adjustments were conducted to account for the correlation among repeated measuresSeparate multiple linear regression models were built for each neurodevelopmental score as a continuous dependent variable, assuming that the score decreased proportionally with increasing exposure to phthalates after receiving TPN solutions. The average of repeated measures of urinary phthalate metabolites was calculated. Each model was adjusted for only birth weight and length of stay in the NICU, owing to the small sample size and for the levels of individual urinary phthalate metabolite measured at admission (before starting TPN). We applied regression imputation to replace the missing urinary phthalate metabolites for ten preterm neonates for whom we could not obtain urine at admission with the mean of each analyte. β regression coefficients and 95% confidence intervals (95% CIs) were calculated to measure the effect of one unit increase of urinary phthalate metabolites on ASQ-3 scores. For ease of interpretation of the results, β regression coefficients were back-transformed and expressed as a percentage change (Δ%) with 95% CIs using the equation: [exp (β) − 1] × 100. We also exponentiated β regression coefficients to calculate the geometric mean ratios (GMRs) and 95%CIs. The R (programming language) was used for the forest plot.
Analyses were considered statistically significant if p < 0.05. We also defined p < 0.10 as a marginally significant effect due to our study's small sample size and exploratory nature. IBM SPSS Statistics for Windows, version 25.0 (IBM Corp., Armonk, NY, USA) was used for statistical analysis and data management.
Ethics approval and consent to participate
Each patient's parent signed an informed consent approved by the King Faisal Specialist Hospital and Research Centre Research Ethics Committee.
Results
Characteristics of the study population
Between January 16, 2020, and March 22, 2021, 33 premature neonates were admitted to NICU and enrolled in this study. There were nine sets of twins. Among enrolled preterm neonates, 10 were born with a birthweight < 1500 g, and common morbidities were retinopathy of prematurity (N = 13), respiratory distress syndrome (N = 21), prolonged rupture of membrane (N = 12), and intraventricular hemorrhage (N = 5), bronchopulmonary dysplasia (N = 2), and necrotizing enterocolitis (N = 2). Out of 33 neonates, 6 (18.2%) were delivered vaginally, and 27 (81.8%) by Cesarean section. There were 15 (45.5%) females and 18 (54.5%) males. All preterm neonates received TPN solutions, with an average of 5.2 received, ranging from 2 to 16 days. DEP, BBzP, DBP, and DEHP were measured in 195 TPN solutions, and all samples had DEP and BBzP above the MDL of 0.05 and 0.047 µg/L, respectively. DBP and DEHP were detected at 25.6% and 82.6% above the MDLs of 0.169 and 0.05 µg/L, respectively.
Table 1 shows the demographics and clinical characteristics of the studied neonates, extracted from the patient's medical chart or measured during their stay.
Phthalates assessment
All urine samples (collected at admission and before discharge) contained MnBP and MEHP above the MDLs of 1.758 and 2.072 µg/L, respectively. MEP, MiBP, MBzP, MECPP, MEHHP, and MEOHP were 99.6%, 97.5%, 70%, 99.3%, 90.1%, and 92.6% above the MDLs of 1.597, 1.508, 0.986, 1.569, 1.619, and 0.605 µg/L, respectively. The ratio of samples (before: after TPN) higher than the MDL were 0:1 (MEP), 20:56 (MiBP), 11:187 (MBzP), 21:260 (MECPP), 16:239 (MEHHP), and 14:248 (MEOHP).
Table 2 presents the urinary levels of MEP, MiBP, MnBP, MBzP, MECPP, MEHHP, MEOHP, and MEHP in preterm neonates at admission and before discharge. The GEE models showed that among the tested phthalates, the means of MBzP (χ2 = 6.85, p = 0.009), MECPP (χ2 = 36.781, p < 0.001), MEHHP (χ2 = 23.033, p < 0.001), MEOHP (χ2 = 30.634, p < 0.001), MEHP (χ2 = 9.791, p = 0.002), ∑4DEHP (χ2 = 36.939, p < 0.001), and ∑3DEHP (χ2 = 33.822, p < 0.001) in the preterm neonates differed significantly at admission and before discharge. Post hoc analysis with a Bonferroni adjustment revealed that preterm neonates before discharge had a significant increase in urinary levels of MBzP [2.024 µg/L (95%CI 1.194, 3.432)], MECPP [12.207 µg/L (95%CI 5.436, 27.413)], MEHHP [7.33 µg/L (95%CI 3.251, 16.544)], MEOHP [11.201 µg/L (95%CI 4.764, 26.364)], MEHP [1.672 µg/L (95%CI 1.212, 2.309)], ∑4DEHP [6.449 µmol/L (95%CI 3.536, 11.763)], and ∑3DEHP [9.767 µmol/L (95%CI 4.531, 21.031)]. The ICC for serial measurements of urinary phthalate metabolites and the molar sum of the metabolites were moderate (< 0.75) for MnBP (0.697) and MEHP (0.612) and excellent (> 0.75) for MEP (0.811), MiBP (0.759), MBzP (0.874), MECPP (0.912), MEHHP (0.867), MEOHP (0.856), ∑4DEHP (0.892), ∑3DEHP (0.890), and ∑DBP (0.828).
Similar results were obtained when urinary phthalate and molar sum metabolites were calculated based on µg/g creat and µmol/g creat, respectively.
Table 3 presents the Spearman rank correlations for phthalates in TPN and their urinary metabolites in preterm neonates individually and on a molar sum basis. The only significant correlation was observed between DEP (a parent plasticizer) and its urinary metabolite MEP (rs = 0.243, p = 0.001). Our results show that correlations between parent compounds and other metabolites are not directly related through the same metabolic pathways. In addition, all the phthalates (except DEP) were inversely correlated with either their metabolites, such as DBP and MiBP (rs = − 0.164) or MnBP (rs = − 0.219), or other metabolites originating from different parent compounds.
Most phthalate metabolites were significantly correlated to various degrees with one another (Table S8). Metabolites from the same parent compound, particularly DEHP, were strongly correlated, with the highest correlation observed between MEOHP and MEHHP (rs = 0.957). A similar pattern was observed between DBP metabolites (MiBP and MnBP), with a correlation of 0.579.
Identification of risk factors associated with levels of phthalates
We looked at the relationship between various cofounders and the levels of phthalate metabolites in urine samples collected from preterm neonates. Inverse correlations were seen between body weight and MECPP (rs = − 0.554, p = 0.001), MEHHP (rs = − 0.403, p = 0.02), MEOHP (rs = − 0.352, p = 0.045), ∑4DEHP (rs = − 0.504, p = 0.003), and ∑3DEHP (rs = − 0.516, p = 0.002). The same metabolites were inversely correlated with head circumference. No gender differences were seen in the levels of phthalates.
We used Spearman rank correlations to examine the relationship between the length of stay in the NICU and body weight before discharge with urinary phthalate metabolites measured in samples collected from the preterm neonates before discharge. Positive relationships were seen between the length of stay in the NICU and MECPP (rs = 0.392, p = 0.024), ∑4DEHP (rs = 0.346, p = 0.049), and ∑3DEHP (rs = 0.349, p = 0.046). Results are presented in Table 4. Similar results were obtained when urinary phthalate and molar sum metabolites were calculated based on µg/g creat and µmol/g creat, respectively.
Neurodevelopment assessment
Table 5 shows that 1, 4, and 1 infants had scores lower than the cutoff of 22.77 (communication), 41.84 (gross motor), and 30.16 (fine motor), respectively. All exhibited low scores in only one domain. We also tried cutoffs based on 1SD/2SD below the mean of each domain. Out of the 30 infants, 80% scored below 1SD, with one infant scoring low in three developmental domains (gross motor, problem-solving, and personal-social) and 7 in two of the five domains. A score below 2SD was seen in 16.7% of infants, with only one scoring low in two domains (gross motor and problem-solving). Among the five domains, problem-solving scores were significantly and positively correlated with birth weight (rs = 0.396, p = 0.03) and crown-heel length (rs = 0.47, p = 0.009). Positive relationships were observed between the mother's age and gross motor (rs = 0.405, p = 0.026) and personal-social scores (rs = 0.389, p = 0.034). None of the five domains showed differences between males and females or was associated with the length of stay in the NICU (p > 0.05).
Bivariate analysis was conducted to test neonatal comorbidies listed in Table 1 with a sufficient number of observations (≥ 5). Only preterm neonates with retinopathy of prematurity had lower personal-solving scores, which was marginally significant (p = 0.08).
Inverse relationships were observed between DEHP in TPN solutions and personal-social scores (rs = − 0.433, p = 0.035). Surprisingly, a moderate positive correlation was seen between DEP and fine motor scores (rs = 0.764, p < 0.001). Results are shown in Table S9.
Crude and multiple regression models
We used linear regression models to evaluate the crude and adjusted effects of phthalate exposure after receiving TPN solutions on infants' neurodevelopmental performance at 2 months. As shown in Table 6, a decrease in ASQ-3 fine motor scores was associated with elevated urinary levels of MiBP (Δ% = − 5.4, 95%CI − 11.6, 1), MECPP (Δ% = − 4.6, 95%CI = − 8.5, − 0.6), MEHP (Δ% = − 5.9, 95%CI = − 11.8, 0.5), ∑4DEHP (Δ% = − 4.7, 95%CI = − 9.2, 0), and ∑3DEHP (Δ% = − 4.1, 95%CI = − 8.6, 0.5) in preterm neonates before discharge. An increase in ASQ-3 problem-solving scores in preterm neonates was associated with higher urinary MEP (Δ% = 12.5, 95%CI 1.3, 25.0), MnBP (Δ% = 13.5, 95%CI 2.2, 26.2), MECPP (Δ% = 4.0, 95%CI 0.3, 7.9), ∑4DEHP (Δ% = 5.1, 95%CI 0.8, 9.6) and ∑3DEHP (Δ% = 5.1, 95%CI 0.9, 9.4). Elevated levels of urinary MEP was associated with 11.5% increase (95%CI 0.8, 25.0) in gross motor scores. All models were adjusted for birth weight and the length of stay in the NICU and respective urinary phthalate metabolite measured at admission. Our findings were also clearly visualized using forest plot displaying based on adjusted GMRs and 95% CIs between ASQ-3 scores and urinary phthalate metabolites (Fig. 2).
Forest plot showing adjusted associations between scores of the five ASQ-3 domains and urinary phthalate metabolites. The estimated effects presented as geometric mean ratio (GMR) and 95% confidence intervals (95% CIs). The vertical red line represent no effect and the horizontal lines represent the 95%CI. Confidence intervals that do not cross the red vertical line are significant at *p < 0.05 or **p < 0.1; n.s.: not significant.
The crude results were roughly similar, although some became either marginally significant or non-significant after adjusting for confounders, such as the association between communication scores and urinary MnBP levels (Table S10).
Further, multiple regression analyses were performed only for urinary phthalate metabolites that exhibited negative association with ASQ-3 fine motor scores after adjusting separately for neonatal comorbidities such as respiratory distress syndrome, intraventricular hemorrhage, prolonged rupture of membrane and retinopathy of prematurity. The results remained the same except the association between MiBP and ASQ-3 fine motor scores became non-significant after adjusting for intraventricular hemorrhage.
Discussion
Main findings of the study
This prospective study was the first to assess the impact of exposure to phthalates through TPN solutions on the neurodevelopment status of 30 preterm neonates when they reached the age of 2 months. Urinary levels of MBzP, MECPP, MEHHP, MEOHP, MEHP, ∑4DEHP, and ∑3DEHP increased significantly in preterm neonates before discharge from the NICU. The daily dose of MEP, DBP, BBzP, and DEHP for our preterm neonates measured from their TPN solutions was much lower than the recommended tolerable daily limit. However, these levels were not correlated with their phthalate parent compounds in the TPN solutions, suggesting other sources of exposure in the NICU, such as devices, tubings, or staff/family members. Urinary DEHP metabolites were associated with reduced head circumference and birth weight. After adjusting for gestational age, the inverse relationships remained significant, suggesting the direct effects of phthalates. Finally, exposure of preterm neonates to DEHP during their stay in the NICU was associated with neurodevelopment scores in infants at the age of 2 months, mainly in fine motor skills. We also observed that increased levels of some phthalate metabolites were positively associated with problem-solving and gross motor skills; although this observation is unexpected, it was reported previously42,43,44.
Exposure of preterm neonates to phthalates during their NICU stay via TPN and its potential association with low neurodevelopment scores at the age of 2 months
We observed a 2- to 23-fold increase in the urinary excretion of MBzP, MECPP, MEHHP, MEOHP, and MEHP in preterm neonates before discharge. Yet, there was no association between these metabolites and their parenteral phthalates (BBzP and DEHP) in TPN solutions, suggesting that TPN might not be the source of exposure in our study. TPN bags used for our preterm neonate were labeled free of DEHP, and phthalate traces were detected in 83% of the TPN solutions, ranging between 0.379 to 4.232 µg/L, reflecting contamination during preparation. The maximum daily intake was 0.272 µg/kg body weight; this is much lower than the European Food Safety Authorities (EFSA) tolerable daily intake (TDI) limit of 50 μg/kg body weight/day45 or the Environmental Protection Agency (EPA) oral reference dose (RfD) of 20 μg/kg body weight/day46. It has been reported that depending on the lipid content added in TPN solutions, infants and children who receive long-term TPN might be regularly exposed to non-negligible amounts of DEHP ranging from 80 to 200 µg per day47. Despite the low content of DEHP in TPN solutions, our preterm neonates had been significantly exposed to DEHP, as shown by the high levels of its four metabolites (MECPP, MEHHP, MEOHP, and MEHP). These levels increased with the length of stay of the preterm neonates in the NICU. Unlike adults, in whom phthalates tend to have a shorter half-life (5–24 h)48, phthalates in preterm neonates are likely to have a longer half-life owing to the 'neonates' immature hepatic and renal clearance49,50. Despite the half-life of phthalates being short enough that they do not persist in the body, one should consider the adverse health effects from repeated, occasional, and long-term exposure. Short and long-term health risks are related to phthalate exposure in NICU, mainly subfertility, bronchopulmonary dysplasia, necrotizing enterocolitis, parenteral nutrition-associated cholestasis, and neurodevelopmental disorders51.
Few researchers have assessed DEHP exposure in NICU patients based on a single metabolite52 or either multiple ones or their sum19,53,54,55. Although our median values of MECPP (191.036 µg/L), MEHHP (58.325 µg/L), MEOHP (48.234 µg/L), and MEHP (30.103 µg/L) were ≥ fourfold higher than Stroustrup et al.19, they were 5–6 times lower than those observed by Su et al.55 and Weuve et al.53, apart from MEHP which was lower in those studies than our value. Demirel et al.23 noted that high excretion of MEHHP in the first urine sample of preterm neonates with birth weight < 1000 g might be associated with renal immaturity.
Nearly 80% of our preterm neonates who received TPN had detectable MBzP in their urine with a maximum concentration of 213.769 µg/L, and its parent compound (BBzP) was found in all solutions at concentrations between 1.075 and 24.952 µg/L. Vinyl flooring in the NICU might represent a hidden source of BBzP exposure to our preterm neonates53,56. The highest dose received by one preterm neonate was 3.226 µg/kg body weight/day, which is much lower than the EFSA's TDI of 500 µg/kg body weight/day57 or the EPA's RfD of 200 µg/kg body weight/day58. Our median MBzP value was almost 5–7 times lower than those reported by Stroustrup et al.19 and Weuve et al.53 at 27.37 and 41 µg/L, respectively.
DEP and ∑DBP are usually used as fixatives in perfumes, cosmetics, and personal care products59,60. All our preterm neonates had detectable MEP (a metabolite of DEP) and MnBP (a metabolite of DnBP) in urine, and 98.8% had MiBP (a metabolite of DiBP). Our median values of MEP (28.31 µg/L), MiBP (19.825 µg/L), and MnBP (34.116 µg/L) in preterm neonates were comparable to the values of 21.13, 15.26, and 36.82 µg/L, respectively, reported by Stroustrup et al.42, but higher than Weuve et al.53 for MnBP (18 µg/L). The highest MEP, MiBP, and MnBP were 541.51, 2035.63, and 268.28 µg/L, respectively. Whereas DEP was detected in all TPN solutions, ranging from 0.209 to 6.68 µg/L, only 20% of the solutions had DBP between 0.026 and 11.04 µg/L. Our data also demonstrated that DEP in TPN solution was significantly associated with urinary MEP levels in our preterm neonates, leading to a daily dose between 0.023 and 0.751 µg/kg body weight. None of our preterm neonates received a daily DEP dose exceeding the RfD of 800 μg/kg body weight61, suggesting that TPN might not be a significant source of exposure in our patients. The contribution of DBP in TPN to our preterm neonates' daily dose ranged between 0.002 and 1.427 µg/kg body weight, much lower than the RfD of 100 µg/kg body weight62.
Our data were 3-, 4-, 14-, 7-, 9-, and 21-fold higher for MnBP, MiBP, MECPP, MEHHP, MEOHP, and MEHP, respectively, than values reported by national surveys such as the National Health and Nutrition Examination Survey in the USA (CDC, 2019). Our findings suggest that phthalates in TPN solutions do not seem to significantly contribute to the high urinary levels of metabolites in preterm neonates. However, DEHP metabolites tended to increase with an extended stay in the NICU. Other possible sources, such as contact with NICU staff or family members who may have applied fragranced personal care products, received medications or blood transfusion, or been in contact with medications, devices, or cleaning materials may be the reason for their high urinary metabolite levels17,19.
In line with other studies, we observed negative associations between phthalate metabolites in the urine of preterm neonates and some birth anthropometric measures63,64. In our study population, smaller head circumference and low birth weight were associated with increased urinary levels of MECPP, MEHHP, MEOHP, ∑4DEHP, and ∑3DEHP. Because anthropometric measurements are expected to influence preterm delivery65, we reexamined these relationships after adjusting for gestational age. Head circumference and birth weight remained significantly and inversely associated only with MECPP, ∑4DEHP, and ∑3DEHP (p < 0.01). Although our study did not measure phthalate metabolites in the cord or maternal blood, phthalates can cross the placenta to the fetus, with a significant correlation between metabolites in the maternal blood and the cord66. In general, phthalates have a short half-life that can reflect the source of exposure in the preceding 1 or 2 days48. Because urine was collected from our preterm neonates immediately after birth, we believe that phthalate metabolites may originate from the mother and be transmitted. It is possible to assume that maternal exposure to phthalates, particularly DEHP, was associated with preterm delivery seen in the population, as reported by others67.
The implications of altered birth anthropometric parameters such as head circumference and birth weight in preterm neonates on child neurodevelopment have been reported68,69,70. Furthermore, exposure to phthalates, particularly DEHP, has been linked with neurodevelopment71, although data are still limited and inconsistent. As we reported earlier, exposure of our preterm neonates to phthalates, mostly DEHP, increased significantly at the end of their NICU stay. In this study, we observed inverse associations between fine motor scores and elevated levels of urinary MiBP, MECPP, MEHP, ∑4DEHP, and ∑3DEHP. Studies have shown that neonatal comorbidity such as respiratory distress, intraventricular hemorrhage, prolonged rupture of membrane, bronchopulmonary dysplasia, and necrotizing enterocolitis can impact neurodevelopmental outcomes72,73,74. The relationship between lower scores in fine motor and some urinary phthalate metabolites remained significant even after controlling for some neonatal comorbidity that we obtained. The risk of neurodevelopmental delay increases as the gestational age at birth decreases75,76. The World Health Organization classified preterm neonates into three categories based on gestational age: extremely preterm (< 28 weeks), very preterm (28–32 weeks), and moderate to late preterm (32 to 37 weeks)77. Since the gestational age of our studied preterm neonates falls within the three categories, we reexamined the inverse association between fine motor scores and MiBP, MECPP, MEHP, ∑4DEHP, and ∑3DEHP after controlling the regression models for gestational age. The results remained the same, where increases in all phthalate metabolites (except for MiBP) were associated with 3.8 to 6.5% decreases in fine motor scores. Because small head circumference predicts abnormal brain volume associated with neurodevelopment delays78,79, we adjusted our regression model for head circumference. Similar results were obtained, and a significant decrease of − 4.9 (95% CI − 8.5, − 1.1), − 5.8 (− 11.8, 0.5), − 5.3 (95%CI − 9.6, − 0.8), and − 4.6 (− 8.7, − 0.3) in fine motor scores was observed with elevated MECPP, MEHP, ∑4DEHP, and ∑3DEHP, respectively. For MiBP, there was a decrease in fine motor scores of − 5.1 (95% CI − 11.1, 1.4) but not significant.
Despite the link between head circumference, intracranial volume, and brain size, its association with cognitive function remains unclear because of risk factors' direct and indirect effects (nutritional, socioeconomic, environmental, and other anthropometric measures)80. For example, a study found the association between prenatal manganese and neurodevelopment scores was mediated by birth length rather than head circumference81. Our results may reflect a complex interplay between phthalate exposure and anthropometric measures that need to be explored to understand better the relationship between exposure and neurodevelopment in infants born prematurely. Impaired fine motor skills may hinder several aspects of daily functions ranging from basics, such as grasp strength, to more complex visual-motor integration that affects learning and writing abilities, subsequently influencing academic performance82. Exposure to endocrine disruptors such as DEHP can alter thyroid hormones critical for brain development83,84, particularly the cerebellum, associated with fine motor coordination85. Preterm neonates are also at high risk of impaired fine motor skills86. Surprisingly, we observed that increased urinary levels of some phthalate metabolites in preterm neonates were associated with better problem-solving and gross motor scores. A similar finding was observed between ∑DEHP and attention/social performance in preterm neonates42. The authors suggested that exposure to phthalates may follow a non-linear trajectory that accelerates the development of specific neural networks. Other researchers related improved behavioral development during the first years of life and prenatal exposure to certain phthalates to gender that might have a modifier role43,44. Although the relationship between maternal exposure to phthalates and neurodevelopment in children has also been examined in human studies71, only one study assessed preterm neonates during their NICU stay Stroustrup et al.42. In general, studies showed an inconsistent pattern in the association between exposure to phthalates and neurodevelopmental outcomes that was related to various factors such as exposure misclassification, critical window of exposure, sex-specific effects, and the effects of phthalate mixtures87,88.
Lastly, we should mention that the non-significant findings in our study might result from low statistical power owing to the small sample size, and additional research is needed using the data of a larger cohort.
Study limitations, strengths, and future direction
The present study's limitations should be acknowledged. First, the small sample size may have limited the statistical power to detect the detrimental effect of certain phthalates. The reasons for not achieving our targeted sample size (N = 150) were partially the outbreak of the SARS-CoV-2 virus and also because some preterm neonates did not require TPN solutions. However, we tried to optimize our study's statistical power by choosing continuous outcome variables rather than dichotomized outcome variables in the regression analyses. This is a valid approach used in epidemiological studies with small samples89,90. Furthermore, a recent study recommended a sample size of ≥ 25 for fixed-effect regression models91. Second, selection bias, including only high-risk preterm neonates, could have affected the results. Third, we tested specific neonatal comorbidities associated with neurodevelopment delay, particularly intraventricular hemorrhage92. No significant relationship was seen because only five preterm neonates were affected, which undermined the validity of intraventricular hemorrhage as a risk factor. Fourth, several potential confounders that might affect exposure and scores, such as genetic susceptibility and socioeconomic and nutritional status, were not considered. Fifth, there was a lack of adjustment in the statistical analysis for risk factors that impact preterm neonates' neurodevelopment, such as glucose and sodium abnormalities93 and metabolic complications related to parenteral nutrition94,95. Sixth, the number of measurements for each patient varied, either because sampling the urine was difficult or because the NICU nurse missed reserving it for the study after usage (TPN units). Seventh, 2 months may be too early to estimate infant neurodevelopment. Eighth, the ASQ-3 has higher sensitivity and specificity in older infants and children96. Ninth, the ASQ-3 is a questionnaire-based assessment for parents that reflects their negative or positive views, and misunderstandings of questions may lead to underestimating or overestimating an infant's developmental scores97. Tenth, residual confounding from unmeasured factors may have led to unexpected associations. Last, co-exposure to other pollutants could also affect the neurodevelopmental outcomes of our infants.
Nonetheless, the study has some strengths, which include (1) its prospective design; (2) exposure assessments being taken at two points (admission and before discharge); (3) daily measurements of phthalate metabolites in the urine of the preterm neonates receiving TPN solutions; (4) daily measurements of four phthalate compounds in TPN solutions received by the preterm neonates during their stay in the NICU; and (5) neurodevelopment assessments at 2 and 18 months (ongoing).
Our current research also examines whether the association between early exposure to phthalates in the NICU and neurodevelopment persists in our infants at the age of 18 months, using a range of neurocognitive tests to ensure an in-depth evaluation of various neurodevelopmental aspects, including autism.
Conclusions
This prospective study is the first to assess the impact of exposure to phthalates through TPN solutions on the neurodevelopment status of 30 preterm neonates at the age of 2 months. Although our sample size was small, our analyses revealed some potentially relevant findings. Urinary levels of MBzP, MECPP, MEHHP, MEOHP, MEHP, ∑4DEHP, and ∑3DEHP increased significantly in preterm neonates before discharge from the NICU, which was not associated with receiving TPN solutions but might have come from other sources. Elevated urinary MECPP, MEHHP, MEOHP, and MEHP metabolites in our preterm neonates indicate intensive use of DEHP devices/materials in the NICU. At 2 months, exposure of preterm neonates to DEHP in the NICU was associated with neurodevelopment scores in infants, mainly in fine motor skills. Some phthalate metabolites were positively associated with gross motor and problem-solving skills. Given the extreme vulnerability of our population, it is critical to minimize their exposure to phthalates during their NICU stay.
Data availability
The data that support the findings of this study are available from King Faisal Specialist Hospital and Research Centre, but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are, however, available from the authors upon reasonable request and with permission of King Faisal Specialist Hospital and Research Centre.
References
Liu, L. et al. Global, regional, and national causes of under-5 mortality in 2000–15: An updated systematic analysis with implications for the Sustainable Development Goals. Lancet 388, 3027–3035. https://doi.org/10.1016/s0140-6736(16)31593-8 (2016).
Kwinta, P. & Pietrzyk, J. J. Preterm birth and respiratory disease in later life. Expert Rev. Respir. Med. 4, 593–604. https://doi.org/10.1586/ers.10.59 (2010).
Lewandowski, A. J. & Leeson, P. Preeclampsia, prematurity and cardiovascular health in adult life. Early Hum. Dev. 90, 725–729. https://doi.org/10.1016/j.earlhumdev.2014.08.012 (2014).
Luyckx, V. A. Preterm birth and its impact on renal health. Semin. Nephrol. 37, 311–319. https://doi.org/10.1016/j.semnephrol.2017.05.002 (2017).
Luu, T. M., Rehman Mian, M. O. & Nuyt, A. M. Long-term impact of preterm birth: Neurodevelopmental and physical health outcomes. Clin. Perinatol. 44, 305–314. https://doi.org/10.1016/j.clp.2017.01.003 (2017).
Spector, L. G. et al. Cancer risk among children with very low birth weights. Pediatrics 124, 96–104. https://doi.org/10.1542/peds.2008-3069 (2009).
Chaudhari, S. & Kadam, S. Total parenteral nutrition in neonates. Indian Pediatr. 43, 953–964 (2006).
Patel, P. & Bhatia, J. Total parenteral nutrition for the very low birth weight infant. Semin. Fetal Neonatal Med. 22, 2–7. https://doi.org/10.1016/j.siny.2016.08.002 (2017).
Zingg, W., Tomaske, M. & Martin, M. Risk of parenteral nutrition in neonates—an overview. Nutrients 4, 1490–1503. https://doi.org/10.3390/nu4101490 (2012).
Sommer, I., Bouchoud, L., Berger-Gryllaki, M., Bonnabry, P. & Sadeghipour, F. Quality and safety of parenteral nutrition for newborn and preterm infants as an on-ward preparation. Eur. J. Hosp. Pharm. Sci. Pract. 27, 292–296. https://doi.org/10.1136/ejhpharm-2018-001788 (2020).
Santos, J., Pearce, S. E. & Stroustrup, A. Impact of hospital-based environmental exposures on neurodevelopmental outcomes of preterm infants. Curr. Opin. Pediatr. 27, 254–260. https://doi.org/10.1097/mop.0000000000000190 (2015).
Faessler, D., McCombie, G., Biedermann, M., Felder, F. & Subotic, U. Leaching of plasticizers from polyvinylchloride perfusion lines by different lipid emulsions for premature infants under clinical conditions. Int. J. Pharm. 520, 119–125. https://doi.org/10.1016/j.ijpharm.2017.01.046 (2017).
Heudorf, U., Mersch-Sundermann, V. & Angerer, J. Phthalates: Toxicology and exposure. Int. J. Hyg. Environ. Health 210, 623–634. https://doi.org/10.1016/j.ijheh.2007.07.011 (2007).
Bernard, L. et al. Analytical methods for the determination of DEHP plasticizer alternatives present in medical devices: A review. Talanta 129, 39–54. https://doi.org/10.1016/j.talanta.2014.04.069 (2014).
Jenkins, R., Ondusko, D., Montrose, L., Forbush, R. & Rozansky, D. Phthalate exposures in the neonatal intensive care unit. Toxics https://doi.org/10.3390/toxics9050090 (2021).
Latini, G., Ferri, M. & Chiellini, F. Materials degradation in PVC medical devices, DEHP leaching and neonatal outcomes. Curr. Med. Chem. 17, 2979–2989 (2010).
Mallow, E. B. & Fox, M. A. Phthalates and critically ill neonates: Device-related exposures and non-endocrine toxic risks. J. Perinatol. 34, 892–897. https://doi.org/10.1038/jp.2014.157 (2014).
Malarvannan, G. et al. Phthalate and alternative plasticizers in indwelling medical devices in pediatric intensive care units. J. Hazard Mater. 363, 64–72. https://doi.org/10.1016/j.jhazmat.2018.09.087 (2019).
Stroustrup, A. et al. Sources of clinically significant neonatal intensive care unit phthalate exposure. J. Expo. Sci. Environ. Epidemiol. 30, 137–148. https://doi.org/10.1038/s41370-018-0069-2 (2020).
Frederiksen, H., Skakkebaek, N. E. & Andersson, A. M. Metabolism of phthalates in humans. Mol. Nutr. Food Res. 51, 899–911. https://doi.org/10.1002/mnfr.200600243 (2007).
Samandar, E., Silva, M. J., Reidy, J. A., Needham, L. L. & Calafat, A. M. Temporal stability of eight phthalate metabolites and their glucuronide conjugates in human urine. Environ. Res. 109, 641–646. https://doi.org/10.1016/j.envres.2009.02.004 (2009).
Liu, L. et al. Infantile phthalate metabolism and toxico/pharmacokinetic implications within the first year of life. Environ. Int. 144, 106052. https://doi.org/10.1016/j.envint.2020.106052 (2020).
Demirel, A. et al. Hidden toxicity in neonatal intensive care units: Phthalate exposure in very low birth weight infants. J. Clin. Res. Pediatr. Endocrinol. 8, 298–304. https://doi.org/10.4274/jcrpe.3027 (2016).
Strommen, K. et al. Increased levels of phthalates in very low birth weight infants with septicemia and bronchopulmonary dysplasia. Environ. Int. 89–90, 228–234. https://doi.org/10.1016/j.envint.2016.01.024 (2016).
Barakat, R. et al. Prenatal exposure to DEHP induces neuronal degeneration and neurobehavioral abnormalities in adult male mice. Toxicol. Sci. 164, 439–452. https://doi.org/10.1093/toxsci/kfy103 (2018).
Kobrosly, R. W. et al. Prenatal phthalate exposures and neurobehavioral development scores in boys and girls at 6–10 years of age. Environ. Health Perspect. 122, 521–528. https://doi.org/10.1289/ehp.1307063 (2014).
Yolton, K. et al. Prenatal exposure to bisphenol A and phthalates and infant neurobehavior. Neurotoxicol. Teratol. 33, 558–566. https://doi.org/10.1016/j.ntt.2011.08.003 (2011).
Rosenfeld, C. S. Bisphenol A and phthalate endocrine disruption of parental and social behaviors. Front. Neurosci. 9, 57. https://doi.org/10.3389/fnins.2015.00057 (2015).
Pak, V. M., Nailon, R. E. & McCauley, L. A. Controversy: Neonatal exposure to plasticizers in the NICU. MCN Am. J. Matern. Child Nurs. 32, 244–249. https://doi.org/10.1097/01.NMC.0000281965.45905.c0 (2007).
von Elm, E. et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. Lancet 370, 1453–1457. https://doi.org/10.1016/s0140-6736(07)61602-x (2007).
Servaes, K. et al. Direct analysis of phthalate ester biomarkers in urine without preconcentration: Method validation and monitoring. J. Chromatogr. A 1294, 25–32. https://doi.org/10.1016/j.chroma.2013.03.054 (2013).
Wang, H. X., Wang, B., Zhou, Y. & Jiang, Q. W. Rapid and sensitive analysis of phthalate metabolites, bisphenol A, and endogenous steroid hormones in human urine by mixed-mode solid-phase extraction, dansylation, and ultra-performance liquid chromatography coupled with triple quadrupole mass spectrometry. Anal. Bioanal. Chem. 405, 4313–4319. https://doi.org/10.1007/s00216-013-6779-3 (2013).
Silva, M. J. et al. Improved quantitative detection of 11 urinary phthalate metabolites in humans using liquid chromatography-atmospheric pressure chemical ionization tandem mass spectrometry. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 789, 393–404 (2003).
Hays, S. M., Aylward, L. L. & Blount, B. C. Variation in urinary flow rates according to demographic characteristics and body mass index in NHANES: Potential confounding of associations between health outcomes and urinary biomarker concentrations. Environ. Health Perspect. 123, 293–300. https://doi.org/10.1289/ehp.1408944 (2015).
Squires, J. & Bricker, D. Ages & Stages Questionnaires (ASQ3): A Parent-Completed Child-Monitoring System (Paul Brookes Publishing Company, 2009).
Hornung, R. W. & Reed, L. D. Estimation of average concentration in the presence of nondetectable values. Appl. Occup. Environ. Hyg. 5, 46–51. https://doi.org/10.1080/1047322X.1990.10389587 (1990).
Lubin, J. H. et al. Epidemiologic evaluation of measurement data in the presence of detection limits. Environ. Health Perspect. 112, 1691–1696. https://doi.org/10.1289/ehp.7199 (2004).
Ferguson, K. K., McElrath, T. F., Ko, Y. A., Mukherjee, B. & Meeker, J. D. Variability in urinary phthalate metabolite levels across pregnancy and sensitive windows of exposure for the risk of preterm birth. Environ. Int. 70, 118–124. https://doi.org/10.1016/j.envint.2014.05.016 (2014).
Johns, L. E., Ferguson, K. K., McElrath, T. F., Mukherjee, B. & Meeker, J. D. Associations between repeated measures of maternal urinary phthalate metabolites and thyroid hormone parameters during pregnancy. Environ. Health Perspect. 124, 1808–1815. https://doi.org/10.1289/ehp170 (2016).
Cole, S. R., Chu, H., Nie, L. & Schisterman, E. F. Estimating the odds ratio when exposure has a limit of detection. Int. J. Epidemiol. 38, 1674–1680. https://doi.org/10.1093/ije/dyp269 (2009).
Rosen, B. Fundamentals of Biostatistics (Duxbury Press, 2005).
Stroustrup, A. et al. Neonatal intensive care unit phthalate exposure and preterm infant neurobehavioral performance. PLoS ONE 13, e0193835. https://doi.org/10.1371/journal.pone.0193835 (2018).
Braun, J. M. et al. Prenatal phthalate, triclosan, and bisphenol A exposures and child visual-spatial abilities. Neurotoxicology 58, 75–83. https://doi.org/10.1016/j.neuro.2016.11.009 (2017).
Téllez-Rojo, M. M. et al. Prenatal urinary phthalate metabolites levels and neurodevelopment in children at two and three years of age. Sci. Total Environ. 461–462, 386–390. https://doi.org/10.1016/j.scitotenv.2013.05.021 (2013).
EFSA. Opinion of the Scientific Panel on food additives, flavourings, processing aids and materials in contact with food (AFC) related to Bis(2-ethylhexyl)phthalate (DEHP) for use in food contact materials. EFSA J. 3, 243. https://doi.org/10.2903/j.efsa.2005.243 (2005).
United States Environmental Protection Agency (USEPA). Di(2-ethylhexyl)phthalate (DEHP) (CASRN 117-81-7): reference dose for chronic oral exposure (RfD). (1987).
Kambia, K. et al. Evaluation of childhood exposure to di(2-ethylhexyl) phthalate from perfusion kits during long-term parenteral nutrition. Int. J. Pharm. 262, 83–91. https://doi.org/10.1016/s0378-5173(03)00335-1 (2003).
Wittassek, M. & Angerer, J. Phthalates: Metabolism and exposure. Int. J. Androl. 31, 131–138. https://doi.org/10.1111/j.1365-2605.2007.00837.x (2008).
Gubhaju, L. et al. Assessment of renal functional maturation and injury in preterm neonates during the first month of life. Am. J. Physiol. Renal Physiol. 307, F149–F158. https://doi.org/10.1152/ajprenal.00439.2013 (2014).
Beath, S. V. Hepatic function and physiology in the newborn. Semin. Neonatol. 8, 337–346. https://doi.org/10.1016/s1084-2756(03)00066-6 (2003).
Fischer, C. J., BickleGraz, M., Muehlethaler, V., Palmero, D. & Tolsa, J. F. Phthalates in the NICU: Is it safe?. J. Paediatr. Child Health 49, E413-419. https://doi.org/10.1111/jpc.12244 (2013).
Green, R. et al. Use of di(2-ethylhexyl) phthalate-containing medical products and urinary levels of mono(2-ethylhexyl) phthalate in neonatal intensive care unit infants. Environ. Health Perspect. 113, 1222–1225. https://doi.org/10.1289/ehp.7932 (2005).
Weuve, J. et al. Exposure to phthalates in neonatal intensive care unit infants: Urinary concentrations of monoesters and oxidative metabolites. Environ. Health Perspect. 114, 1424–1431. https://doi.org/10.1289/ehp.8926 (2006).
Calafat, A. M., Needham, L. L., Silva, M. J. & Lambert, G. Exposure to di-(2-ethylhexyl) phthalate among premature neonates in a neonatal intensive care unit. Pediatrics 113, e429-434 (2004).
Su, P. H. et al. Exposure to di(2-ethylhexyl) phthalate in premature neonates in a neonatal intensive care unit in Taiwan. Pediatr. Crit. Care Med. 13, 671–677. https://doi.org/10.1097/PCC.0b013e3182455558 (2012).
Just, A. C. et al. Vinyl flooring in the home is associated with children’s airborne butylbenzyl phthalate and urinary metabolite concentrations. J. Expo Sci. Environ. Epidemiol. 25, 574–579. https://doi.org/10.1038/jes.2015.4 (2015).
EFSA et al. Update of the risk assessment of di-butylphthalate (DBP), butyl-benzyl-phthalate (BBP), bis(2-ethylhexyl)phthalate (DEHP), di-isononylphthalate (DINP) and di-isodecylphthalate (DIDP) for use in food contact materials. EFSA J. 17, e05838. https://doi.org/10.2903/j.efsa.2019.5838 (2019).
United States Environmental Protection Agency (USEPA). Butyl benzyl phthalate (BBP) CASRN 85-68-7|DTXSID3020205 (1989).
Koch, H. M. et al. Identifying sources of phthalate exposure with human biomonitoring: Results of a 48h fasting study with urine collection and personal activity patterns. Int. J. Hyg. Environ. Health 216, 672–681. https://doi.org/10.1016/j.ijheh.2012.12.002 (2013).
Parlett, L. E., Calafat, A. M. & Swan, S. H. Women’s exposure to phthalates in relation to use of personal care products. J. Expo Sci. Environ. Epidemiol. 23, 197–206. https://doi.org/10.1038/jes.2012.105 (2013).
United States Environmental Protection Agency (USEPA). Diethyl phthalate (DEP)-CASRN 84-66-2. (1987).
United States Environmental Protection Agency (USEPA). Dibutyl phthalate; CASRN 84-74-2. (1987).
Darvishmotevalli, M. et al. Association between prenatal phthalate exposure and anthropometric measures of newborns in a sample of Iranian population. Environ. Sci. Pollut. Res. Int. 28, 50696–50706. https://doi.org/10.1007/s11356-021-14182-0 (2021).
Bloom, M. S. et al. Association between gestational phthalate exposure and newborn head circumference; impacts by race and sex. Environ. Res. 195, 110763. https://doi.org/10.1016/j.envres.2021.110763 (2021).
Karna, P., Brooks, K., Muttineni, J. & Karmaus, W. Anthropometric measurements for neonates, 23 to 29 weeks gestation, in the 1990s. Paediatr. Perinat. Epidemiol. 19, 215–226. https://doi.org/10.1111/j.1365-3016.2005.00641.x (2005).
Li, X. et al. Distribution of phthalate metabolites between paired maternal-fetal samples. Environ. Sci. Technol. 52, 6626–6635. https://doi.org/10.1021/acs.est.8b00838 (2018).
Wu, Y. et al. Maternal exposure to endocrine disrupting chemicals (EDCs) and preterm birth: A systematic review, meta-analysis, and meta-regression analysis. Environ. Pollut. 292, 118264. https://doi.org/10.1016/j.envpol.2021.118264 (2022).
Ranke, M. B., Krägeloh-Mann, I. & Vollmer, B. Growth, head growth, and neurocognitive outcome in children born very preterm: Methodological aspects and selected results. Dev. Med. Child Neurol. 57, 23–28. https://doi.org/10.1111/dmcn.12582 (2015).
Selvanathan, T. et al. Head circumference, total cerebral volume and neurodevelopment in preterm neonates. Arch. Dis. Childhood Fetal Neonatal Ed. 107, 181–187. https://doi.org/10.1136/archdischild-2020-321397 (2022).
Longo, S. et al. Neurodevelopmental outcome of preterm very low birth weight infants admitted to an Italian tertiary center over an 11-year period. Sci. Rep. 11, 16316. https://doi.org/10.1038/s41598-021-95864-0 (2021).
Minatoya, M. & Kishi, R. A review of recent studies on bisphenol A and phthalate exposures and child neurodevelopment. Int. J. Environ. Res. Public Health 18, 3585. https://doi.org/10.3390/ijerph18073585 (2021).
Meller, C. H., Carducci, M. E., CerianiCernadas, J. M. & Otaño, L. Preterm premature rupture of membranes. Archivos argentinos de pediatria 116, e575–e581. https://doi.org/10.5546/aap.2018.eng.e575 (2018).
Tan, H. et al. Neurodevelopmental outcomes in preterm infants with retinopathy of prematurity. Surv. Ophthalmol. 66, 877–891. https://doi.org/10.1016/j.survophthal.2021.02.012 (2021).
Chung, E. H., Chou, J. & Brown, K. A. J. T. P. Neurodevelopmental outcomes of preterm infants: A recent literature review. Transl. Pediatr. 2019, S3–S8 (2019).
Jois, R. S. Understanding long-term neurodevelopmental outcomes of very and extremely preterm infants: A clinical review. Austral. J. Gen. Pract. 48, 26–32. https://doi.org/10.31128/ajgp-04-18-4545 (2019).
Larsen, M. L. et al. The effect of gestational age on major neurodevelopmental disorders in preterm infants. Pediatr. Res. 91, 1906–1912. https://doi.org/10.1038/s41390-021-01710-4 (2022).
Blencowe, H. et al. National, regional, and worldwide estimates of preterm birth rates in the year 2010 with time trends since 1990 for selected countries: A systematic analysis and implications. Lancet 379, 2162–2172. https://doi.org/10.1016/s0140-6736(12)60820-4 (2012).
Bartholomeusz, H. H., Courchesne, E. & Karns, C. M. Relationship between head circumference and brain volume in healthy normal toddlers, children, and adults. Neuropediatrics 33, 239–241. https://doi.org/10.1055/s-2002-36735 (2002).
Dupont, C. et al. The predictive value of head circumference growth during the first year of life on early child traits. Sci. Rep. 8, 9828. https://doi.org/10.1038/s41598-018-28165-8 (2018).
Nicolaou, L. et al. Factors associated with head circumference and indices of cognitive development in early childhood. BMJ Glob. Health https://doi.org/10.1136/bmjgh-2020-003427 (2020).
Lee, J. J. et al. Growth parameters at birth mediate the relationship between prenatal manganese exposure and cognitive test scores among a cohort of 2- to 3-year-old Bangladeshi children. Int. J. Epidemiol. 47, 1169–1179. https://doi.org/10.1093/ije/dyy069 (2018).
Escolano-Pérez, E., Herrero-Nivela, M. L. & Losada, J. L. Association between preschoolers’ specific fine (but not gross) motor skills and later academic competencies: Educational implications. Front. Psychol. 11, 1044. https://doi.org/10.3389/fpsyg.2020.01044 (2020).
Koibuchi, N. The role of thyroid hormone on functional organization in the cerebellum. Cerebellum 12, 304–306. https://doi.org/10.1007/s12311-012-0437-8 (2013).
Zhang, X. et al. Di(2-ethylhexyl) phthalate (DEHP) and thyroid: Biological mechanisms of interference and possible clinical implications. Environ. Sci. Pollut. Res. Int. 29, 1634–1644. https://doi.org/10.1007/s11356-021-17027-y (2022).
Burr, P. & Choudhury, P. StatPearls (StatPearls Publishing LLC, 2022).
Bos, A. F., Van Braeckel, K. N., Hitzert, M. M., Tanis, J. C. & Roze, E. Development of fine motor skills in preterm infants. Dev. Med. Child Neurol. 55(Suppl 4), 1–4. https://doi.org/10.1111/dmcn.12297 (2013).
Radke, E. G., Braun, J. M., Nachman, R. M. & Cooper, G. S. Phthalate exposure and neurodevelopment: A systematic review and meta-analysis of human epidemiological evidence. Environ. Int. 137, 105408. https://doi.org/10.1016/j.envint.2019.105408 (2020).
Selevan, S. G., Kimmel, C. A. & Mendola, P. Identifying critical windows of exposure for children’s health. Environ. Health Perspect. 108(Suppl 3), 451–455. https://doi.org/10.1289/ehp.00108s3451 (2000).
Bhandari, M., Lochner, H. & Tornetta, P. 3rd. Effect of continuous versus dichotomous outcome variables on study power when sample sizes of orthopaedic randomized trials are small. Arch. Orthop. Trauma Surg. 122, 96–98. https://doi.org/10.1007/s004020100347 (2002).
Altman, D. G. & Royston, P. The cost of dichotomising continuous variables. BMJ 332, 1080. https://doi.org/10.1136/bmj.332.7549.1080 (2006).
Jenkins, D. G. & Quintana-Ascencio, P. F. A solution to minimum sample size for regressions. PLoS ONE 15, e0229345. https://doi.org/10.1371/journal.pone.0229345 (2020).
Triplett, R. L. & Smyser, C. D. Neuroimaging of structural and functional connectivity in preterm infants with intraventricular hemorrhage. Semin. Perinatol. 46, 151593. https://doi.org/10.1016/j.semperi.2022.151593 (2022).
Yang, H. Y. et al. Neurodevelopment of preterm infants with glucose and sodium abnormalities. Pediatr. Neonatol. 62, 647–654. https://doi.org/10.1016/j.pedneo.2021.06.008 (2021).
Terrin, G. et al. Energy-enhanced parenteral nutrition and neurodevelopment of preterm newborns: A cohort study. Nutrition 89, 111219. https://doi.org/10.1016/j.nut.2021.111219 (2021).
Boscarino, G. et al. Neonatal hyperglycemia related to parenteral nutrition affects long-term neurodevelopment in preterm newborn: A prospective cohort study. Nutrients https://doi.org/10.3390/nu13061930 (2021).
Steenis, L. J. P., Verhoeven, M., Hessen, D. J. & van Baar, A. L. Parental and professional assessment of early child development: The ASQ-3 and the Bayley-III-NL. Early Hum. Dev. 91, 217–225. https://doi.org/10.1016/j.earlhumdev.2015.01.008 (2015).
Kendall, S. et al. Evaluating the use of a population measure of child development in the Healthy Child Programme Two Year Review. 1–134 (2014).
Acknowledgements
The corresponding author extends appreciation to King Salman Center for Disability Research for funding this work through Research Group no KSRG-2022-082. The investigators gratefully acknowledge the Neonatal Intensive Care Unit [Raquel Salindong, Flor Ainah Banayat, Jamaica Sanchez, Wejdan Otayf, Norah Albashir, and Samia Handoum] and the parents of preterm neonates for their participation in this study. The forest plot was done by Jude Aljoysir using R (programming language).
Funding
This study was supported by King Salman Center for Disability Research (RG-2022-082).
Author information
Authors and Affiliations
Contributions
I.A.—conceptualization/writing—original draft/writing—review and editing/formal analysis/visualization/project administration/supervision/funding acquisition; RE- Methodology/validation; H.A.—methodology/coordinating; H.A.—neurodevelopment evaluation; E.A.—neurodevelopment assessment; A.B., A.H., F.A., M.J.—resources (neonatologist); G.M.—statistical advise/interpretation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Al-Saleh, I., Elkhatib, R., Alnuwaysir, H. et al. Exposure of preterm neonates receiving total parenteral nutrition to phthalates and its impact on neurodevelopment at the age of 2 months. Sci Rep 13, 6969 (2023). https://doi.org/10.1038/s41598-023-33715-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-33715-w
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.