Abstract
The aim of our research was to identify the factors that most strongly determine the C, N and P cycles in the deadwood—soil system in mountains forest ecosystems. We assumed that the climatic conditions resulting from the location in the altitude gradient and rate of deadwood decomposition most strongly determine the C/N/P stoichiometry. A climosequence approach comprising north (N) and south (S) exposure along the altitudinal gradient (600, 800, 1000 and 1200 m a.s.l.) was set up. Spruce logs at different decomposition stages (III, IV and V) were selected for the analysis in Babiogórski National Park (southern Poland). We calculated the C/N/P stoichiometry for deadwood and soil samples to reflect the nutrient availability. Our research indicates a very strong influence of the location conditions in the altitude gradient on the C/N/P stoichiometry. The GLM analysis confirmed the importance of high elevation in shaping the C, N and P content. A strong correlation was confirmed between P content, N content and C/N ratio. A higher C/N/P ratio was found in deadwood compared to soil, regardless of location. Decaying wood is an important source of N and P and the degree of decomposition made a significant contribution to explaining the variability of C, N and P content. The obtained results indicate the need to leave deadwood in forest ecosystems in order to improve biogeochemical cycles. Deadwood, by having a beneficial effect on many components of the forest ecosystem, will improve its biodiversity and, consequently, its stability.
Similar content being viewed by others
Introduction
Deadwood occurs in every forest ecosystem and performs many functions on which their proper functioning depends1,2. The key functions of deadwood include increasing biodiversity, shaping microhabitats and soil properties, storing nutrients and water3,4,5. Deadwood plays an important role in the cycle of carbon, nutrients and hydrological cycles6,7,8. Decaying deadwood is an important component of biodiversity in European forests and is used as an indicator in assessing and monitoring the biodiversity of forest ecosystems.
Deadwood can vary in type and degree of decay. The most deadwood can be divided into coarse woody debris (CWD) and fine woody debris (FWD)9. Deadwood includes dead woody plant material, for example standing dead trees, lying dead trees, snags, stumps and branches10. In studies of deadwood it is important to assessing the degree of deadwood decomposition and this is determined using a five-stage classification based on the characteristics of the wood, such as the presence of bark, structure and color of the wood11. Depending on the degree of decomposition, wood affects the soil environment12. More heavily decomposed wood has a greater impact on physical, chemical and biochemical properties compared to less decomposed wood13. The rate of decomposition of deadwood depends on the type of wood, species and conditions in which the decomposition process takes place14. Thermal conditions and moisture have a very strong impact on the rate of decomposition in forest ecosystems15,16,17. According to Liu et al.18 warming affects the circulation of C, N and P in the forest ecosystem. The decomposition process is important for nutrient cycling in terrestrial ecosystems and is influenced not only by climate but also by microorganisms19,20. Mountain forest ecosystems growing in different climate conditions provide the opportunity to conduct research on the rate of decomposition depending on climatic conditions20,21. In addition to the temperature along the elevation gradient. The water regime changes, which has a direct impact on the rate of decomposition22.
The rate of decomposition of organic remains in forest ecosystems determines the cycle of C, N and P. Ecological stoichiometry is used to represent the balance between many elements, with particular emphasis on C, N and P23. C/N/P stoichiometry is closely related to elementary processes such as photosynthesis, respiration or mineralization of organic matter24. According to Liu et al.25 the C/N/P stoichiometry strongly correlates with thermal conditions and the amount of precipitation. In the research of Chen et al.26 the climatic factor made a strong contribution to explaining the C/N/P stoichiometry of soils. Deadwood is an important element of forest ecosystems, it affects the C, N and P cycles and thus recognition the patterns of ecological stoichiometry in the deadwood—soil system will allow for a better understanding of nutrient cycling, ecosystem dynamics and mechanisms of the biogeochemical cycle. So far, there have been no research on C/N/P stoichiometry in the deadwood-soil system which would consider the influence of climatic conditions.
In our study, we used the climosequence approach in determining patterns of ecological stoichiometry in system of deadwood and soil. The aim of our research was to identify the factors that most strongly determine the C, N and P cycles in the deadwood—soil system. We assumed that the climatic conditions resulting from the location in the altitude gradient most strongly determine the C/N/P stoichiometry. We assume that the greatly decomposed wood in the 5th stage of decomposition strongly affects the circulation of C, N and P.
Materials and methods
Study area
The research was carried out in the Babiogórski National Park in southern Poland. (49°35′18′′N; 19°32′23′′E). The study included three transects with study plots on the southern and northern slopes with an inclination of 15°. A climosequence approach comprising north (N) and south (S) exposure along the altitudinal gradient (600, 800, 1000 and 1200 m a.s.l.) was set up. In each variant of the elevation, in each transect research plots were designated separately for deadwood in different decay rate. Lying dead trees of spruce in the form of logs at different decomposition stages (III, IV and V) were selected for the analysis. Our study covered logs in the III-V degree of decomposition with visible signs of the decomposition process. Logs in the first and second stages of decomposition are not included in this study as previous studies have confirmed that they have no or only a small impact on soil characteristics27,28. The decay classes (DC) of logs were estimated according to the classification of dead trees in Maser et al.29 which was used in the previous study4,5. The tested soils were characterized by the similar texture (average sand content was 52%, silt 42% and clay 6%). The sensors (5TM, Sensor, Decagon) were used to temperature test during the year. The average annual temperature of the S exposure for the study plots at 600 m a.s.l. it was 6.8 °C, for the study plots of 800 m a.s.l. it was 5.6 °C, at an elevation of 1000 m a.s.l. it was 4.5 °C and at an elevation of 1200 m a.s.l. it amounted to 3.4 °C. The average annual temperature of the N exposure for the study plots at 600 m a.s.l. was 6.0 °C, for the study plots of 800 m a.s.l. was 5.1 °C, at an elevation of 1000 m a.s.l. was 4.0 °C and at an elevation of 1200 m a.s.l. was 3.1 °C. Regardless of the exposure, the lowest locations (600 m a.s.l.) are characterized by lower average annual precipitation (about 1200 mm), while at an altitude of 1200 m a.s.l. the average annual precipitation is about 1400 mm30. The study plot was the area with the log covered by the analysis with the soil sampling area around it. We selected logs with diameter between 25 and 35 cm to ensure direct comparability of observations. The study covered 72 logs of deadwood located in different location condition (2 exposure × 4 points in altitude gradient × 3 degrees of decomposition × 3 repetitions = 72 logs). Wood samples measuring 7 × 7 × 7 cm for laboratory analysis were taken from the midpoint of each log. In our study we analyzed 144 soil samples. The soil samples were collected directly under the log, the soil was sampled from 0 to 10 cm depth using a small spade. Additionally soil samples (background) were taken 1 m from logs from 0 to 10 cm depth. In total, 216 wood and soil samples were taken for laboratory tests. In the study, we included samples from decaying logs (D), soil samples lying under deadwood (UD) and soil samples taken 1 m from the logs of decaying wood (C). The field study was realized in 2021.
Laboratory analysis
Total nitrogen and carbon content was determined using a LECO CNS True Mac Analyser (Leco, St. Joseph, MI, USA). We used the ICP analysis (ICP-OES Thermo iCAP 6500 DUO, Thermo Fisher Scientific, Cambridge, U.K.) to determine the P content. We determined the levels of P after mineralisation of the mixture with concentrated nitric and perchloric acids at a ratio of 3:1 and calculated the C/N, C/P and N/P ratios on a molecular level.
Statistical analysis
The Spearman correlation coefficient between the C, N and P content and C/N/P stoichiometry of deadwood and soil was calculated. The Shapiro–Wilk test was used to assess normality and Levene’s test was used to check the homogeneity of variances. Regression models were developed for the N, P content and C/N ratio with a division into three types of samples (wood, soil under wood and control). In order to show the relationship between the examined variables, principal component analysis (PCA) was performed. The GLM analysis was used to determine the significance of the influence of altitude, exposure, samples type and the degree of decomposition of deadwood on the C/N/P stoichiometry. Differences with p < 0.05 were considered to be statistically significant. All statistical analyses were performed using R statistical software (R Core Team 2020), R Studio (RStudio Team 2020), and Statistica 10 software (2010).
Results
The GLM analysis confirmed the influence of altitude, decomposition degree and type of sample on the content of N, C and P (Table 1). In the case of the P content, the exposure also had a significant impact on its content. For the N and C content, the exposure had no significant effect (Table 1).
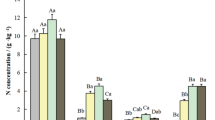
The highest nitrogen content was recorded in soils affected by the most strongly decomposed wood in the 5th stage of decomposition at an altitude of 600 m a.s.l. at the N and S exposure (19.35 g kg-1 and 15.59 g kg-1 respectively). Regardless of the location and the climate conditions prevailing there, the soil affected by the impact of decaying wood is characterized by a higher content of N and C (Fig. 1). Wood in the 5th degree of decomposition is characterized by the highest content of N regardless of the location (Fig. 1). At the N and S exposure, the soil affected by the wood had a higher C content compared to the control soil. The content of C in soils under the influence of wood in the 5th degree of decomposition at the N exposure ranged from 157.6 to 542.5 g kg-1, and in the samples of control soils the carbon content ranged from 61.9 to 152.1 g kg-1. The content of C in soils affected by wood in the 5th degree of decomposition at the S exposure ranged from 243.5 to 397.9 g kg-1, and in the samples of control soils, the content of C ranged from 91.7 to 136.5 g kg-1. In most locations, the impact of decaying wood on shaping the phosphorus content in soils influenced by wood can be seen. In most cases, the content of phosphorus in soils affected by decaying wood was higher than in control soils. At the N exposure at the altitude of 600 m a.s.l. lower P content was noted in the soils affected by the decaying wood compared to the S exposure (Fig. 1).
The conducted GLM analysis confirmed the importance of the degree of decomposition in shaping the C/N ratio. In the case of C/P, the location conditions were more important than the degree of decomposition (Table 1). Wood samples, regardless of the degree of decomposition, were characterized by higher C/N and C/P ratios compared to the soil samples (Fig. 2). The average C/N of less decayed wood (III DC) was in the range of 225.9–541.7, the C/N of wood in IV DC was from 108.6 to 464.8, and the C/N of the most heavily decayed wood in the V degree of decay was in the range of 61.8–277.4. As in the case of the C/N ratio, the C/P ratio decreased in more strongly decomposed wood, regardless of the location. Soils affected by decaying wood had higher C/N and C/P ratios compared to the control. In the case of the N/P ratio, the GLM analysis confirmed the importance of the location conditions and the degree of decomposition in shaping its value. Regardless of the location, the N/P ratio was higher in the wood samples and in the soil affected by decaying wood compared to controls (Fig. 2).
The conducted correlation analysis indicates a strong positive relationship between the content of N and P (r = 0.87) (Fig. 3). P content negatively correlated with C content, C/N ratio and C/P ratio (r = − 0.60, r = − 0.78, r = − 0.78 respectively). The C/N ratio strongly positively correlated with the C/P and N/P ratios (r = 0.95 and r = 0.61, respectively). In addition, there was a strong positive relationship between the C/P and N/P ratios (r = 0.78) (Fig. 3).
Figure 4 shows the relationship between the content of N and P in relation to the type of sample. In the case of both wood and soil samples, the P content increases with increasing N content. In the case of wood samples, a strong negative relationship was noted between the C/N ratio and the P content (Fig. 4).
The performed PCA analysis confirmed the relationship between the studied properties (Fig. 5). Two main factors contributed to the observed variance (89.4%): factor 1 accounts for 66.7% of the variance while factor 2 explains 22.7% of the variance. Factor 1 is related to P content and C/P and C/N stoichiometry, while factor 2 is related to N content and N/P ratio. The performed PCA analysis confirms the positive relationship between the P and N content and the positive relationship between the C content and the C/N, C/P and N/P stoichiometry. The PCA analysis confirmed that the wood samples are characterized by a higher C/N/P stoichiometry compared to the soil samples. In terms of the examined properties, the wood samples form a separate set. The highest content of N and P was recorded in the soil samples affected by decaying wood.
Discussion
The conducted research confirmed the differences between decaying wood and the soil environment in terms of C:N:P stoichiometry. Decaying wood is very rich in C and at the same time poor in terms of N and P. The consequence of this is wide C/N and C/P ratios in decaying wood. The obtained results confirm previous research31. In the soil environment, the situation is reversed. The surface horizons of humus accumulation reach high carbon contents with relatively high nitrogen and phosphorus content. As a result of the above, the average C/N ratios are 10–20 times lower in soil compared to wood, and the C/P ratios are even 50–100 times lower than those found in wood. The differences found in the stoichiometry of wood in relation to the soil environment coincide with the earlier research12,31. The very wide C:N:P ratio in decaying wood is due to the chemical composition of the wood substance itself. Raw wood composed mainly of cellulose and lignin contains C in the amount of approx. 50%, while N and P in raw wood are only about 1.0–1.5% and 0.15–0.20% respectively32. The C:N:P proportions depend to some extent on the wood species31. Our experiment was limited to the use of decomposing spruce wood and its impact on the soil environment was analysed. With the advancement of the wood decomposition process, its chemical composition changes27,33,34. As a result of the decomposition of polysaccharides, the amount of carbon decreases, while the amount of nitrogen and phosphorus increases relatively35. An increase in the content of nitrogen and phosphorus in decomposed wood may be related to the transport of these nutrients by mycelial hyphae from the soil environment adjacent to deadwood36,37. Specialized groups of bacteria with the ability to bind atmospheric N also participate in the process of wood decomposition38,39.
The experiment confirmed the significant influence of decaying wood on the stoichiometry of soil affected by deadwood. Decaying wood releases large amounts of dissolved carbon, which moves to the underlying soil with fallout40,41,42. Along with leachates from decaying wood, large amounts of nitrates and phosphates are released, which feed the soil under the wood5. In our experiment, the most decomposed wood (in V DC) had the strongest impact on the soil, which is consistent with the results of previous studies41. The significantly higher nitrogen content in the soil, compared to the control variant (without the influence of wood) was found in all localization conditions under decaying wood in V DC. In the case of phosphorus, we did not find such an unambiguous effect. More phosphorus than controls accumulated under the V DC wood on most of the tested surfaces but there were exceptions to this rule. At the cooler exhibition, such exceptions occurred at an altitude of 800 and 1000 m above sea level, at the highest altitude at the warmer exposure. Phosphorus is considered to be an ingredient limiting the development of microorganisms decomposing organic matter43,44. There are reports of a higher accumulation of this macroelement in cooler locations, where the growing season is shorter and probably the entire P pool is not used by soil microorganisms45,46.
In our research, the GLM analysis confirmed the importance of the influence of altitude and slope exposure on the formation of the C:N:P stoichiometry. In mountainous areas, the climatic conditions change with the altitude, the temperature decreases and the amount of precipitation increases at the same time. According to Allison et al.47,48, temperature could influence decomposition of organic material by changing the activity of the decomposer community and also by changing plant species composition and litter chemistry. Climatic factors slow down biological activity and they explain the morphology of the humus forms and their variations49. In the area of our experiments, in the highest locations (1200 m above sea level), the vegetation period is shorter by 50 days, and rainfall is on average 200 mm higher than in the lowest locations per year30,50. In the highest locations, a significant slowdown in the rate of decomposition of deadwood can be expected and, at the same time, increased processes of washing the decomposition products into the soil beneath the wood. In our study, we found an expansion of the C:P ratio in wood in III and IV DC with increasing altitude, culminating at 1000 m above sea level. In the case of the most decomposed wood, the C:P ratio decreased with increasing altitude. On the other hand, in soil influenced by wood, the C:P ratio tends to increase, as in less decomposed wood. In wood, in the earlier stages of decomposition, we observed a decrease in the content of phosphorus with increasing altitude, which proves its role as a component occurring in the minimum, determining the occurrence of biochemical processes in the soil, including the processes of decomposition of organic matter43,44. It does not indicate the presence of an excess of this macroelement in high positions as suggested51. At the same time, in the most decomposed wood, the content of phosphorus increases, which can be explained by the differences in the time needed to reach a certain stage of decomposition in different climatic conditions. Herrmann and Bauhus35 proved that in the case of spruce logs, after the loss of approx. 40% of mass, the P content increases rapidly and is the greater the longer the decomposition process. For the N:P ratio, we found very similar ranges in the wood and in the affected soil. In most cases, they are in the range of 40–80. In deadwood, they show an upward trend with height up to 1000 m above sea level, a downward trend is observed in higher areas. In the soil, N:P first narrows, then after exceeding the position of 800–1000 m above sea level, it shows an upward trend again. It is difficult to explain the observed trends clearly. In studies carried out in various regions of Germany, significantly lower N/P ratios were found in spruce CWD35. In the initial stages of decomposition, this ratio was 13–23, while spruce wood, more strongly decomposed, increased the N/P ratio to the value of approx. 30, with wood decomposing mainly in lowlands and upland areas.
Conclusions
Our research confirmed the importance of the location conditions and the degree of decomposition in shaping the C/N/P stoichiometry in the deadwood-soil system. Deadwood, regardless of the degree of decomposition, was characterized by a higher C/N/P stoichiometry compared to soils. The C, N and P stoichiometry of mountain soils was most strongly affected by wood in the 5th degree of decomposition, regardless of location. With the height of a.s.l. the C/N and C/P ratio increases, which is associated with a change in climate conditions. The obtained results indicate a positive effect of decaying wood on the C/N/P stoichiometry. The research provides justification for leaving wood to natural decomposition in forest ecosystems in order to improve biogeochemical cycles. The positive impact of decaying wood on soil properties will result in improved stability of forest ecosystems.
Data availability
Data will be made available on request. In order to receive data from this study, please contact Ewa Błońska: ewa.blonska@urk.edu.pl.
References
Persiani, A. M. et al. Linking taxonomical and functional biodiversity of saproxylic fungi and beetles in broad-leaved forests in southern Italy with varying management histories. Plant Biosyst. 144(1), 250–261. https://doi.org/10.1080/11263500903561114 (2010).
Stokland, J., Siitonen, J., Jonsson, B. G. Biodiversity in Deadwood (Cambridge University Press, Cambridge, 2012) https://doi.org/10.1017/CBO9781139025843
Ciach, M. Dead and dying trees in forest ecosystem – quantity, quality, and diversity. Stud. Mater. CEPL Rogow. 2, 186–199 (2011).
Błońska, E., Kacprzyk, M. & Spólnik, A. Effect of deadwood of different tree species in various stages of decomposition on biochemical soil properties and carbon storage. Ecol. Res. 32(2), 193–203. https://doi.org/10.1007/s11284-016-1430-3 (2017).
Lasota, J., Błońska, E., Piaszczyk, W. & Wiecheć, M. How the deadwood of different tree species in various stages of decomposition affected nutrient dynamics?. J. Soil Sedim. 18(8), 2759–2769. https://doi.org/10.1007/s11368-017-1858-2 (2017).
Strukelj, M. et al. Chemical transformations in downed logs and snags of mixed boreal species during decomposition. Can. J. For. Res. 43(9), 785–798. https://doi.org/10.1139/cjfr-2013-0086 (2013).
Piaszczyk, W., Lasota, J., Gaura, G. & Błońska, E. Effect of deadwood decomposition on the restoration of soil cover in landslide areas of the Karpaty Mountains Poland. Forests 12(2), 237. https://doi.org/10.3390/f12020237 (2021).
Błońska, E., Piaszczyk, W. & Lasota, J. Emissions of CO2 from downed logs of different species and the surrounding soil in temperate forest. Ann. For. Res. 65(2), 47–56. https://doi.org/10.15287/afr.2022.2386 (2022).
Harmonn, M. E., Sexton, J. Guidelines for Measurements of Woody Detritus in Forest Ecosystems, vol. 20. VS LTER Network Office, Seattle (W.A). (1996).
Puletti, N. et al. A dataset of forest volume deadwood estimates for Europe. Ann. For. Sci. 76(3), 68. https://doi.org/10.1007/s13595-019-0832-0 (2019).
Hunter, M. L. Wildlife, Forests, and Forestry: Principles of Managing Forests for Biological Diversity 270 (Prentice and Hall, Englewood Cliffs, 1990).
Piaszczyk, W., Błońska, E. & Lasota, J. Soil biochemical properties and stabilization soil organic matter in relation to deadwood of different species. FEMS Microbiol. Ecol. 95(3), fiz011. https://doi.org/10.1093/femsec/fiz011 (2019).
Piaszczyk, W., Lasota, J. & Błońska, E. Effect of organic matter released from deadwood at different decomposition stages on physical properties of forest soil. Forests 11(1), 24. https://doi.org/10.3390/f11010024 (2020).
Lombardi, F. et al. Investigating biochemical processes to assess deadwood decay of beech and silver for in Mediterranean mountains forests. Ann. For. Sci. 70, 101–111. https://doi.org/10.1007/s13595-012-0230-3 (2013).
Allison, S. D. & Vitousek, P. M. Extracellular enzyme activities and carbon chemistry as drivers of tropical plant litter decomposition. Biotropica 36, 285–296. https://doi.org/10.1111/j.1744-7429.2004.tb00321.x (2004).
Wu, Q., Yue, K., Wang, X., Ma, Y. & Li, Y. Differential responses of litter decomposition to warming, elevated CO2, and changed precipitation regime. Plant Soil 455, 155–169. https://doi.org/10.1007/s11104-020-04675-1 (2020).
Moinet, G. Y. K. et al. Temperature sensitivity of decomposition decreases with increasing soil organic matter stability. Sci. Total Environ. 704, 135460. https://doi.org/10.1016/j.scitotenv.2019.135460 (2020).
Liu, X. et al. Soil warming delays leaf litter decomposition but exerts no effect on litter nutrient release in a subtropical natral forest over 450 days. Geoderma 427, 116139. https://doi.org/10.1016/j.geoderma.2022.116139 (2022).
Prescott, C. E. Litter decomposition: What controls it and how can we alter it to sequester more carbon in forest soils?. Biogeochemistry 101, 133–149. https://doi.org/10.1007/s10533-010-9439-0 (2010).
Berger, T. W. et al. Decomposition of beech (Fagus sylvatica) and pine (Pinus nigra) litter along on Alpine elevation gradient: Decay and nutrient release. Geoderma 251, 92–104. https://doi.org/10.1016/j.geoderma.2015.03.024 (2015).
Vitousek, P. M. & Matson, P. A. Nitrogen transformations in a range of tropical forest soils. Soil Biol. Biochem. 20(3), 361–367. https://doi.org/10.1016/0038-0717(88)90017-X (1988).
Davidson, E. A. & Janssens, I. A. Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature 440, 165–173. https://doi.org/10.1038/nature04514 (2006).
Elser, J. J. et al. Biological stoichiometry from genes to ecosystems. Ecol. Lett. https://doi.org/10.1111/j.1461-0248.2000.00185.x (2000).
Batterman, S. A., Wurzburger, N. & Hedin, L. O. Nitrogen and phosphorus interact to control tropical symbiotic N2 fixation: a test in Inga punctata. J. Ecol. 101(6), 1400–1408. https://doi.org/10.1111/1365-2745.12138 (2013).
Liu, J., Gou, X., Zheng, F., Bian, R. & Yin, D. Spatial patterns in the C:N: P stoichiometry in Qinghai spruce and the soil across the Qilian Mountains, China. CATENA 196, 104814. https://doi.org/10.1016/j.catena.2020.104814 (2021).
Chen, B. et al. C: N: P stoichiometry of plant, litter and soil along an elevational gradient in subtropical forests of China. Forests 13(3), 372. https://doi.org/10.3390/f13030372 (2022).
Ganjegunte, G. K., Condron, L. M., Clinton, P. W., Davis, M. R. & Mahieu, N. Decomposition and nutrient release from radiate pine (Pinus radiate) coarse woody debris. For. Ecol. Manage. 187, 197–211. https://doi.org/10.1016/S0378-1127(03)00332-3 (2004).
Petrillo, M. et al. Decomposition of Norway spruce and European larch coarse woody debris (CWD) in relation to different elevation and exposure in a an alpine setting. iForest 9, 154–164. https://doi.org/10.3832/ifor1591-008 (2015).
Maser, C., Anderson, R. G., Cromack, K. Jr., Williams, J. T. & Martin, R. E. Woody Material. Wildl. Habitats Managed For. Blue Mt. Oregon Wash. 553, 78 (1979).
Obrębska-Starklowa, B., Hess, M., Olecki, Z., Trepińska, J., Kowanetz, L. Klimat. In: Karpaty polskie, przyroda, człowiek i jego działalność; Warszyńska, J. (red.), Uniwersytet Jagielloński 31–47 (1995).
Piaszczyk, W., Błońska, E., Lasota, J. & Lukac, M. A comparison of C:N: P stoichiometry in soil and deadwood at an advanced decomposition stage. CATENA 179, 1–5. https://doi.org/10.1016/j.catena.2019.03.025.Doi:10.1016/j.catena.2019.03.025 (2019).
Rodzkin, A., Khroustalev, B., Kundas, S., Chernenok, E. & Krstic, B. Potential of energy Willow plantations for biological reclamation of soils polluted by 137Cs and heavy metals, and for control of nutrients leaking into water systems. Environ. Clim. Technol. 23(3), 43–56. https://doi.org/10.2478/rtuect-2019-0078 (2019).
Kahl, T., Mund, M., Bauhus, J. & Detlef, S. E. Dissolved organic carbon from European beech logs: patterns of input to and retention by surface soil. Ecoscience 19, 1–10. https://doi.org/10.2980/19-4-3501 (2012).
Zalamea, M., González, G., Ping, C. L. & Michaelson, G. Soil organic matter dynamics under decaying wood in a subtropical wet forest: effect of tree species and decay stage. Plant Soil 296, 173–185. https://doi.org/10.1007/s11104-007-9307-4 (2007).
Herrmann, S. & Bauhus, J. Nutrient retention and release in coarse woody debris of three important central European tree species and the use of NIRS to determine deadwood chemical properties. For. Ecosyst. 5, 22. https://doi.org/10.1186/s40663-018-0140-4 (2018).
Lindahl, B. D. & Finlay, R. D. Activities of chitinolytic enzymes during primary and secondary colonization of wood by basidiomycetous fungi. New Phytol. 169, 389–397. https://doi.org/10.1111/j.1469-8137.2005.01581.x (2006).
Palviainen, M. et al. Carbon and nitrogen release from decomposing Scots pine, Norway spruce and silver birch stumps. For. Ecol. Manage. 259, 390–398. https://doi.org/10.1016/j.foreco.2009.10.034 (2010).
Tláskal, V. et al. Complementary roles of wood-inhabiting fungi and bacteria facilitate deadwood decomposition. mSystems 6(1), e0107820. https://doi.org/10.1128/mSystems.01078-20 (2021).
Tláskal, V. & Baldrian, P. Deadwood-inhabiting bacteria show adaptations to changing carbon and nitrogen availability during decomposition. Front. Microbiol. 12, 685303. https://doi.org/10.3389/fmicb.2021.685303 (2021).
Rinne-Garmston, K. T. et al. Carbon flux from decomposing wood and its dependency on temperature, wood N2 fixation rate, moisture and fungal composition in a Norway spruce forest. Glob. Change Biol. 25, 1852–1867. https://doi.org/10.1111/gcb.14594 (2019).
Błońska, E., Lasota, J. & Piaszczyk, W. Dissolved carbon and nitrogen release from deadwood of different tree species in various stages of decomposition. Soil Sci. Plant Nutr. 65(1), 100–107. https://doi.org/10.1080/00380768.2018.1545517 (2019).
Shannon, V. L., Vanguelova, E. I., Morison, J. I. L., Shaw, L. J. & Clark, J. M. The contribution of deadwood to soil carbon dynamics in contrasting temperate forest ecosystems. Eur. J. For. Res. 141, 241–252. https://doi.org/10.1007/s10342-021-01435-3 (2022).
Bueis, T., Bravo, F., Pando, V., Kissi, Y. A. & Turrión, M. B. Phosphorus availability in relation to soil properties and forest productivity in Pinus sylvestris L. plantations. Ann. For. Sci. 76, 1–13. https://doi.org/10.1007/s13595-019-0882-3 (2019).
Oliverio, A. M. et al. The role of phosphorus limitation in shaping soil bacterial communities and their metabolic capabilities. MBio 11, e01718-e1720. https://doi.org/10.1128/mBio.01718-20 (2020).
Treseder, K. K. & Vitousek, P. M. Effects of soil nutrient availability on investment in acquisition of N and P in Hawaiian rain forests. Ecology 82(4), 946–954. https://doi.org/10.1890/0012-9658(2001)082[0946:EOSNAO]2.0.CO;2 (2001).
Wallenstein, M. et al. Controls on the temperature sensitivity of soil enzymes: a key driver of in situ enzyme activity rates. In (eds. Shukla, G, Varma, A.) Soil Enzymology 245–258 (Springer, Berlin, 2010) https://doi.org/10.1007/978-3-642-14225-3_13
Allison, S. D. et al. Low levels of nitrogen addition stimulate decomposition by boreal forest fungi. Soil Biol. Biochem. 41, 293–302. https://doi.org/10.1016/j.soilbio.2008.10.032 (2009).
Allison, S. D. et al. Microbial abundance and composition influence litter decomposition response to environmental change. Ecology 94, 714–725. https://doi.org/10.1890/12-1243.1 (2013).
Bütler, R., Patty, L., Le Bayon, R. C., Guenat, C. & Schlaepfer, R. Log decay of Picea abies in the Swiss Jura Mountains of central Europe. For. Ecol. Manage. 242, 791–799. https://doi.org/10.1016/j.foreco.2007.02.017 (2007).
Durło, G. B. Forest vegetation period in the Beskid Śląski Mountains. Sylwan 154(8), 577–584. https://doi.org/10.26202/sylwan.2009252 (2010).
Shaw, A. N. & Cleveland, C. C. The effects of temperature on soil phosphorus availability and phosphatase enzyme activities: A cross-ecosystem study from the tropics to the Arctic. Biogeochemistry 151, 113–125. https://doi.org/10.1007/s10533-020-00710-6 (2020).
Acknowledgements
The research was financed by the National Science Centre, Poland: decision no. DEC 2020/39/B/NZ9/00372. In these studies, no material was collected from endangered animal and plant species. The conducted studies were in accordance with the relevant national guidelines and legislation. Consents for research have been obtained from the Ministry of the Environment and the National Park.
Author information
Authors and Affiliations
Contributions
E.B., J.L. conceived the study and obtained funding for this research. W.P. performed sample processing and provided samples for this study. E.B., W.P., J.L. developed the research questions, analysed the data and drafted the manuscript. All authors contributed substantially to writing and revisions.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Błońska, E., Piaszczyk, W. & Lasota, J. Patterns and driving factors of ecological stoichiometry in system of deadwood and soil in mountains forest ecosystem. Sci Rep 13, 5676 (2023). https://doi.org/10.1038/s41598-023-32946-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-32946-1
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.