Abstract
Macroalgae is an under-utilised tool as a bioindicator of anthropogenic nitrogen loading to the coastal environment in the UK. This study compared two island systems—Jersey (Channel Islands) and St Mary’s (Isles of Scilly) to assess how differing sewerage infrastructure affects nitrogen loading. A total of 831 macroalgae samples of Fucus vesiculosus and Ulva sp. were analysed for nitrogen isotopes (δ15N). Elevated δ15N values were recorded for Jersey (> 9‰) in St Aubin’s Bay—caused by the outflow of the Bellozanne Sewerage Treatment Works (STW). δ15N isoplots maps indicate low diffusion of nitrogen out of St Aubin’s Bay. St Mary’s produced a varied δ15N isoplot map in comparison. δ15N was typically lower and is attributed to a smaller population and inefficient STW. Outflow of sewage/effluent at Morning Point, Hugh Town and Old Town produced elevated δ15N values in comparison to the island average. St Mary’s inefficient sewerage treatment and reliance on septic tanks/soakaways complicates δ15N interpretation although it still indicates that nitrogen pollution is an island-wide issue. Future sewerage development and upgrades on islands are required to prevent similar effluent environmental issues as recorded in St Aubin’s Bay. This study advocates the use of macroalgae as a bioindicator of nitrogen effluent in the marine environment.
Similar content being viewed by others
Introduction
Anthropogenic activity has altered the modern nitrogen cycle primarily through the increased influx of sewage to marine environments1. It has been estimated that biologically available nitrogen to the ocean has doubled in rate between 1960 and 1990 and hence, is no longer a limiting factor on marine ecosystem productivity2,3,4. Sewage outfalls have been linked to multiple instances of eutrophication, whereby enhanced nutrient uptake by algae results in blooms and hypoxia in the marine environment5. Monitoring sewage pollution extent, severity and point source is critical to minimise the risk to public and environmental health1,5.
Traditional nitrogen isotope monitoring techniques rely on analysing the dissolved inorganic nitrogen content of seawater, which is time-consuming and expensive in comparison to nitrogen isotope analysis of macroalgae6. Nitrogen isotope ratios (expressed as δ15N) from macroalgae have been shown to accurately record the nitrogen isotope composition of seawater1,7. In fact, nitrogen uptake in macroalgae has successfully traced the influence of sewage/effluent up to 24 km from a point source3. The ability to distinguish nitrogen pollution sources come from the premise that sewage/effluent δ15N exhibits more elevated values compared to chemical industry sources7,8. This is primarily due to physical, chemical, and biological processes (denitrification9) that occur during the treatment of sewage at wastewater treatment plants. The process of denitrification preferentially removes 14N from the effluent, hence producing a discharge with more positive δ15N values > + 8‰3,7,10. When sewage/effluent is released without any denitrification processes the δ15N will reflect the source (e.g., artificial fertiliser, animal/human sewage; see below for discussion)8. Typically, coastal environments that are affected by sewage range between + 4‰ to + 19‰8,11.
Savage and Elmgren12 recommend that values below ~ + 4 ‰ reflect artificially produced nitrate that uses atmospheric nitrogen (~ 0‰) as its nitrogen source. Most agricultural fertilisers use artificially produced products and thus, typically exhibit low or negative δ15N values7. Therefore, macroalgae in a coastal environment dominantly affected through fertilizer runoff will exhibit near zero or negative δ15N values. When animal and/or human sewage/effluent reaches the coastal environment unprocessed (i.e., raw) or processed (denitrification), the δ15N value of coastal macroalgae will be elevated (since herbivore/human δ15N values for the British Isles range between + 4‰ to + 8‰13) in comparison to artificial fertilizers and/or background marine signatures (e.g., artificial fertilizers < + 4‰, unpolluted/background + 4‰ to + 6‰, sewage/effluent/manure polluted = > + 6‰). However, coastal and/or estuarine environments influenced by wastewater treatment plants will exhibit much higher δ15N values (e.g., > + 10‰3) in comparison to untreated sewage (e.g., > + 6‰). Large-scale, coastal macroalgae δ15N isoplot maps will help differentiate and determine point sources of pollution, which can then influence societal practices and policies in that region14.
The macroalgae, Fucus vesiculosus (bladder wrack) and Ulva sp. are commonly used in nitrogen isotope studies of the coastal environment15,16. Ulva sp. are more opportunistic and will bloom when concentrations of nitrogen in the water column become elevated17. Both macroalgae are found around the coastlines of Europe, with F. vesiculosus commonly found in the intertidal zone of rocky shores, while Ulva sp. prefers sheltered habitats18,19. Dissolved inorganic nitrogen is incorporated within 13–19 days in F. vesiculosus, whereas in Ulva sp. it has been reported to be within as little as 48 h6,11,16,20. Although the use of nitrogen isotopes in macroalgae is cheaper and quicker, it is not widely used15 and there are only a handful of studies on UK coastlines6,11,15. This is surprising considering the exponential increase of unregulated release of sewage/effluent into UK rivers since leaving the European Union (i.e., Brexit)21.
In this study, we performed nitrogen isotope analysis of F. vesiculosus and Ulva sp. (simply referred to as Fucus and Ulva hereafter) from two contrasting island environments in terms of wastewater treatment processes: Jersey has a centralised wastewater treatment facility that is currently being upgraded, and St Mary’s (Isles of Scilly) has no major wastewater treatment facility but relies on continuous outflow, septic tanks and soakaway systems.
Study sites
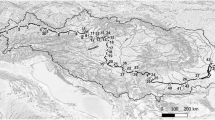
Jersey is the largest of the Channel Islands located 14 miles off the coast of Normandy, France. As of 2019 the population was 106,800 increasing by ~ 1200 persons per year, and the majority of the population resides in St Helier and St Aubin’s Bay (Fig. 1A)22. Jersey relies solely on the Bellozanne Sewerage Treatment Work (STW) (Fig. 1A) for all sewage treatment on the island. Prior to its commission in 1959, untreated sewage was discharged directly into the sea. The STW was built for a population of 57,000 and so continuous improvements have been required as the population has grown and environmental standards have changed23. Despite continued improvements, surveys have shown that St Aubin’s Bay continues to exhibit high levels of nutrient loading from effluent24,25. Trophic status reports carried out by the Centre for Research into Environmental Health (CREH) in 1997 suggest winter hyper-nutrification in St Aubin’s Bay; despite the predicted 2% to 21% reduction in chlorophyll concentration from nutrient removal by the Bellozanne STW25. A 2010 reassessment shows considerable reduction in nitrogen load compared to 1997 values, and modelling indicates no potential eutrophication in the bay26. However, the STW has continued to fail the Total Nitrogen limit set at 10 mg/l, instead recording between 11 and 63 mg/l with an average of 31.3 mg/l between 2009 and 201527,28. No further upgrades are possible for the Bellozanne STW and it cannot support the Jersey population as it currently stands. Sewage effluent entering the bay is causing increased nitrogen loading that is resulting in substantial Ulva growth and eutrophication in St Aubin’s Bay27,29; the Jersey Government report that Ulva growth in St Aubin’s Bay will always persist29. The construction of a new STW at Bellozanne began in 2018 and will be operational in 2023 and aims to reduce discharge of partially treated sewage by 97%30. The new STW will support a population size of 118,000 and aims to also reduce nitrogen loading to St Aubin’s Bay by an additional 10–15% compared to the current STW outflow.
Map of the islands investigated in this study: (A) Jersey, Channel Islands; (B) St Mary’s, Isles of Scilly. Sample locations are indicated by numbered grey circles. Key locations discussed in text are also indicated. SAB St Aubin’s Bay, SMP St Mary’s Pool, PC Porth Cressa, OTB Old Town Bay. Note different scales for each island.
The Isles of Scilly (IOS) are a group of islands situated 27 miles southwest off the coast of the UK (Fig. 1B). The islands sit at the end of the North Atlantic Current and the Gulf Stream producing a milder climate compared to mainland UK, with winter surface temperatures of ~ 10 °C31. Two tidal jets transport water around the isles, the primary operates in a clockwise direction transporting water south, the secondary current transports water northwards32. St Mary’s has an area of 6 square miles and is the largest island in the IOS. The permanent population is ~ 1800, although this increases to > 6000 in summer months33. Most of the island’s population lives in the southwest in Hugh Town (Fig. 1B). The northwest supports small-scale agriculture (6–7 hectares) with no intensive agriculture, and as such, the coastal waters are generally classified as pristine34. There is no wastewater treatment facility on St Mary’s—instead the island relies on several smaller wastewater infrastructures. There is a 3 km sewerage network in Hugh Town supporting ~ 800 properties and connects to the Morning Point outflow (Fig. 1B). Morning Point is the only permitted outflow on the IOS where untreated raw sewage is discharged into the Atlantic35. Two emergency outflows intermittently discharge sewage into the Hugh Town harbour in the case of flood events (Fig. 1B)35. Old Town is serviced by a combination of a Bio-Bubble facility (i.e., an advanced aeration system, https://www.bio-bubble.com) connected a soak-away draining to Old Town Bay, the Morning Point network, and three septic tanks33,35. Septic tanks serve as the main sewage treatment process for properties outside of Hugh Town and Old Town. The Environment Agency does not provide a full list of all septic tanks on the island; thus, it must be noted that more septic tanks than those presented in Fig. 1B are likely to be in operation36.
The private company South West Water (SWW) has taken over responsibility for sewage treatment on the IOS from the local council. SWW aim to reduce sewage pollution by 2030 and and report that St Mary’s sewerage network is in need of major repair35,37. Poor record keeping by the IOS council has resulted in a lack of information regarding sewerage leaks and septic tank conditions across the island35. By 2030, SWW aims to build a resilient treatment facility and upgrade the current Bio-Bubble37. Initial plans by SWW35 considered closing the Morning Point outflow, with discharge redirected to the emergency outflow into St Mary’s Pool (Fig. 1B). The emergency outflow pipeline would be upgraded, and households encouraged to connect to the sewerage network and thus, reducing the reliance on septic tanks37.
Materials and methods
Macroalgae from Jersey was collected during December 2020 and January 2021 by one of us (LPW) during the Covid pandemic. 372 samples were collected from 47 sites around Jersey (Fig. 1A) consisting of 336 Fucus and 36 Ulva samples. Macroalgae from the IOS was collected by one of us (NB) during the same time period. 459 macroalgae samples were collected from 62 sites (Fig. 1B) consisting of 429 Fucus and 30 Ulva samples. Sample sites from both locations were selected primarily on the presence of macroalgae and ease of accessibility for collecting. The outermost non-fertile tip of Fucus was sampled from several different specimens, whereas a 4 cm-square area of Ulva was sampled and compressed to remove all seawater. Each sample was placed in a small brown envelope, labelled and subsequently dried in their envelopes soon after collection in a domestic oven set between 45 and 60 °C. Sub-samples of compressed Ulva and the margin of the non-fertile tip of Fucus were weighed into tin capsules for subsequent stable isotope analysis following the protocols outlined in Gröcke et al.6 and Bailes and Gröcke11. QGIS software was used to produce the isoplot maps of δ15N around Jersey and St Mary’s.
Results
The average δ15N value from Jersey for Fucus is + 7.0‰ ± 1.9‰ (n = 336) and + 7.1‰ ± 1.3‰ (n = 36) for Ulva, which are above natural background levels reported between + 4‰ and + 6‰8,12. δ15N values of Fucus range between + 2.9‰ (Sites 1 and 13) and + 12.1‰ (Site 36) showing elevated δ15N in St Aubin’s Bay for both Fucus and Ulva (Fig. 2A). Site 40 recorded the greatest elevation in δ15N for Fucus (+ 11.0‰ ± 0.6‰, n = 7) and the five most elevated δ15N (> + 9.0‰) sites (33, 34, 36, 38 and 40) are found in St Aubin’s Bay. Ulva recorded a δ15N range between + 4.3‰ and + 9.8‰ (Fig. 2A). Lower average δ15N values were recorded in Fucus for the north coastline (+ 5.1‰ ± 1.3‰, n = 57), the west coastline (+ 5.7‰ ± 1.4‰, n = 78) and for the east coastline (+ 6.5‰ ± 1.2‰, n = 69) in comparison to the southern coastline (+ 8.3‰ ± 1.8‰, n = 160). Although there are less data for Ulva it recorded higher δ15N average values for each coastline compared to Fucus for the north, east and west coastlines (+ 5.6‰ ± 1.1‰, n = 7; + 6.7‰ ± 0.5‰, n = 10; and + 7.1‰ ± 0.6‰, n = 6, respectively). However, the south coast showed a depleted δ15N average (+ 7.8‰ ± 1.4‰, n = 14) compared to Fucus. Macroalgae in St Aubin’s Bay produced a significantly more elevated δ15N signature in comparison to the whole island average; this is observed in both Fucus (+ 9.5‰ ± 1.1‰, n = 85) and Ulva sp. (+ 8.3‰ ± 1.2‰, n = 8) (with a p value < 0.05; see Supplementary Fig. 1).
Box and whiskers plots of macroalgae δ15N from Jersey (A) and St Mary’s (B). All numbered sites represent Fucus results. All Ulva results for the islands have been grouped as one dataset and are presented on the farthest right of each graph. δ15N values above the dashed red line represent coastal environments that are affected by sewage/effluent. Macroalgae δ15N elevation is recorded for St Aubin’s Bay (Sites 30–43) linked to the Bellozanne STW outflow.
From the IOS, Fucus recorded an average δ15N value of + 5.8‰ ± 1.2‰ (n = 429), and Ulva recorded an average δ15N value of + 6.2‰ ± 0.7‰ (n = 30). IOS seagrass has an average δ15N value of ~ + 5‰ which is on the low side of our dataset38. Fucus ranged between + 2.2‰ (Site 25) and + 9.0‰ (Site 41), whereas Ulva varied between + 4.1‰ and + 7.7‰ (Fig. 2B). δ15N values are slightly skewed towards more elevated δ15N values along the southern (+ 5.9‰ ± 1.4‰, n = 99), western (+ 5.8‰ ± 1.2‰, n = 99) and northern coastlines (+ 5.8‰ ± 1.0‰, n = 140) compared to the eastern coastline (+ 5.5‰ ± 1.3‰, n = 90). Site 46 recorded the most elevated average δ15N value from the IOS (+ 7.7‰ ± 0.7‰, n = 10), which is located at Porth Cressa. The three other most elevated δ15N (> 7.0‰) sites are also in the southwest of the island (Sites 40, 41, 51) (Fig. 2B). Average δ15N values decrease in distance away from Morning Point (see Supplementary Fig. 2). No correlation exists between site average δ15N values and proximity to septic tanks (see Supplementary Figs. 3 and 4). Sites 30 and 56 recorded the lowest δ15N average value for Fucus (+ 3.8‰ ± 0.4‰, n = 10 and + 3.8‰ ± 0.8‰, n = 10, respectively). Ulva was found to have the lowest δ15N values along the east coastline (+ 5.6‰ ± 0.4‰), which is in line with results from Fucus.
Discussion
Jersey: focused point-source effluent pollution
The δ15N values from macroalgae around the Jersey coast demonstrate a clear geospatial pattern with more elevated δ15N values in St Aubin’s Bay compared to the remainder of the island (Fig. 3A). Multiple elevated δ15N values > + 6.0‰ indicate nitrogen loading from an anthropogenic effluent source1,12,39. The position of the Bellozanne STW corresponds with these elevated δ15N sites (Fig. 3A for Fucus and Fig. 3B for Ulva). Such elevated δ15N values are in line with previous studies of macroalgae near STW1,39. δ15N of groundwater sampled from Jersey in 1995 also show elevated values (+ 11.8‰ to + 18.4‰ in the St Aubin’s Bay coastal margin40. These elevated δ15N values correspond to the lowest nitrate concentrations in the groundwater and are interpreted as a result of denitrification processes in the deep groundwater system. Quantitative information on the mixing zone between groundwater and seawater is not available for St Aubin’s Bay and thus, we are uncertain of the influence of deep groundwater δ15N on macroalgae δ15N. Although data on Total Dissolved Solids for Jersey indicate that saline intrusion occurs around St Aubin’s Bay and other low-lying regions, and thus would imply little influence from deep groundwater41.
δ15N isoplot map for the macroalgae, Fucus (A) and Ulva (B) around Jersey, Channel Islands. Colour range represent distinct δ15N value ranges, while the size of the circle represents the standard deviation of δ15N from each site. Elevated macroalgae δ15N values are recorded for St Aubin’s Bay (Sites 30–43) and is associated to the Bellozanne STW outflow. Normal background macroalgae δ15N values are located on the west and north coastlines (blue circles). For abbreviations refer to Fig. 1.
The lack of elevated δ15N values elsewhere around Jersey strongly suggests that the release of sewage effluent nitrogen in St Aubin’s Bay is not transported around the island through oceanic currents. St Aubin’s Bay has a large tidal range (on the order of 10 m) and is sheltered by the westerlies and Atlantic swell, thus causing reduced mixing with the open ocean. Limited water exchange with the open ocean reduces the capacity for effluent diffusion as well as reducing δ15N via ammonia volatisation3,42,43. Macroalgae δ15N data from Jersey shows that the geographic position of STWs and the placement of outflow pipes require a thorough environmental assessment, considering—at least—prevailing wind and oceanic and tidal currents prior to any planning and development. If such investigations were conducted in Jersey, St Aubin’s Bay may not be the environmental disaster it has become44,45. The north, east and west coastlines of Jersey reflect δ15N values typical of natural background levels, suggesting minimal anthropogenic nitrogen sources (effluent, fertilizer, industrial sources) (Fig. 3A) and/or that oceanic currents, tidal ranges and bay openness are dissipating any nitrogen loading into the open ocean.
St Mary’s: dispersed point-source effluent pollution
In comparison to Jersey, St Mary’s in the IOS exhibits more complex nitrogen loading and geospatial variation in δ15N around the coastline. The main difference causing this complexity is the difference in sewerage treatment approaches between the islands: a single, principal sewage treatment plant versus a spatial array of septic tanks and two isolated sewerage systems in disrepair around the island. Several sites around St Mary’s coastline record elevated δ15N values in comparison to normal, healthy marine values: Sites 41, 46 and 51 are located in three separate bays (Fig. 4A). Nitrogen pollution point sources on St Mary’s can be separated into three main infrastructures: Morning Point discharge, septic tanks/soakaways and the Hugh Town emergency outflow. Figure 4A reveals that the two most elevated δ15N sites correspond to Morning Point discharge and the Hugh Town emergency outflow. Site 46 has the most elevated δ15N values on the island and is the closest sampled point to the sewage outflow at Morning Point, discharging untreated and semi-treated sewage into Porth Cressa (Fig. 4A,B)33. A steady decline in δ15N values is observed across a 160 m profile at Porth Cressa, indicating diffusion of nitrogen loading from the Morning Point source into the bay: even with limited data this pattern is also reflected in Ulva (see Supplementary Fig. 2). A lowering of macroalgae δ15N away from a source outflow would also suggest that Porth Cressa is not well-mixed, allowing the opportunity for the effluent to be absorbed by the local environment (e.g., macroalgae, microalgae, microflora, sediment). The relationship between decreasing δ15N and distance from a nitrogen source has been recorded in other macroalgae investigations1,12,20.
δ15N isoplot map for the macroalgae, Fucus (A) and Ulva (B) around St Mary’s, Isles of Scilly. Colour range represent distinct δ15N value ranges, while the size of the circle represents the standard deviation of δ15N from each site. In comparison to Jersey, macroalgae δ15N values are not as elevated (e.g., with dark orange or red circles) on St Mary’s: most likely related to the lower population size, release of untreated sewage/effluent and/or the islands reliance on a septic tank/soakaway system. Elevated macroalgae δ15N values are recorded for Morning Point, Hugh Town and Old Town regions. For abbreviations refer to Fig. 1.
A significant proportion of the population (residential and tourist) on St Mary’s rely on septic tanks and soakaways as a method for sewage processing, storage and release. As a result of sewage storage in septic tanks, the septic tank environment would start with nitrification processes (e.g., ammonia oxidisation to nitrate) in an oxygenated empty tank and then shift to anaerobic denitrification once oxygen becomes depleted; at that point denitrification would further elevate the δ15N signature of the sludge and effluent46,47. Reduced regulation and monitoring of septic tanks will lead to their disrepair, allowing them to leak nutrients (nitrogen and phosphorus) into the groundwater system and eventually to the coastal environment.
Old Town has a soakaway delivering effluent from the Bio-Bubble to Old Town Bay. Although this effluent may have slightly elevated δ15N (e.g., animal/human signature13), the aeration process (i.e., nitrification processes) would not elevate it further. Site 41 at Old Town Bay has the highest individual δ15N value (+ 9.0‰) for the entire island. Therefore, elevated δ15N at Old Town Bay may be attributed instead to the three septic tanks located nearby (see Fig. 4). Sites around St Mary’s that are located near septic tanks exhibit large standard deviations (> 0.7‰), but there is no trend with distance from the source (Sites 39–41, 56–64) (see Supplementary Figs. 3 and 4). Although legislation on St Mary’s does not allow for the discharge of effluent from septic tanks the maintenance of these tanks is not regulated: septic tanks are meant to be emptied and the contents transported to the mainland of England for processing33,48—although it is known by one of us (NB) that emptied tanks are not being transported back to the mainland. Farmers may be managing their own septic tank systems, such as those on Bryher, IOS21. In addition, many septic tanks on the island have soak-aways that may have some degree of contact with oxygen causing nitrification processes to occur (e.g., lowering δ15N values)12. Elevated δ15N values recorded at the above-named sites may be caused from poorly maintained septic tank systems or from groundwater denitrification processes at depth producing less distinct source point pollution areas with elevated δ15N signatures40,47,48.
Another issue that complicates the interpretation of macroalgae δ15N around St Mary’s is the geographic position of the island and surrounding islands, the structure of the coastline and ocean circulation/currents. Viana and Bode43 showed that open coastal waters along the coast of Spain recorded lower δ15N values in macroalgae compared to enclosed bays. For example, elevated δ15N values at Sites 31–35 for Fucus and Ulva are potentially caused by the shape of the bay restricting water movement, causing 15N enrichment. There are no obvious sewage/effluent sources to the eastern coastline. A direct comparison between our macroalgae δ15N data with IOS seagrass δ15N data38 is not possible since latitudinal information was not provided for the seagrass data. However, the seagrass δ15N data is typically < + 5‰, suggesting that the nitrogen isotope signature of effluent is not reaching the seagrass community further out from the coastline. Sewage nitrogen trapped in bays has a longer time to become isotopically fractionated, and therefore could lead to an elevated δ15N signature that is subsequently incorporated into macroalgae3. The enclosed nature of Porth Cressa, may also be preserving and/or enhancing the elevated δ15N values in macroalgae at this site. Another complicating factor is the clockwise movement of oceanic water around the IOS, which has the potential for transporting effluent-contaminated waters away from the north-west coastline producing less elevated macroalgae δ15N values on the isoplot map32.
To treat or not to treat: effluent pollution in island environments
As shown in this study, a single, operational wastewater treatment plant for an entire island has the potential to concentrate effluent release to a single point/area, resulting in elevated coastal macroalgae δ15N values. The principal issue in Jersey is that the outflow of the Bellazonne STW is directed solely into St Aubin’s Bay. This causes elevated nitrogen loading in the bay which has a direct impact on the health of the ecosystem. The nature of the oceanic conditions in the region and the fact that coastal bays are natural traps means that nitrogen loading is intensified, causing significant environmental issues—for example, green tides, high levels of nitrogenous compounds and obvious water discolouration44,45,49,50. In direct contrast, St Mary’s has no centralised wastewater treatment plant and therefore effluent is either/or; (1) directly released into the ocean with no and/or limited treatment, and unregulated emptying of septic tanks; and/or (2) an antiquated, sewerage system in need of significant investment to upgrade it. The lack of adequate wastewater treatment on St Mary’s may be the cause behind lower macroalgae δ15N values compared to Jersey. Although the δ15N values may be lower, indicating less denitrification, this does not equate to less nitrogen loading in the coastal environment around St Mary’s. In fact, groundwater nitrate concentrations from 1995 on St Mary’s averaged 58 mg/l51, which exceeds the World Health Organisation guideline value: no current nitrate or Total Nitrogen is publicly available for St Mary’s. Irrespectively, nitrogen chemical analyses (i.e., nitrogen pollution) would need to be monitored around the entire island of St Mary’s, compared to Jersey which could focus on the one region (i.e., St Aubin’s Bay), to monitor effluent contamination. Therefore, despite the significantly smaller population size on St Mary’s nitrogen loading may equally be having an impact on the coastal environment and ecosystems (e.g., on eelgrass, Zostera marina, populations and genetic diversity around the IOS52).
In the future, wastewater treatment plants require more thorough development and planning on islands, specifically in terms of a more comprehensive environmental assessment that considers oceanic circulation, modelling the effect of swell, geography and bathymetry of the coastline, tidal ranges, and any other ocean–atmosphere environmental conditions; this must all occur prior to placement of an outflow pipe. St Aubin’s Bay clearly demonstrates a lack of foresight when positioning the outflow, since it is now causing environmental issues in that bay all-year round27,28,29,44,45. The Morning Point outflow on St Mary’s is also positioned incorrectly, as evidenced by the elevated δ15N values at Porth Cressa: this beach also encounters problems associated with abundant macroalgae and Ulva sp. reflecting nitrogen-rich waters. A more confusing geospatial pattern in macroalgae δ15N values around St Mary’s is thought to be caused by the island’s reliance on managed and unregulated septic tanks, and an inadequate sewerage system.
Conclusions
Nitrogen isotopes of macroalgae around island coastlines can offer valuable insight into sewerage practices and effluent pollution affecting the region. This study shows that Jersey, which relies on an inadequate STW facility is unable to cope with current and projected population demands. The effluent from this STW facility drains into St Aubin’s Bay which has led to nitrogen overloading and its current environmental issue: blooms of opportunistic macroalgae smothering the beaches of that bay (e.g., Ulva sp.). Other regions around Jersey do not show significant elevation in macroalgae δ15N indicating a lack of effluent pollution and therefore, nitrogen loading. This may be the result of oceanic currents, geography and bathymetry of the coastline and/or simply a lack of effluent release to those coastline regions. On the other hand, St Mary’s, IOS, records a complex pattern resulting from an array of unmonitored sewage/effluent sources from all over the island and not from a single source, into a single region (e.g., St Aubin’s Bay, Jersey). Future planning and development of any wastewater treatment plant on St Mary’s requires a thorough investigation of the marine system, especially oceanic currents, tidal effects, marine chemistry, and ecological assessments as standard practice. Case studies on the δ15N value of macroalgae from other islands should be conducted to assess the influence and impact of effluent discharge. The use of macroalgae δ15N can help identify point source effluent discharges and thus, impact policy changes to mitigate current and future environmental problems/disasters along our coastlines. This study shows that islands are not immune to the environmental issues caused by effluent pollution. This idea was aptly conveyed by John W. Gardner53, p. 108 when he wrote, “we cannot have islands of excellence in a sea of slovenly indifference to standards”.
Data availability
All data generated or analysed during this study are included in this published article [and its supplementary information files].
References
Costanzo, S. D., Oõdonohue, M. J., Dennison, W. C., Loneraganà, N. R. & Thomas, M. A new approach for detecting and mapping sewage impacts. Mar. Pollut. Bull. 42, 149–156 (2001).
Howarth, R. W. Coastal nitrogen pollution: A review of sources and trends globally and regionally. Harmful Algae 8, 14–20 (2008).
Savage, C. Tracing the influence of sewage nitrogen in a coastal ecosystem using stable nitrogen isotopes. AMIBO J. Hum. Environ. 34, 145–150 (2005).
Vitousek, P. M. et al. Human alteration of the global nitrogen cycle: Sources and consequences. Ecol. Appl. 7, 737–750 (1997).
Howarth, R. et al. Coupled biogeochemical cycles: Eutrophication and hypoxia in temperate estuaries and coastal marine ecosystems. Front. Ecol. Environ. 9, 18–26 (2011).
Gröcke, D. R., Racionero-Gómez, B., Marschalek, J. W. & Greenwell, H. C. Translocation of isotopically distinct macroalgae: A route to low-cost biomonitoring?. Chemosphere 184, 1175–1185 (2017).
Heaton, T. H. E. Isotopic studies of nitrogen pollution in the hydrosphere and atmosphere: A review. Chem. Geol. (Isot. Geosci. Sect.) 59, 87–102 (1986).
Xue, D. et al. Present limitations and future prospects of stable isotope methods for nitrate source identification in surface- and groundwater. Water Res. 43, 1159–1170 (2009).
Lee, H. S. & Liao, B. Anaerobic membrane bioreactors for wastewater treatment: Challenges and opportunities. Water Environ. Res. 93, 993–1004 (2021).
Lemesle, S., Erraud, A., Mussio, I., Rusig, A. M. & Claquin, P. Dynamics of δ15N isotopic signatures of different intertidal macroalgal species: Assessment of bioindicators of N sources in coastal areas. Mar. Pollut. Bull. 110, 470–483 (2016).
Bailes, I. R. & Gröcke, D. R. Isotopically labelled macroalgae: A new method for determining sources of excess nitrogen pollution. Rapid Commun. Mass Spectrom. 34, e8951 (2020).
Savage, C. & Elmgren, R. Macroalgal (Fucus vesiculosus) δ15N values trace decrease in sewage influence. Ecol. Appl. 14, 517–526 (2004).
Bird, M. I., Haig, J., Ulm, S. & Wurster, C. A carbon and nitrogen isotope perspective on ancient human diet in the British Isles. J. Archaeol. Sci. 137, 105516 (2022).
Howarth, L. M. et al. Using macroalgal bioindicators to map nutrient plumes from fish farms and other sources at a bay-wide scale. Aquac. Environ. Interact. 11, 671–684 (2019).
García-Seoane, R., Fernández, J. A., Villares, R. & Aboal, J. R. Use of macroalgae to biomonitor pollutants in coastal waters: Optimization of the methodology. Ecol. Indic. 84, 710–726 (2018).
Viana, I. G., Bode, A., Bartholomew, M. & Valiela, I. Experimental assessment of the macroalgae Ascophyllum nodosum and Fucus vesiculosus for monitoring N sources at different time-scales using stable isotope composition. J. Exp. Mar. Biol. Ecol. 466, 24–33 (2015).
Roleda, M. Y. & Hurd, C. L. Seaweed nutrient physiology: Application of concepts to aquaculture and bioremediation. Phycologia 58, 552–562 (2019).
Kapkov, V., Shoshina, E. & Belenikina, O. Ecological characteristic of brown algae communities—Potential aquaculture objects. In International Applied Research Conference: Biological Resources Development and Environmental Management (KnE Life Sciences, 2020). https://doi.org/10.18502/kls.v5i1.6068.
Pizzolla, P. Ulva lactuca: Sea lettuce. Tyler-Walters and Hiscock K. Marine Life Information Network: Biology and Sensitivity Key Information Reviews. Plymouth: Marine Biological Association of the United Kingdom https://www.marlin.ac.uk/species/detail/1467 (2008).
Orlandi, L. et al. The effects of nitrogen pollutants on the isotopic signal (δ15N) of Ulva lactuca: Microcosm experiments. Mar. Pollut. Bull. 115, 429–435 (2017).
House of Lords. Sewage Disposal in Rivers and Coastal Waters. Hansard vol. 823. https://hansard.parliament.uk/lords/2022-07-07/debates/D682D219-EF91-413E-B9C9-03274416B890/SewageDisposalInRiversAndCoastalWaters (2022).
Statistics Jersey. Jersey Resident Population 2019 Estimate. (2019).
Transport and Technical Services Department. Jersey’s Waste Water Strategy. (2013).
Stapleton, C. M., Kay, D., Jackson, G. F. & Wyer, M. D. Estimated inorganic nutrient inputs to the coastal waters of Jersey from catchment and waste water sources. Water Resour. 34, 787–796 (1999).
Centre for Research into Environment and Health. Trophic Status of St Aubin’s Bay. Jersey. (1997).
Centre for Research into Environment and Health. Reassessment of the trophic status of St Aubin’s Bay, Jersey. (2010).
WCA Environment Limited. The environmental status of St Aubin’s Bay, Jersey according to the requirements of the water framework directive. Data Management and Assessment for Monitoring Programmes: Monitoring Programme Results and Status Assessments (2015).
Government of Jersey. St Aubin’s Bay and nitrate reports (FOI). https://www.gov.je/government/freedomofinformation/pages/foi.aspx?ReportID=991 (2016).
Jersey Evening Post. Green bay is here to stay, but tech will help ‘limit nuisance weed’, says minister. Jersey Evening Post (2022).
Government of Jersey. Bellozanne Sewage Treatment Works Replacement. (2018).
Petzold, J. Social adaptability in ecotones: Sea-level rise and climate change adaptation in flushing and the Isles of Scilly, UK. Island Stud. J. 13, 101–118 (2018).
Pingree, R. D. & Mardell, G. T. Coastal tidal jets and tidal fringe development around the Isles of Scilly. Estuar. Coast Shelf Sci. 23, 581–594 (1986).
Council of the Isles of Scilly. Draft Isles of Scilly Local Plan 2015–2030 Including Minerals and Waste. https://www.scilly.gov.uk/sites/default/files/planning-apps/Infrastructure%20Capacity%20Assessment%20Topic%20Paper%202018.pdf (2018).
Farr, H. & Rogers, A. Tourism and the environment on the Isles of Scilly: conflict and complementarity. Landsc. Urban Plan. 29, 1–17 (1994).
South West Water. Isles of Scilly Business Plan. (2019).
Environment Agency. Sewage-related contaminants in Isles of Scilly groundwater: Concentrations, spatial distribution, sources, and risk. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/938382/Withdrawn_Final_IOS_report_V2_-_2_Part1.pdf (2016).
South West Water. Our emerging drainage and wastewater management plan: Isles of Scilly. https://www.southwestwater.co.uk/siteassets/document-repository/business-plan-2020-2025/sww-draft-dwmp-l2-isles-of-scilly-v1.0.pdf (2022).
Jones, B. L., Cullen-Unsworth, L. C. & Unsworth, R. K. F. Tracking nitrogen source using δ15N reveals human and agricultural drivers of seagrass degradation across the British Isles. Front. Plant Sci. 9, 133 (2018).
Munksgaard, N. C., Warnakulasooriya, K. N., Kennedy, K., Powell, L. & Gibb, K. S. Enrichment of 15N/14N in wastewater-derived effluent varies with operational performance of treatment systems: Implications for isotope monitoring in receiving environments. Environ. Monit. Assess. 189, 36 (2017).
Green, A. R., Feast, N. A., Hiscock, K. M. & Dennis, P. F. Identification of the source and fate of nitrate contamination of the Jersey bedrock aquifer using stable nitrogen isotopes. Geol. Soc. Spec. Publ. 130, 23–35 (1998).
Robins, N. S. & Smedley, P. L. Hydrogeology and hydrogeochemistry of a small, hard-rock island-the heavily stressed aquifer of Jersey. J. Hydrol. (Amst.) 3, 249–269 (1994).
Jersey Planning and Environment Committee. Countryside Character Appraisal: Marine Intertidal Zone. (1999).
Viana, I. G. & Bode, A. Stable nitrogen isotopes in coastal macroalgae: Geographic and anthropogenic variability. Sci. Total Environ. 443, 887–895 (2013).
ITV News. Murky brown water in St Aubin’s Bay is ‘compliant’ with regulations. ITV News (2020).
Bailiwick Express. ‘Shocking’ pollution claims in St Aubin’s Bay. Bailiwick Express (2016).
Aravena, R., Evans, M. L. & Cherry, J. A. Stable isotopes of oxygen and nitrogen in source identification of nitrate from septic systems. Groundwater 31, 180–186 (1993).
Steffy, L. Y. & Kilham, S. S. Elevated δ15N in stream biota in areas with septic tank systems in an urban watershed. Ecol. Appl. 14, 637–641 (2004).
House of Lords. Amendment 75. Environment Bill—Report (3rd Day) (Continued). Hansard vol. 75. https://www.theyworkforyou.com/lords/?id=2021-09-13b.1228.1 (2022).
Government of Jersey. Causes of sea lettuce. https://www.gov.je/News/2015/pages/sealettuceinformation.aspx (2015).
Government of Jersey. St Aubin water safety assurance. https://www.gov.je/news/2016/pages/StAubinWater.aspx (2016).
Banks, D., Skarphagen, H., Reimann, C. & Watkins, D. The comparative hydrochemistry of two granitic island aquifers: The Isles of Scilly, U.K. and the Hvaler Islands, Norway. (Report 97070) (1997).
Alotaibi, N. M., Kenyon, E. J., Cook, K. J., Börger, L. & Bull, J. C. Low genotypic diversity and long-term ecological decline in a spatially structured seagrass population. Sci. Rep. 9, 18387 (2019).
Gardner, J. W. Excellence: Can We Be Equal And Excellent Too? (Harper & Row, 1961).
Acknowledgements
We want to thank all family and friends who helped collect samples during the Covid pandemic. Funding for this project was provided by the Stable Isotope Biogeochemistry Laboratory (SIBL) at Durham. Three anonymous reviewers helped to improve the manuscript.
Author information
Authors and Affiliations
Contributions
D.R.G. developed the concept and designed the study. L.P.W. and N.B. collected the samples and provided background information and notes on the island environments. F.A. & C.Y.L. conducted the nitrogen isotope analyses under the direction and supervision of D.R.G. The QGIS maps were generated by F.A. The manuscript was written by F.A. and D.R.G., and all authors have provided contributions to the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Alldred, F.C., Gröcke, D.R., Leung, C.Y. et al. Diffuse and concentrated nitrogen sewage pollution in island environments with differing treatment systems. Sci Rep 13, 4838 (2023). https://doi.org/10.1038/s41598-023-32105-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-32105-6
This article is cited by
-
Anthropogenic modifications: impacts and conservation strategies
Scientific Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.