Abstract
Abundant evidence shows that consolidated memories are susceptible to modifications following their reactivation. Processes of memory consolidation and reactivation-induced skill modulation have been commonly documented after hours or days. Motivated by studies showing rapid consolidation in early stages of motor skill acquisition, here we asked whether motor skill memories are susceptible to modifications following brief reactivations, even at initial stages of learning. In a set of experiments, we collected crowdsourced online motor sequence data to test whether post-encoding interference and performance enhancement occur following brief reactivations in early stages of learning. Results indicate that memories forming during early learning are not susceptible to interference nor to enhancement within a rapid reactivation-induced time window, relative to control conditions. This set of evidence suggests that reactivation-induced motor skill memory modulation might be dependent on consolidation at the macro-timescale level, requiring hours or days to occur.
Similar content being viewed by others
Introduction
Consolidation is a crucial process in the formation of memories, occurring after the initial encoding of information or a skill, and resulting in memory stabilization. While previously thought to be an irreversible process1,2, evidence from studies in rodents3,4,5, further supported by human studies6,7,8 indicates that even fully consolidated memories can become unstable again upon their reactivation. Such reactivation can result in deterioration7,8,9,10 or in enhancement of the memory (11, for a review see12). Similar to previous reports across multiple memory domains13,14, spanning from fear memory3,4,15 to perceptual memories11, evidence for such modification of memories following their reactivation was recently demonstrated in motor skill memories as well: reactivations were shown to protect memories from future interference16 and even induce learning of a motor skill17,18.
At the neural level, the post-reactivation time window is known to involve protein synthesis-dependent processes3, requiring hours or days to occur19,20,21. While similar timescales were reported for post-encoding consolidation as well22,23,24,25, it has been shown that a rapid form of consolidation occurs even in early stages of learning, at a micro-timescale of minutes26. Thus, while consolidated memories often show offline learning gains between sessions, such gains were evident between trials in early stages of a single encoding session, at a micro-timescale level.
Motivated by this set of evidence, we asked whether encoded skill memories would be susceptible to modifications within a rapid 'micro-timescale' window following their reactivation. In a set of experiments, we collected crowdsourced motor sequence learning27 data from 459 participants recruited from the Amazon Mechanical Turk platform (MTurk), to test whether post-reactivation interference and enhancement are evident at the micro-timescale of minutes, according to the established human reconsolidation criteria14. Since memories were previously shown to be susceptible to interference following reactivation in the macro-timescale of 24 h between sessions8,28, Experiment 1 tested whether encoded memories are susceptible to interference within a micro-timescale window induced by brief reactivations. Participants underwent rapid micro-timescale consolidation26 of the motor sequence task. Then, an interfering sequence was presented interleaved with brief reactivations of the original sequence. Retest was compared to a control group without reactivations (see Fig. 1b). Since reactivations were recently shown to induce learning in motor skills17, Experiment 2 tested if memories can benefit from brief reactivations to produce enhanced learning gains even following rapid consolidation, and not a full, macro-timescale consolidation. Following micro-timescale consolidation, the memory was reactivated repeatedly to induce learning. Retest was then compared to a control group without reactivations (see Fig. 1c).
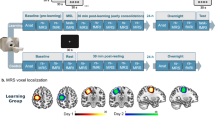
Experimental design. (a) The sequence tapping task required participants to tap a sequence of numbers (either sequence A: 4 1 3 2 4 or sequence B: 4 2 3 1 4) which was constantly presented on the screen during trials. (b) Experimental design of Experiment 1. Each purple square represents a trial of the main sequence A, and each grey square represents a trial of the new sequence B. (c) Experimental design of Experiment 2.
Materials and methods
Participants
Participants were recruited from the Amazon Mechanical Turk Platform (MTurk). Qualifications for registered MTurk workers to participate in the experiments were: > 95% approval rate on all previous MTurk assignments, location in the United States, right-handedness and no previous participation in any sequence learning task offered by our lab. All procedures were in accordance with a protocol approved by the Tel Aviv University Ethics committee, and all methods were performed in accordance with the relevant guidelines and regulations approved by committee. Informed consent was obtained from all participants, declaring via button press that they agree to participate and acknowledge the outline and purpose of the study, their voluntary participation, the time commitment and payment, their mandatory age above 18 years old, information on data safety and being given contact information.
Sample sizes for each experiment were estimated based on power analysis of pilot studies of motor sequence learning. Sample sizes were 230 participants for Experiment 1 (95 female, mean ± sd age 33.9 ± 7.2), and 229 for Experiment 2 (110 female, age 34.7 ± 8.8). These sample sizes represent the total number of participants after exclusion of assignments demonstrating incomplete adherence to task instructions. Participants were paid 1.5$ for the session, equivalent to > 8$ per hour. The time of the task being posted was midday on weekdays for each experiment. Tasks were posted at batches of 30–50 workers each time for administrative (monetary) reasons.
Task
Participants practiced the motor skill learning task29 in which they were asked to tap, as fast and as accurate as they could, a five-digit sequence (Experiment 1: main sequence 4–1–3–2–4, new sequence 4–2–3–1–4; Experiment 2: 4–1–3–2–4; see Fig. 1a) on the numeric keys of participants’ computer keyboards with the pinky finger corresponding to button # 1, the ring finger to # 2, middle finger to # 3, and index finger to # 4. During each trial, the sequence was presented constantly on a computer screen. The task was performed with the left nondominant hand in all trials. Each trial lasted 10 s, during which feedback was provided in the form of a star displayed at the top portion of the screen, appearing immediately after each keypress regardless of correctness17,26. Stimuli were programmed, presented and responses recorded using the Pavlovia.org platform.
Experimental procedure
All subjects performed identical encoding consisting of five trials lasting 10 s each with 10 s breaks in between, based on pilot experiments showing that participants reached 95% performance by trial 5. Experiment 1 tested whether early learning memories are susceptible to interference within a reactivation-induced time window. Accordingly, encoding was followed by a 30 s break, during which a countdown was consistently presented on the screen to maintain subjects’ engagement with the task. Following the break, subjects in the Interference-Reactivations group (N = 115) performed five reactivations trials, in which they performed the main sequence (4–1–3–2–4), each immediately followed by a trial of the new sequence (4–2–3–1–4, 5 new sequence trials in total), while subjects in the Interference-NoReactivations group (N = 115) performed the five new sequence trials with 10 s breaks in between, without reactivations. Immediately following these trials, all participants completed a retest consisting of seven trials of the main sequence (see Fig. 1b). Experiment 2 tested if early learning memories can benefit from brief reactivations to produce enhanced learning gains. Accordingly, participants in the Reactivations group (N = 127) performed 5 reactivations trials following the 30 s break and encoding of the skill memory, followed by a retest identical to experiment 1, while participants in the NoReactivations group (N = 102) performed encoding and retest without reactivations, and with a break of the same length as the duration between test and retest for the Reactivations group (120 s; see Fig. 1b). Of note, the number of trials used in test and retest sections was identical in both Experiment 1 and Experiment 2 to maintain consistency throughout the study.
Data analysis
Behavioral data were analyzed with Matlab R2019b and SPSS statistics 27. Due to the experimental setting offered via crowdsourcing platforms, data registered from each participant were checked for correct implementation and adherence to task instructions. Incorrect implementation of instructions or adherence was defined by (i) completion of the task with the right hand (documented by given answer to question after the task), (ii) completion of only one sequence repetition except for trial 1 (in both experiments) and reactivation trials in Experiment 1, (iii) keypresses consistently different from instructed sequence, (iv) response times until the first key tap longer than 2 s, indicative of lack of attention to the screen, (v) no registered responses during at least one trial. Subjects with incorrect implementation (according to at least one of the above) were excluded from analyses.
To test for baseline differences between groups, a repeated measures ANOVA was conducted with the five test trials as a within subject factor, and group as a between subject factor. Test–retest improvements were evaluated with a repeated measures ANOVA with two performance levels (mean 5 test trials, mean 7 retest trials), and group as a between subject factor, Bonferroni corrected. In experiment 1, to test for gradual improvements in the new sequence, as well as gradual decrease in performance of the original sequence during reactivations, two repeated measures ANOVA were conducted with five trials (5 new sequence trials, or 5 reactivation trials) as a within subject factor, Bonferroni corrected. Each null result in an ANOVA test was further confirmed with a Bayesian approach, by calculating the Bayes factor of comparing total learning across the two conditions. Bayesian analyses were performed with JASP version 0.16.
Results
To test whether memories express increased susceptibility to interference during early learning within micro-timescale reactivation-induced time windows, Experiment 1 compared the effect of interference in motor sequence learning between two groups who experienced interference either with or without reactivations. Both Interference-Reactivation and Interference-NoReactivation groups first performed five trials of skill acquisition. A repeated measures ANOVA with five test trials and two group levels showed no baseline differences between the groups (no group effect F1,228 = 0.07, p = 0.79 and no group × trial interaction F4,912 = 0.80, p = 0.52; see Fig. 2a), with a complimentary Bayesian analysis confirming that there was substantial support in favor of the null hypothesis of no difference between groups relative to the alternative hypothesis for the group effect (BF01 = 7.10) and the group x trial interaction (BF01 = 127.31). Test–retest improvements showed a main effect for time (F1,228 = 207.69, p < 0.001), but no effect for group (F1,228 = 0.11, p = 0.74; BF01 = 3.17) nor a significant interaction (F1,228 = 3.40, p = 0.07; see Fig. 2b,c), suggesting that micro-timescale reactivation-induced time windows did not modulate the susceptibility to interference at early stage of skill learning. Of note, while retest performance was not significantly different between groups (F1,228 = 0.76, p = 0.39), post-hoc pairwise comparisons showed that the first retest trial was better in the Interference-Reactivation group, compared to the Interference-NoReactivation group (t228 = 2.96, p = 0.003). However, this difference diminished in the following retest trials and was not significant in any of the following retest trials (significance level: p = 0.37, p = 0.97, p = 0.53, p = 0.95, p = 0.64, p = 0.58 for trials 2–7 respectively; Bonferroni corrected).
Micro-timescale reactivations do not enhance memory interference. (a) Single-trial performance (Interference-Reactivations group in blue and Interference-NoReactivations group in pink). (b) Test versus retest single-subject performance presented in a scatterplot along a unit slope line (y = x) where each point reflects a participant11,16,30. Data accumulating above the unit line reflect subjects who improved from test to retest, expressing less interference, while data points below the line indicate degraded retest performance, expressing stronger interference. (c) Colored bars reflect the mean performance in test and retest sessions (corresponding to the left y-axis), dashed black bars (corresponding to the right y-axis) reflect the percentage of participants on each side of the unit slope line in (b). (d) Mean performance of the Interference-Reactivations group in both sequences executed alternatingly (main sequence A as circles, interfering sequence B as triangles). Error bars represent SEM.
Of note, while performance in trials of the new memory showed gradual improvements (F4,456 = 40.42, p < 0.001, Greenhouse–Geisser corrected; see triangles in Fig. 2d), performance in the reactivation trials showed gradual decrease (F4,456 = 12.54, p < 0.001, Greenhouse–Geisser corrected; see circles in Fig. 2d), suggesting that in early stages of learning, two memories can be encoded in parallel, converging performance to a shared similar level.
To further investigate whether reactivation-induced skill modulation operates at a micro-timescale level, Experiment 2 tested whether such immediate reactivations of the memory induce learning, as recently reported between-days17. Thus, Experiment 2 maintained similar test and retest as in Experiment 1, with either 5 reactivations trials (Reactivation group) between test and retest, or without reactivations (NoReactivation group). A repeated measures ANOVA with 5 test trials and 2 group levels showed comparable baseline performance across groups during acquisition and test trials (no group effect F1,227 = 1.16, p = 0.28, BF01 = 4.16 and no group x trial interaction F4,908 = 0.44, p = 0.78, BF01 = 155.20; see Fig. 3a). Test–retest improvements showed a main effect for time (F1,227 = 538.91, p < 0.001), but no effect for group (F1,227 = 1.42, p = 0.24, BF01 = 2.51) nor a significant interaction (F1,227 = 0.10, p = 0.76, BF01 = 4.71; see Fig. 3b,c), suggesting that micro-timescale reactivation-induced time windows did not induce learning gains.
Micro-timescale reactivations do not induce learning. (a) Single-trial performance (Reactivations group in yellow and NoReactivation group in black). (b) Test versus retest single-subject performance presented in a scatterplot along a unit slope line (y = x) where each point reflects a participant11,16,30. Data accumulating above the unit line reflect subjects who improved from test to retest, expressing learning gains, while data points below the line indicate degraded retest performance. (c) Colored bars reflect the mean performance in test and retest sessions (corresponding to the left y-axis), dashed black bars (corresponding to the right y-axis) reflect the percentage of participants on each side of the unit slope line in (b). Error bars represent SEM.
Discussion
In a set of crowdsourced experiments, with data collected from hundreds of participants, this study examined whether reactivation-induced modulation of motor skill memory operates at a micro-timescale level. To address this question, two main behavioral aspects of memory modulation were tested: memory interference and performance enhancement. Based on multiple studies reporting modified effects of memory interference if a new memory is presented during the reactivation-induced time windows8,16,28, Experiment 1 tested whether interference with reactivated skill memory is enhanced immediately following encoding. However, results showed that the effect of interference was comparable regardless of whether the new memory was reactivated or not. In addition, since memory reactivation was recently reported to induce learning in motor skills17,18, Experiment 2 tested whether uninterrupted reactivations following early skill learning enhance performance. Results showed that these reactivations did not enhance skill performance, with performance at retest comparable to the NoReactivations group who did not undergo reactivations between test and retest. In sum, these results show that memory modulation effects reported in both human and animal studies, do not apply at the micro-timescale level, and thus do not increase the susceptibility of memories to modulations in early stages of learning.
Interestingly, rapid consolidation was reported to occur in early stages of motor skill learning, during rest periods between trials, lasting only 10 s26. Moreover, a recent study suggested that micro-timescale consolidation might be induced by fast neural replay occurring during wakeful rest periods between trials31. Even though consolidation is evident at the micro-timescale, the current study did not find evidence for an analogous form of rapid memory modulation occurring at the micro-timescale level. The reason for this might stem from the state of the memory trace itself. Consolidation processes transform unstable newly acquired memories to a stable state, in which they will be less prone to modifications, while following memory reactivation, memories are transformed from a stable state back to an unstable state. Thus, since consolidation affects unstable memories, it may be effective for newly acquired skills at early stages of learning, but reactivation-induced modulation, operating on stable memory traces, might not be effective in early stages of learning, and therefore does not operate successfully in a rapid form at the micro-timescale level.
When a second skill memory was presented during the rapid reactivation-induced time windows in Experiment 1, performance of both memories gradually converged to a common level, which was better than the starting level of the new memory, but worse than the starting level of the original memory. This notion might suggest that in early stages of learning, when the memory is still flexible enough, two memories can be acquired simultaneously, with a small detriment in performance of the original memory. Moreover, the convergence of performance towards a similar level of skill execution might imply that these memories are stored in a shared manner in the brain as a single merged memory trace, consistent with the concept of neural engrams32, which were shown to overlap when learning two linked memories33.
These findings may provide additional evidence for the long-known discussion on the fundamental dissociation between consolidation and reconsolidation, occurring during reactivation-induced time windows34, to be further tested in future research. A number of studies have tested the molecular differences between the two, with some studies reporting similarities at the molecular level of the underlying mechanisms3,35, and some studies reporting differences (for example36,37,38,39). In line with these studies, the results of the current study point to a dissociation between consolidation and reconsolidation, with a behavioral separation between consolidation, that indeed occurs at a rapid form within minutes26,40, and reconsolidation that does not operate in such a micro-timescale. Nevertheless, it should be noted that the current study is limited to specific reactivation and interference protocols, based on previous studies. Future studies could further investigate reactivation-induced skill modulation via alternative protocols. For example, while interference in Experiment 1 was performed by interleaved trials of both skill memories16, future studies could induce interference in blocked practice41,42 following single-trial reactivation and by incorporating a prediction-error43 as done in additional memory domains. In addition, studies can evaluate reactivation-induced skill modulation by testing interference between hands instead of alternating sequences12,44.
In sum, the findings of the current study provide robust evidence that contrary to the regular timescales of reactivation-induced memory modulation, interference (Experiment 1) and learning (Experiment 2) are not enhanced within reactivation-induced time windows. Thus, findings of both experiments converge to suggest that reactivation-induced modulation does not operate at a micro-timescale level. Revealing the boundaries and time frames of memory modulation can have implications on future strategies geared to modulate learning and memory.
Data availability
The datasets generated and analyzed during the current study are publicly available at https://osf.io/mrh9d/. This manuscript was deposited as a preprint in PsyArXiv under license CC-By Attribution 4.0 International: https://doi.org/10.31234/osf.io/8da5f.
References
Nettersheim, A., Hallschmid, M., Born, J. & Diekelmann, S. The role of sleep in motor sequence consolidation: Stabilization rather than enhancement. J. Neurosci. 35, 6696–6702 (2015).
McGaugh, J. L. Memory—A century of consolidation. Science (80-) 287, 248–251 (2000).
Nader, K., Schafe, G. E. & Le Doux, J. E. Fear memories require protein synthesis in the amygdala for reconsolidation after retrieval. Nature 406, 722–726 (2000).
Suzuki, A. et al. Memory reconsolidation and extinction have distinct temporal and biochemical signatures. J. Neurosci. 24, 4787–4795 (2004).
Lee, J. L. C. Memory reconsolidation mediates the strengthening of memories by additional learning. Nat. Neurosci. 11, 1264–1266 (2008).
Herz, N. et al. Neuromodulation of visual cortex reduces the intensity of intrusive memories. Cereb. Cortex https://doi.org/10.1093/cercor/bhab217 (2021).
Censor, N., Horovitz, S. G. & Cohen, L. G. Interference with existing memories alters offline intrinsic functional brain connectivity. Neuron 81, 69–76 (2014).
Walker, M. P., Brakefield, T. & Hobson, J. A. Dissociable stages of human memory consolidation and reconsolidation. Nature 425, 616–620 (2003).
Gabitov, E. et al. Re-stepping into the same river: competition problem rather than a reconsolidation failure in an established motor skill. Sci. Rep. 7, 9406 (2017).
Shmuel, D. et al. Early visual cortex stimulation modifies well-consolidated perceptual gains. Cereb. Cortex 31, 138–146 (2021).
Amar-Halpert, R., Laor-Maayany, R., Nemni, S., Rosenblatt, J. D. & Censor, N. Memory reactivation improves visual perception. Nat. Neurosci. 20, 1325 (2017).
Herszage, J. & Censor, N. Modulation of learning and memory: A shared framework for interference and generalization. Neuroscience 10, 270–280 (2018).
Dudai, Y. Reconsolidation: The advantage of being refocused. Curr. Opin. Neurobiol. 16, 174–178 (2006).
Elsey, J. W. B., Van Ast, V. A. & Kindt, M. Human memory reconsolidation: A guiding framework and critical review of the evidence. Psychol. Bull. 144, 797 (2018).
Schiller, D. et al. Preventing the return of fear in humans using reconsolidation update mechanisms. Nature 463, 49–53 (2010).
Herszage, J. & Censor, N. Memory reactivation enables long-term prevention of interference. Curr. Biol. 27, 1529-1534.e2 (2017).
Herszage, J., Sharon, H. & Censor, N. Reactivation-induced motor skill learning. Proc. Natl. Acad. Sci. 118, 2102242118 (2021).
Wymbs, N. F., Bastian, A. J. & Celnik, P. A. Motor skills are strengthened through reconsolidation. Curr. Biol. 26, 338–343 (2016).
Stickgold, R. & Walker, M. P. Memory consolidation and reconsolidation: What is the role of sleep?. Trends Neurosci. 28, 408–415 (2005).
Duvarci, S. & Nader, K. Characterization of fear memory reconsolidation. J. Neurosci. 24, 9269–9275 (2004).
Monfils, M.-H., Cowansage, K. K., Klann, E. & LeDoux, J. E. Extinction-reconsolidation boundaries: Key to persistent attenuation of fear memories. Science (80-) 324, 951–955 (2009).
Brashers-Krug, T., Shadmehr, R. & Bizzi, E. Consolidation in human motor memory. Nature 382, 252–255 (1996).
Press, D. Z., Casement, M. D., Pascual-Leone, A. & Robertson, E. M. The time course of off-line motor sequence learning. Cogn. Brain Res. 25, 375–378 (2005).
Walker, M. P. & Stickgold, R. Sleep-dependent learning and memory consolidation. Neuron 44, 121–133 (2004).
Lugassy, D., Herszage, J., Pilo, R., Brosh, T. & Censor, N. Consolidation of complex motor skill learning: evidence for a delayed offline process. Sleep 41, zsy123 (2018).
Bönstrup, M. et al. A rapid form of offline consolidation in skill learning. Curr. Biol. 29, 1346–1351 (2019).
Karni, A. et al. Functional MRI evidence for adult motor cortex plasticity during motor skill learning. Nature 377, 155–158 (1995).
de Beukelaar, T. T., Woolley, D. G. & Wenderoth, N. Gone for 60 seconds: Reactivation length determines motor memory degradation during reconsolidation. Cortex 59, 138–145 (2014).
Karni, A. et al. The acquisition of skilled motor performance: Fast and slow experience-driven changes in primary motor cortex. Proc. Natl. Acad. Sci. USA 95, 861–868 (1998).
Gelstein, S. et al. Human tears contain a chemosignal. Science (80-) 331, 226–230 (2011).
Buch, E. R., Claudino, L., Quentin, R., Bönstrup, M. & Cohen, L. G. Consolidation of human skill linked to waking hippocampo-neocortical replay. Cell Rep. 35, 109193 (2021).
Josselyn, S. A., Köhler, S. & Frankland, P. W. Finding the engram. Nat. Rev. Neurosci. 16, 521–534 (2015).
Kastellakis, G., Silva, A. J. & Poirazi, P. Linking memories across time via neuronal and dendritic overlaps in model neurons with active dendrites. Cell Rep. 17, 1491–1504 (2016).
Dudai, Y. The restless engram: Consolidations never end. Annu. Rev. Neurosci. 35, 227–247 (2012).
Bang, J. W. et al. Consolidation and reconsolidation share behavioural and neurochemical mechanisms. Nat. Hum. Behav. 2, 507–513 (2018).
Lee, J. L. C., Everitt, B. J. & Thomas, K. L. Independent cellular processes for hippocampal memory consolidation and reconsolidation. Science (80-) 304, 839–843 (2004).
von Hertzen, L. S. J. & Giese, K. P. Memory reconsolidation engages only a subset of immediate-early genes induced during consolidation. J. Neurosci. 25, 1935–1942 (2005).
Li, Y. et al. Learning and reconsolidation implicate different synaptic mechanisms. Proc. Natl. Acad. Sci. 110, 4798–4803 (2013).
Censor, N., Buch, E. R., Nader, K. & Cohen, L. G. Altered human memory modification in the presence of normal consolidation. Cereb. Cortex 26, 3828–3837 (2016).
Bönstrup, M., Iturrate, I., Hebart, M. N., Censor, N. & Cohen, L. G. Mechanisms of offline motor learning at a microscale of seconds in large-scale crowdsourced data. NPJ Sci. Learn. 5, 7 (2020).
Magill, R. A. & Hall, K. G. A review of the contextual interference effect in motor skill acquisition. Hum. Mov. Sci. 9, 241–289 (1990).
Schmidt, R. A. & Bjork, R. A. New conceptualizations of practice: Common principles in three paradigms suggest new concepts for training. Psychol. Sci. 3, 207–217 (1992).
Fernández, R. S., Boccia, M. M. & Pedreira, M. E. The fate of memory: Reconsolidation and the case of prediction error. Neurosci. Biobehav. Rev. 68, 423–441 (2016).
Censor, N. Generalization of perceptual and motor learning: A causal link with memory encoding and consolidation?. Neuroscience 250, 201–207 (2013).
Acknowledgements
The study was supported by the Israel Science Foundation (grant 526/17), the United States-Israel Binational Science Foundation (grant 2016058), and the European Research Council (grant ERC-2019-COG 866093).
Author information
Authors and Affiliations
Contributions
Conceptualization, J.H., L.G.C., and N.C.; Methodology, J.H., M.B., L.G.C., and N.C.; Data Collection and Analysis, J.H..; Writing and Editing, J.H., L.G.C., and N.C.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Herszage, J., Bönstrup, M., Cohen, L.G. et al. Reactivation-induced motor skill modulation does not operate at a rapid micro-timescale level. Sci Rep 13, 2930 (2023). https://doi.org/10.1038/s41598-023-29963-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-29963-5
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.