Abstract
Human T-lymphotropic viruses 1 and 2 (HTLV-1/2) have a worldwide distribution. HTLV-1 has been associated with several diseases, including an aggressive malignant disease known as adult T-cell leukemia/lymphoma and a chronic inflammatory neurological disease called HTLV-1-associated myelopathy, while HTLV-2 has not been definitively associated with diseases. HTLV-2 is most prevalent in specific groups such as injecting drug users and the indigenous population. In Brazil, most studies about HTLV in indigenous are carried out in indigenous communities from the north of the country. Mato Grosso do Sul (MS), Central Brazil, has the second-largest indigenous population in Brazil. However, there is no available data about HTLV infection in this group. We conducted the first investigation of HTLV-1/2 infection prevalence in the indigenous population from Jaguapiru and Bororó villages in Dourados City, MS, to provide the prevalence and molecular characterization of HTLV. For that, a total of 1875 indigenous participated in the study. All the serum samples were screened by an enzyme-linked immunosorbent assay commercial kit for the presence of anti-HTLV-1/2 antibodies. Positive samples were confirmed by HTLV-1/2 Western Blot assay. The HTLV-1 5’LTR region was detected by nested PCR amplification and sequenced by Sanger. Most of the study population declared belonging to Guarani-Kaiowá ethnicity (69.18%), 872 (46.51%), and 1003 (53.49%) were from Jaguapiru and Bororó villages, respectively. The median age of participants was 31 years, and 74.24% were females. Two individuals were detected with HTLV-1 (0.1%; CI 95% 0.1–0.2). The phylogenetic analysis revealed that isolates belong to the Cosmopolitan subtype and the Transcontinental subgroup (HTLV-1aA). The low HTLV-1 prevalence found in this study is similar to that observed among blood donors, and pregnant populations from Mato Grosso do Sul. The absence of HTLV-2 infection among these Brazilian indigenous communities would suggest a distinct behavior pattern from other indigenous populations in Brazil.
Similar content being viewed by others
Introduction
The indigenous population in Brazil shows a high prevalence of sexually or vertically transmitted infections and a complex epidemiological profile1,2,3,4,5. Lower socioeconomic and educational levels associated with cultural aspects of some ethnicities as cross-breastfeeding, polygamy, unprotected sex, and interaction with non-indigenous society, may expose them to risk factors that lead to an increase in the detection rate of infectious diseases1,3,6. Mato Grosso do Sul (MS), Central Brazil, is the second state with the largest indigenous population in Brazil7. The Guarani-Kaiowá, KiniKinawa, Kadiwéu, Terena, Guató, Atikum and Ofaié are the ethnicities present in the state8. This indigenous reserve, with about 18,000 people and located in Dourados city, 3474.50 ha, is represented mainly by the Guarani-Kaiowá and Terena ethnicities living in the Bororó and Jaguapiru villages9,10.
The Human T-lymphotropic virus 1 (HTLV-1) is a retrovirus associated with adult T-cell leukemia/lymphoma (ATL), HTLV-1-associated myelopathy (HAM), and other inflammatory diseases11,12. It is estimated that 5–10 million people are infected by HTLV-1 worldwide. South Japan, sub-Saharan Africa, Caribbean islands, Melanesia, and South America are considered endemic regions for HTLV-1 infections13. HTLV-2 has no clinical manifestations well-defined and has been observed as prevalent in specific groups such as American indigenous populations and people who inject drugs (PWID)14,15. HTLV-1/2 are transmitted by sexual relations, transfusion/transplantation of contaminated blood/organs, and from mother to child, mainly by breastfeeding16.
HTLV-2 is predominant in the Brazilian indigenous population, which the prevalence can reach rates greater than 30%, mainly in the Kayapo Indigenous communities from the north of the country13,17,18,19. However, HTLV-1 infection can be found in this group in lower prevalence or in isolated cases13,17,20.
Once remaining gaps in the knowledge about HTLV infection in the country, especially among these indigenous people, this cross-sectional study aimed to investigate the prevalence, risk factors, and molecular characterization of HTLV among this second-largest indigenous reserve in Brazil located in Mato Grosso do Sul.
Materials and methods
Study population
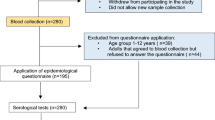
The study population was conducted among indigenous individuals in Dourados City, MS, Central Brazil, from Bororó and Jaguapiru villages (Fig. 1).
From September 2017 to March 2020, 2190 indigenous persons were invited, and 1875 agreed to participate in the study. Participants underwent an interview with a standardized questionnaire containing sociodemographic and sexual behavior information. Blood samples were collected from all subjects to perform serological tests. The inclusion criteria were being an indigenous community member from Bororó or Jaguapiru villages and aged over 18 years, unless unable to give informed written consent. All subjects gave their written informed consent to participate in the study. This study was approved by the Comissão Nacional de ética em Pesquisa (CONEP) under protocol number 2.000.496. All research was performed following relevant guidelines and regulations.
Serological tests
All the serum samples were screened by an enzyme-linked immunosorbent assay (ELISA) commercial kit for the presence of anti-HTLV-1/2 antibodies (GOLD ELISA HTLV-I/II—REM), following the manufacturer’s instructions. Positive samples were repeatedly tested and confirmed by HTLV-1/2 Western Blot (WB) assay (MP Diagnostics HTLV BLOT 2.4–Singapore). HTLV infection was defined as a repeatedly positive ELISA and positive WB.
Molecular analysis
The HTLV molecular characterization of ELISA and WB positive samples was performed by nested polymerase chain reaction (nested PCR). To this end, DNA was extracted from the whole blood of the anti-HTLV positive participants using the QIAamp DNA Blood mini kit (QIAgen), according to the manufacturer’s instructions. Then, the amplification of a 646 bp fragment of the HTLV-1 5’LTR region was performed by nested PCR as previously reported21. Furthermore, the amplicons were purified using a QIA Quick Purification kit (Qiagen Inc., Maryland, USA), according to the manufacturer’s instructions. The fragments were sequenced using BigDye Terminator Cycle Sequencing Ready Reaction Kit and ABI 3500XL (Applied Biosystems, Foster City, CA, United States) by Sanger’s method.
The HTLV-1 isolates were subjected to an analysis in the BLAST (Basic Local Alignment Search Tool) after nucleotide sequencing to identify the subtype and subgroup. Nucleotide sequences were aligned and compared with 50 published HTLV-1 sequences available from GenBank with reference sequences used as outgroup, and sequences were searched with filter HTLV-1 and Transcontinental and Brazil using MAFFT v.7 (Multiple Alignment Program for Amino acid or nucleotide sequences). Phylogenetic analysis was performed using Maximum likelihood with 1000 bootstrap replicates with RAxML program and Bayesian method based on appropriate models determined by JModelTest (TIM2 + G model) with MR-base program on CYPRES Science Gateway. The phylogenetic tree was constructed by use of the FigTree v.1.4.4 program. The haplotype network was developed with NETWORK v.10.2 using the Median Joining algorithm.
The GenBank nucleotide sequences accession number included in the phylogenetic analysis: Z32527, L02534, L76310, Y17014, KM023757, KM023765, D13784, AF033817, DQ235699, DQ235698, Y16481, AF054627, J02029, KY510690, KY510691, KY490575, KY490581, GQ443755-57, L36905, JF271836-38, JF271840-42, AY499185, AY920503, DQ471187-97, DQ70891-92, EU392159-60, FJ853490-91, OK247616-17, KM023763-62.
Statistical analysis
The variables of interest were analyzed using STATA 13.0 software (Stata Corporation, College Station, TX, USA). Prevalence for serological markers of HTLV infection was estimated with 95% confidence intervals. Categorical variables were presented by absolute and percentage frequency. Continuous variables were expressed as mean, standard deviation, median, and range. The chi-square test was used to evaluate differences between proportions.
Ethics statement
The study involving human participants was reviewed and approved by the Comissão Nacional de Ética em Pesquisa (CONEP), an ethics Committee on Human Research, protocol number 2.000.496. The participants provided their written informed consent to participate in this study.
Results
A total of 1875 individuals were enrolled in the study. The median age of participants was 31 years, including 483 males (25.76%) and 1392 females (74.24%). Regarding the village, 872 (46.51%) and 1003 (53.49%) were from Jaguapiru and Bororó, respectively. Most of them declared belonging to Guarani-Kaiowá ethnicity (69.18%). In relation to formal education, 66.28% had more than 5 years of study.
History of alcohol, drug, and sexually transmitted infections was reported by 27.68%, 3.73%, and 2.88% of the participants, respectively. None of them reported injectable drug use history. Other risk behaviors such as multiple sexual partners, irregular condom use, history of blood transfusion, tattoos, sharing sharp objects, and history of surgery are shown in Table 1.
Anti-HTLV was detected in two individuals (0.1%; CI 95% 0.1–0.2). The two ELISA reactive samples were further tested by WB assay and confirmed anti-HTLV reactivity with complete profiles in the WB test for HTLV-1. The test results were subsequently referred to the health authorities responsible for this study population.
The HTLV-1 5’LTR region was detected by nested PCR amplification in the two anti-HTLV-1 positive samples, which were successfully sequenced. The phylogenetic analysis revealed that both isolates were classified as belonging to the Cosmopolitan (1a) subtype and the Transcontinental (A) subgroup (Fig. 2). The GenBank accession numbers for the sequences were: OM863789 (ID-137) and OM863790 (ID-763). Due to low variation between the sequences, a haplotype network with HTLV-1aA sequences (Supplementary Fig. S1) was performed to show the differences between sequences.
Phylogenetic tree of HTLV-1 subtypes from different groups. Phylogenetic tree constructed based on Maximum likelihood and Bayesian method with sequences of HTLV-1 5’LTR region from indigenous of this study (highlighted) and using 50 sequences from GenBank. Support for the branching was determined by 1000 bootstrap replicates and only values of 70% or superior was shown for Maximum likelihood and 0.9 or superior of posterior probability for Bayesian analyses. ID-137 (OM863789), ID-763 (OM863790) clustered with different population groups.
Table 2 shows the sociodemographic characteristics and risk behaviors of the two anti-HTLV positive individuals. One of the positive individuals, a 42-year-old female (ID-763), was married twice. No information was available on the HTLV-1 status of her first husband, with whom she had 6 children. Also, she had 1 daughter with her current husband. Of these 6 older children, 3 were found at the time of our visit and referred to the health authorities responsible. The current husband did not agree to participate in the study. Besides, their daughter was under 2 years old and was being breastfed by her mother. She reported having breastfed four of her children for more than 1 year and the other two children for less than 6 months. She also reported having normal labor and that her tested offspring were negative for the presence of anti-HTLV-1/2 by the ELISA method.
The other positive individual was an 81-year-old man (ID-137). Both individual ID-137 and ID-763 did not report any risk behaviors such as injection drug use, unprotected sexual intercourse, and multiple sexual partners. Both positive individuals were born in Dourados city, lived in Bororó village, and declared themselves Guarani-Kaiwá ethnicity.
Discussion
This is the first investigation of HTLV-1/2 infection prevalence carried out among the indigenous population from Central Brazil. The prevalence of HTLV infection found in this study (0.1%; CI 95% 0.1–0.2) is similar to the prevalence observed in the blood donors, and pregnant population from Mato Grosso do Sul (0.2% and 0.1%, respectively)22,23. However, the results of this present study contrast with most of the studies conducted among indigenous populations in Brazil.
Several studies carried out among the indigenous population from the Brazilian Amazon region were developed, and HTLV-2 infection was shown to be prevalent, which is a change in the epidemiological pattern of this HTLV type in this specific group1,24,25. The high HTLV-2 prevalence in the indigenous population in Brazil usually differs between communities (from 1.4% in the Wayampi community on Amapá to 57.9% in the Kayapo community on Pará)1,13,17,24,26. Unlike these studies conducted in Brazil, the present study, no individuals infected with HTLV-2 were found.
The introduction of HTLV-1 in the Mato Grosso do Sul State probably occurred during the human migratory process from the slave trade from Africa and Japanese migration in the previous centuries27,28,29. A prevalence study conducted among several Brazilian indigenous populations has already identified HTLV-1 infection, and all of them were from the North region of the country. Ishak et al.17 observed HTLV-1 infection prevalence of 2.94%, 0.67%, and 0.48% among Yanomami, Galibi, and Aukre communities, respectively. Besides, a prevalence of 0.62% was found in the Wayampi community in 2002, and one case was reported in the same community in 200720,30. The absence of more recent HTLV-1 studies in a Brazilian indigenous group may be due to the cultural characteristics of the studied communities since most studies are carried out in closed communities27. The geographic isolation of these villages limits contact with urban centers or other indigenous groups24. However, the Jaguapiru and Bororó villages are located 5 km from Dourados city, the second-largest city of Mato Grosso do Sul state, therefore geographically accessible to the urban center7,31,32. HTLV-1 infection was recently reported among Warao indigenous refugees living in urban area of Belém city33. Also, HTLV-1 is already reported in Indigenous people from other South American countries, such as Chile and Venezuela13,33,34,35. There was no difference of positivity for anti-HTLV (0.10% vs. 0.20%; p > 0.30) between female and male participants, respectively. These results are in accordance with studies conducted among the indigenous population in which no difference between genders was found17,36.
HTLV-1 testing has been included in the routine antenatal screening of the state of Mato Grosso do Sul by the Program for Protection of Pregnant Woman since 200223. The positive female individual (ID-763) reported that she was evaluated during her youngest daughter's pregnancy by the antenatal screening. She was diagnosed with positive anti-HTLV-1/2 and was recommended the interruption of breastfeeding since that time. The Brazilian Ministry of Health recommends the interruption of breastfeeding and the provision of milk formula substitutes feeding for those babies born from HTLV-1/2 seropositive mothers to avoid mother-to-child transmission16. Although these measures, adequate counseling with information about HTLV-1 transmission or clinical evaluation to identify signs or symptoms of HTLV-1 associated diseases was not offered to the ID-763 participant. This demonstrates the need to spread knowledge about HTLV infection and the public health policies that are already implemented in Brazil to properly provide counseling for positive patients.
The HTLV-1aA sequences present in this study diverge from the previous studies conducted in Brazil among indigenous populations that found mainly HTLV-2. The sequences obtained from the present study grouped next with other isolates from various non-indigenous populations and geographical regions in Brazil, such as people who use drugs, Afro-descendants, pregnant women, blood donors, and people who live with HIV (PLHIV), suggesting a possible HTLV-1 transmission from these non-indigenous individuals. Unfortunately, sequences from other worldwide indigenous populations belonging to the Transcontinental subgroup (HTLV-1aA) were not found registered on Genbank.
Some limitations must be acknowledged. First, underreporting of risk behaviors and reporting bias are some of the limitations of this cross-sectional study. Because some of them may have been omitted during the interviews due to the distrust of discrimination and stigma. In addition, individuals from the micro-areas of indigenous health care were enrolled by convenience sampling, with greater acceptance of the study among residents of the Bororó village, resulting in unequal sample distribution between them. Because of the low number of HTLV infected individuals in this study, regression analysis could not be performed. Despite these limitations, this study is representative of the indigenous population of Central Brazil since we used a large sample size. Moreover, our study has important information which would contribute to closing the gaps in HTLV epidemiology and controlling public policies.
The results of the study show the presence and circulation of HTLV-1 in indigenous populations from Jaguapiru and Bororó villages in Dourados City, MS. A better understanding of the patterns of occurrence of HTLV-1 in the indigenous population of Mato Grosso do Sul is critical to guiding the development of public health initiatives to reduce transmission. Although a higher risk of infection has been supposed for this specific study population, the prevalence rate of HTLV-1 infection found in this study was similar to that observed among Brazilian blood donors (0.48%)37. Also, the absence of HTLV-2 infection among these Brazilian indigenous communities would suggest a behavior pattern distinct from other indigenous populations in Brazil.
Data availability
All the relevant and original data presented in the study are included in the article. The datasets generated and/or analyzed during the current study are not included in a publicly available repository due to restrictions applied to the availability of these data, such as possible identification of the participants, being a restricted group, but are available from the corresponding author on reasonable request. DNA sequences were registered into the GenBank repository under accession numbers OM863789 (ID-137) and OM863790 (ID-763).
References
Braço, I. et al. High prevalence of human T-lymphotropic virus 2 (HTLV-2) infection in villages of the Xikrin tribe (Kayapo), Brazilian Amazon region. BMC Infect. Dis. 19(1), 459. https://doi.org/10.1186/s12879-019-4041-0 (2019).
Garnelo, L. Saúde Indígena: Uma introdução ao tema. Luiza Garnelo; Ana Lúcia Pontes (2012). Brasília: MEC-SECADI, 280 p. il. Color. (Coleção Educação para Todos) ISBN 978-85-7994-063-7.
Graeff, S. V., Pícolli, R. P., Arantes, R., Castro, V. & Cunha, R. Epidemiological aspects of HIV infection and AIDS among indigenous populations. Rev. Saude Publica 53, 71. https://doi.org/10.11606/S1518-8787.2019053000362 (2019).
Russell, N. K. et al. HIV, syphilis, and viral hepatitis among Latin American indigenous peoples and Afro-descendants: A systematic review. Rev. Panam. Salud Publica Pan Am. J. Public Health 43, 17. https://doi.org/10.26633/RPSP.2019.17 (2019).
Benzaken, A. S. et al. HIV and syphilis in the context of community vulnerability among indigenous people in the Brazilian Amazon. Int. J. Equity Health 16(1), 92. https://doi.org/10.1186/s12939-017-0589-8 (2017).
Vallinoto, A. C. et al. Molecular epidemiology of human T-lymphotropic virus type II infection in Amerindian and urban populations of the Amazon region of Brazil. Hum. Biol. 74(5), 633–644. https://doi.org/10.1353/hub.2002.0059 (2002).
BRASIL. IBGE. Instituto Brasileiro de Geografia Estatística. Os indígenas no Censo Demográfico 2010 primeiras considerações com base no quesito cor ou raça (Rio de Janeiro, 2012). https://indigenas.ibge.gov.br/images/indigenas/estudos/indigena_censo2010.pdf.
SECID/MS. Subsecretaria Especial de Cidadania. Governo do Estado de Mato Grosso do Sul. https://www.secid.ms.gov.br/comunidades-indigenas-2/.
de Alcantara, M. L. B., Moure, W., Trajber, Z., Machado, I. R., Equipe de Jovens da Ação dos Jovens Indígenas de Dourados-MS. A percepção do suicídio como inseparável das outras formas de violência segundo os/as jovens indígenas: Um estudo de caso da Reserva Indígena de Dourados. Rev. Med. 99(3), 305–318 (2020).
Mota, J. G. B. & Cavalcante, T. L. V. Reserva Indígena de Dourados: Histórias e Desafios Contemporâneos, 285 (Karywa, 2019). ISBN: 978–85–68730–38–6.
Bangham, C. R., Araujo, A., Yamano, Y. & Taylor, G. P. HTLV-1-associated myelopathy/tropical spastic paraparesis. Nat. Rev. Dis. Primers 1, 15012. https://doi.org/10.1038/nrdp.2015.12 (2015).
Hermine, O., Ramos, J. C. & Tobinai, K. A review of new findings in adult T-cell leukemia-lymphoma: A focus on current and emerging treatment strategies. Adv. Ther. 35(2), 135–152. https://doi.org/10.1007/s12325-018-0658-4 (2018).
Paiva, A. & Casseb, J. Origin and prevalence of human T-lymphotropic virus type 1 (HTLV-1) and type 2 (HTLV-2) among indigenous populations in the Americas. Rev. Inst. Med. Trop. Sao Paulo 57(1), 1–13. https://doi.org/10.1590/S0036-46652015000100001 (2015).
Martinez, M. P., Al-Saleem, J. & Green, P. L. Comparative virology of HTLV-1 and HTLV-2. Retrovirology 16(1), 21. https://doi.org/10.1186/s12977-019-0483-0 (2019).
Oliveira-Filho, A. B. et al. Human T-lymphotropic virus 1 and 2 among people who used illicit drugs in the state of Pará, northern Brazil. Sci. Rep. 9(1), 14750. https://doi.org/10.1038/s41598-019-51383-7 (2019).
Rosadas, C. et al. Blocking HTLV-1/2 silent transmission in Brazil: Current public health policies and proposal for additional strategies. PLoS Negl. Trop. Dis. 15(9), e0009717. https://doi.org/10.1371/journal.pntd.0009717 (2021).
Ishak, R. et al. Identification of human T cell lymphotropic virus type IIa infection in the Kayapo, an indigenous population of Brazil. AIDS Res. Hum. Retrovir. 11(7), 813–821. https://doi.org/10.1089/aid.1995.11.813 (1995).
Menna-Barreto, M. et al. Human T-cell lymphotropic virus type II in Guaraní Indians, Southern Brazil. Cad. Saude Publica 21(6), 1947–1951. https://doi.org/10.1590/s0102-311x2005000600045 (2005).
Vallinoto, A. C. R. & Ishak, R. HTLV-2: Uma infecção antiga entre os povos indígenas da Amazônia brasileira. Rev. Pan Amaz. Saude Ananindeua 8(2), 7–9. https://doi.org/10.5123/s2176-62232017000200001 (2017).
Shindo, N. et al. Human retroviruses (HIV and HTLV) in Brazilian Indians: Seroepidemiological study and molecular epidemiology of HTLV type 2 isolates. AIDS Res. Hum. Retrovir. 18(1), 71–77. https://doi.org/10.1089/088922202753394736 (2002).
de Oliveira, E. H. et al. Human T-cell lymphotropic virus in patients infected with HIV-1: Molecular epidemiology and risk factors for transmission in Piaui, Northeastern Brazil. Curr. HIV Res. 10(8), 700–707. https://doi.org/10.2174/1570162x11209080700 (2012).
Freitas, G. M. B. Estudo Clínico e Epidemiológico da Infecção pelos Vírus Linfotrópicos de Células-T Humanas (HTLV-I/II) em Doadores de Sangue em Campo Grande-MS (1994–2001). (Doctoral dissertation, Dissertação de mestrado, Fundação Oswalddo Cruz, Rio de Janeiro, 2002).
Dal Fabbro, M. M. F. J. et al. HTLV 1/2 infection: Prenatal performance as a disease control strategy in State of Mato Grosso do Sul. Rev. Soc. Bras. Med. Trop. 41(2), 148–151. https://doi.org/10.1590/s0037-86822008000200003 (2008).
Ishak, R., Vallinoto, A. C., Azevedo, V. N. & Ishak, M. Epidemiological aspects of retrovirus (HTLV) infection among Indian populations in the Amazon Region of Brazil. Cad. Saúde Publica 19(4), 901–914. https://doi.org/10.1590/s0102-311x2003000400013 (2003).
Machado, L. et al. The epidemiological impact of STIs among general and vulnerable populations of the Amazon Region of Brazil: 30 years of surveillance. Viruses 13(5), 855. https://doi.org/10.3390/v13050855 (2021).
Fujiyoshi, T. et al. Characteristic distribution of HTLV type I and HTLV type II carriers among native ethnic groups in South America. AIDS Res. Hum. Retrovir. 15(14), 1235–1239. https://doi.org/10.1089/088922299310124 (1999).
Ishak, R. et al. HTLV in South America: Origins of a silent ancient human infection. Virus Evol. 6(2), veaa053. https://doi.org/10.1093/ve/veaa053 (2020).
Bandeira, L. M. et al. High prevalence of HTLV-1 infection among Japanese immigrants in non-endemic area of Brazil. PLoS Negl. Trop. Dis. 9(4), e0003691. https://doi.org/10.1371/journal.pntd.0003691 (2015).
Nascimento, L. B. et al. Prevalência da infecção pelo HTLV-1, em remanescentes de quilombos no Brasil Central [Prevalence of infection due to HTLV-1 in remnant quilombos in Central Brazil]. Rev. Soc. Bras. Med. Trop. 42(6), 657–660. https://doi.org/10.1590/s0037-86822009000600009 (2009).
Kanzaki, L. I. B. & Casseb, J. Unusual finding of HTLV-I infection among Amazonian Amerindians. Arch. Med. Res. 38(8), 897–900. https://doi.org/10.1016/j.arcmed.2007.05.002 (2007).
BRASIL. Instituto Brasileiro de Geografia e Estatística (Cidades e Estados Dourados, 2022). https://www.ibge.gov.br/cidades-e-estados/ms/dourados.html.
Oliveira, G. F. et al. Prevalence of obesity and overweight in an indigenous population in central Brazil: A population-based cross-sectional study. Obes. Facts 8(5), 302–310. https://doi.org/10.1159/000441240 (2015).
Abreu, I. N. et al. HTLV-1 and HTLV-2 infection among Warao indigenous refugees in the Brazilian Amazon: Challenges for public health in times of increasing migration. Front. Public Health 10, 833169. https://doi.org/10.3389/fpubh.2022.833169 (2022).
Zanella, L. et al. Phylogenetic dating analysis of HTLV-1 from Chile suggests transmissions events related to ancient migrations and contemporary expansion. Int. J. Infect. Dis. 99, 186–189. https://doi.org/10.1016/j.ijid.2020.07.037 (2020).
Zanella, L. et al. Genome analysis suggests HTLV-1aA introduction in Chile related to migrations of ancestral indigenous populations. Virus Res. 311, 198687. https://doi.org/10.1016/j.virusres.2022.198687 (2022).
Black, F. L., Biggar, R. J., Neel, J. V., Maloney, E. M. & Waters, D. J. Endemic transmission of HTLV type II among Kayapo Indians of Brazil. AIDS Res. Hum. Retrovir. 10(9), 1165–1171. https://doi.org/10.1089/aid.1994.10.1165 (1994).
Catalan-Soares, B., Carneiro-Proietti, A. B., Proietti, F. A., Interdisciplinary HTLV Research Group. Heterogeneous geographic distribution of human T-cell lymphotropic viruses I and II (HTLV-I/II): Serological screening prevalence rates in blood donors from large urban areas in Brazil. Cad. Aúde Publica 21(3), 926–931. https://doi.org/10.1590/s0102-311x2005000300027 (2005).
Acknowledgements
We would like to acknowledge all indigenous participants and the staff members of the DSEI/MS for their support during the study period. A special thanks for the important contribution in this study to the indigenous nurse Cassiano de Souza Ribeiro (in memory) who worked in the Primary Health Care of the Special Indigenous Health District (DSEI), of the Special Secretariat for Indigenous Health (Sesai), at the Dourados base pole. Our appreciation also involves the staff from the GPBMM/UFGD research group for the partnership during the study.
Funding
This study was supported by Governo do Estado de Mato Grosso do Sul, Fundação de Apoio ao Desenvolvimento do Ensino, Ciência e Tecnologia do Estado de Mato Grosso do Sul—FUNDECT/MS (Grant Number: No. 03/2016, TO 021/2017 FUNDECT/DECIT-MS/CNPq/SES-PPSUS-MS) and Conselho Nacional de Desenvolvimento Científico e Tecnológico—CNPQ (Grant Number: Nos. 442522/2019-3, 30295/2021-5). This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001.
Author information
Authors and Affiliations
Contributions
Conceptualization: S.S., J.H.R.C. and A.R.C.M.C.; methodology: L.M.B., C.A., G.A.C., S.M.S.W.T., T.S.O.T., I.R.M., S.S., F.B.F., E.C.S.S., and A.R.C.M.C.; analyzed the data: L.M.B., C.A., and A.R.C.M.C.; contributed reagents/materials/analysis tools: C.C.M.G., F.B.F., A.C.R.V., S.S. and A.R.C.M.C., writing—original draft preparation, review, and editing: L.M.B, C.A., S.M.S.W.T., S.S., A.C.R.V., A.R.C.M.C., supervision: A.R.C.M.C. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Amianti, C., Bandeira, L.M., Cesar, G.A. et al. HTLV infection in Brazil’s second-largest indigenous reserve. Sci Rep 12, 16701 (2022). https://doi.org/10.1038/s41598-022-21086-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-21086-7
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.