Abstract
The direct impact of chronic hepatitis B and hepatitis C on neurocognition remains elusive due to the frequent comorbidities, and the domains of the neurocognitive functions affected have rarely been investigated comprehensively. We cross-sectionally assessed the neurocognitive functions of the individuals with chronic hepatitis B, chronic hepatitis C, treated chronic hepatitis C with a sustained virologic response, and their healthy control counterparts. Laboratory examinations were used to investigate the impact of inflammation on neurocognition, exclude the medical conditions that could interfere with neurocognition assessment, and assess liver function and fibrotic severity of the liver of the participants. This study found the detrimental impact of chronic hepatitis B on language and executive functions. In contrast, individuals with chronic hepatitis C showed deficits in executive functions, psychomotor speed, memory, and attention. Successful elimination of hepatitis C resulted in improved liver function, but not neuropsychological test performance. Moreover, erythrocyte sedimentation rate level was found to mediate the deficits in the attention of individuals with chronic hepatitis C. These results demonstrate the neurocognitive deficits and the difference in the profiles of neurocognitive deficits in individuals with chronic hepatitis B and chronic hepatitis C. Our study also provided results suggesting the mediation by systemic inflammation on the attention deficit in individuals with chronic hepatitis C.
Similar content being viewed by others
Introduction
According to the global hepatitis report 2017 by the World Health Organization, in 2015, the global prevalence of HBV and HCV infection in the general population was estimated to be 3.5% and 1%, respectively1. In addition to the life-threatening complications, including cirrhosis and hepatocellular carcinoma, both HBV and HCV cause extrahepatic manifestations. More importantly, these extrahepatic manifestations cause an impaired quality of life and economic burden on patients with viral hepatitis2. Among the extrahepatic manifestations, the neuropsychiatric symptoms found in individuals with HCV have been reported previously. However, with depression, cirrhosis, thyroid disorders, cerebrovascular diseases, and end-stage renal diseases as common comorbidities associated with HCV and significantly interfering with the accurate evaluation of neurocognitive functions, the inclusion of patients with the common comorbidities associated with HCV in the previous studies prevents a clear demonstration of the direct effect of HCV infection on neurocognitive function changes. In addition, while the negative impact of HCV infection on neurocognitive functions has been shown across several studies, the investigation into the effects of HBV on cognition has rarely been reported. As HBV has an even higher prevalence than HCV and is still without the treatment for cure, an investigation to understand the long-term impact of HBV on neurocognitive functions is warranted.
Furthermore, several mechanisms through which HCV causes neurocognitive deficits have been proposed, but a definite conclusion remains to be explored due to the limited evidence available. Meanwhile, although the treatment regimens for HCV infection have advanced significantly, whether the neurocognitive deficit of patients with HCV is reversible and whether successful elimination of HCV infection causes improvement in neurocognitive functions remains unknown. Finally, as the concept of "neurocognitive functions" is a broad term and covers a wide range of complex mental processes, the elucidation of the neurocognitive functions attributable to specific conditions requires comprehensive assessments covering different domains of each ability area and a careful combination of tasks to reveal patterns of performance3. However, a thorough evaluation of the neurocognitive functions covering all the major neurocognitive domains of patients with chronic viral hepatitis has rarely been investigated.
Taiwan used to have a high prevalence of chronic HBV and HCV. The prevalences of HBV and HCV among the general Taiwanese population were previously reported to be around 15–20% and 4.1%, respectively4,5. The prevalence was even reported to be up to 17.2% in Kaohsiung County, the southern part of Taiwan6. Although the prevalences of HBV and HCV have significantly decreased in recent years, the impact of chronic viral hepatitis on the generation born before the nationwide HBV vaccination program in 1986 has been long-lasting and remained not entirely clear. The situation is similar for those infected with HCV before the wide availability of effective treatment against HCV. Therefore, in the current study, we comprehensively evaluated the neurocognitive functions (summarized in Table 1) of healthy participants without viral hepatitis and those with chronic HBV or HCV to understand the long-term impact and potential mechanisms of the neurocognitive deficit caused by chronic HBV and HCV infection.
Results
Patients with chronic viral hepatitis showed different profiles of neurocognitive deficit across different domains
To examine whether chronic viral hepatitis causes neurocognitive deficits and assess which neurocognitive domains were affected, the participants were divided into four groups: healthy volunteers, participants with HBV, participants carrying HCV, and participants with a sustained virologic response (SVR) after HCV treatment. All the participants with HBV had a chronic infection as determined by HBsAg positivity for six months or more or the presence of HBV DNA by polymerase chain reaction six months or more after the initial HBsAg positivity. The number of participants in the phase of HBeAg positive chronic infection, HBeAg positive chronic hepatitis, HBeAg negative chronic infection, and HBeAg positive chronic hepatitis were 1, 7, 27, and 36, respectively, and 43 of the participants with HBV were taking antiviral medication against HBV. All the participants with untreated HCV had chronic HCV as determined by the persistent presence of HCV RNA by polymerase chain reaction for six months or more until the neuropsychological test. The definition of SVR was based on the absence of HCV RNA by polymerase chain reaction six months after the end of anti-HCV therapy before the assessment with the neuropsychological test. The demographic data, including age, sex, education, hypertension (HTN), diabetes mellitus (DM), signs of polyneuropathy, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), alanine transaminase (ALT), and FIB-4 index of the participants within each study group are shown in Table 2. There was no significant difference in the percentage of participants with HTN, DM, or signs of polyneuropathy observed among the different study groups. There were significant differences in age, sex ratio, education years, ESR, and ALT between the study groups. The difference in age and sex ratio between the different study groups may be due to the difference in the epidemiological profile of HBV and HCV among Taiwanese, and the difference in the education years may be secondary to the difference in age and sex ratio. As ESR is closely associated with age and sex, which are variables significantly different between the study groups, we used Quade's test to compare ESR and CRP levels between the study groups to eliminate age and sex's confounding effect. After controlling the effect of age and sex, no statistically significant difference was observed between the study groups, and the results are shown in supplementary table 1. Although the ALT level of the untreated HCV group is significantly higher than those of the HBV and SVR group, the mean values of the ALT in the three groups were all below the twofold upper limit of the normal ALT reference range, indicating that most of the patients are not in the acute exacerbation phase. Furthermore, there was no statistically significant difference in the FIB-4 among the study groups. The mean values of the FIB-4 in the three groups were all lower than 3.25 (the value over which can be suggestive of significant cirrhosis), indicating that most of the participants recruited did not have significant cirrhosis.
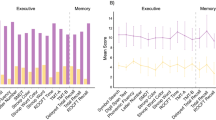
The profiles of the neurocognitive functions of each study group's participants are shown in Table 3, and the existence of the differences in neurocognitive functions between different study groups was examined with the Kruskal–Wallis test. Statistically significant differences between the study groups were noted in (1) both the immediate and delayed recall of logical memory and the immediate recall of visual production in Wechsler Memory Scale-III (WMS-III); (2) full-scale intelligence quotient, verbal comprehension index, perceptual reasoning index, similarities, information, block design, matrix reasoning, digit span total score, longest digit span forward, digit symbol substitution, and symbol search of Wechsler Adult Intelligence Scale-IV (WAIS-IV); (3) Paced Auditory Serial Addition Test (PASAT); (4) semantic association of category verbal fluency; (5) trial 2 of Color Trails Test (CTT). The results of the post-hoc analysis identifying the significant between-group differences with the Dunn's test after the Kruskal–Wallis test are shown in Supplementary Table 2.
However, as shown in Table 2, significant differences exist in the demographic characteristics between the study groups. Thus, the significant differences in the task performance, as shown in Table 3 and Supplementary Table 2, may be secondary to the differences in the demographic characteristics between the study groups, such as age, sex, and education. To eliminate the effect of the potential confounding factors, we used Quade's test and set age, sex, and education years as covariates to compare the participants' neurocognitive functions within each study group. As shown in Table 4, after controlling the potential effect of age, sex, and education years with the Quade test, patients with HBV showed significantly worse performance in verbal comprehension index, information, and matrix reasoning of WAIS-IV than their healthy control counterparts. Besides, patients with HBV showed significantly worse performance in the language function than those in the SVR group (with a borderline significant difference compared with the HCV group). These results suggest the negative impact of HBV on the neurocognitive domains of language function and executive function.
Meanwhile, the patients with HCV showed significantly worse performance in perceptual reasoning and similarities of WAIS-IV and the trial 1 of CTT than the healthy control counterparts. The results suggest that HCV causes a deficit in executive functions and psychomotor speed. For the SVR group, the patients showed significantly worse performance in the immediate recall of visual production in WMS-III and verbal comprehension index of WAIS-IV than their healthy control counterparts. Compared with the patients with HBV, the SVR group still showed worse performance in both the immediate and delayed recall of the logical memory of WMS-III and shorter longest digit span forward. The results indicate that HCV patients have deficits in memory and attention, and the worse performance in memory and attention can still be observed in those after successful treatment of HCV obtaining SVR. No statistically significant difference was observed when comparing the HCV patients with those whose HCV had been treated and obtained SVR. The results suggest that successful HCV treatment does not significantly improve the neurocognitive function deficit of individuals with viral hepatitis. However, a significant improvement in hepatic injury was observed, as evidenced by the ALT level.
The neurocognitive deficit in individuals with viral hepatitis seems to be mediated by systemic inflammation, and attention is the most sensitive neurocognitive domain affected
In addition, we hypothesize that the systemic inflammation induced by HCV may be the mediator for developing neurocognitive deficits, which was examined with a regression-based analysis carried out with PROCESS macro on SPSS. The mediation effects of ESR and CRP on the neurocognitive functions affected by HCV compared with HBV are shown in Table 5 and supplementary table 3, respectively. As shown in Table 5, the significant mediation by ESR on the neurocognitive functions affected by HCV is demonstrated in serial 7 of Mini-Mental State Examination (MMSE), full-scale intelligence, working memory, block design, letter-number sequencing, and digit span of WAIS-IV, PASAT, and interference score of Modified Wisconsin Card Sorting Test (M-WCST). The statistically significant mediation by ESR in serial 7 of MMSE and working memory and digit span of WAIS-IV is still observed after correcting the potential confounders, including age, sex, and education years. These results suggest that the neurocognitive function deficit found in individuals with HCV is mediated partially through systemic inflammation, and attention is the most sensitive domain to the detrimental effect of systemic inflammation. Besides, the impact of systemic inflammation on neurocognitive functions is more closely associated with mechanisms affecting ESR, but not CRP.
Discussion
Previous studies rarely investigated the neurocognitive deficit of individuals with chronic HBV or HCV infection in parallel with a detailed neuropsychological test battery covering all the major neurocognitive domains. In the present study, we found neurocognitive deficits in both individuals with chronic HBV and chronic HCV with the comprehensive neuropsychological test battery, and the deficits seem to have different patterns. Patients with chronic HBV were found to have significantly worse performance in verbal comprehension index, information, and matrix reasoning of WAIS-IV than the healthy control group and worse language function assessed with MMSE than the HCV group. These results suggest that chronic HBV causes a neurocognitive deficit in the domains of language function and executive function. Although the negative effect of chronic HBV infection on neurocognitive functions has never been definitely reported, the extrahepatic manifestation of HBV involving the central nervous system has been suggested by previous studies. A retrospective cohort study reported the association between HBV and Parkinson's disease (PD), with a standardized rate ratio of PD following HBV infection found to be 1.76 based on 44 observed compared with 26 expected cases7. In one study applying single-cell laser capture methods to assess the full range of microglia-specific expression, HBV was found to be expressed in the microglia. Besides, with immunohistochemical analysis using HBV-core antibody as HBV-positive control, HBV immunoreactivity was found to be increased from amnestic mild cognitive impairment to HBV-positive AD cases as disease pathology increased8.
Moreover, in one study investigating the influence of HCV on cognition where HBV was recruited as the control group, the impairment was similar between HBV and HCV in verbal learning/memory when assessed with Rey Auditory Verbal Learning Test9. One recent study showed the progressive impairment of dynamic functional network connectivity in patients with HBV-related cirrhosis, although whether the impairment is due to HBV or cirrhosis remains unknown10. These previous studies suggest the detrimental impact of HBV on CNS degeneration. The current study demonstrates the effect of chronic HBV infection on neurocognitive functions, with a deficit in language and executive functions.
In addition to the detrimental impact of chronic HBV on language and executive functions, the current study also found that patients with chronic HCV infection showed poorer performance in the tasks associated with executive functions, psychomotor speed, memory, and attention, while visuospatial and language functions remain unaffected. Previous studies had attempted to investigate the impact of HCV on neurocognitive functions, but the findings diverged and varied across different studies. Several studies showed worse performance in tasks associated with attention and executive functions in HCV patients11,12, while other studies found the detrimental effect in tasks associated with memory and psychomotor speed9,13,14,15. There were also several studies revealing no definite changes in neurocognitive functions that could be attributable to HCV16,17,18. The conflicting results across the different studies may be due to the difference in the participants' demographic characteristics (such as age and education levels) and the different sensitivity in the tasks used to assess neurocognitive functions.
Moreover, although common comorbidities associated with HCV significantly influence neurocognitive functions, few studies have excluded the patients with depression, cirrhosis, thyroid disorders, cerebrovascular diseases, and end-stage renal diseases in one study to examine the direct impact of HCV on neurocognitive functions19. Meanwhile, HCV has also been shown to increase the risk of kidney injury20, thyroid disorder21, and cerebrovascular events22, all of which are associated with neurocognitive impairment. In the current study, to investigate the direct impact of chronic HCV infection on neurocognitive function, we excluded the participants with a history of cerebrovascular disease, brain surgery, thyroid disorder, end-stage renal disease, signs suggestive of decompensated liver cirrhosis, psychiatric disorders, and illicit recreational drug usage. Our results showing the worse performance in tasks associated with executive functions, psychomotor speed, memory, and attention suggest the direct impact of chronic HCV infection on cognition.
We also compared the neurocognitive functions of patients with chronic HCV and those successfully treated and obtained SVR. In the current study, significant improvement in the ALT level, but not the neurocognitive function profiles, was noted, suggesting that the successful elimination of chronic HCV caused improvement in liver function but not neurocognitive functions. However, the results do not necessarily rule out the potentially beneficial effect of successfully eliminating HCV in decreasing the neurocognitive decline rate after the successful treatment. Long-term follow-up is needed to understand the beneficial impact of HCV treatment on neurocognitive function.
Furthermore, the current study found systemic inflammation seems to partially mediate the neurocognitive deficit of individuals with chronic HCV. The significant mediation effect of ESR on the performance in tasks associated with attention suggests that chronic HCV patients' attention deficit is mediated by systemic inflammation. Several mechanisms have been proposed for the neurocognitive deficit associated with HCV. One proposed mechanism suggests that HCV infects the microglia/macrophages in the central nervous system, and the release of proinflammatory cytokines from the infected microglia/macrophages results in neurocognitive deficits23. Moreover, as systemic inflammation has been suggested to cause neurocognitive deficits24,25,26, systemic inflammation induced by HCV was also proposed to be involved in the decline of neurocognitive functions. However, no direct evidence has been provided except for the current study27. Besides, the mediation effect of ESR on the cognitive domains of attention revealed in the present study may also be in agreement with the pathogenic mechanisms of chronic hepatitis as immune complex diseases28,29. Last but not least, HBV- and HCV-infected individuals had been shown to have dysbiosis and change in the composition of intestinal bacteria30,31,32,33, and the inflammatory responses induced by the translocated microbial products, such as lipopolysaccharide (LPS), were proposed to be one of the pathogenic mechanisms associated with HBV or HCV infection34. The association between LPS and the inflammation-mediated attention deficit observed in this study may need further investigation to clarify the liver-gut-brain association.
Meanwhile, CRP was not shown to mediate the neurocognitive dysfunction found in HCV patients in the current study. The dissociation between ESR and CRP in mediating the detrimental effect of HCV suggests the decline in attention associated with HCV is more closely related to the inflammatory conditions that cause elevation of ESR without elevation of CRP. One condition for such dissociation was observed in autoimmune diseases35 when interferon-α suppresses the release of CRP. Whether the decline in attention induced by HCV is also associated with pathways related to interferon-α needs further investigation.
Also noteworthy is the difference in the profiles of the neurocognitive function impairment of chronic HBV and chronic HCV patients. While chronic HBV causes deficits in the domains of language and executive functions, chronic HCV was found to cause functional decline in executive functions, psychomotor speed, memory, and attention. The different deficit profiles revealed in the neuropsychological tests may also reflect the potential difference in the mechanisms causing the neurocognitive impairment in chronic HBV or chronic HCV patients. Based on previous studies' results, the deficits found in chronic HBV patients in the language functions in the current study may be associated with cortical dementia syndromes. In contrast, chronic HCV patients' deficits in attention and memory may be more closely related to subcortical dementia syndromes36. The results suggest the diverging pathogenic mechanisms of chronic HBV and HCV on neurocognitive functions. In the present study, we focused on the long-term effect of the chronic infections of HBV and HCV on neuropsychological function decline. The neurocognitive function declines found in the chronic HBV and HCV patients and the mediation effect by systemic inflammation shown in the present study suggest a gradual process, in which the waxing and waning disease activity of chronic viral hepatitis causes systemic inflammation that drives the neurodegeneration.
The present study has potential limitations. The significant differences in age and sex ratio between the study groups may cause selection bias. The difference in the education years may interfere with the results of the neurocognitive profile assessment. However, the difference may be inevitable and may reflect the difference in the epidemiological profile of chronic HBV and HCV among Taiwanese, with the prevalence of HBV and HCV in Taiwan being higher in males and females, respectively37,38,39. More importantly, we have used Quade's test to eliminate the effect of the potential confounding factors. The other limitation of the current study is the lack of the laboratory results of ESR and CRP of the healthy control group, which prevents the detailed exploration of the mechanism for the cause of the neurocognitive deficit induced by viral hepatitis, especially HBV. The lack of the healthy control group's laboratory results also prevented the exclusion of patients with end-stage renal diseases and thyroid disorders and may underestimate the detrimental impact of viral hepatitis. However, such underestimation would not cause a significant difference in the main results of the present study.
The other limitation of this study is the lack of laboratory results confirming the absence of human immunodeficiency virus in the participants recruited due to the strict regulations in screening human immunodeficiency virus (HIV) in Taiwan to prevent confidentiality breaches. As the detrimental impact of HIV on neurocognitive functions is significant and well-established40,41, and the coinfection of HIV and viral hepatitis is frequent in some areas around the world42. However, the transmissions of HBV and HCV in the Taiwanese generation older than 50 years old were mainly due to vertical transmission and iatrogenic factors, respectively4,5, while the major risk groups for HIV are those with a history of illegal intravenous drug usage or homosexual practices with men. The prevalence of HIV in the participants recruited in the present study should be low, especially after excluding those with a history of illegal recreational drug usage or same-sex relationship or those with abnormalities suggestive of HIV coinfection during the evaluation by physicians, based on history taking, physical examinations, and laboratory examinations during the clinical follow-up. Therefore, the impact of potential HIV coinfection may be minor, although such a possibility could not be totally excluded in the present study.
Besides, the wide availability of direct-acting antiviral agents against HCV in Taiwan has significantly reduced the number of untreated HCV patients recruited in the present study. Meanwhile, the strict inclusion criteria for participant recruitment in the present study to minimize confounding factors limited the number of participants in the current study and precluded the comparison of the neurocognitive functions across the different disease spectrums. However, the sample size did not cause restriction in the power detecting the neurocognitive deficits and the potential mechanism found in the present study, which focuses on the long-term effect of the chronic infections of HBV and HCV. Future studies with a larger sample size are warranted to validate the detrimental impact of chronic HBV and HCV infection and compare the difference in the neurocognitive deficit conferred across different disease spectrums.
In conclusion, the present study found the detrimental effect of chronic HBV on language and executive functions and showed the negative impact of chronic HCV on executive functions, psychomotor speed, memory, and attention. Also, the neurocognitive function deficit was not reversible after obtaining SVR. Besides, systemic inflammation was found to mediate the deficits in attention in chronic HCV patients. The present study enables a more in-depth understanding of the extrahepatic manifestations of viral hepatitis and also provides evidence for the potential mechanisms of the neurocognitive function deficit induced by viral hepatitis. Lastly, the findings will also raise the awareness of the detrimental impact of viral hepatitis, especially the previously unknown HBV, on neurocognitive functions.
Methods
Participants and assessment of clinical presentations
This study cross-sectionally recruited a total of 199 participants, including 39 healthy volunteers, 72 patients diagnosed with HBV, and 88 patients diagnosed with HCV between the 21st of September 2017 to the 24th of March 2020. Out of the 88 HCV patients, 65 have obtained SVR for more than six months before recruitment in the study. Among the 23 participants without SVR, 22 participants were treatment naïve except for one participant who had received peginterferon and ribavirin 17 years ago. Among the 65 participants obtaining SVR, 35 obtained SVR with peginterferon and ribavirin, and 30 obtained SVR after direct-acting antiviral agents. The decision to initiate antiviral therapy for HBV was primarily based on the presence or absence of cirrhosis, the ALT level, and the HBV DNA level following the EASL 2017 guidelines for HBV infection43. All participants were between 40 and 80 years old with an education level equal to or more than six years to fully understand the content of the neuropsychological assessment. We excluded patients with a history of cerebrovascular disease, brain surgery, thyroid disorder, malignancy, alcohol abuse, end-stage renal disease, signs suggestive of decompensated cirrhosis (including ascites, jaundice, esophageal variceal bleeding, or hepatic encephalopathy), psychiatric disorders, and current pregnancy or breastfeeding. We also excluded patients with signs suggestive of organic brain lesions after a neurological examination by a board-certified neurologist, including 1 individual with HBV, 2 individuals with untreated HCV, and 6 individuals with treated HCV before the recruitment. Participants with HIV or a history of illicit recreational drug usage and tattooing were excluded to minimize the possibility of recruiting patients with HIV in the study. Meanwhile, individuals in same-sex relationships were also not recruited. Participants with end-stage renal disease were excluded based on the estimated glomerular filtration rate < 30 mL/min/1.73m2 calculated with CKD-EPI Creatinine Equation. The exclusion of participants with decompensated cirrhosis was based on history taking and clinical assessment by the hepatologists referring the patients.
The participants' written informed consent was obtained before enrollment, following the ethical standards outlined in the 1975 Declaration of Helsinki. All study procedures were approved by the ethical research committee of Kaohsiung Medical University Hospital (KMUHIRB-G(I)-20170022), and National Cheng Kung University Hospital (-/B-ER-104-082), and all methods were performed following the approved guidelines.
Neurological examinations
All participants in the disease groups were assessed with a neurological examination. The neurological examination performed by a board-certified neurologist is composed of the examinations of cranial nerves, motor functions, sensory functions, coordination, and reflexes. Participants with signs suggestive of organic brain lesions were not recruited for further evaluation to exclude the presence of stroke44,45 or intracranial lesions46 that are associated with viral hepatitis. Participants with signs suggestive of peripheral nervous system disorders were not excluded as the degeneration of the peripheral nervous system and the neurocognition may share common pathogenic mechanisms47.
Neuropsychological assessment
All study participants were assessed with a comprehensive neuropsychological battery composed of tests evaluating all the major neurocognitive domains, including memory, psychomotor speed, attention, visuospatial, executive function, and language function, similar to our previous investigation on the cognitive profiles of patients with PD48. The disease state of chronic viral hepatitis and the state of sustained virologic response had been confirmed with laboratory tests before the evaluation of neuropsychological assessment. Logical Memory-I&II and Visual Reproduction-I&II in the WMS-III were used to assess memory49. Trial 1 of CTT50, Digit Symbol Substitution, and Symbol Search were used for psychomotor speed evaluation. Stroop Color and Word Test51, Digit Span of WAIS-IV52, PASAT53, and serial seven of MMSE54 were used to evaluate attention. Block Design of WAIS-IV was used for visuospatial function evaluation. Similarities, Matrix Reasoning, Information, Letter-Number Sequencing of WAIS-IV, Semantic Association of category verbal fluency55, trial 2 of CTT, and M-WCST56 were used to assess executive function. Naming, repetition, verbal comprehension, and writing of MMSE were used for language function evaluation. The raw scores of the neuropsychological tests were used for the analysis. The neuropsychological tests used for the assessment of the major neurocognitive domains are summarized in Table 1.
Laboratory evaluation
The diagnosis of HBV was based on the serological test positive for HBsAg, and the diagnosis of HCV was based on the serological test positive for the anti-HCV antibody. The presence of the HCV virus in the HCV patients was confirmed with polymerase chain reactions. The laboratory tests used to diagnose viral hepatitis were collected between the 11th of April 2001 and the 8th of May 2019. The creatinine levels of all the participants in the study group of HBV, HCV, or SVR were also examined to rule out the possibility of neurocognitive impairment attributable to end-stage renal disease. Levels of the ALT of chronic hepatitis patients were examined to evaluate their liver function. In addition, aspartate transaminase (AST) and platelet count of chronic hepatitis patients were examined to obtain the FIB-4 index, as an indicator of cirrhosis. Levels of ESR and CRP were also examined among the participants with HBV, HCV, or SVR to evaluate systemic inflammation status. The laboratory examinations for ALT, creatinine, ESR, and CRP were collected within the one-month period from the date of neuropsychological tests. The date of the sample collection for the laboratory results used to calculate FIB-4, including AST, ALT, and platelet count, was within the six-month period from the date of neuropsychological tests. There were two participants in the SVR group whose ESR and CRP were not examined.
Statistical analysis
Similar to our previous publication57, proportions were calculated for qualitative variables, and means and standard deviations were calculated for quantitative variables. We used the Kolmogorov–Smirnov test for the normality test. Afterward, we examined quantitative variables with the Kruskal–Wallis test and qualitative variables with a chi-square test. We controlled the impact of confounding variables (age, sex, and education years) with Quade's test58. Statistical significance was predetermined with an alpha level less than 0.05. The statistical analysis was performed by a commercially available software program (IBM Corp. Released 2012. IBM SPSS Statistics for Windows, Version 21.0. Armonk, NY: IBM Corp.). A regression-based analysis was carried out with PROCESS macro on SPSS59 to calculate the mediation effect of systemic inflammation on neurocognitive functions in hepatitis C patients. Helmert coding was used for the grouping of patients with HBV (d1 = −2/3; d2 = 0), HCV (d1 = 1/3; d2 = −1/2), and treated HCV with SVR (d1 = 1/3; d2 = 1/2). Bias-corrected bootstrap confidence intervals for the effect based on 5000 bootstrap samples were calculated and used to determine whether ESR or CRP mediated the neurocognitive function changes in hepatitis C patients. All the associated data not provided within the paper are available on request from Rwei-Ling Yu.
References
World Health, O. Global hepatitis report 2017. 83 p. (World Health Organization, 2017).
Younossi, Z., Park, H., Henry, L., Adeyemi, A. & Stepanova, M. Extrahepatic manifestations of hepatitis C: A meta-analysis of prevalence, quality of life, and economic burden. Gastroenterology 150, 1599–1608. https://doi.org/10.1053/j.gastro.2016.02.039 (2016).
Harvey, P. D. Domains of cognition and their assessment. Dialogues Clin. Neurosci. 21, 227–237. https://doi.org/10.31887/DCNS.2019.21.3/pharvey (2019).
Wang, H.-H. et al. Risk of HBV infection among male and female first-time blood donors born before and after the July 1986 HBV vaccination program in Taiwan. BMC Publ. Health 21, 1831. https://doi.org/10.1186/s12889-021-11846-x (2021).
Sun, C. A. et al. Transmission of hepatitis C virus in Taiwan: Prevalence and risk factors based on a nationwide survey. J. Med. Virol. 59, 290–296 (1999).
Yang, J. F. et al. Viral hepatitis infections in southern Taiwan: A multicenter community-based study. Kaohsiung J. Med. Sci. 26, 461–469. https://doi.org/10.1016/s1607-551x(10)70073-5 (2010).
Pakpoor, J. et al. Viral hepatitis and Parkinson disease: A national record-linkage study. Neurology 88, 1630–1633. https://doi.org/10.1212/wnl.0000000000003848 (2017).
Mastroeni, D. et al. Laser-captured microglia in the Alzheimer’s and Parkinson’s brain reveal unique regional expression profiles and suggest a potential role for hepatitis B in the Alzheimer’s brain. Neurobiol. Aging 63, 12–21. https://doi.org/10.1016/j.neurobiolaging.2017.10.019 (2018).
Karaivazoglou, K. et al. Neuropsychological function in Greek patients with chronic hepatitis C. Liver Int.: Off. J. Int. Assoc. Study Liver 27, 798–805. https://doi.org/10.1111/j.1478-3231.2007.01486.x (2007).
Lin, S. et al. Progressive disruption of dynamic functional network connectivity in patients with hepatitis B virus-related cirrhosis. J. Magn. Reson. Imag. https://doi.org/10.1002/jmri.27740 (2021).
Ibrahim, I. et al. Hepatitis C virus antibody titers associated with cognitive dysfunction in an asymptomatic community-based sample. J. Clin. Exp. Neuropsychol. 38, 861–868. https://doi.org/10.1080/13803395.2016.1168780 (2016).
Huckans, M. et al. The cognitive effects of hepatitis C in the presence and absence of a history of substance use disorder. J. Int. Neuropsychol. Soc.: JINS 15, 69–82. https://doi.org/10.1017/s1355617708090085 (2009).
Quarantini, L. C. et al. A neuropsychological study comparing patients infected with HCV and HBV without psychiatric comorbidities. J. Med. Virol. 81, 1184–1188. https://doi.org/10.1002/jmv.21508 (2009).
Lowry, D., Coughlan, B., McCarthy, O. & Crowe, J. Investigating health-related quality of life, mood and neuropsychological test performance in a homogeneous cohort of Irish female hepatitis C patients. J. Viral Hepatitis 17, 352–359. https://doi.org/10.1111/j.1365-2893.2009.01188.x (2010).
Fontana, R. J. et al. Cognitive function in hepatitis C patients with advanced fibrosis enrolled in the HALT-C trial. J. Hepatol. 43, 614–622. https://doi.org/10.1016/j.jhep.2005.04.006 (2005).
Hilsabeck, R. C., Perry, W. & Hassanein, T. I. Neuropsychological impairment in patients with chronic hepatitis C. Hepatology (Baltimore, MD) 35, 440–446. https://doi.org/10.1053/jhep.2002.31257 (2002).
Cordoba, J. et al. Quality of life and cognitive function in hepatitis C at different stages of liver disease. J. Hepatol. 39, 231–238. https://doi.org/10.1016/s0168-8278(03)00189-2 (2003).
Abrantes, J., Torres, D. S. & de Mello, C. E. Patients with hepatitis C infection and normal liver function: An evaluation of cognitive function. Postgrad. Med. J. 89, 433–439. https://doi.org/10.1136/postgradmedj-2012-131185 (2013).
Abrantes, J., Torres, D. S. & Brandao-Mello, C. E. The many difficulties and subtleties in the cognitive assessment of chronic hepatitis C infection. Int. J. Hepatol. 2020, 9675235. https://doi.org/10.1155/2020/9675235 (2020).
Petta, S. & Craxì, A. Extrahepatic manifestations of chronic viral C hepatitis. Gastroenterol. Clin. North Am. 49, 347–360. https://doi.org/10.1016/j.gtc.2020.01.012 (2020).
Antonelli, A. et al. Thyroid disorders in chronic hepatitis C virus infection. Thyroid 16, 563–572. https://doi.org/10.1089/thy.2006.16.563 (2006).
Enger, C. et al. Thromboembolic events among patients with hepatitis C virus infection and cirrhosis: A matched-cohort study. Adv. Ther. 31, 891–903. https://doi.org/10.1007/s12325-014-0138-4 (2014).
Wilkinson, J., Radkowski, M., Eschbacher, J. M. & Laskus, T. Activation of brain macrophages/microglia cells in hepatitis C infection. Gut 59, 1394–1400. https://doi.org/10.1136/gut.2009.199356 (2010).
Komulainen, P. et al. Serum high sensitivity C-reactive protein and cognitive function in elderly women. Age Ageing 36, 443–448. https://doi.org/10.1093/ageing/afm051 (2007).
Jefferson, A. L. et al. Inflammatory biomarkers are associated with total brain volume: The Framingham Heart Study. Neurology 68, 1032–1038. https://doi.org/10.1212/01.wnl.0000257815.20548.df (2007).
Beydoun, M. A. et al. Systemic inflammation is associated with longitudinal changes in cognitive performance among urban adults. Front. Aging Neurosci. 10, 313–313. https://doi.org/10.3389/fnagi.2018.00313 (2018).
Senzolo, M. et al. Neuropsychological alterations in hepatitis C infection: The role of inflammation. World J. Gastroenterol. 17, 3369–3374. https://doi.org/10.3748/wjg.v17.i29.3369 (2011).
Salam, K. A. et al. Binding of Free and immune complex-associated hepatitis C virus to erythrocytes is mediated by the complement system. Hepatology (Baltimore, MD) 68, 2118–2129. https://doi.org/10.1002/hep.30087 (2018).
Gower, R. G., Sausker, W. F., Kohler, P. F., Thorne, G. E. & McIntosh, R. M. Small vessel vasculitis caused by hepatitis B virus immune complexes. Small vessel vasculitis and HBsAG. The J. Allergy Clin. Immunol. 62, 222–228. https://doi.org/10.1016/0091-6749(78)90211-7 (1978).
Preveden, T., Scarpellini, E., Milic, N., Luzza, F. & Abenavoli, L. Gut microbiota changes and chronic hepatitis C virus infection. Expert Rev. Gastroenterol. Hepatol. 11, 813–819. https://doi.org/10.1080/17474124.2017.1343663 (2017).
Inoue, T. et al. Gut dysbiosis associated with hepatitis C virus infection. Clin. Infect. Dis. 67, 869–877. https://doi.org/10.1093/cid/ciy205 (2018).
Aly, A. M., Adel, A., El-Gendy, A. O., Essam, T. M. & Aziz, R. K. Gut microbiome alterations in patients with stage 4 hepatitis C. Gut Pathog. 8, 42. https://doi.org/10.1186/s13099-016-0124-2 (2016).
Zeng, Y. et al. Gut microbiota dysbiosis in patients with hepatitis B virus-induced chronic liver disease covering chronic hepatitis, liver cirrhosis and hepatocellular carcinoma. J. Viral Hepatitis 27, 143–155. https://doi.org/10.1111/jvh.13216 (2020).
Sandler, N. G. et al. Host response to translocated microbial products predicts outcomes of patients with HBV or HCV infection. Gastroenterology 141, 1220–1230. https://doi.org/10.1053/j.gastro.2011.06.063 (2011).
Feldman, M. et al. C-reactive protein and erythrocyte sedimentation rate discordance: Frequency and causes in adults. Transl. Res. 161, 37–43. https://doi.org/10.1016/j.trsl.2012.07.006 (2013).
Salmon, D. P. & Filoteo, J. V. Neuropsychology of cortical versus subcortical dementia syndromes. Semin. Neurol. 27, 7–21. https://doi.org/10.1055/s-2006-956751 (2007).
Hu, Y.-C. et al. Seroprevalence of hepatitis B virus in Taiwan 30 years after the commencement of the national vaccination program. PeerJ 6, e4297–e4297. https://doi.org/10.7717/peerj.4297 (2018).
Su, F. H. et al. Seroprevalence of Hepatitis-B infection amongst Taiwanese university students 18 years following the commencement of a national Hepatitis-B vaccination program. J. Med. Virol. 79, 138–143. https://doi.org/10.1002/jmv.20771 (2007).
Wu, G.H.-M. et al. The epidemiological profile of chronic hepatitis C with advanced hepatic fibrosis regarding virus genotype in Taiwan: A nationwide study. J. Formos. Med. Assoc. 120, 1444–1451. https://doi.org/10.1016/j.jfma.2021.01.005 (2021).
Jadhav, S. & Nema, V. HIV-associated neurotoxicity: The interplay of host and viral proteins. Mediat. Inflamm. 2021, 1267041. https://doi.org/10.1155/2021/1267041 (2021).
Fields, J. et al. HIV-1 Tat alters neuronal autophagy by modulating autophagosome fusion to the lysosome: Implications for HIV-associated neurocognitive disorders. The J. Neurosci.: The Off. J. Soc. Neurosci. 35, 1921–1938. https://doi.org/10.1523/jneurosci.3207-14.2015 (2015).
Lacombe, K. & Rockstroh, J. HIV and viral hepatitis coinfections: Advances and challenges. Gut 61, i47–i58. https://doi.org/10.1136/gutjnl-2012-302062 (2012).
European Association for the Study of the Liver. Electronic address, e. e. e. & European Association for the Study of the, L. EASL. Clinical practice guidelines on the management of hepatitis B virus infection. J. Hepatol. 67(370–398), 2017. https://doi.org/10.1016/j.jhep.2017.03.021 (2017).
Tseng, C. H., Muo, C. H., Hsu, C. Y. & Kao, C. H. Increased risk of intracerebral hemorrhage among patients with hepatitis C virus infection. Medicine 94, e2132. https://doi.org/10.1097/MD.0000000000002132 (2015).
Liao, C. C., Su, T. C., Sung, F. C., Chou, W. H. & Chen, T. L. Does hepatitis C virus infection increase risk for stroke? A population-based cohort study. PLoS ONE 7, e31527. https://doi.org/10.1371/journal.pone.0031527 (2012).
Moretti, R. et al. Hepatitis C virus-related central and peripheral nervous system disorders. Brain Sci. https://doi.org/10.3390/brainsci11121569 (2021).
Brenowitz, W. D., Robbins, N. M., Strotmeyer, E. S. & Yaffe, K. Associations of lower extremity peripheral nerve impairment and risk of dementia in black and white older adults. Neurology 98, e1837–e1845. https://doi.org/10.1212/WNL.0000000000200185 (2022).
Yu, R. L. et al. Neuropsychological profile in patients with early stage of Parkinson’s disease in Taiwan. Parkinsonism Relat. Disord. 18, 1067–1072. https://doi.org/10.1016/j.parkreldis.2012.06.002 (2012).
Hua, M. et al. Wechsler memory scale. 3rd ed. Taipei: Chinese Behavioral Science Corp. (2005).
D'Elia, L. & Satz, P. Color trails test. (Psychological Assessment Resources, 1996).
Golden, C. J. & Freshwater, S. M. Stroop color and word test. Age 15, 90 (1978).
Wechsler, D. Wechsler adult intelligence scale–Fourth Edition (WAIS–IV). San Antonio, TX: NCS Pearson 22, 1 (2008).
Tombaugh, T. N. A comprehensive review of the Paced Auditory Serial Addition Test (PASAT). Arch. Clin. Neuropsychol. 21, 53–76. https://doi.org/10.1016/j.acn.2005.07.006 (2006).
Tombaugh, T. N. & McIntyre, N. J. The mini-mental state examination: A comprehensive review. J. Am. Geriatr. Soc. 40, 922–935. https://doi.org/10.1111/j.1532-5415.1992.tb01992.x (1992).
Benton, A. L. Differential behavioral effects in frontal lobe disease. Neuropsychologia 6, 53–60 (1968).
Nelson, H. E. A modified card sorting test sensitive to frontal lobe defects. Cortex 12, 313–324 (1976).
Lin, C. Y., Yu, R. L., Wu, R. M. & Tan, C. H. Effect of ALDH2 on sleep disturbances in patients with Parkinson’s disease. Sci. Rep. 9, 18950. https://doi.org/10.1038/s41598-019-55427-w (2019).
Quade, D. Rank analysis of covariance. J. Am. Stat. Assoc. 62, 1187–1200. https://doi.org/10.1080/01621459.1967.10500925 (1967).
Hayes, A. F. Introduction to mediation, moderation, and conditional process analysis: A regression-based approach. (Guilford Press, 2013).
Vallet-Pichard, A. et al. FIB-4: an inexpensive and accurate marker of fibrosis in HCV infection. comparison with liver biopsy and fibrotest. Hepatology 46, 32–36 (2007).
Acknowledgements
The authors are grateful to the participants involved in this study. The study was supported by grants from the Ministry of Science and Technology (MOST), Taipei, Taiwan (MOST 109-2628-B-037-016 -, MOST 108-2320-B-037 -034 -MY3, and MOST 110-2628-B-006-020 -), and Kaohsiung Medical University Hospital (KMUH109-9R73).
Author information
Authors and Affiliations
Contributions
All authors fulfilled the criteria of authorship, and no one who met the criteria was excluded. C.H. Tan and R.L. Yu had the idea and designed the experiments. C.H. Tan, M.C. Chang, W.F. Tsai, W.L. Chuang, J.F. Huang, Z.Y. Lin, C.Y. Dai, and M.L. Yeh were involved in sample collection. C.H. Tan, W.F. Tsai, C.T. Li, and R.L. Yu performed data analysis. C.H. Tan and R.L. Yu wrote the paper and took responsibility for the interpretation of the results. All authors critically reviewed drafts and approved the final version of this article. R.L. Yu accepted full responsibility for the work and controlled the decision to publish.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tan, CH., Chang, MC., Tsai, WF. et al. Different profiles of neurocognitive impairment in patients with hepatitis B and C virus infections. Sci Rep 12, 10625 (2022). https://doi.org/10.1038/s41598-022-14736-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-14736-3
This article is cited by
-
Prediction of conversion from mild cognitive impairment to Alzheimer’s disease and simultaneous feature selection and grouping using Medicaid claim data
Alzheimer's Research & Therapy (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.