Abstract
Understanding the impacts of microplastics on living organisms in aquatic habitats is one of the hottest research topics worldwide. Despite increased attention, investigating microplastics in underwater environments remains a problematic task, due to the ubiquitous occurrence of microplastic, its multiple modes of interactions with the biota, and to the diversity of the synthetic organic polymers composing microplastics in the field. Several studies on microplastics focused on marine invertebrates, but to date, the benthic sea slugs (Mollusca, Gastropoda, Heterobranchia) were not yet investigated. Sea slugs are known to live on the organisms on which they feed on or to snack while gliding over the sea floor, but also as users of exogenous molecules or materials not only for nutrition. Therefore, they may represent a potential biological model to explore new modes of transformation and/or management of plastic, so far considered to be a non-biodegradable polymer. In this study we analysed the stomachal content of Bursatella leachii, an aplysiid heterobranch living in the Mar Piccolo, a highly polluted coastal basin near Taranto, in the northern part of the Ionian Sea. Microplastics were found in the stomachs of all the six sampled specimens, and SEM/EDX analyses were carried out to characterize the plastic debris. The SEM images and EDX spectra gathered here should be regarded as a baseline reference database for future investigations on marine Heterobranchia and their interactions with microplastics.
Similar content being viewed by others
Introduction
Despite increased attention the unceasing accumulation of plastic materials in aquatic environments represents a critical issue and emergent threat worldwide—boosted by an increasing global plastic production and the improper disposal of plastic waste. The impacts of plastic on marine organisms are widely reported1,2,3 and the list of threatened organisms is constantly increasing, as witnessed by the high number of recent scientific papers on this topic4,5,6. Plastic items can be categorized according to their size as: mega- (> 1 m diameter), macro- (between 2.5 cm and < 1 m), meso- (between 5 mm and < 2.5 cm), micro- (between 0.1 μm and < 5 mm) and nanoplastics (< 0.1 μm)7,8,9,10,11,12,13. Focusing on microplastic, a great effort is currently being made in collecting data on its distribution, quantification, and the occurrence in biological taxa, in order to assess how advanced is the level of contamination of food webs in natural environments. In this framework, several protocols have been developed to optimize the extraction of microplastics from different marine environments and substrates (surface waters, water column, and seabed at different depths) or from different animal taxa, organs, and tissues (e.g., stomach, liver, kidney, gills, lungs, gonads, and specific tissues)14,15. To date, vertebrates have been the primary focus of investigation for the detection of plastic ingestion16,17,18,19,20,21. However, smaller plastic items are commonly ingested by a wider range of (smaller) organisms7,22,23,24. Major groups of invertebrates, such as annelids (Polychaeta), crustaceans (Cirripedia, Amphipoda) and echinoderms (Holothuroidea), are known to ingest microplastic particles during laboratory trials25,26. The common destiny of this ingested plastic seems to be expulsion within faecal material25,27,28,29,30.
The characterization of collected microplastics can take place using different techniques, but most used is Scanning Electron Microscopy (SEM) for morphological investigation, associated with Energy Dispersive X-ray spectroscopy (EDX) for elemental microanalysis. The combination of morphological and elemental composition analyses has proved an effective tool to identify plastic materials extracted from the stomach contents and the tissues of fishes, marine invertebrates, and sediments31,32,33,34. These two coupled techniques seem to be highly effective when ‘in situ studies’ (i.e., based on organisms freshly collected in the field, not subject to laboratory experiments) are carried out, and when other contaminants are present. Indeed, synthetic molecules and materials of anthropogenic origin can interact with the microplastics under investigation, leading to detection of emission spectra not comparable with those available in reference libraries. This is the main drawback of other techniques, such as the Fourier transform infrared spectroscopy analysis (FTIR), requiring comparative screening of reference spectra to identify the chemical nature of the sample. This limitation does not apply to SEM/EDX, which is also a faster technique with the potential to analyse many samples in a relatively short time33,35,36,37. Conversely, procedures of FTIR spectroscopy are longer, leading to analysis of just a sub-sample of the total extracted microplastics31,38,39, with a consequent decrease of accuracy (e.g., due to wrong identifications and/or underestimation).
The Mediterranean Sea is known as a hotspot of biodiversity, with high rate of cryptic diversity and new species being continuously revealed and described40,41,42,43,44. Unfortunately, it is also particularly sensitive to microplastic pollution, with contamination levels almost four times greater than the North Pacific Ocean, comparable to the five top accumulation zones known for subtropical ocean gyres45,46. A significant land-based plastic input in the semi-closed Mediterranean Sea is also combined with a proportion of floating plastics originated outside the basin45 and transported by a constant inflow of superficial waters from the Atlantic Ocean47,48. The patchy spatial distribution of floating plastics in the Mediterranean Sea suggests that shelf areas near river estuaries and population centres may be related to local plastic accumulation in short term, but the overall Mediterranean distribution of plastics seems largely dependent on the pattern of the surface circulation45,48. Eventually, nearly 94% of fragmented plastics accumulate on the seabed with a large proportion occurring as microplastics, up to 2175 items per Kg of sediment in areas subject to high anthropogenic pressures, such as the Venice Lagoon49.
The Mar Piccolo of Taranto (Apulian, Ionian Sea, Central Mediterranean Sea) is a semi closed basin divided in two sheltered and interconnected inlets, a natural site subjected to heavy human pressure and environmental pollution, as the consequence of the high number of industries and intensive farming of mussels and fish operating in the area. The Mar Piccolo contains more than 30 natural submarine springs of brackish water, and it receives some surface creeks from the surrounding territory50. This water input somewhat opposes the entrance of marine water, which is also limited due to a very weak tide oscillation (maximum 30 cm of daily excursion). Such negligible introduction of open sea water minimizes the load of any foreign litter and/or plastics51,52,53, so restricting plastic pollution to local and easily identifiable input sources54. Due to its geomorphological peculiarities, Mar Piccolo can be used as a model system, a natural laboratory where to observe and predict the negative effects that may occur on a larger geographical scale53,54,55, and even to propose localized recovery/remediation interventions.
Various papers have been recently published on the presence and the identification of microplastics from molluscs, but these were focused mainly on filter feeding animals such as bivalves and/or on a few detritivore organisms such as some gastropod species32,56,57,58,59. Unexpectedly, no studies focused so far on the occurrence of microplastics in marine Heterobranchia (Mollusca, Gastropoda), better known as sea slugs, usually living on the organisms on which they feed or gliding around on the sea floor in search for food. As far as we know, the only published paper on microplastics and sea slugs deals with laboratory experiments which did not involve analytical methods of microplastics analysis24. Marine Heterobranchia are generally small and characterized by a reduced or completely lost shell, a very variable body plan and ecological patterns, and a highly specialized diet60,61,62,63. They are known to have a unique tendency to use non-food ingested particles for other purposes, mainly defensive chemical strategies64,65,66,67,68,69,70. In fact, this group of molluscs shows a wide range of sophisticated defensive stratagems that evolved in response to the reduction or completely loose of the shell. They are able to acquire, accumulate and/or modify chemical compounds and/or entire intracellular organelles, extracting from their stomach contents and retaining them in a non-feeding role (e.g., for defensive purposes)71,72,73,74. This raises the question of whether these delicate slugs may have the potential to detect, handle and possibly modify traditional plastic polymers (e.g., non-biodegradable ones). Indeed, Heterobranchia are able not only to recover and accumulate chemical compounds (with different properties) from their diet but can also manipulate these chemicals by modifying them into new ones75,76. This capability also proved to be interesting for potential applications in pharmacology77.
Among heterobranchs, the family Aplysiidae (order Aplysiida) is characterized by relatively large species with a reduced or completely absent shell in adults. They have a more or less selective diet consisting mainly of plant organisms and accidentally of small animals living in the detritus78. Even if the interactions between microplastics and Aplysiidae are almost unknown, several studies have been done on other biological aspects like the ecological, behavioural, chemical, developmental biology and molecular, focused on this interesting heterobranch group79,80.
A resident population of the Aplysiidae species Bursatella leachii Blainville, 1817 lives in the Mar Piccolo of Taranto (Ionian Sea, Apulia, Italy) and it is characterized by seasonal demographic expansions perhaps due to an increase in the trophic availability, a trait shared with other heterobranchs species living in analogous habitats81. This species contains several chemical compounds with different biological activity82,83,84,85, may reach up to 10 cm in length and shows flexibility in prey selection even if it is far from being considered as a ‘non-selective’ bottom feeder86. Given the large availability of plastic litter in the Mar Piccolo sediments, we aimed to: (1) investigate the potential occurrence of microplastics in the stomach contents of B. leachii, (2) collect data on the possible interaction with microplastics, (3) characterize the detected microplastics using SEM/EDX analyses for the first time on Heterobranchia, and (4) create a first preliminary repository of SEM/EDX’s images and microplastic spectra obtained from environmental samples that is ‘non-virgin microplastics’. These data will help to deliver a reference baseline of microplastic contamination for future monitoring studies, not only in areas under severe contamination levels as in the Mar Piccolo of Taranto, but also in less polluted areas under chronical, long-time exposure.
Materials and methods
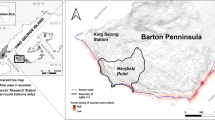
The sampling locality was in the Mar Piccolo (40° 48′ N, 17° 25′ E) in Taranto (Apulian, Ionian Sea, Central Mediterranean Sea), a semi closed highly polluted basin, hosting a high number of industries and intensive farming of mussels and fish (Fig. 1).
Sampling locality objects of the present study. (a) Map of the sampled area in Apulian Ionian Sea with the Mar Piccolo highlighted by the red circle (Taranto, Southern Italy). Map was obtained using Microsoft Paint 3D Version 6.2105.4017.0. (b) External photo of the buoy indicating the submerged underwater site (40° 29′ 05.1′′ N 17° 15′ 08.3′′ E). (c) Fishing nets along the beach. (d) Polluted environment characteristic of Mar Piccolo (9 m depth). (e) Fishing net with the sea slug Dendrodoris limbata (Cuvier, 1804) crawling on it. (f) Sessile tunicate, Clavelina lepadiformis (Müller, 1776), living on a fishing net in the sampling site. Red arrows indicate the plastics present in the studied area.
Six specimens of Bursatella leachii (Fig. 2) were collected by scuba diving at 10 m depth (Fig. 1a,b) and identified according to the external morphological diagnostic characters87,88.
Each sample was observed in situ and in laboratory, photographed using a stereomicroscope and a microscope, preserved in 95% ethanol for future analysis and deposited as voucher at the Department of Science of the Roma Tre University (Rome, Italy). To reduce possible contamination of samples, with the consequent overestimation of microplastic detected, preventive measures were applied. In particular, specimens were manipulated underwater without using gloves, wrapped in aluminium foil before having been placed in a tank and finally transferred to the laboratory where they were suddenly stored in alcohol and analysed. Furthermore, in each step of the laboratory analyses, only glass materials washed with micro-filtered water were used.
Anatomical dissection
Analyses of the internal anatomy of the collected specimens were carried out by anatomical dissection under the stereo microscope at different magnification levels. The digestive system, from the oesophagus (taken right at the end of the gizzard) to the terminal anus, was isolated from the rest of the body and prepared for the next microplastic extraction protocol. Stomachal content was observed at the stereomicroscope and the ingested particles that were undoubtedly not plastics were separated for further detailed observations. The rest of the stomachal content, including visible fibres and microplastic debris, was placed in a separate 50 ml tube.
Microplastic extraction and samples preparation
Prior to carrying out the microplastics extraction and characterization, the digestive system of each specimen was rinsed with pre-filtered (0.22 μm) deionized water and centrifuged to eliminate alcohol used to store them. Subsequently each pellet was incubated with 10% of KOH (w/v) solution prepared using KOH pellets (Sigma-Aldrich, Saint-Quentin-Fallavier, France) and double-distilled water. Then they were placed on an agitation plate (IKA RT15, Staufen, Germany) set at 300 rpm and 60 ± 1 °C for 24 h. After digestion, all samples were filtered on 90 mm diameter GF/C glass microfibre filters (Whatman, Velizy-Villacoublay, France) using a vacuum system. Filters were then placed in closed Petri dishes until subsequent analysis. For the first characterisation, filters were observed under a stereomicroscope (Nikon SMZ25, Tokyo, Japan), allowing the identification of plastic particles. Items with characteristics similar to plastic polymers were characterized by size (< 100 µm; 100–500 µm; ˃500 µm) and colour. Filtered fragments and fibres were then rinsed in distilled water and mounted on double-sided adhesive carbon tabs on aluminium SEM stubs for successive SEM/EDX analyses.
SEM/EDX analyses
SEM/EDX analysis was conducted on individual candidate microplastics selected by optical microscopy from the glass microfibre filters through which the treated B. leachii guts were filtered. SEM/EDX allowed many potential microplastic particles to be screened in a relatively short time. SEM/EDX screening utilized surface morphology and elemental composition to determine whether each particle was potentially a plastic. The analyses were conducted using two different microscopes: The JSM-6480LV Scanning Electron Microscope (JEOL Ltd., Tokyo, Japan) with a Sirius SD Energy Dispersive X-ray Spectometer (iXRF Systems Inc., Houston, USA) (hereafter as SEM-JEOL) and the Sigma 300 VP Field Emission Scanning Electron Microscope (ZEISS, Oberkochen, Germany) (hereafter as FESEM-ZEISS). The former was used to the preliminary morphological assessment and to carry out the detailed microanalysis while the latter to obtain high resolution images useful to the in-depth observations of the morphological details. After checking that there was no sample charging under the electron beam, to avoid the contamination due to chemical artifacts introduced by the metal coating of the samples, these latter were initially analysed without the gold-coating step. The SEM-JEOL provided low resolution imaging of particle surface structures (not shown), as well as elemental composition signatures. Spectra of the chemical composition of the debris analysed were then compared with those already present in literature and related not only to microplastics but also to other organic compounds that cannot be removed by treatment with the KOH solution (like for example cellulose and chitin) but that could anyway be ingested by B. leachii.
To obtain high resolution SEM images, fragments and fibres previously mounted on the aluminum stubs and analysed with EDX, were afterwards gold coated in an Emitech K550x sputter unit, and finally examined by FESEM-ZEISS up to × 5000 magnification. The integration between results obtained by SEM and EDX methods and taking into consideration that samples were previously digested with 10% of KOH (w/v) solution which means that a lot of organic compounds were consequently excluded from the dataset under investigation, allows establishing if the analysed samples were effectively plastics or not.
Compliance with ethical standards
All applicable international, national and/or institutional guidelines for sampling, care and experimental use of organisms for the study have been followed and all necessary approvals have been obtained.
Results
Digestive systems from six individuals of B. leachii (Fig. 2a,b) from the Mar Piccolo of Taranto (Fig. 1b,c) were dissected and isolated (Fig. 3) for preliminary microscopic analyses. Microplastics were found in the stomach contents of all specimens (Fig. 4) and sorted according to their morphologies into fragments and fibres and by size and colour. Fibre particles were fewer than the number of irregular fragments in five out of six analysed stomach contents (Table 1).
Anatomical dissection of Bursatella leachii. (a) Dorsal view of the body after removal of the internal visceral mass. (b) The digestive apparatus (oesophagus, gizzard, stomach, intestine, anus) separated from the rest of the body. (c,d) Images showing the stomach content where are visible the small microplastic debris. an anus, g gizzard, h head, i.b.c. internal body cavity, in intestine, oe oesophagus.
Relative abundance (%) of microplastics detected on glass microfibre filters after digestion and filtration of gastrointestinal tracts of six Nudibranchia and classified according to (a) colour; (b) size and shape and (c) size for each colour; (d) images observed by stereomicroscope of different shapes of MPs found in specimens. Scale bar = 100 μm.
Microplastic extraction and analyses
In the gastrointestinal tract of six B. leachii individuals, many microplastics were detected, visible onto the glass microfilters (Fig. 4a-d). The number and type of microplastics detected in each analysed sample were reported in Table 1. The microplastics size distribution is presented in Fig. 4b. The size class distribution revealed a marked prevalence for particles smaller than 100 µm (54%), followed by particles with a range 100–500 µm (24%) and particles over 500 µm (22%). A total of 11 chromatic components were observed. The black colour dominated with 45.60% of particles found in specimens (Fig. 4a). Other colours representing important proportions were transparent (4.75%), white (11.87%), brown (10.67%), orange (9.92%), blue (6.65%), bi-colour (5.46%), green (4.27%) and small percentage of the other colours. In addition, we reported the size distribution for each colour, as shown in Fig. 4c. The particle sizes varied for every colour, without a predominance of size in the case of the most common colours. Only the small percentage of pink fibres (0.23%) observed on the filter had a length over 500 µm, while the brown particles reached mostly 500 µm.
SEM/EDX analyses
Surface texture created by environmental exposure is one of the primary characteristics that can be used to screen for microplastics by electron microscopy. Morphological analysis of the microplastic particle surfaces often revealed degradation and abrasion signs, suggesting mechanical weathering processes89,90, which were observed in this study. SEM/EDX analyses provided high resolution pictures of the particles surface structure of the fibres and fragments analysed, as well as their elemental composition signatures. This information was used to screen for likely microplastics and rule out non-plastics. Since it is known from literature that the most common kind of plastics, as Polypropylene (PP) and Polyethylene (PE), show a strong Carbon EDX peak90, and considering that we are dealing with plastics extracted from an open environment, possibly affected by all the variety of existing plastics, we searched for spectra showing a significant concentration of Carbon to be the possible candidates for microplastics. Resultant spectra were compared with some reported by published studies carried out in laboratory and using previously known plastics polymers that were used as reference. Examples of spectra from canonical microplastic fragments and fibres are shown in Fig. 5 as well as spectra from different kind of plastic (Fig. 6) where it is easy to see the Carbon peak (C) and other few additional elements characteristic of other plastic types. Spectra obtained were characterized by a high variability that perfectly reflects the huge variety of plastic currently available from natural environment. SEM/EDX analysis from fragments and fibres which were determined to be non-plastic (such as natural fibres, mollusc’s shell fragments and debris of plant organisms) were also shown (Fig. 7).
SEM/EDX images and microanalysis from different kind of plastics. SEM images (a,b,d,e,g,h) and spectra (c,f,i) showing the different kind of microplastics obtained from Bursatella leachii analysed. (a–c) Fibreglass fibre containing Carbon and Oxygen as the most abundant elements. (d–f) fibReglass fibre showing the Silicon peak and Carbon, Oxygen and several other minor elements. (g–i) Fibreglass debris with the Silicon peak followed by Carbon and Oxygen and several other minor elements.
SEM/EDX images and microanalysis from non-plastic materials. SEM images (a,b,d,e,g,h) and spectra (c,f,i) of non-plastic materials obtained from the stomachal content of collected Bursatella leachii specimens. (a–c) natural cotton fibre. (d–f) Part of a mollusc shell made mainly of Calcium Carbonate. (g–i) Piece of plant organism with the cellulose as the main component.
Discussion
Morphological and elemental composition analyses of the stomachal content of Bursatella leachii from a highly polluted environment revealed the presence of microplastic fibres and fragments in all the studied individuals. Despite increased international attention, investigating microplastic in environmental samples is a difficult task, because of its wide range of possible interactions with the living biota and because microplastic includes different organic polymers which can be chemically and mechanically modified by environmental factors like the weather, hydrodynamic forces, solar radiation, the presence of other contaminants, the occurrence of biofouling, etc.91,92,93,94,95,96. The complexity of the processes of cause/effect characterizing microplastic implies the use of different protocols each of them optimized for a specific target of study. Additionally, even if studying microplastic is nowadays of a central importance and several standardized protocols of extraction and analysis have been published97,98,99,100,101,102, the continuous search for the most effective or performing one is still ongoing103,104,105. Furthermore, even though considerable research effort focused on several target species, (mainly vertebrates from fish to mammals)17,19,21, few invertebrates were investigated so far, and, among them, filter-feeder molluscs (bivalves) were mainly studied15,31,39,57. To date, interactions between environmental microplastics and Heterobranchia remained neglected, perhaps due to the difficulties of studying small and infrequent animals that are characterized by soft and very delicate internal anatomy; however, bridging the knowledge gap on these benthic consumers—known to unceasingly explore soft and hard bottoms on the seafloor in search of food and with different trophic preferences—is indeed a highly promising challenge. In fact, the potential of marine Heterobranchia is high since these gastropods are characterized by unique defensive strategies like the ability to accumulate and, in most of the cases, modify, chemical active compounds obtained from the diet. Considering that microplastic is everlasting due to the absence of known multicellular organisms able to digest it, the capacity showed by marine Heterobranchia to modify foreign chemical molecules may be of great interest under a potential biotechnological perspective.
In this framework, we investigated stomachal content from Bursatella leachii, an Aplysiidae living in Mar Piccolo of Taranto (Ionian Sea, Mediterranean Sea), a coastal aquatic environment under high anthropogenic pressures, particularly exposed to plastic pollution (Fig. 1). The combination of the high-resolution SEM morphological observations with the EDX elemental composition of the debris was very useful to investigate and identify microplastics in the digestive trait of B. leachii. In fact, considering that microplastic can bind with other pollutants already present in the environment and taking into consideration that Mar Piccolo of Taranto host several anthropogenic pollutants, it can be hypothesised that the collected microplastic may show an atypical chemical composition, reflected by a non-canonical spectrum. This characteristic would eventually affect results from other kind of techniques like for example the FTIR analyses, since it is based on the perfect match between the spectra investigated and the canonical spectra of plastic already available in the reference public libraries. Being extracted from individuals living in a natural environment and not from ‘in laboratory study’, EDX spectra obtained were characterized by a high variability that perfectly reflects the vast diversity of plastic polymers now recorded in natural environments48.
The data presented here can be considered as preliminary, as they are based on dissection and analysis of six specimens only: a larger sampling will be required to corroborate and enlarge the value of these initial observations. However, the information gathered so far seems to be indicative of a consistent pattern. Firstly, fibres and small fragment particles were abundant in all the Bursatella stomachs analysed. This could be due to a selection made by the individuals with the exclusion of larger fragments and in favour of smaller ones or fibres stuck to the algae they feed on. Anyway, the dominance of fragments instead of fibre particles is of outmost interest and unexpected, since almost all previous studies on benthic animals report the opposite condition. This finding may corroborate the hypothesis of an active choice made by B. leachii, ingesting size- and shape-specific plastic debris and not just the most abundant ones, as usually happen for filter-feeders and/or non-selective detritivore organisms. In fact, fibres are expected to be copious in Mar Piccolo sediments, where they derive from abundant textile waste, degrading fishing lines, and particularly from the plastic nest nets widely used in mussel aquaculture (Fig. 1c–f), a major source of local contamination24. These preliminary data will represent baseline information for future comparative studies. Indeed, the analyses of high-res SEM images, together with the corresponding EDX spectra of the analysed fragments and fibres, highlighted the variability of the pool of debris that could be found in the field, with reference to already published information on compounds belonging to both natural and plastic materials. In fact, there are some natural materials that may resist the KOH digestion and therefore may require a future, in-depth analysis to distinguish them from the microplastic dataset and to avoid misidentifications.
Regarding the microplastic debris found in B. leachii, the canonical spectra that could be identified as plastic are reported in Fig. 5 both related to fragments and fibres. These spectra are consistent with the ones from literature as microplastic90,106. Among these, an important class is that of fibreglass which includes different kind of plastic all of them characterized by Carbon (C), Oxygen (O), and Silicon (Si)107,108 (https://www.nrc.gov/docs/ML0530/ML053040493.pdf) where the glass fibres are commonly added to reinforce plastic structures109,110. This specific kind of plastic is part of the components detected in the present study (Fig. 6). Apart from the elements discussed till now, other interesting and unusual elements were found like Titanium (Ti), Barium (Ba) and Zinc (Zn) (Fig. 6). These elements are used as additives in some types of plastics thus driving a more precise identification111,112. SEM/EDX microanalysis from fragments and fibres which were found to be non-plastic were also shown to be useful reference for further in situ studies focused on animals in natural environments (Fig. 7). In fact, among the samples that were non-plastic, there was a natural cellulosic fibre (cotton) characterized by a typical twisted morphology and the presence of peaks resembling those reported for cellulose (Fig. 7a–c)113,114,115.
Also, we detected debris containing a high concentration of calcium carbonate (CaCO3) (Fig. 7d–f), which is coherent with spectra reported for bivalves and gastropod shells31,32,116,117, tubes of sedentary polychaete worms118 and therefore not associated with plastic. The almost equal concentration of Carbon and Oxygen (C and O) elements detected in a single fragment (Fig. 7g–i), together with the characteristic external morphology, is related to the cellulose and, indeed, to plant organisms and not to plastic. Another important result for consideration must be the absence of Nitrogen (N) in the EDX spectra of our samples. This element in fact is one of the main components of biological compounds (containing proteins and/or peptide bonds) such as egg capsules of the gastropod Rapana venosa (Valenciennes, 1846)119 and eggshells of the Ascarididae nematode Ophidascaris baylisi Baylis (1921)120.
Investigating the variability among plastic materials is essential to fill the gap of knowledge still existing to date, avoiding misidentifications and errors in quantification of microplastic in the environments or into living organisms91,92,93,94,95,96,121. This last point is crucial and constitutes one of the main limits of the recent studies. In fact, most of them are carried out in laboratory and have used already known kinds of plastics which are therefore characterized by a known specific composition, while many other papers focused on plastic obtained by in situ studies but analysed using reference spectra from virgin plastic with known composition and eliminating those which are far from that reference. Anyway, the plastic debris found in a natural environment, and even more in a very polluted one, is not virgin plastic but instead a mixture of organic polymers plus a lot of different other elements that are added as additives or that are independently attracted by plastic due to chemical properties of the plastic itself. In fact, the microplastics found in the stomachs of B. leachii living in Mar Piccolo represent strong evidence of the high level of plastic contamination acting in this semi closed basin and the ease with which it enters the food chain as it is ingested by organisms that largely select the nutrients they eat. Interestingly, none of the stomachs analysed contained detritus, confirming that B. leachii is not a detritivore but selects plant organisms and a lot of other living organisms, in some way selecting them. We cannot exclude, therefore, that the microplastics were deposited on the surface of the prey ingested by the sea slugs. Anyway, it is interesting to note that once in the marine environment, microplastics are colonised by the ‘plastisphere’ as are collectively called the wide variety of microbial communities coating plastic debris and forming biofilms121,122. Considering that it was recently demonstrated the higher preference of some marine filter-feeders to eat microplastics coated with microbial biofilms instead of virgin microplastics, we can speculate the possible trophic preference of B. leachii for microplastics covered by cyanobacteria or other microbial biofilms. Anyway, this important ecological aspect is to date poorly known since most of the studies investigating the impacts of microplastics ingestion by aquatic organisms have currently used virgin plastic particles, which, however, do not reflect the real conditions of the sea121 as the results here reported have also demonstrated. Another consideration can be made regarding the fact that plastic was found in all the specimens analysed independently if they had a lot of food in the digestive apparatus or not. This could be evidence of the possible persistence of plastic that would not be excreted with faeces. Taking into consideration that Heterobranchia have the capability to accumulate exogenous material and chemically modify it for defensive purposes, this observation becomes even more interesting. Further in-depth studies are needed to fulfil the gap of knowledge on this intriguing biological capability with potential highly innovative applications.
In conclusion, this work constitutes a baseline study useful for future in-depth investigations on the diet and faecal contents and for future comparisons with plastics contents from digestive traits of other taxa and with EDX spectra from other materials of difficult identification. Furthermore, it could be a reference database on the composition of locally originated plastics in the Mar Piccolo (due to its typical hydrology) that will be useful to simulate possible future scenarios on other polluted coastal areas and to predict the potential outcome of negative impacts on the biota.
Data availability
All data generated or analysed during this study are included in this published article (and its Supplementary Information files).
References
Derraik, J. G. The pollution of the marine environment by plastic debris: A review. Mar. Pollut. Bull. 44(9), 842–852 (2002).
Gregory, M. R. Environmental implications of plastic debris in marine settings—Entanglement, ingestion, smothering, hangers-on, hitch-hiking and alien invasions. Philos. Trans. R. Soc. B Biol. Sci. 364(1526), 2013–2025 (2009).
Claessens, M., Van Cauwenberghe, L., Vandegehuchte, M. B. & Janssen, C. R. New techniques for the detection of microplastics in sediments and field collected organisms. Mar. Pollut. Bull. 70(1–2), 227–233 (2013).
Auta, H. S., Emenike, C. U. & Fauziah, S. H. Distribution and importance of microplastics in the marine environment: A review of the sources, fate, effects, and potential solutions. Environ. Int. 102, 165–176 (2017).
Zobkov, M. B. & Esiukova, E. E. Microplastics in a Marine Environment: Review of Methods for Sampling, Processing, and Analyzing Microplastics in Water, Bottom Sediments, and Coastal Deposits (2018).
Coyle, R., Hardiman, G. & O’Driscoll, K. Microplastics in the marine environment: A review of their sources, distribution processes, uptake and exchange in ecosystems. Case Stud. Chem. Environ. Eng. 2, 100010 (2020).
Barnes, D. K., Galgani, F., Thompson, R. C. & Barlaz, M. Accumulation and fragmentation of plastic debris in global environments. Philos. Trans. R. Soc. Lond. B Biol. Sci. 364, 1985–1998 (2009).
GESAMP. Sources, Fate and Effects of Microplastics in the Marine Environment: Part 2 of a Global Assessment. A Report to Inform the Second United Nations Environment Assembly, 220 (Joint Group of Experts on the Scientific Aspects of Marine Environmental Protection, 2016).
Kroon, F. J., Motti, C. E., Jensen, L. H. & Berry, K. L. Classification of marine microdebris: A review and case study on fish from the Great Barrier Reef, Australia. Sci. Rep. 8(1), 1–15. https://doi.org/10.1038/s41598-018-34590-6 (2018).
Cole, M., Lindeque, P., Halsband, C. & Galloway, T. Microplastics as contaminants in the marine environment: A review. Mar. Pollut. Bull. 62(12), 2588–2597 (2011).
Cole, M. A novel method for preparing microplastic fibers. Sci. Rep. 6(1), 1–7. https://doi.org/10.1038/srep34519 (2016).
Costa, M. et al. On the importance of size of plastic fragments and pellets on the strandline: A snapshot of a Brazilian beach. Environ. Monit. Assess. 168, 299–304 (2010).
Kershaw, P. J. et al. (eds) GESAMP Guidelines or the Monitoring and Assessment of Plastic Litter and Microplastics in the Ocean, Rep. Stud. GESAMP No. 99 130 (IMO/FAO/UNESCO-IOC/UNIDO/WMO/IAEA/UN/UNEP/UNDP/ISA Joint Group of Experts on the Scientific Aspects of Marine Environmental Protection, 2019).
Lusher, A. L., Welden, N. A., Sobral, P. & Cole, M. Sampling, isolating and identifying microplastics ingested by fish and invertebrates. Anal. Methods 9, 1346 (2017).
Lusher, A., Bråte, I. L. N., Hurley, R., Iversen, K. & Olsen, M. Testing of Methodology for Measuring Microplastics in Blue Mussels (Mytilus spp) and Sediments, and Recommendations for Future Monitoring of Microplastics (R & D-project) (2017).
Laist, D. W. Impacts of marine debris: Entanglement of marine life in marine debris including a comprehensive list of species with entanglement and ingestion records. In Marine debris, 99–139 (Springer, 1997).
Denuncio, P. et al. Plastic ingestion in Franciscana dolphins, Pontoporia blainvillei (Gervais and d’Orbigny, 1844), from Argentina. Mar. Pollut. Bull. 62(8), 1836–1841 (2011).
Do Sul, J. A. I., Santos, I. R., Friedrich, A. C., Matthiensen, A. & Fillmann, G. Plastic pollution at a sea turtle conservation area in NE Brazil: Contrasting developed and undeveloped beaches. Estuar. Coasts 34(4), 814–823 (2011).
Lazar, B. & Gračan, R. Ingestion of marine debris by loggerhead sea turtles, Caretta caretta, in the Adriatic Sea. Mar. Pollut. Bull. 62(1), 43–47 (2011).
Poppi, L. et al. Post-mortem investigations on a leatherback turtle Dermochelys coriacea stranded along the Northern Adriatic coastline. Dis. Aquat. Org. 100(1), 71–76 (2012).
Van Franeker, J. A. et al. Monitoring plastic ingestion by the northern fulmar Fulmarus glacialis in the North Sea. Environ. Pollut. 159(10), 2609–2615 (2011).
Betts, K. Why Small Plastic Particles May Pose a Big Problem in the Oceans 8995–8995 (ACS Publications, 2008).
Cefas, L. Programme 8: Bass gillnet selectivity. Fish. Sci. 09 (2008).
Priscilla, V., Sedayu, A. & Patria, M. P. Microplastic abundance in the water, seagrass, and sea hare Dolabella auricularia in Pramuka Island, Seribu Islands, Jakarta Bay, Indonesia. J. Phys. Conf. Ser. 1402, 033073. https://doi.org/10.1088/1742-6596/1402/3/033073 (2019).
Graham, E. R. & Thompson, J. T. Deposit-and suspension-feeding sea cucumbers (Echinodermata) ingest plastic fragments. J. Exp. Mar. Biol. Ecol. 368(1), 22–29 (2009).
Thompson, R. C. et al. Lost at sea: Where is all the plastic? Science 304(5672), 838–838 (2004).
Hämer, J., Gutow, L., Köhler, A. & Saborowski, R. Fate of microplastics in the marine isopod Idotea emarginata. Environ. Sci. Technol. 48(22), 13451–13458 (2014).
Setälä, O., Fleming-Lehtinen, V. & Lehtiniemi, M. Ingestion and transfer of microplastics in the planktonic food web. Environ. Pollut. 185, 77–83 (2014).
Cole, M. et al. Microplastics alter the properties and sinking rates of zooplankton faecal pellets. Environ. Sci. Technol. 50(6), 3239–3246 (2016).
Gutow, L., Eckerlebe, A., Giménez, L. & Saborowski, R. Experimental evaluation of seaweeds as a vector for microplastics into marine food webs. Environ. Sci. Technol. 50(2), 915–923 (2016).
Naji, A., Nuri, M. & Vethaak, A. D. Microplastics contamination in molluscs from the northern part of the Persian Gulf. Environ. Pollut. 235, 113–120 (2018).
Ding, J. et al. Detection of microplastics in local marine organisms using a multi-technology system. Anal. Methods 11(1), 78–87 (2019).
Gniadek, M. & Dąbrowska, A. The marine nano-and microplastics characterisation by SEM-EDX: The potential of the method in comparison with various physical and chemical approaches. Mar. Pollut. Bull. 148, 210–216 (2019).
Dąbrowska, A. A roadmap for a plastisphere. Mar. Pollut. Bull. 167, 112322 (2021).
Ebere, E. C. & Ngozi, V. E. Microplastics, an emerging concern: A review of analytical techniques for detecting and quantifying microplatics. Anal. Methods Environ. Chem. J. 2(2), 13–30 (2019).
Mariano, S., Tacconi, S., Fidaleo, M., Rossi, M. & Dini, L. Micro and nanoplastics identification: Classic methods and innovative detection techniques. Front. Toxicol. https://doi.org/10.3389/ftox.2021.636640 (2021).
Ferrante, M. et al. Microplastics in fillets of Mediterranean seafood. A risk assessment study. Environ. Res. 204, 112247 (2022).
Li, J. et al. Characterization, source, and retention of microplastic in sandy beaches and mangrove wetlands of the Qinzhou Bay, China. Mar. Pollut. Bull. 136, 401–406 (2018).
Liu, J. et al. Pollution characteristics of microplastics in mollusks from the coastal Area of Yantai. China. Bull. Environ. Contamin. Toxicol. 107, 1–7 (2021).
Tarjuelo, I., Posada, D., Crandall, K., Pascual, M. & Turon, X. Cryptic species of Clavelina (Ascidiacea) in two different habitats: Harbours and rocky littoral zones in the northwestern Mediterranean. Mar. Biol. 139(3), 455–462 (2001).
Brunetti, R. & Mastrototaro, F. Botrylloides pizoni, a new species of Botryllinae (Ascidiacea) from the Mediterranean Sea R. Zootaxa 3258(1), 28–36 (2012).
Beli, E. et al. The zoogeography of extant rhabdopleurid hemichordates (Pterobranchia: Graptolithina), with a new species from the Mediterranean Sea. Invertebr. Syst. 32(1), 100–110 (2018).
Chimienti, G., Angeletti, L., Furfaro, G., Canese, S. & Taviani, M. Habitat, morphology and trophism of Tritonia callogorgiae sp. nov., a large nudibranch inhabiting Callogorgia verticillata forests in the Mediterranean Sea. Deep Sea Res. I Oceanogr. Res. Pap. 165, 103364 (2020).
Furfaro, G. & Mariottini, P. A new Dondice Marcus Er. 1958 (Gastropoda: Nudibranchia) from the Mediterranean Sea reveals interesting insights into the phylogenetic history of a group of Facelinidae taxa. Zootaxa 4731(1), 1–22. https://doi.org/10.11646/zootaxa.4731.1.1 (2020).
Cózar, A. et al. Plastic accumulation in the Mediterranean Sea. PLoS ONE 10(4), e0121762. https://doi.org/10.1371/journal.pone.0121762 (2015).
Sharma, S., Sharma, V. & Chatterjee, S. Microplastics in the Mediterranean Sea: Sources, pollution intensity, sea health, and regulatory policies. Front. Mar. Sci. 8, 634934. https://doi.org/10.3389/fmars.2021.634934 (2021).
Pinardi, N. & Masetti, E. Variability of the large scale general circulation of the Mediterranean Sea from observations and modelling: A review. Palaeogeogr. Palaeoclimatol. Palaeoecol. 158(3–4), 153–173 (2000).
Suaria, G. et al. The Mediterranean Plastic soup: Synthetic polymers in Mediterranean surface waters. Sci. Rep. 6(1), 1–10 (2016).
Vianello, A. et al. Microplastic particles in sediments of Lagoon of Venice, Italy: First observations on occurrence, spatial patterns and identification. Estuar. Coast. Shelf. Sci. 130, 54–61. https://doi.org/10.1016/j.ecss.2013.03.022 (2013).
Parenzan, P. Il Mar Piccolo di Taranto. Ciem. Comm. Taranto (1984).
Cavallo, R. A. & Stabili, L. Presence of vibrios in seawater and Mytilus galloprovincialis (Lam.) from the Mar Piccolo of Taranto (Ionian Sea). Water Res. 36(15), 3719–3726 (2002).
Cardellicchio, N. et al. Organic pollutants (PAHs, PCBs) in sediments from the Mar Piccolo in Taranto (Ionian Sea, Southern Italy). Mar. Pollut. Bull. 55(10–12), 451–458 (2007).
Cardellicchio, N., Annicchiarico, C., Di Leo, A., Giandomenico, S. & Spada, L. The Mar Piccolo of Taranto: An interesting marine ecosystem for the environmental problems studies. Environ. Sci. Pollut. Res. 23(13), 12495–12501 (2016).
Tursi, A. et al. Mega-litter and remediation: The case of Mar Piccolo of Taranto (Ionian Sea). Rendiconti Lincei. Sci. Fisiche e Nat. 29(4), 817–824 (2018).
Mastrototaro, F. et al. Benthic diversity of the soft bottoms in a semi-enclosed basin of the Mediterranean Sea. Marine Biological Association of the United Kingdom. J. Mar. Biol. Assoc. U.K. 88(2), 247 (2008).
Li, J. et al. Using mussel as a global bioindicator of coastal microplastic pollution. Environ. Pollut. 244, 522–533 (2019).
Corami, F. et al. Evidence of small microplastics (< 100 μm) ingestion by Pacific oysters (Crassostrea gigas): A novel method of extraction, purification, and analysis using Micro-FTIR. Mar. Pollut. Bull. 160, 111606 (2020).
De-la-Torre, G. E., Apaza-Vargas, D. M. & Santillán, L. L. Microplastic ingestion and feeding ecology in three intertidal mollusk species from Lima, Peru. Rev. Biol. Mar. Oceanogr. 55(2), 167–171 (2020).
Jiang, Y. et al. A review of microplastic pollution in seawater, sediments and organisms of the Chinese coastal and marginal seas. Chemosphere 286, 131677 (2021).
Haszprunar, G. The heterobranchia—A new concept of the phylogeny of the higher Gastropoda. J. Zool. Syst. Evol. Res. 23(1), 15–37 (1985).
Wägele, H., Klussmann-Kolb, A., Vonnemann, V. & Medina, M. Heterobranchia I: The Opisthobranchia. In Phylogeny and Evolution of the Mollusca (eds Ponder, W. F. & Lindberg, D.) 385–408 (University of California Press, 2008).
Prkic, J. et al. First record of Calma gobioophaga Calado and Urgorri, 2002 (Gastropoda: Nudibranchia) in the Mediterranean Sea. Mediterr. Mar. Sci. 15(2), 423–428 (2014).
Furfaro, G., Trainito, E., De Lorenzi, F., Fantin, M. & Doneddu, M. Tritonia nilsodhneri Marcus Ev., 1983 (Gastropoda, Heterobranchia, Tritoniidae): First records for the Adriatic Sea and new data on ecology and distribution of Mediterranean populations. Acta Adriat. 58, 2 (2017).
Thompson, T. E. Studies on ontogeny of Tritonia hombergi Cuvier (Gastropoda: Opisthobranchia). Philos. Trans. R. Soc. Lond. B 245, 171–218. https://doi.org/10.1098/rstb.1962.0009 (1962).
Cattaneo-Vietti, R., Angelini, S. & Bavestrello, G. Skin and gut spicules in Discodoris atromaculata (Bergh, 1880) (Mollusca: Nudibranchia). Bollettino Malacol. 28, 173–180 (1993).
Cattaneo-Vietti, R., Angelini, S., Gaggero, L. & Lucchetti, G. Mineral composition of nudibranch spicules. J. Molluscan Stud. 61(3), 331–337. https://doi.org/10.1093/mollus/61.3.331 (1995).
Garese, A., García-Matucheski, S., Acuña, F. H. & Muniain, C. Feeding behavior of Spurilla sp. (Mollusca: Opisthobranchia) with a description of the kleptocnidae sequestered from its sea anemone prey. Zool. Stud. 51(7), 905–912 (2012).
Braga, T. et al. Bursatella leachii from Mar Menor as a source of bioactive molecules: Preliminary evaluation of the nutritional profile, in vitro biological activities and fatty acids contents. J. Aquat. Food Prod. Technol. 26(10), 1337–1350 (2017).
Willis, T. J. et al. Kleptopredation: A mechanism to facilitate planktivory in a benthic mollusc. Biol. Let. 13, 20170447. https://doi.org/10.1098/rsbl.2017.0447 (2017).
Goodheart, J. A. et al. Comparative morphology and evolution of the cnidosac in Cladobranchia (Gastropoda: Heterobranchia: Nudibranchia). Front. Zool. 15(1), 1–18. https://doi.org/10.1186/s12983-018-0289-2 (2018).
Marin, A. & Ros, J. Chemical defenses in Sacoglossan Opisthobranchs: Taxonomic trends and evolutive implications. Sci. Mar. 67(Suppl. 1), 227–241 (2004).
Wägele, H., Ballestero, M. & Avila, C. Defensive glandular structures in opisthobranch molluscs—From histology to ecology. Oceanogr. Mar. Biol. Annu. Rev. 44, 197–276 (2006).
Pavlik, J. R. Antipredatory defensive roles of natural products from marine invertebrates. In Handbook of Marine Natural Products Vol. 12 (eds Fattorusso, E. et al.) 677–710 (Springer, 2012).
Avila, C., Nuñez-Pons, L. & Moles, J. From the tropics to the poles chemical defense strategies in sea slugs (Mollusca: Heterobranchia). In Chemical Ecology: The Ecological Impact of Marine Natural Products (eds Puglisi, M. P. & Becerro, M. A.) 93 (CRC Press, 2018).
Capper, A., Tibbetts, I. R., O’Neil, J. M. & Shaw, G. R. The fate of Lyngbya majuscula toxins in three potential consumers. J. Chem. Ecol. 31(7), 1595–1606 (2005).
Dean, L. J. & Prinsep, M. R. The chemistry and chemical ecology of nudibranchs. Nat. Prod. Rep. 34(12), 1359–1390 (2017).
Simmons, T. L., Andrianasolo, E., McPhail, K., Flatt, P. & Gerwick, W. H. Marine natural products as anticancer drugs. Mol. Cancer Ther. 4(2), 333–342 (2005).
Klussmann-Kolb, A. Phylogeny of the Aplysiidae (Gastropoda, Opisthobranchia) with new aspects of the evolution of seahares. Zool. Scr. 33, 439–462 (2004).
Willan, R. C. Phylogenetic systematics of the Notaspidea (Opisthobranchia) with reappraisal of families and genera. Am. Malacol. Bull. 5, 215–241 (1987).
Medina, M. & Walsh, P. J. Molecular systematics of the order Anaspidea based on mitochondrial DNA sequences (12S, 16S, and COI). Mol. Phylogenet. Evol. 15, 41–58 (2000).
Furfaro, G., De Matteo, S., Mariottini, P. & Giacobbe, S. Ecological notes of the alien species Godiva quadricolor (Gastropoda: Nudibranchia) occurring in Faro Lake (Italy). J. Nat. Hist. 52(11–12), 645–657 (2018).
Appleton, D. R., Sewell, M. A., Berridge, M. V. & Copp, B. R. A new biologically active malyngamide from a New Zealand collection of the sea hare Bursatella leachii. J. Nat. Prod. 65(4), 630–631 (2002).
Rajaganapathi, J., Kathiresan, K. & Singh, T. P. Purification of anti-HIV protein from purple fluid of the sea hare Bursatella leachii de Blainville. Mar. Biotechnol. 4(5), 447–453 (2002).
Suntornchashwej, S., Chaichit, N., Isobe, M. & Suwanborirux, K. Hectochlorin and morpholine derivatives from the Thai Sea Hare, Bursatella leachii. J. Nat. Prod. 68(6), 951–955 (2005).
Dhahri, M. et al. Extraction, characterization, and anticoagulant activity of a sulfated polysaccharide from Bursatella leachii viscera. ACS Omega 5(24), 14786–14795 (2020).
Clarke, C. L. The population dynamics and feeding preferences of Bursatella leachii (Opisthobranchia: Anaspidea) in northeast Queensland, Australia. Rec. West. Austral. Museum Suppl. 69, 11–21 (2006).
Blainville, H. M. D. de. Bursatella, p. 138, in: Dictionnaire des Sciences Naturelles (F. Cuvier, ed.), Vol. 5, Supplément. Levrault, Strasbourg & Le Normant, Paris (1817).
Trainito, E. & Doneddu, M. Nudibranchi del Mediterraneo 2nd edn, 192 (Il Castello, 2014).
Zbyszewski, M., Corcoran, P. L. & Hockin, A. Comparison of the distribution and degradation of plastic debris along shorelines of the Great Lakes, North America. J. Great Lakes Res. 40(2), 288–299 (2014).
Wang, Z. M., Wagner, J., Ghosal, S., Bedi, G. & Wall, S. SEM/EDS and optical microscopy analyses of microplastics in ocean trawl and fish guts. Sci. Total Environ. 603, 616–626 (2017).
Gewert, B., Plassmann, M. & MacLeod, M. Pathways for degradation of plastic polymers floating in the marine environment. Environ. Sci. Process. Impacts 17, 1513–1521 (2015).
Gewert, B., Plassmann, M., Sandblom, O. & MacLeod, M. Identification of chain scission products released to water by plastic exposed to ultraviolet light. Environ. Sci. Technol. Lett. 5, 272–276 (2018).
Lang, M. et al. Fenton aging significantly affects the heavy metal adsorption capacity of polystyrene microplastics. Sci. Total Environ. 722, 137762 (2020).
Ding, L., Mao, R., Ma, S., Guo, X. & Zhu, L. High temperature depended on the ageing mechanism of microplastics under different environmental conditions and its effect on the distribution of organic pollutants. Water Res. 174, 115634 (2020).
Wang, F. et al. The influence of polyethylene microplastics on pesticide residue and degradation in the aquatic environment. J. Hazard. Mater. 394, 122517 (2020).
Ouyang, Z. et al. The aging behavior of polyvinyl chloride microplastics promoted by UV-activated persulfate process. J. Hazard. Mater. 424, 127461 (2022).
Dehaut, A. et al. Microplastics in seafood: Benchmark protocol for their extraction and characterization. Environ. Pollut. 215, 223–233 (2016).
Besley, A., Vijver, M. G., Behrens, P. & Bosker, T. A standardized method for sampling and extraction methods for quantifying microplastics in beach sand. Mar. Pollut. Bull. 114(1), 77–83 (2017).
Karami, A. et al. A high-performance protocol for extraction of microplastics in fish. Sci. Total Environ. 578, 485–494 (2017).
Caron, A. G. et al. Ingestion of microplastic debris by green sea turtles (Chelonia mydas) in the Great Barrier Reef: Validation of a sequential extraction protocol. Mar. Pollut. Bull. 127, 743–751 (2018).
Piarulli, S. et al. Microplastic in wild populations of the omnivorous crab Carcinus aestuarii: A review and a regional-scale test of extraction methods, including microfibres. Environ. Pollut. 251, 117–127 (2019).
Pfohl, P. et al. Microplastic extraction protocols can impact the polymer structure. Microplast. Nanoplast. 1(1), 1–13 (2021).
Qiu, Q. et al. Extraction, enumeration and identification methods for monitoring microplastics in the environment. Estuar. Coast. Shelf Sci. 176, 102–109 (2016).
Lusher, A. L., Munno, K., Hermabessiere, L. & Carr, S. Isolation and extraction of microplastics from environmental samples: An evaluation of practical approaches and recommendations for further harmonization. Appl. Spectrosc. 74(9), 1049–1065 (2020).
Bellasi, A., Binda, G., Pozzi, A., Boldrocchi, G. & Bettinetti, R. The extraction of microplastics from sediments: An overview of existing methods and the proposal of a new and green alternative. Chemosphere 278, 130357 (2021).
Essa, A. M. & Khallaf, M. K. Antimicrobial potential of consolidation polymers loaded with biological copper nanoparticles. BMC Microbiol. 16(1), 1–8 (2016).
Etcheverry, M., Ferreira, M. L., Capiati, N. J., Pegoretti, A. & Barbosa, S. E. Strengthening of polypropylene–glass fiber interface by direct metallocenic polymerization of propylene onto the fibers. Compos. A Appl. Sci. Manuf. 39(12), 1915–1923 (2008).
Ivanič, A., Kravanja, G., Kidess, W., Rudolf, R. & Lubej, S. The influences of moisture on the mechanical, morphological and thermogravimetric properties of mineral wool made from basalt glass fibers. Materials 13(10), 2392 (2020).
Kavad, B. V., Pandey, A. B., Tadavi, M. V. & Jakharia, H. C. A review paper on effects of drilling on glass fiber reinforced plastic. Procedia Technol. 14, 457–464 (2014).
Alsayed, S. H., Al-Salloum, Y. A. & Almusallam, T. H. Performance of glass fiber reinforced plastic bars as a reinforcing material for concrete structures. Compos. B Eng. 31(6–7), 555–567 (2000).
Fries, E. et al. Identification of polymer types and additives in marine microplastic particles using pyrolysis-GC/MS and scanning electron microscopy. Environ. Sci. Process Impacts 15(10), 1949–1956 (2013).
Turner, A. & Filella, M. The influence of additives on the fate of plastics in the marine environment, exemplified with barium sulphate. Mar. Pollut. Bull. 158, 111352 (2020).
Barathi, M., Kumar, A. S. K. & Rajesh, N. Efficacy of novel Al–Zr impregnated cellulose adsorbent prepared using microwave irradiation for the facile defluoridation of water. J. Environ. Chem. Eng. 1(4), 1325–1335 (2013).
Bahsis, L. et al. Cellulose-copper as bio-supported recyclable catalyst for the clickable azide-alkyne [3+2] cycloaddition reaction in water. Int. J. Biol. Macromol. 119, 849–856 (2018).
Ibrahim, N. A., Eid, B. M., Abd El-Aziz, E., Abou Elmaaty, T. M. & Ramadan, S. M. Multifunctional cellulose-containing fabrics using modified finishing formulations. RSC Adv. 7(53), 33219–33230 (2017).
Van, H. T., Le Sy, H., Nguyen, T. M. L. & Nguyen, D. K. Application of Mussell-derived biosorbent to remove NH 4+ from aqueous solution: Equilibrium and Kinetics. SN Appl. Sci. 3(4), 1–12 (2021).
Lakshmanna, B. et al. Data on Molluscan Shells in parts of Nellore Coast, southeast coast of India. Data Brief 16, 705–712 (2018).
Taylor, P. D., Vinn, O., Kudryavtsev, A. & Schopf, J. W. Raman spectroscopic study of the mineral composition of cirratulid tubes (Annelida, Polychaeta). J. Struct. Biol. 171(3), 402–405 (2010).
Schröder, V. et al. Micromorphological details and identification of chitinous wall structures in Rapana venosa (Gastropoda, Mollusca) egg capsules. Sci. Rep. 10(1), 1–13 (2020).
Ngamniyom, A., Wongroj, W., Karnchaisri, K. & Siriwattanarat, R. Ophidascaris baylisi (Nematoda: Ascarididae): Scanning electron microscopic study of the adult surface with ultrastructure and chemical composition analysis of eggshells. Sci. Technol. Asia 26, 189–198 (2021).
Fabra, M. et al. The plastic Trojan horse: Biofilms increase microplastic uptake in marine filter feeders impacting microbial transfer and organism health. Sci. Total Environ. 797, 149217 (2021).
Jacquin, J. et al. Microbial ecotoxicology of marine plastic debris: A review on colonization and biodegradation by the “Plastisphere”. Front. Microbiol. 10, 865 (2019).
Acknowledgements
The Authors are grateful to Bernard Picton (Northern Ireland, UK) help with the English language editing of the manuscript. GF wish to thank the Italian Ministry of Education, University and Research (MIUR, PON 2014-2020, Grant AIM 1848751-2, Linea 2) for support. The authors would like to thank the anonymous reviewers who helped to improve the quality of the paper.
Funding
GF is supported by funds from the Italian Ministry of Education, University and Research (MIUR, PON 2014-2020, Grant AIM 1848751-2, Linea 2).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by G.F., M.D. and S.M. The first draft of the manuscript was written by Giulia Furfaro and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Furfaro, G., D’Elia, M., Mariano, S. et al. SEM/EDX analysis of stomach contents of a sea slug snacking on a polluted seafloor reveal microplastics as a component of its diet. Sci Rep 12, 10244 (2022). https://doi.org/10.1038/s41598-022-14299-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-14299-3
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.