Abstract
Women with cardiovascular disease may be more susceptible to concomitant mental health problems, such as depression and cognitive decline. Exercise training has beneficial effects on the cardiovascular system as well as on mental functions. Aim of the present study was to study the effects of exercise training on heart, brain and behavior in the isoproterenol (ISO) model in middle-aged female rats. Twelve months old female Wistar rats were submitted to ISO injections (70 mg/kg s.c., on two consecutive days) or received saline. One week later, rats were assigned to either exercise training (treadmill running) or control handling for five weeks. During the last 7 days, tests were performed regarding depressive-like behavior and cognitive function. Then, rats were sacrificed and heart and brains were dissected for (immuno)histochemistry. ISO-induced cardiac effects were eminent from cardiac fibrosis and declined cardiac function. Exercise training reversed cardiac damage and partly restored ISO-induced cardiac dysfunction. However, ISO treatment could not be associated with neuroinflammation, nor impaired hippocampal neurogenesis or neuronal function. Accordingly, no cognitive impairment or depressive-like behavior were observed. Actually, hippocampal microglia hyper-ramification was observed after ISO. Exercise left neuroinflammation and behavior merely unaltered, and even reduced neuronal function. Our data indicated that the cardiac damage after ISO in middle-aged female rats, and the subsequent beneficial effects of five weeks exercise training on the heart, were not reflected in changes in the brain nor in altered behavior.
Similar content being viewed by others
Introduction
Cardiovascular disease is often associated with mental health problems, such as depression and cognitive decline. Major depression was found in 15–20% of cardiovascular disease patients, while even up to 65% reported symptoms of depression1,2. Similarly, cognitive decline or dementia was increased (odds ratio of 1.45) in patients with coronary artery disease3. Moreover, women were indicated at higher risk for heart-failure-associated depression4,5, but higher prevalence of cognitive decline is reported for both women6, and men5. This reduced mental health is not an innocent bystander in cardiovascular disease since it is associated with increased morbidity and mortality7,8. However, the pathophysiology of the heart-brain interaction4,5 is far from fully understood. We hypothesize that the inflammatory response necessary for infarct healing can become derailed and reflected in the brain as neuroinflammation, associated with reduced mental health9. Although evidence supported a key role for inflammation-neuroinflammation10, efficacy of anti-inflammatory treatment so far is poor.
Animal studies have contributed to build up a better understanding of this heart-brain interaction. We11 and others12,13 reported depressive-like behavior in rodents after coronary artery ligation-induced heart failure, which was sensitive to cardiovascular14 as well as different types of brain-targeted treatment15,16,17,18,19. In addition, decline of cognitive performance was observed in this model20. However, the coronary artery ligation model requires major thoracic surgery, and we previously showed that the outcome in this model could be attributed to the combined effect of surgery for coronary artery ligation and effect of the ligation itself20. Therefore, in the present study, the isoproterenol (ISO)-induced myocardial infarction model was used21. Not many behavioral studies have been performed in this model, but reduced exploratory behavior22 and cognitive impairment23 suggest relevant behavioral consequences. In a recent study, decreased open field exploration and declined sucrose preference were observed shortly after ISO24. In these studies, an imbalanced pro- and anti-oxidant system22 or mitochondrial function23, rather than neuroinflammation, were observed as underlying mechanism. Moreover, all three behavioral studies were only performed in male rats.
Exercise training is well-known for its positive impact on physical and mental well-being25, and this beneficial influence could be associated with anti-inflammatory mechanisms26, as well as increased brain derived neurotrophic factor (BDNF) expression27,28. The anti-inflammatory properties of exercise could balance the activated inflammation-neuroinflammation and thereby improve cognition and mood29. Similarly, exercise may improve cognitive function and mood by counteracting a declined BDNF expression30. In the ISO model, beneficial effects of physical exercise were observed31,32, but studies mainly focused on cardiac aspects. Nevertheless, exercise training could prevent upregulation of 18 cytokines after ISO32, supporting anti-inflammatory potential of exercise training in this model. Exercise training from 12 weeks before ISO injections to 7 days after ISO reduced responses of proinflammatory TNFα and IL6, while IL10 response was increased33. However, effects of exercise training on mood and cognition are fairly unexplored in the ISO model.
Aim of the present study was to evaluate the effects of exercise training on heart, brain and behavior in the ISO model in middle-aged female rats.
Methods
Animals and experimental design
Forty-seven middle-aged (12 months old) female Wistar rats were obtained from the breeding colony of the University of Physical Education, Hungary. Animals were housed in groups of 2 or 3 in cages of 30 * 42 * 20 cm with sawdust as bedding. Rats were kept in the conventional animal facility of University of Physical Education, Hungary in a room with 22 ± 2 °C and humidity of 50 ± 10%. Light was provided from 7 am to 7 pm CEST. Standard rodent chow (LT/R, Innovo Ltd., Gödöllő, Hungary) and tap water were provided ad libitum. All methods were performed in accordance with the ARRIVE guidelines. All experiments were performed in accordance with relevant guidelines and regulations/legislations. The experiments were conducted under and approved by the general license for animal experiments of the laboratory of Physical Education, University of Budapest, Hungary, under license number TE-KEB/No3/2020.
Rats were randomized to 4 experimental groups; rats were treated with isoproterenol (n = 27) to induce heart lesions, or received saline injections (n = 20), and after one week of recovery, half of the survivors were subjected to five weeks of treadmill running while the other halve received control handling (sedentary) (n = 10 per group). Exploratory behavior and cognitive performance were assessed at the last 7 days of the training period. After completion of all tests, animals were anesthetized and echocardiographic measurements of cardiac function were obtained. Subsequently, rats were sacrificed, and heart and brain tissues were collected for further analyses of cardiac damage, neuroinflammation and neuronal function.
Cardiac damage
Cardiac damage was chemically induced by isoproterenol hydrochloride (C11H7NO3·HCl: ISO). ISO is a non-selective β-adrenoceptor agonist that mimics the histological, physical and endocrinological events of human myocardial infarction presumably by myocardial hyperactivity induced ischemia and energy depletion34. Rats were injected subcutaneously with ISO (Tokyo Chemical Industry Co., Ltd., Tokyo, Japan) in a dose of 70 mg/kg dissolved in 1 ml/kg saline. Control animals received 1 ml/kg saline. Both groups received 2 injections with 24 h in between. The ISO protocol was based on previous studies23,35. Two injections of ISO with 24 h in between increased cytokine production leading to cardiac fibrosis36, left ventricular hypertrophy and dilatation, and ultimately heart failure37.
Tread-mill running
Both control and ISO treated rats were randomly assigned to treadmill running or sedentary controls. Running was performed on a six-lane rat treadmill (Tartonik Elektronika, Italy) with individual lanes of 12 * 54 * 13 cm. The rats were stimulated to stay on the treadmill by cardboard pushers. No electric shocks were used to motivate the animals. The training program lasted for 5 weeks, 5 times per week on each weekday. On the first week of the training program rats were habituated to running: on the first day, rats started with 10 min of running with a maximal speed of 10 m/min which was gradually increased to 30 min and maximal speed of 18 m/min (moderate intensity; approximately 65% of VO2max) by the fifth day. For the following four weeks each running session lasted 30 min35.
Behavior
Behavioral tests were carried out during the last 7 days of the intervention. Interest in environment and anxiety/depressive like behavior was assessed with open field exploration (OF). Regarding cognition, short-term memory was tested in the novel object recognition (NOR) and the novel location recognition test (NLR). All tests were carried out in a quiet and clean test room (between 10 and 12 a.m.) with the same temperature and humidity parameters as the housing room.
All tests were recorded with a digital video camera (Canon Legria HFR106, Canon Inc., Tokyo, Japan) and stored on a memory card for later off-line analyses using Eline software (University of Groningen, the Netherlands).
Open field
Open field (OF) exploration test was performed to assess exploratory and anxiety related behavior11. A round shaped arena (diameter of 80 cm) was divided into an inner circle (diameter of 32 cm; center area), and an outer annulus (wall area) by black circular lines, and surrounded by a 45 cm tall wall. Animals were placed in the arena and allowed to explore for 5 min. After each animal the arena was cleaned with 70% ethanol to remove smell cues. Locomotor activity was estimated from the number of line crossings. Time spent in the wall area was obtained using Eline software (University Groningen, the Netherlands). More locomotor activity was regarded as more exploration and therefore more interest in the environment. More time in the wall area was regarded as choice for the safe area and therefore sign of anxious-depressive like behavior.
Novel object and novel location recognition
The novel object recognition test (NOR) was performed to assess short term visual memory, which depends primarily on prefrontal cortex function38, while the novel location recognition test (NLR) determined short term spatial memory, associated with hippocampal activity38. The two memory tests were combined in one protocol38 and took place in a black box of 45 * 55 * 50 cm. The combined test consisted of 4 phases, each of 3 min, with 1 min in between: (1) in the habituation phase the animal was placed in the test box and allowed 3 min to get accustomed to the settings, (2) in the exploration phase the rat was presented two identical objects, (3) in the novel location phase the two identical objects were presented again but one of them on a different location than in the previous phase, (4) in the novel object phase one of the two identical objects were replaced by a different object and put to the same location as in the preceding phase. Between the phases, the objects were removed and cleaned with 70% ethanol to remove smell cues. After each animal the test box and objects were also cleaned with 70% ethanol. All phases were recorded by a digital video camera (Canon Legria HFR106, Canon Inc., Tokyo, Japan). Time spent with exploring the objects were measured using Eline software (University of Groningen, the Netherlands). Preference for the novel location or the novel object was calculated by dividing the time spent exploring the novel location or novel object by the time spent exploring both objects, while 50% indicated chance level = no recognition. Animals who did not explore the objects or only one of them were excluded at the final statistical analysis.
Cardiac function
Cardiac function was evaluated with transthoracic echocardiography in a subgroup of rats, randomly chosen from each experimental group, because of limited access to the ECHO machine. Rats were anesthetized with pentobarbital (60 mg/kg, ip), and placed in supine position on heating pads (37 °C core temperature). Standard two-dimensional and M-mode long- and short-axis images at the midpapillary level were acquired using a 13 MHz linear transducer (12L-RS; GE Healthcare, Horten, Norway) connected to a commercially available system (Vivid i; GE Healthcare). Images were analyzed using dedicated software (EchoPac v113; GE Healthcare). Heart rate was obtained. From the images acquired, stroke index (stoke volume indexed to body weight), cardiac index (cardiac output indexed to body weight = stroke index times heart rate) and left ventricular ejection fraction were calculated.
Tissue collection and processing
At the end of the experiment rats were terminally anaesthetized with 6% sodium pentobarbital solution injected intraperitoneally (2 ml/kg) and perfused transcardially with heparinized (1 ml/l) 0.9% saline. Heart and brain tissues were dissected. Brain and heart tissue was immersion fixated in 4% buffered formaldehyde freshly depolymerized from paraformaldehyde. After 4 days, tissue was washed in 0.01 M phosphate buffered saline (PBS), dehydrated using a 30% sucrose solution, and subsequently quickly frozen in liquid nitrogen and stored at -80 °C until further processing. Microscopical sections were cut. Heart sections were placed on glass immediately after cutting, and processed for histochemical staining of collagen. For brain tissue, free floating sections were stored in 0.01 M (PBS) containing 0.1% sodium azide at 4 °C till further processing for immunohistochemistry. In a subgroup of rats (n = 5–6 per group), randomly chosen from each experimental group, immunohistochemistry staining was performed to visualize microglia, immature neurons (double cortin positive cells) and brain derived neurotrophic factor expression, as has been described previously38.
Immunohistochemistry
Cardiac collagen
Since ISO was anticipated to cause focal myocardial infarcts, percentage collagen was used to measure cardiac damage. For that, 25 µm thick transverse slices at mid-ventricular level of the heart were stained with Sirius red (Sigma, Aldrich) and fast green as counterstaining20. Colour pictures were taken. Image analysis (Image Pro plus, USA) was used to measure the collagen positive (red) area and was expressed as percentage of total left ventricular tissue area.
Microglia
Microglia activity was used as measure for neuroinflammation. To visualize microglia, immunohistochemical staining of ionized calcium binding adaptor molecule 1 (IBA-1) was performed, as described in detail previously39. Briefly, after pretreated with 0.3% H2O2 for 20 min., sections were incubated for 3 days with 1:2,500 rabbit-anti IBA-1 (Wako, Neuss, Germany) in 2% bovine serum albumin, 0.1% triton X-100 at 4 °C, followed by a 1 h incubation with 1:500 goat-anti rabbit secondary antibody (Jackson, Wet Grove, USA) at room temperature. The sections were then incubated for 2 h with avidin–biotin peroxidase complex (Vectastain ABC kit, Vector, Burlingame, USA) at room temperature. Labeling was visualized by using a 0.075 mg/mL diaminobenzidine (DAB) solution activated with 0.1% H2O2. All dilutions were made in 0.01 mol/L PBS. All sections were thoroughly rinsed 4 times with 0.01 mol/L PBS between staining steps. Sections were mounted onto glass slides in a 1% gelatin solution and dehydrated through gradients of ethanol and xylol solutions. Photographs were taken from the prefrontal cortex (Pfc), the paraventricular nucleus of the hypothalamus (PVN), and the dorsal hippocampus (hippocampus; CA1, CA3, Dentate Gyrus and Hilus areas) at 200 × magnification. Microglia morphology was analyzed (Image Pro Plus, USA) according to our previous publication39, regarding coverage, density, cell size, cell body area and processes area. Microglia activity was calculated as cell body area/total cell size39.
Neurogenesis
Double Cortin (DCX) staining was used to obtain a measure for hippocampal neurogenesis, as described in detail elsewhere38. Images of DCX stained sections of the dentate gyrus of the hippocampus were taken at 50 × magnification. The number of labeled neuronal cell bodies was counted manually by two independent researchers blinded for the experimental groups, and corrected for the length of the DG (Image Pro Plus, USA).
Brain-derived neurotrophic factor (BDNF)
For brain function, brain slices were stained with Brain Derived Neurotrophic Factor (BDNF) antibody (Alomone Labs, Israel). In the different areas of the dorsal hippocampus, CA1, CA3, Dendate Gyrus and Hilus, BDNF expression was obtained as corrected optical density (Image-J, USA) compared to an underlying reference area, as described previously38.
Data analysis and presentation
The study has been reported in accordance with ARRIVE guidelines. All reports were performed in accordance with relevant guidelines and regulations/legislations. Data are presented as mean and standard error of mean (SEM), unless indicated otherwise. Results outside twice the standard deviation of its group were considered outliers and were excluded before analyses (maximally 1 per experimental group). Results were compared using two-way analysis of variance (ANOVA) with least square difference (LSD) post-hoc tests, with saline/ISO (indicated as treatment = T) and sedentary/runner (indicated as intervention = I) as factors. Statistically significant outcomes of the two-way ANOVA are presented in the text, while results of post-hoc testing are presented within the figures, indicating significant differences between specific groups. Sample size (n) is mentioned in the figure legends. Association between selected parameters were measured with Pearson linear correlation. For the Novel Object /Novel Location Recognition tests, outcomes were also tested against change level (= 50%), using a single sample t-test. A p-value of < 0.05 was considered statistically significant and presented as *. Potentially relevant tendencies (p < 0.1) were mentioned as well.
Results
General
ISO-induced mortality was 26%, and occurred within 3 days after ISO treatment; that is before separating the groups according to sedentary/exercise intervention. Body weight at the end of the protocol was slightly higher in rats that had performed exercise, independent of saline or ISO treatment (saline sedentary: 253 ± 7 g; saline exercise: 266 ± 9 g; ISO sedentary: 243 ± 7 g and ISO exercise: 264 ± 7 g; n = 10 per group; ANOVA: p = 0.141; effect of treatment F1,39 = 0.560, p = 0.459; effect of intervention F1,39 = 4.939, p = 0.033; interaction F3,39 = 0.320, p = 0.575).
Effects on the heart
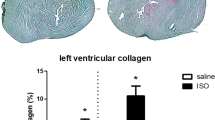
Heart weight to body weight ratio did not differ between groups (saline sedentary: 0.33 ± 0.03%; saline exercise: 0.38 ± 0.01%; ISO sedentary: 0.38 ± 0.02% and ISO exercise: 0.35 ± 0.02%; ANOVA, p = 0.237; effect of treatment F1,39 = 0.351, p = 0.557; effect of intervention F1,39 = 0.355, p = 0.555; interaction F3,39 = 3.724, p = 0.062). Figure 1 presents effects on cardiac damage, measured as percentage of collagen at mid-ventricular level (effect of treatment F1,38 = 3.310, p = 0.077; effect of intervention F1,38 = 0.158, p = 0.693; interaction F3,38 = 6.369, p = 0.016). ISO significantly increased cardiac collagen, which was significantly reversed by exercise.
(A) and (B): typical pictures of sections of the left ventricle, stained for collagen with Sirius red (purple) and fast green as counterstaining. (A) section of a saline sedentary rat; (B): section of an ISO treated sedentary rats. Arrows point to areas with increased collagen. (C): Measured percentage of collagen in the left ventricle as Sirius Red positive area, at mid-ventricular level, in saline or isoproterenol (ISO) treated rats, under sedentary conditions or after five weeks of exercise training (n = 10, 10, 10, 9 for saline sedentary, saline exercise, ISO sedentary and ISO exercise rats, respectively). *: significant difference between groups (p < 0.05).
Effects on cardiac function were measured by echocardiography. Figure 2 presents the main results. Two-way ANOVA revealed a significant higher heart rate in exercise versus sedentary rats, which appeared most prominent in the ISO treated rats (ANOVA p = 0.011; effect of treatment F1,18 = 0.034, p = 0.856; effect of intervention F1,18 = 8.482, p = 0.011; interaction F3,18 = 0.287, p = 0.600). Although analysis of stroke index did not reach statistical significance in the two–way ANOVA (p = 0.089), the indicated reduced stroke index after exercise training (post-hoc analysis; p = 0.035) may be of physiological relevance (effect of treatment F1,18 = 0.914, p = 0.354; effect of intervention F1,18 = 6.465, p = 0.023; interaction F3,18 = 0.940, p = 0.348). The product of heart rate and stroke index, cardiac index, was preserved in all groups. Left ventricular ejection fraction, as measure for left ventricular function, was significantly declined by exercise training as well as by ISO, but may be partly reversed by combined ISO plus exercise; the latter being not significantly lower than sedentary saline controls anymore (effect of treatment F1,18 = 2.745, p = 0.118; effect of intervention F1,18 = 0.1.931, p = 0.185; interaction F3,18 = 8.673, p = 0.010). Underlying measures of cardiac dimensions are summarized in Supplementary Table 1 None of the cardiac function parameters correlated with collagen percentage.
Echocardiographic measures of cardiac function in saline or isoproterenol (ISO) treated rats, under sedentary conditions or after five weeks of exercise training, (n = 4, 4, 6, 5 for saline sedentary, saline exercise, ISO sedentary and ISO exercise rats, respectively). *: significant difference between groups (p < 0.05); + : tendency (ANOVA p = 0.089; subsequent post-hoc test < 0.05).
Effects on the brain
Neuroinflammation was obtained from morphological changes of microglia, indicated as microglia activity; cell body to cell size ratio. Prefrontal cortex microglia activity was not affected by ISO, nor by exercise training (saline sedentary 8.0 ± 0.6; saline exercise 8.6 ± 0.6; ISO sedentary 7.5 ± 0.7; ISO exercise 8.3 ± 1.2). Similarly, no significant effects were observed in the paraventricular nucleus of the hypothalamus microglia activity (saline sedentary 9.5 ± 1.1; saline exercise 7.5 ± 0.2; ISO sedentary 7.1 ± 0.8; ISO exercise 8.0 ± 1.7). Moreover, none of the parameters of microglia morphology in these areas appeared affected by either ISO, or exercise, or showed interaction. Nevertheless, microglia activity in the PVN seemed reduced by about 50% after ISO. Similarly, ISO substantially reduced microglia activity in the hippocampus; an effect that was not affected by exercise (Fig. 3) (effect of treatment F1,19 = 6.290, p = 0.023; effect of intervention F1,19 = 0.063, p = 0.805; interaction F3,19 = 0.435, p = 0.519). Overall hippocampal parameters for microglia morphology were not different between groups, as ISO increased coverage by only 5%, but microglia cell size increased by 44%, which was mainly attributable to increased processes (47%), as cell bodies increased only 15%. Exercise training did not affect these observations.
Microglia activity in the hippocampus, in saline or isoproterenol (ISO) treated rats, under sedentary conditions or after five weeks of exercise training, (n = 4, 4, 6, 6 for saline sedentary, saline exercise, ISO sedentary and ISO exercise rats, respectively). (A): Overall microglia activity in the hippocampus; (B): typical pictures of microglia in the CA1 in saline sedentary and ISO sedentary rats (tile size: 0.1mm2); (C): Morphological parameters underlying the calculated microglia activity in the CA1 area, as the area best reflecting overall effects on the hippocampus. + : tendency (ANOVA p < 0.10; subsequent post-hoc test < 0.05).
Since the hippocampus plays a role in mood as well as cognition, effects on the different hippocampal areas were analyzed as well to evaluate local differences (see Fig. 3 and Table 1). What was indicated for overall values of the hippocampus, appeared merely reflected in the CA1 area (Fig. 3). ISO decreased microglia density (ANOVA p = 0.043; effect of treatment F1,18 = 3.823, p = 0.069; effect of intervention F1,18 = 0.000, p = 1.000; interaction F3,18 = 3.839, p = 0.069), and increased the size of the microglia (ANOVA p = 0.010; effect of treatment F1,18 = 15.290, p = 0.001; effect of intervention F1,18 = 0.311, p = 0.585; interaction F3,18 = 0.484, p = 0.497), hence preserving coverage. Increased cell size could be attributed to increased processes (ANOVA p = 0.014; effect of treatment F1,18 = 14.259, p = 0.002 effect of intervention F1,18 = 259, p = 0.623; interaction F3,18 = 0.387, p = 0.543), as cell body size remained unaltered; microglia hyper-ramification. In the other hippocampal areas, coverage increased in exercise after ISO, compared to exercise after saline treatment, without significantly altering morphology of the individual microglia.
No significant effects were observed on neurogenesis, nor on overall hippocampal or DG BDNF expression (Supplementary Table 2). However, although 2-way ANOVA appeared to show a trend in the CA1 area (p < 0.1), but not in other areas, BDNF was significantly decreased by exercise in ISO rats (Fig. 4) (effect of treatment F1,19 = 0.386, p = 0.543; effect of intervention F1,19 = 0.922, p = 0.351; interaction F3,19 = 5.506, p = 0.032).
Expression of BDNF in the CA1 region of the hippocampus in saline or isoproterenol (ISO) treated rats, under sedentary conditions or after five weeks of exercise training, (n = 4, 4, 6, 6 for saline sedentary, saline exercise, ISO sedentary and ISO exercise rats, respectively). OD = optical density; *: significant difference between groups (p < 0.05); +: p = 0.052.
Effects on behavior
Effects on short-term memory were obtained from performance in the NOR and NLR tests (Fig. 5). For the NOR test, data of six rats were excluded because they did not meet the degree of exploration criteria (four with no exploration and 2 with exploration of only one object), resulting in 8–9 rats per experimental group. For the NLR test, four rats were excluded for only exploring one object, resulting in 8–10 rats per group. The rats that were excluded did not happen to be the most anxious rats in the open field. No significant differences were observed between the experimental groups in performances in these cognitive tests. Twelve months old female rats were well capable of recognizing the novel object in the NOR test, irrespective of saline/ISO treatment or sedentary/exercise training. In the NLR test, control saline sedentary rats seemed unable to recognize the relocated object. Exercise training may improve that, as saline-treated exercise rats performed above random level. However, exercise in ISO-treated rats did not improve NLR performance.
Short-term memory tested as recognizing a novel object (NOR) or a novel location (NLR) in saline or isoproterenol (ISO) treated rats, under sedentary conditions or after five weeks of exercise training, (NOR: n = 9, 9, 8, 8 for saline sedentary, saline exercise, ISO sedentary and ISO exercise rats, respectively; NLR: n = 10, 9, 8, 9 for saline sedentary, saline exercise, ISO sedentary and ISO exercise rats, respectively). #: significantly different from random performance (= 50%; dashed line).
Effects on mood were indicated by spatial exploration in the OF (Fig. 6). Locomotor activity was not affected by ISO, nor by exercise. Spatial exploration in the OF, measured as wall time, revealed no significant effects of ISO, nor of exercise.
Discussion
General
Aim of the present study was to evaluate the effects of exercise training on heart, brain and behavior in the ISO model in middle-aged female rats. As expected, ISO induced cardiac damage and declined cardiac function. However, these effects could not be associated with neuroinflammation, reduced hippocampal neurogenesis or impaired neuronal function. Accordingly, no signs of cognitive impairment or depressive-like behavior were observed after ISO. Exercise training reversed cardiac damage and partly restored ISO-induced cardiac dysfunction, but left neuroinflammation and behavior merely unaltered. Hence, our data indicated that the cardiac damage after ISO in middle-aged female rats, and the subsequent beneficial effects of five weeks exercise training on the heart, were not reflected in changes in the brain nor in altered behavior.
Effects of ISO
The ISO model has a long history as a method to induce focal cardiac damage and consequently cardiac dysfunction40. Similar to our set-up, two injections of ISO given 24 h apart mimicked acute sympathetic stress, activating the innate immune system, resulting in focal cardiac necrosis shown by collagen deposition32,36, that over a period of weeks developed into heart failure37. In contrast to Grant et al.41, other researchers indicated sex difference in the ISO model, as male rats had a lower survival rate, developed more massive necrosis and displayed slower repair compared to female rats34,42. Accordingly, when compared to our previous study in male rats of the same age35, in the female rats in the present study cardiac fibrosis and mortality were relatively mild. Nevertheless, cardiac fibrosis was eminent and caused long-term cardiac dysfunction, suggesting activation of inflammation as repair mechanism. However, these effects could not be associated with neuroinflammation (microglia activation) or associated behavioral changes, such as cognitive decline or depressive-like behavior 5 weeks later. In fact, in contrast to our previous study in male rats35, in female rats microglia activity in the hippocampus indicated reduced, rather than elevated neuroinflammation after ISO. Usually, injury evokes “classical” pro-inflammatory activation of microglia, with morphological changes that include shortened dendrites with increased cell body size43. However, alternatively activated microglia show opposite morphological changes, and are associated with non-pathological stimuli44,45. In our study, we observed a significant increase in total microglial cell size, that could be attributed to increased dendrite area of microglia cells, as cell body area remained unchanged. This finding indicated that the microglia converted into the alternatively activated phenotype, associated with microglia priming; microglia sensitization to become hyperactive once (re)activated43.
Since women seem more susceptible to develop depression after myocardial infarction, we anticipated that our female rats also may develop signs of depressive-like behavior after ISO. However, we did not observe altered OF behavior; neither less locomotion, nor more time in the safe wall area, hence no signs of depressive-like behavior in our female ISO rats. Although depressive-like behavior has been described in literature for male rats, and shortly after ISO-induced damage22,24, in our previous study 5 weeks after ISO in male rats35, if anything, ISO seemed to reduce depressive-like behavior.
To our knowledge, no other female rat studies on behavior are available in that regard. Nevertheless, the older studies of Wexler and coworkers46,47 clearly indicated sex differences in the response of peripheral parameters, that could be linked to behavioral changes, such as depression and cognition48.
Effects of exercise
Exercise training is well recognized for it is beneficial effect on physical as well as mental health25. Indeed, exercise training reversed the ISO-induced scar formation in the heart, thereby partly restoring left ventricular function in our female rats. However, in our previous study in male rats, no cardiac effects of exercise were observed35. Actually, in the only study we are aware of that started endurance training after-, instead of before ISO, exacerbated cardiac damage was reported after four weeks of training49. Differences with the present study may be attributed to the age; 8–10 weeks versus 12 months, and sex of the rats, male- versus female rats, as well as the time point of start of the exercise training; two days after ISO, compared to the one-week delay in our study. Especially the latter aspect could provide an explanation. Our study was set-up to intervene when cardiac damage was eminent, at one week after ISO, whereas the study of Jazi et al.49 started exercise during the inflammatory healing phase, with no cardiac fibrosis present yet32. In the present study exercise intervention could then have been started when the acute healing process was merely complete, hence interfering with the more chronic inflammatory state; timing is crucial.
Exercise training is indicated to exert it beneficial effects, amongst others, by its anti-inflammatory action26. Since we hypothesized that depressive-like behavior9 as well as cognitive impairment20 in association with myocardial infarction, are at least in part attributable to a derailed peripheral inflammation-neuroinflammatory response, we anticipated increased neuroinflammation after ISO, which would be reversed by exercise training. Since we did not observe increased neuroinflammation, measured by microglia activity, nor depressive-like behavior or cognitive decline after ISO, accordingly, we were unable to determine potential reversal of these effects by exercise training. Although neuronal function, as indicated by altered BDNF levels, was not declined after ISO, results seemed to indicate a decline after exercise in ISO rats. These observations are in contrast to our expectations; that were a decline after ISO, that could be reversed by exercise. Brain-derived neurotrophic factor (BDNF) has an important role in regulating maintenance, growth and survival of neurons. It is long known that exercise training increases BDNF expression in the hippocampus50, playing a beneficial role in learning and memory51. However, circulating BDNF also increases after exercise, which could be mainly attributed to the increased brain expression (hippocampus and cortex)52. Moreover, exercise training after myocardial infarction in male rats also induced BDNF in skeletal muscle and the non-infarct area of the left ventricle, which may contribute to improvement of muscle function as well as cardiac function53. The declined BDNF expression in the hippocampus (CA1 area) in the present study, in contrast to the increase seen in male rats in our previous study35, would then be contradictory to the improved cardiac aspects after exercise in ISO rats, and may well related to the female sex of the rats.
Timing
An important aspect of intervention is timing. As we aimed to intervene with the ISO-induced cardiac disease when cardiac fibrosis was eminent, we started exercise training one week after ISO. According to Alemasi et al.32 cardiac inflammation is progressing 3 days after ISO, while cardiac fibrosis seemed not present before seven days after ISO. All referred studies on exercise intervention that had started exercise training before ISO, showed improvement of cardiac parameters, potentially interfering with the inflammatory and/or metabolic responses early after ISO. Our previous study on exercise intervention starting one week after ISO in male rats, showed no effect on cardiac collagen percentage35. However, the one study that started exercise training two days after ISO49, that is within the inflammation phase32, even showed deterioration of cardiac outcome. These results may indicate that optimal timing of exercise intervention in relation to the time course of the inflammatory/metabolic response to ISO seemed crucial. Since in the present study, we showed a positive effect of exercise on the heart, without clear consequences for brain function, this may add to sex differences in the ISO-induced pathology. Striking finding was the indicated microglia hyper-ramification after ISO in female rats. Acute ISO administration produced tachycardia associated with relative ischemia due to imbalance between increased myocardial oxygen demand and reduced coronary blood supply37. Another study, evaluating microglia activity four days after an acute imbalance between oxygen supply and demand due to anesthesia, also showed microglia hyper-ramification, that was explained as a prolonged state of priming54. Hence, it could well be that one week after ISO, the acute response to ISO is merely over, leaving only limited scope for effects of exercise training. The microglia hyper-ramification six weeks after ISO may then represent long-term alertness, that is still present after exercise intervention. Alternatively, the time course of the inflammatory response on microglia activation, as we have observed after a different inflammatory event (surgery38;) could provide an explanation for our results; early increased microglia activity, followed by normalization, and subsequent even significantly declined microglia activity. Therefore, in our opinion, in order to obtain beneficial effects of exercise intervention after ISO, timing of intervention with respect to the time course of cardiac pathology, is crucial.
Limitations
Although the set-up was chosen carefully, each study has its limitations. Since we used inflammation-neuroinflammation-depression/cognitive impairment as our working hypothesis, in hindsight it would have been wise to have collected timed blood samples, to further elucidate on the underlying mechanism of our findings, based on plasma markers, TNFα and IL633, or lipocalin55. The same holds true for measurements of cardiac inflammatory markers at sacrifice. Although the initial event, the cardiac damage, as well as the ultimate behavioral consequences were measured in all rats, the in between factors were only obtained in subgroup of rats, limiting power of the conclusions concerning mechanisms.
The dose of ISO was selected based on effect (mortality) in our previous study35. Actually, this dose appeared in between the medium-dose and high-dose of ISO as reviewed by Nichtova et al.37, with potentially mixed effects described for these dose ranges.
Finally, although the ISO model is mainly used to induce cardiac damage, one has to keep in mind that ISO-effects may not be limited to the heart, as indicated by changes in adrenal gland weight and corticosterone levels as well47.
Conclusion
Our data indicated that five weeks of exercise training in middle-aged female rats, starting when ISO-induced cardiac damage was eminent, decreased cardiac damage and partly restored cardiac dysfunction. However, since ISO could not be associated with neuroinflammation, nor with depressive-like behavior and cognitive impairment, anticipated reversal by exercise could not be observed. Hence, our data indicated that the cardiac damage after ISO in middle-aged female rats, and the subsequent beneficial effects of five weeks exercise training on the heart, were not reflected in changes in the brain nor in altered behavior.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
Abbreviations
- BDNF:
-
Brain-derived neurotrophic factor
- CA:
-
Cornu ammonis
- EF:
-
Ejection fraction
- ISO:
-
Isoproterenol
- Iba-1:
-
Ionized-binding adaptor protein 1
- LSD:
-
Least square difference
- NLR:
-
Novel location recognition test
- NOR:
-
Novel object recognition test
- OF:
-
Open field
- SEM:
-
Standard error of the mean
- VO2max:
-
Maximal oxygen uptake capacity
References
Carney, R. M., Freedland, K. E., Sheline, Y. I. & Weiss, E. S. Depression and coronary heart disease: A review for cardiologists. Clin. Cardiol. 20, 196–200 (1997).
Meijer, A. et al. Prognostic association of depression following myocardial infarction with mortality and cardiovascular events: A meta-analysis of 25 years of research. Gen. Hosp. Psychiatry 33, 203–216 (2011).
Deckers, K. et al. Coronary heart disease and risk for cognitive impairment or dementia: Systematic review and meta-analysis. PLoS ONE 12, e0184244 (2017).
Gottlieb, S. S. et al. The influence of age, gender, and race on the prevalence of depression in heart failure patients. J. Am. Coll. Cardiol. 43, 1542–1549 (2004).
Eastwood, J. A. et al. Commonalities and differences in correlates of depressive symptoms in men and women with heart failure. Eur. J. Cardiovasc. Nurs. 11, 356–365 (2012).
Ghanbari, A. et al. The study of cognitive function and related factors in patients with heart failure. Nurs. Midwifery Stud. 2, 34–38 (2013).
Meijer, A. et al. Adjusted prognostic association of depression following myocardial infarction with mortality and cardiovascular events: individual patient data meta-analysis. Br. J. Psychiatry 203, 90–102 (2013).
Gharacholou, S. M. et al. Cognitive impairment and outcomes in older adult survivors of acute myocardial infarction: Findings from the translational research investigating underlying disparities in acute myocardial infarction patients’ health status registry. Am. Heart J. 162, 860-869.e1 (2011).
Liu, H., Luiten, P. G., Eisel, U. L., Dejongste, M. J. & Schoemaker, R. G. Depression after myocardial infarction: TNF-alpha-induced alterations of the blood-brain barrier and its putative therapeutic implications. Neurosci. Biobehav. Rev. 37, 561–572 (2013).
Angermann, C. E. & Ertl, G. Depression, anxiety, and cognitive impairment : Comorbid mental health disorders in heart failure. Curr. Heart Fail. Rep. 15, 398–410 (2018).
Schoemaker, R. G. & Smits, J. F. Behavioral changes following chronic myocardial infarction in rats. Physiol. Behav. 56, 585–589 (1994).
Frey, A. et al. Experimental heart failure causes depression-like behavior together with differential regulation of inflammatory and structural genes in the brain. Front. Behav. Neurosci. 8, 376 (2014).
Wang, Y. et al. The effects of apoptosis vulnerability markers on the myocardium in depression after myocardial infarction. BMC Med. 11, 32–32 (2013).
Schoemaker, R. G., Kalkman, E. A. & Smits, J. F. “Quality of life” after therapy in rats with myocardial infarction: dissociation between hemodynamic and behavioral improvement. Eur. J. Pharmacol. 298, 17–25 (1996).
Bah, T. M., Kaloustian, S., Rousseau, G. & Godbout, R. Pretreatment with pentoxifylline has antidepressant-like effects in a rat model of acute myocardial infarction. Behav. Pharmacol. 22, 779–784 (2011).
Bah, T. M. et al. Escitalopram reduces circulating pro-inflammatory cytokines and improves depressive behavior without affecting sleep in a rat model of post-cardiac infarct depression. Behav. Brain Res. 225, 243–251 (2011).
Grippo, A. J., Moffitt, J. A., Beltz, T. G. & Johnson, A. K. Reduced hedonic behavior and altered cardiovascular function induced by mild sodium depletion in rats. Behav. Neurosci. 120, 1133–1143 (2006).
Grippo, A. J., Francis, J., Weiss, R. M., Felder, R. B. & Johnson, A. K. Cytokine mediation of experimental heart failure-induced anhedonia. Am. J. Physiol. Regul. Integr. Comp. Physiol. 284, 666 (2003).
Ito, K., Hirooka, Y. & Sunagawa, K. Brain sigma-1 receptor stimulation improves mental disorder and cardiac function in mice with myocardial infarction. J. Cardiovasc. Pharmacol. 62, 222–228 (2013).
Hovens, I. B., van Leeuwen, B. L., Mariani, M. A., Kraneveld, A. D. & Schoemaker, R. G. Postoperative cognitive dysfunction and neuroinflammation; Cardiac surgery and abdominal surgery are not the same. Brain Behav. Immun. 54, 178–193 (2016).
Balakumar, P., Singh, A. P. & Singh, M. Rodent models of heart failure. J. Pharmacol. Toxicol. Methods 56, 1–10 (2007).
Tkachenko, V., Kovalchuk, Y., Bondarenko, N. & Bondarenko capital O C, Ushakova G, Shevtsova A,. The cardio- and neuroprotective effects of corvitin and 2-oxoglutarate in rats with pituitrin-isoproterenol-induced myocardial damage. Biochem. Res. Int. 2018, 9302414 (2018).
Ravindran, S., Gopalakrishnan, S. & Kurian, G. A. Beneficial effect of sodium thiosulfate extends beyond myocardial tissue in isoproterenol model of infarction: Implication for nootropic effects. J. Biochem. Mol. Toxicol. 34, e22606 (2020).
Hu, Y. et al. Behavioral and biochemical effects of KXS on postmyocardial infarction depression. Front. Pharmacol. 11, 561817 (2020).
Pedersen, B. K. & Saltin, B. Exercise as medicine—Evidence for prescribing exercise as therapy in 26 different chronic diseases. Scand. J. Med. Sci. Sports 25(Suppl 3), 1–72 (2015).
Petersen, A. M. & Pedersen, B. K. The anti-inflammatory effect of exercise. J. Appl. Physiol. 98, 1154–1162 (1985).
Sleiman, S. F. et al. Exercise promotes the expression of brain derived neurotrophic factor (BDNF) through the action of the ketone body beta-hydroxybutyrate. Elife https://doi.org/10.7554/eLife.15092 (2016).
El Hayek, L. et al. Lactate mediates the effects of exercise on learning and memory through SIRT1-dependent activation of hippocampal brain-derived neurotrophic factor (BDNF). J. Neurosci. 39, 2369–2382 (2019).
Svensson, M., Lexell, J. & Deierborg, T. Effects of physical exercise on neuroinflammation, neuroplasticity, neurodegeneration, and behavior: What we can learn from animal models in clinical settings. Neurorehabil. Neural. Repair 29, 577–589 (2015).
Kobilo, T. et al. Running is the neurogenic and neurotrophic stimulus in environmental enrichment. Learn. Mem. 18, 605–609 (2011).
Ma, X. et al. Cardiac fibrosis alleviated by exercise training is AMPK-dependent. PLoS ONE 10, e0129971 (2015).
Alemasi, A. et al. Exercise attenuates acute beta-adrenergic overactivation-induced cardiac fibrosis by modulating cytokines. J. Cardiovasc. Transl. Res. 12, 528–538 (2019).
Serra, A. J. et al. Exercise training inhibits inflammatory cytokines and more than prevents myocardial dysfunction in rats with sustained beta-adrenergic hyperactivity. J. Physiol. 588, 2431–2442 (2010).
Wexler, B. C., Judd, J. T. & Kittinger, G. W. Myocardial necrosis induced by isoproterenol in rats. Changes in serum protein, lipoprotein, lipids and glucose druing active necrosis and repair in arteriosclerotic and nonarteriosclerotic animals. Angiology 19, 665–682 (1968).
Toth, K., Oroszi, T., van der Zee, E. A., Nyakas, C. & Schoemaker, R. G. Effects of exercise training on behavior and brain function after high dose isoproterenol-induced cardiac damage. Sci. Rep. 11, 23576-z (2021).
Adamcova, M. et al. Relations between markers of cardiac remodelling and left ventricular collagen in an isoproterenol-induced heart damage model. J. Physiol. Pharmacol. https://doi.org/10.26402/jpp.2019.1.08 (2019).
Nichtova, Z., Novotova, M., Kralova, E. & Stankovicova, T. Morphological and functional characteristics of models of experimental myocardial injury induced by isoproterenol. Gen. Physiol. Biophys. 31, 141–151 (2012).
Hovens, I. B. et al. Postoperative cognitive dysfunction: Involvement of neuroinflammation and neuronal functioning. Brain Behav. Immun. 38, 202–210 (2014).
Hovens, I. B., Nyakas, C. & Schoemaker, R. G. A novel method for evaluating microglial activation using ionized calcium-binding adaptor protein-1 staining: Cewll body to cell size ratio. Neuroimmunol. Neuroinflamm. 1, 82–88 (2014).
Beznak, M. & Hacker, P. Hemodynamics during the chronic stage of myocardial damage caused by isoproterenol. Can. J. Physiol. Pharmacol. 42, 269–274 (1964).
Grant, M. K. O., Abdelgawad, I. Y., Lewis, C. A., Seelig, D. & Zordoky, B. N. Lack of sexual dimorphism in a mouse model of isoproterenol-induced cardiac dysfunction. PLoS ONE 15, e0232507 (2020).
Wexler, B. C., Willen, D. & Greenberg, B. P. Progressive electrocardiographic changes in male and female arteriosclerotic and non-arteriosclerotic rats during the course of isoproterenol-induced myocardial infarction. Cardiovasc. Res. 8, 460–468 (1974).
Eggen, B. J., Raj, D., Hanisch, U. K. & Boddeke, H. W. Microglial phenotype and adaptation. J. Neuroimmune Pharmacol. 8, 807–823 (2013).
Hellwig, S. et al. Altered microglia morphology and higher resilience to stress-induced depression-like behavior in CX3CR1-deficient mice. Brain Behav. Immun. 55, 126–137 (2016).
Beynon, S. B. & Walker, F. R. Microglial activation in the injured and healthy brain: What are we really talking about? Practical and theoretical issues associated with the measurement of changes in microglial morphology. Neuroscience 225, 162–171 (2012).
Wexler, B. C. Metabolic and histopathologic changes in arteriosclerotic versus nonarteriosclerotic rats following isoproterenol-induced myocardial infarction with superimposed diabetes. Metabolism 24, 1321–1337 (1975).
Wexler, B. C. & Greenberg, B. P. Effect of gonadectomy on isoproterenol-induced myocardial infarction. Angiology 30, 377–394 (1979).
Keller, J. et al. HPA axis in major depression: Cortisol, clinical symptomatology and genetic variation predict cognition. Mol. Psychiatry 22, 527–536 (2017).
Azamian Jazi, A., Abdi, H., Haffezi Ahmadi, M. R. & Cheraghi, J. Effect of endurance exercise training on morphological changes in rat heart tissue following experimental myocardial infarction. J. Bas. Res. Med. Sci. 4, 8–16 (2017).
Neeper, S. A., Gomez-Pinilla, F., Choi, J. & Cotman, C. W. Physical activity increases mRNA for brain-derived neurotrophic factor and nerve growth factor in rat brain. Brain Res. 726, 49–56 (1996).
Vaynman, S., Ying, Z. & Gomez-Pinilla, F. Hippocampal BDNF mediates the efficacy of exercise on synaptic plasticity and cognition. Eur. J. Neurosci. 20, 2580–2590 (2004).
Rasmussen, P. et al. Evidence for a release of brain-derived neurotrophic factor from the brain during exercise. Exp. Physiol. 94, 1062–1069 (2009).
Lee, H. W., Ahmad, M., Wang, H. W. & Leenen, F. H. Effects of exercise training on brain-derived neurotrophic factor in skeletal muscle and heart of rats post myocardial infarction. Exp. Physiol. 102, 314–328 (2017).
Tasbihgou, S. R. et al. Brain changes due to hypoxia during light anaesthesia can be prevented by deepening anaesthesia: A study in rats. PLoS ONE 13, e0193062 (2018).
Gouweleeuw, L., Hovens, I. B., Liu, H., Naude, P. J. W. & Schoemaker, R. G. Differences in the association between behavior and neutrophil gelatinase-associated lipocalin in male and female rats after coronary artery ligation. Physiol. Behav. 163, 7–16 (2016).
Acknowledgements
We thank Kunja Slopsema and Jan Keijser for their technical assistance in (immuno)histochemistry and image analyses of the photographed slices, respectively.
Author information
Authors and Affiliations
Contributions
K.T.: conception and design of the work; acquisition and interpretation of data T.O.: acquisition and interpretation of data, revision of the manuscript E.Z.: conception and design of the work, revision of the manuscript C.N.: conception and design of the work, revision of the manuscript R.S.: conception and design of the work, acquisition and interpretation of data, drafted the work and substantively revised it. All authors have approved the submitted version (and any substantially modified version that involves the author's contribution to the study), and have agreed both to be personally accountable for the author's own contributions and to ensure that questions related to the accuracy or integrity of any part of the work, even ones in which the author was not personally involved, are appropriately investigated, resolved, and the resolution documented in the literature.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tóth, K., Oroszi, T., van der Zee, E.A. et al. The effects of exercise training on heart, brain and behavior, in the isoproterenol-induced cardiac infarct model in middle-aged female rats. Sci Rep 12, 10095 (2022). https://doi.org/10.1038/s41598-022-14168-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-14168-z
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.