Abstract
The thalassemia of Hemoglobin H-Constant Spring disease (HbH-CS) is the most common type of Thalassemia in non-transfusion thalassemia. Interestingly, the clinical manifestations of the same genotype of thalassemia can be vastly different, likely due to epigenetic regulation. Here, we used microarray technology to reveal the epigenetic regulation of m6A in modifiable diseases and demonstrated a role of BCL2A1 in disease regulation. In this study, we revealed that methylating enzyme writers including METTL16, WTAP, CBLL1, RBM15B, and ZC3H13 displayed low expression and the demethylating enzyme ALKBH5, along with reader proteins including IGF2BP2 and YTHDF3 exhibited high expression. In addition, BCL2A1 was hypo-methylated and showed low expression. We also revealed that the BCL2A1 methylation level and IGF2BP2 expression were negatively correlated. Additionally, the mRNAs expression between ALKBH5 and IGF2BP2 were positively correlated. In HbH-CS, most genes were hypo-methylated. This included BCL2A1, which may play an important role in the process of red blood cell differentiation and development of HbH-CS. Moreover, the mRNA-M6A methylation status may be regulated by the demethylating enzyme ALKBH5 via IGF2BP2.
Similar content being viewed by others
Introduction
Thalassemia is a serious genetic hemolytic anemic disease that destroys human health and brings about disability and/or death. Various forms of this disease are caused by a defect in the Globin gene, which reduces or completely ceases globin chain synthesis, thereby creating an imbalance in the chain/non-chain ratio of hemoglobin formation1. Thalassemia is one of the most common single gene diseases in the world, accounting for more than 5% of cases worldwide2. The severity of this disease is judged by the need for transfusion. Patients with thalamassia, is therefore, grouped into one of two categories: transfusion dependent thalassemia (TDT) and non-transfusion dependent thalassemia (NTDT)2,3,4,5. NTDT can further be broken into 3 clinically discrete categories: β-thalassemia intermedia, hemoglobin E/β-thalassemia (mild and moderate forms), and α-thalassemia intermedia (otherwise known as, α-thalassemia or hemoglobin H disease)5. Annually, ~ 10,000 births carry the α-thalassemia intermedia form of NTDT6,7. Among them, the Hemoglobin H Constant Spring (HbH-CS) is the most common non-deletion form of the Haemoglobin H disease. Moreover, it is more severe in nature, as compared to thalassemia alone2. In view of the severe clinical phenotype of HbH-CS and its related complications, there is a growing consensus that HbH-CS is a disease with poor prognosis. Similar to the absence of HbH, HbH-CS requires special attention and perfomalized therapy4. Unfortunately, historically, thalassemia intermedia, was deemed as a milder form of NTDT. Hence, patients with HbH-CS were administered few to no transfusion and thereby had little to no iron chelation. However, with evidences from multiple long-term clinical studies, it is now clear that NTDT, which do not require blood transfusions early in life, can develop life threatening complications later in life and must, therefore, be monitored and managed with care8. The clinical manifestations of HbH-CS thalassemia greatly vary in severity and cannot be explained solely by the causative genes related to thalassemia intermedia3. Mild patients have only mild anemia, normal growth and development, and do not require blood transfusion and splenectomy. In contrast, severe patients have moderate to severe anemia, jaundice, hepatosplenomegaly, thalassemia appearance, backward growth and development, low resistance, can be complicated with infection, iron overload, cholelithiasis, folic acid deficiency, fracture, etc. In addition, oxidative drugs and infection can induce hemolytic crisis9. Hence, the effect of epigenetic modification on HbH-CS must be considered.
Epigenetics, which includes DNA, RNA, and protein modifications is often used to link genetic modifications to disease phenotypes10,11. Recently, DNA methylation was reported to be involved in thalassemia or hematopoietic diseases12,13,14. However, there are no reports on whether RNA methylation plays a role in thalassemia. Nevertheless, RNA methylation has been implicated in other circulatory diseases15,16,17. Hence, in this study, we analyzed the link between epigenetics and the HbH-CS phenotype, using the human m6A-mRNA and lncRNA epitranscriptomic microarray data from the immature erythrocytes of 5 HbH-CS thalassemia (T) and 5 normal healthy volunteers (N). Our goal was to elucidate the underlying mechanism behind HbH-CS pathogenesis.
Results
M6A-mRNA in HbH-CS and healthy volunteers
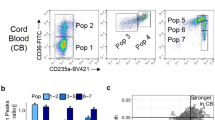
We observed no discernible differences in the age or gender of the HbH-CS thalassemia (T) and healthy volunteers (N) cohorts (Table 1). Using flow cytometry we determined that our samples were primarily composed of immature RBCs (Fig. 1). Based on our m6A-mRNA and lncRNA epitranscriptomic microarray analysis, there were 8981 up-regulated RNA and 6606 RNA that were either down-regulated or showed no differential expression between the T versus N samples (Fig. 2A). Moreover, comparing differential RNA methylation between the two cohorts, we observed 126 RNAs that were heavily methylated, 61 RNAs that had very little methylation, 5971 RNAs that had slight elevation in methylation, and 5043 RNAs that exhibited a slight reduction in methylation (Fig. 2B).
Differential expression of genes in T versus N
Based on our analysis, we selected 8 differentially expressed mRNAs, based on their m6A level, and confirmed their expression patterns in T (n = 15) and N (n = 16) samples, using RT-qPCR. The selected mRNAs were Mettl16, WTAP, CBLL1, RBM15B, ZCH3H13, IGF2BP2, YTHDF3, and ALKBH5. As depicted in Fig. 3A, these genes expressed differently in T versus N. However, their expression patterns were consistent with the epitranscriptomic microarray sequences (Fig. 3B), thereby confirming the reliability of the microarray technique. The primers for our RT-qPCR work are presented in Table 2.
Gene ontology enrichment and pathway analysis
To explore the functional correlations between the differentially m6A-methylated and differentially expressed mRNA, we enriched select gene ontological functions and GO terms (http://www.geneontology.org) and used the bioconductor top GO package for analysis. Among the top 10 GO were biological process (BP), cellular component (CC), and molecular function (MF). Moreover, we determined significance using Fisher Exact test p-value and established the enrichment score via -log10 (p) formula. Figures 4A illustrates the differentially m6A hypo-methylated mRNA. In addition, we conducted metabolic pathway analysis, using KEGG pathways, on the differentially m6A hypo-methylated mRNAs, with statistical analysis as described before. As depicted in Fig. 4B, the pathway enrichment analysis showed involvement of essential pathways like the Herpes simplex virus 1 infection, sphingolipid signaling pathway, NF-kappa B axis, Th17 cell differentiation, B cell receptor axis, Viral myocarditis, Yersinia infection, osteoclast differentiation, phospholipase D axis, and the AGE-RAGE axis.
The top ten enrichment scores of significant enrichment hypo-methylated genes ontology (in bar format) depicted genes from the biological process, cell component, and molecular function (A). Significance was established at p-value 0.05. The top ten enrichment scores of significant enrichment hypo-methylated genes pathway (− log10 (Pvalue)) (B), in dot format. The degree of red corresponds to significance. The size of the dots corresponds to mRNA entities that are directly linked to the noted Pathway IDs.
BCL2A1 expression is regulated by m6A modification
The “m6A methylation level” for all transcripts was calculated as the percentage of Modified RNA (% Modified) in all RNAs based on the immunoprecipitated (IP) (Cy5-labelled) and supernatant (Sup) (Cy3-labelled) normalized intensities:
% Modified = (modified RNA)/(Total RNA) = IP/ (IP + Sup) = 〖IP〗_(Cy5 normalized intensity)/(〖IP〗_(Cy5 normalized intensity) + 〖Sup〗_(Cy3 normalized intensity) ).
The top 20 differentially hypo-methylated m6A mRNAs in T versus N are listed in Table 3 and 20 differentially hypo-methylated m6A non- mRNAS including lncRNA and other small RNAs are listed in Table 4. Here, we evaluated m6A levels of BCL2A between T (n = 10) and N (n = 10). The primary methylated sites were in the CDS and 5’UTR regions21. Figure 5A illustrates the methylated mRNA and positions of methylation. Based on our data, we demonstrated that the mRNA expression and m6A levels of BCL2A1 were markedly down-regulated in T versus N (Fig. 5B,C). The primers used for BCL2A1 identification were 5′AGAATCTGAAGTCATGCTTGGA3′and 5′CTCCTTTTCCATCACTTGGTTG3. In addition, using KEGG analysis, we showed that the methylation of BCL2A1 was linked to IGF2BP2 mRNA expression (Fig. 5D). Figure 5E showed that ALKBH5 is positively correlated with IGF2BP2. To investigate the the potential roles of IGF2BP2, we examined the effects of IGF2BP2 knockdown in K562 cells and confirmed that IGF2BP2 was knocked down by using siRNA sequences (Fig. 5F,I). We found that knockdown of IGF2BP2 significantly inhibited the ALKBH5 and BCL2A1 expression of k562 cells (Fig. 5G–I).
BCL2A1 mRNA levels are modulated by m6A associations. (A) An illustration of m6A sites in BCL2A121 (http://m6avar.renlab.org/) (B) The m6A-BCL2A1 association was severely hypo-regulated (P < 0.0001). (C) BCL2A1 transcription was markedly reduced in T versus N (P = 0.0085). (D) BCL2A1 methylation status and IGF2BP2 levels were negatively correlated (r = − 0.7015, p = 0.0006). (E) Transcriptional relationship between ALKBH5 and IGF2BP2 (r = 0.6989, p < 0.0001). P-values were derived from Student's t-test and Correlation Analysis. (F–H) qRT-PCR analysis of IGF2BP2, ALKBH5 and BCL2A1 in K562 transfected with siRNA negative control (siCtrl), IGF2BP2 siRNA. (I) western blot analysis of IGF2BP2, ALKBH5 and BCL2A1 in K562 transfected with siRNA negative control (siCtr1), IGF2BP2 siRNA.
Discussion
In recent years, it became evident that people carrying the same Globin genotype can have vastly different phenotypes. Moreover, thalassemia can aggravate in presence of stressors like pregnancy, infection, surgery and so on. Based on these facts, it is possible that thalassemia may be affected by other, non-linked genes19. Multiple reports have confirmed that cis-regulatory elements like DNA methylation, histone acetylation, and micro RNAs can regulate pathogenesis and clinical heterogeneity of thalassemia19,20,21. In addition, other modifiers were also shown to affect thalassemia phenotype, such as mutations in the molecular chaperone haemoglobin stabilizing protein (AHSP) and transcription regulator GATA1. These emerging reports offer novel view into the therapeutics and prevention of thalassemia, including prevention of the disease in thalassemia heterozygote carriers22.
Recent studies on m6A-mediated RNA modification demonstrated the dynamic reversibility of RNA modification in regulating RNA metabolism, processing, and directional differentiation of stem cells23. Moreover, the m6A methylation was shown to modulate the coordinated efforts of transcription and post-transcriptional expression24. This includes events like mRNA splicing, export, localization, translation, and stability. M6A-mediated methylation of mRNA can potentially enable interaction with modulatory proteins which can regulate downstream gene expression. Emerging evidences suggest a crucial role of m6A-mediated mRNA modification in driving mRNA translation25.
To determine whether m6A modification is involved in producing the thalassemia phenotype, we assessed immature erythrocytes from the peripheral blood of 15 T and 16 N. We identified that methylating enzymes like METTL16, WTAP, CBLL1, RBM15B, and ZC3H13 were scarcely expressed and the demethylating enzyme ALKBH5, along with reading protein-related genes, such as, IGF2BP2 and YTHDF3, were highly expressed in T versus N. In addition, we detected differential regulation of other m6A-methylated RNAs like RBM15, YTHDF3, IGF2BP1, IGF2BP3, and multiple mRNAS, micRNA, and LNC RNA, which were hypo-methylated in T versus N. Given these evidences, we believe that the m6A-mediated methylation is crucial for the pathogenesis of HbH-CS.
BCL2A1 (B cell lymphoma 2 related A1), from the BCL2 (B cell lymphoma 2) protein family26, counters cell apoptosis via prevention of cytoplasmic accumulation of cytochrome and prevents the subsequent stimulation of the internal apoptotic axis. This protein is often highly expressed in advanced cancers and denotes poor prognosis27. In this study, we verified that the BCL2A1 mRNA was hypo-m6A-methylated and had low expression in T versus N. However, the underlying mechanism is yet to be determined. M6A methylation can stabilize target mRNA, thereby affecting downstream gene expression28,29. M6A methylation is generally common in protein-coding transcripts and are localized in the 3′UTRs29 and 5′UTR30 regions. Interestingly, the methylation sites of BCL2A1 are located within the CDS21 and 5'UTR regions18.
Based on our data, we propose that alterations in the mRNA methylation status contributes to thalassemia. Due to the severe down-regulation of BCL2A1 in HbH-CS patients, apoptosis remains uninhibited, leading to hemolytic anemia. In thalassemia, erythropoiesis is finely regulated by a complex network of transcription factors, including those involved in erythropoiesis like the erythropoietin receptor EPOR, glycophorin, and Globin peptide chains. In addition, the anti-apoptotic protein BCL-x, induced by the transcription factors STAT5 and GATA1, are activated by erythropoietin EPO31. When BCL2A1 levels diminish during erythroid development, further erythrocytes production ceases, thereby aggravating thalassemia. Based on our analysis, we detected BCL2A1 in 3 separate signaling pathways, namely, NF-KappaB, Acute Myeloid Leukemia and transcriptional misregulation in cancer (Supplemental Information). Interestingly, we also discovered that the m6A methylation status of BCL2A1 was negatively correlated with IGF2BP2 gene expression. However, m6A methylation was not directly affected by the M6A-associated enzymes. Alternately, ALKBH5 is positively correlated with IGF2BP2. Collectively, based on our analysis, we propose that ALKBH5 regulates RBC differentiation and development by altering the methylation status of BCL2A1 via IGF2BP2. However, this requires further study and confirmation.
Conclusion
M6A plays a significant role in thalassemia. In this study, we have, for the first time, explored the m6A RNA methylation status and its regulation of RNA stability in HbH-CS patients. Moreover, using m6A-RIP-seq and RNA-seq data, we established a profile of differentially methylated mRNA in T versus N samples. Lastly, we proposed involvement of m6A-regulated BCL2A1 in the pathogenesis of thalassemia. To confirm our preliminary data, more investigation is necessary to explore the methylation status of relevant RNAs and modulation of target downstream genes during erythroid differentiation.
Our work had certain deficiencies. Firstly, we discovered that there were two m6A methylation sites in BCL2A1. However, we did not elucidate which site holds more significance to thalassemia. Secondly, the underlying mechanism behind RNA methylation of BCL2A1 was not examined, and needs further exploration.
Materials and methods
Participants and samples
We collected samples from 16 HbH-CS thalassemia patients and 15 healthy volunteers, between March and July 2020. The HbH-CS patients had differing degrees of moderate anemia and hepatosplenomegaly and did not receive any blood transfusion in the past 3 months. The HGB range, among the HbH-CS patients, were between 56 and 103 g/l. In addition, Doppler's ultrasound of HbH-CS patients revealed splenomegaly ranging from Grade 1 to Grade 3. HbH-CS thalassemia and healthy volunteers will hereby be referred to as group T and group N, respectively. We conducted the Arraystar Human m6A-mRNA and lncRNA epitranscriptomic microarray analysis of 5 pairs of immature erythrocytes, particularly, 5 from HbH-CS thalassemia (T) and 5 from healthy volunteers (N). Our works received ethical approval from the First Affiliated Hospital of Guangxi Medical University. All participants were recruited from the same university and agreed to sign informed consent forms. If subjects were under 18, the informed consents were signed by their parent and/or legal guardian. All methods were performed in accordance with the Declaration of Helsinki. All HbH-CS patients were confirmed of their diagnosis with blood routine, haemoglobin electrophoresis, and DNA analysis32,33. All N groups were without any blood-related diseases. All participants were between the ages of 2 and 50. Patient demographics are provided in Table 1.
Sorting of immature red blood cells (RBCs)
Immature RBCs were collected by sorting peripheral blood from the T and N cohorts, using a positive CD71 (CD71 Microbeads human, Miltenyi Biotec GmbH, Germany) selection method with a magnetic shelf (MiniMACS Starting Kit, Miltenyi Biotec GmbH, Germany), followed by confirmation with flow cytometry34,35. The flow cytometry results are depicted in Fig. 1. The sorted RBC samples were subsequently maintained in TRIZOL at − 80 °C for further analysis.
Total RNA isolation and RT-qPCR
Total RNA was extracted with RNAiso Plus (Takara) and cDNA was synthesized with Prime Script TMRT reagent Kit with GDNA Eraser (Perfect Real-Time; Takara Bio, Shiga, Japan). Next, real time polymerase chain reaction (RT-qPCR) was conducted with the QuantiNova SYBR Green PCR Kit (QIAGEN, Product of Germany). Lastly, β-Actin was employed for normalization of gene expression.
M6A Immunoprecipitation (MeRIP)
1–3 μg of total RNA and m6A spike-in control were introduced to 300 μL 1 × IP buffer (50 mM Tris–HCl, pH7.4, 150 mM NaCl, 0.1% NP40, 40U/μL RNase Inhibitor) with 2 μg of anti-m6A rabbit polyclonal Ab (Synaptic Systems). The solution was then maintained with head-over-tail rotation at 4 °C for 2 h. Meanwhile, 20μL (per sample) of Dynabeads™ with M-280 Sheep Anti-Rabbit IgG suspension was blocked with freshly made 0.5% BSA at 4 °C for 2 h, rinsed thrice with 300 μL 1 × IP buffer, and resuspended in the total RNA-antibody mixture described above. RNA was allowed to bind to the m6A-Ab beads during head-over-tail rotations at 4 °C for 2 h. Next, the beads were rinsed thrice with 500 μL 1 × IP buffer and twice with 500 μL wash buffer (50 mM Tris–HCl, pH7.4, 50 mM NaCl, 0.1% NP40, 40 U/μL RNase Inhibitor). Finally, the enriched RNA was eluted with 200 μL elution buffer (10 mM Tris–HCl, pH7.4, 1 mM EDTA, 0.05% SDS, 40U Proteinase K) at 50 °C for 1 h, before extraction and precipitation with acid phenol–chloroform and ethanol, respectively.
Two-color RNA labeling and array hybridization
The modified RNAs were eluted from the immunoprecipitated magnetic beads as “IP” (immunoprecipitated, Cy5-labelled). The unmodified RNAs were recovered from the supernatant as “Sup” (supernatant, Cy3-labelled). The “IP” and “Sup” RNAs were then labeled with Cy5 and Cy3 respectively, in separate reactions, using Arraystar Super RNA Labeling Kit. The RNAs were combined together and hybridized onto Arraystar Human mRNA & lncRNA Epitranscriptomic Microarray (8 × 60 K, Arraystar). After washing the slides, the arrays were scanned in the two-color channels by an Agilent Scanner G2505C.
Cell culture and transfection
K562 cell lines (Procell Biotechnology Co., Ltd, WUHAN, CHINA) were cultured as grown in RPMI medium 1640 basic (Gibco) supplemented with 10% fetal bovine serum (FBS) (Sijiqing, Zhejiang, China), and 1% penicillin–streptomycin (Solarbio, Beijing, China). Cells were transfected with siRNAs (final concentration: 20 nM) by riboFECT™ CP Reagent (RiboBio, GuangZhou, China) according to the manufacturer’s instructions. All siRNAs were obtained from Tsingke Biotechnology Co.Ltd. (nanning, China) shown below: IGF2BP2 sense: GAAGUGAUCGUCAGAAUUATT, antisense: UAAUUCUGACGAUCACUUCTT; siRNA Negative control (siCtrl), Sense: UUCUCCGAACGUGUCACGUTT, Antisense: ACGUGACACGUUCGGAGAATT.
IGF2BP2、ALKBH5 and BCL2A1 expression were confirmed by RT-Qpcr and Immunoblot.
Immunoblotting
Cells were lysed using in RIPA buffer (Beyotime, Jiangsu, China). Proteins were electrophoretically resolved on 10% or 15% SDS–polyacrylamide gels (40 μg per lane), and transferred onto nitrocellulose membranes (Bio-Rad, Hercules, CA, USA). and electroblotted onto polyvinylidene difluoride (PVDF) membranes. After blocking with 5% BSA Blocking Buffer (Solarbio, Beijing, China)The membranes were washed 3 times with TBST (Solarbio, Beijing, China) with 1% Tween20. After incubation with appropriate first antibody 4 °C overnight and HRP-coupled secondary antibodies at room teperature for 1 h. Target proteins were detected in Gel and chemiluminescence dual imaging system (FluouChem HD2, Santa Clara, CA USA)and developed with BeyoECL Plus (Beyotime, Shanghai, China) and images analyzed with ImageJ (version 1.51j).
Reagents
Antibodies used for experiment were as follows: GAPDH (60004-1-1 g, 1:10,000)、 ALKBH5 (16837-AP, 1:2000), IGF2BP2 (11,601–1-AP, 1:2000) and HRP-coupled secondary antibodies (SA00001-1,1:2000) were from Proteintech Group, Inc (Wuhan, China). BCL2A1 (A0134,1:500) antibody was from Abclonal (Wuhan, China).
Data analysis
The Agilent Feature Extraction software (version 11.0.1.1) was employed for the analysis of acquired array images. Raw intensities of Modified RNA (Cy5-labelled) and Unmodified RNA (Cy3-labelled) were normalized with the mean of log2-scaled Spike-in RNA intensities. Next, signals with Present (P) or Marginal (M) QC flags in a minimum of 1 in 10 samples were marked as “All Targets Value” in Excel for further “m6A methylation level”, “m6A quantity” and “expression level” calculations. The “m6A methylation level” was measured via the percentage of modification based on sample normalized intensities. The “m6A quantity” was assessed from the Modified RNA (Cy5-labelled) normalized intensities. The “RNA expression level” was analyzed according to the normalized intensities of all RNA. Moreover, we performed additional quartile normalization using the limma package to ascertain array expression, before probe flag screening. Differentially m6A-methylated RNAs or differentially expressed RNAs between the HbH-CS and healthy volunteers were recognized by fold change and statistical significance (p-value) values. Lastly, hierarchical clustering was done to show degrees of m6A-methylation within samples. P value < 0.05 denotes significance.
Ethics approval and consent to participate
Our works received ethical approval from the First Affiliated Hospital of Guangxi Medical University. All participants agreed to sign informed consent forms.
Consent for publication
All authors agree to the publication of this article.
Data availability
The following information was supplied regarding data availability. The sequencing data are available in the Supplemental Files.
Change history
10 November 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41598-021-01793-3
Abbreviations
- HbH-CS:
-
Hemoglobin H-constant spring disease
- TDT:
-
Transfusion dependent thalassemia
- NTDT:
-
Non-transfusiondependent thalassemia
- BP:
-
Biological process
- CC:
-
Cellular component
- MF:
-
Molecular function
- IP:
-
Immunoprecipitated
- Sup:
-
Supernatant
- siCtrl:
-
siRNA Negative control
References
Frédéric, B., Piel, D. J. & Weatherall, M. D. The α-Thalassemias. N. Engl. J. Med. 371, 1908–1916 (2014).
Ali, T. T., David, J. W. & Maria, D. C. Thalassaemia. Lancet 391, 155–167 (2018).
Danjou, F., Anni, F. & Galanello, R. Betathalassemia: From genotype to phenotype. Haematologica 96, 1573–1575 (2011).
Taher, A., Vichinsky, E., Musallam, K., Cappellini, M. D. & Viprakasit, V. Guidelines for the management of non transfusion dependent thalassaemia (NTDT) (Thalassaemia International Federation, 2013).
Weatherall, D. J. The definition and epidemiology of non-transfusion-dependent thalassemia. Blood Rev. 26(1), S3-6 (2012).
Weatherall, D. J. The inherited diseases of hemoglobin are an emerging global health burden. Blood 115(22), 4331–4336 (2010).
Modell, B. & Darlison, M. Global epidemiology of haemoglobin disorders and derived service indicators. Bull. World Health organ. 86(6), 480–487 (2008).
Lal, A., Goldrich, M. L., Haines, D. A. & Vichinsky, E. P. Heterogeneity of hemoglobin H disease in childhood. N. Engl. J. Med. 364(8), 710–718 (2011).
Lal, A. et al. Heterogeneity of hemoglobin H disease in childhood. N. Engl. J. Med. 364(8), 710–718 (2011).
Bao, X., Zhang, X., Wang, L. & Xu, X. Epigenetic inactivation of ERF reactivates γ-globin expression in β-thalassemia. Am. J. Hum. Genet. 108(4), 709–721 (2021).
Gong, Y., Zhang, X., Zhang, Q. & Xu, X. A natural DNMT1 mutation elevates the fetal hemoglobin level via epigenetic derepression of the γ-globin gene in β-thalassemia. Blood 137(12), 1652–1657 (2021).
Mimouni, N. E. H., Paiva, I., Barbotin, A. L. & Giacobini, P. Polycystic ovary syndrome is transmitted via a transgenerational epigenetic process. Cell Metab. 33(3), 513-530.e8 (2021).
Seddon, A. R., Liau, Y., Pace, P. E. & Stevens, A. J. Genome-wide impact of hydrogen peroxide onmaintenance DNA methylation in replicating cells. Epigenet. Chrom. 14(1), 17 (2021).
Weinberg, D. N., Papillon-Cavanagh, S., Chen, H. & Lu, C. The histone mark H3K36me2 recruits DNMT3A and shapes the intergenic DNA methylation landscape. Nature 573(7773), 281–286 (2019).
Huff, S., Tiwari, S. K., Gonzalez, G. M. & Rana, T. M. m6A-RNA demethylase FTO inhibitors impair self-renewal in glioblastoma stem cells. ACS Chem Biol. 16(2), 324–333 (2021).
Huang, H., Wang, Y., Kandpal, M. & Matei, D. FTO-dependent N6-methyladenosine modifications inhibit ovarian cancer stem cell self-renewal by blocking cAMP signaling. Cancer Res. 80(16), 3200–3214 (2020).
Huang, Y., Su, R., Sheng, Y. & Yang, C. G. Small-molecule targeting of oncogenic FTO demethylase in acute myeloid leukemia. CancerCell. 35(4), 677–691 (2019).
Zheng, Y. Y. et al. m6AVar: A database of functional variants involved in m6A modification. Nucleic Acids Res. 46, 139–145 (2018).
Bradner, J. E. et al. Chemical genetic straregy identifies histone deadetulase1 (HDAC1) and HDAC2 as therapeutic targets in sickle cell disease. Proc. Natl. Acad. Sci. U.S.A. 107(28), 12617–12622 (2010).
Zhang, L., Sankaran, V. G. & Lodish, H. F. MicroRNAs in erythroid and megakaruocytic differentiation and megakaryocyte-erythroid progenitor lineage commitment. Leukemia 26(11), 2310–2316 (2012).
Fu, Y. F. et al. Mir-q44 selectively regulates embryonic alpha-hemoglobin synthesis during primitive erythropoiesis. Blood 113(6), 1340–1349 (2009).
Surapolchai, P. et al. A molecular study on the role of alpha-hemoglobin-stabilizingprotein in hemoglobin H disease. Ann. Hematol. 96(6), 1005–1014 (2017).
Yang, Y., Hsu, P. J., Chen, Y. S. & Yang, Y. G. Dynamic transcriptomic m6 A decoration: Writers, erasers, readers and functions in RNA metabolism. Cell Res. 28(6), 616–624 (2018).
Frye, M., Harada, B. T., Behm, M. & He, C. RNA modifications modulate gene expression during development. Science 361(6409), 1346–1349 (2018).
Yang, Y., Hsu, P. J., Chen, Y. S. & Yang, Y. G. Dynamic transcriptomic m(6)A decoration: Writers, erasers, readers and functions in RNA metabolism. Cell Res. 28, 616–624 (2018).
Lionnard, L. et al. TRIM17 and TRIM28 antagonistically regulate the ubiquitination and anti-apoptotic activity of BCL2A1. Cell Death Differ. 26(5), 902–917 (2019).
Yu, B., You, W., Chen, G., Yu, Y. & Yang, Q. MiR-140-5p inhibits cell proliferation and metastasis by regulating MUC1 via BCL2A1/MAPK pathway in triple negative breast cancer. Cell Cycle 18, 2641–2650 (2019).
Vogler, M. BCL2A1: the underdog in the BCL2 family. Cell Death Differ. 19, 67–74 (2012).
Huang, H. et al. Recognition of RNA N6 -methyladenosine by IGF2BP proteins enhances mRNA stability and translation. Nat. Cell. Biol. 20(3), 285–295 (2018).
Zhou, J. et al. Dynamic m6 A mRNA methylation directs translational control of heat shock response. Nature 526(7574), 591–594 (2015).
Gregory, T. et al. GATA-1 and erythropoietin cooperatr to promote erythroid cell survival by regulating bcl-Xl expression. Blood 94(1), 87–96 (1999).
Tmusallam, K. M., Cappellini, M. D., Viprakasit, V. & Taher, A. T. Revisiting the non-transfusion-dependent (NTDT) vs transfusion-dependent (TDT) thalassemia classification 10 years later. Am. J. Hematol. 96(2), E54–E56 (2021).
Khaled, M., Musallam, M. D., Stefano, R., Ali, T. & Taher, M. D. Management of non-transfusion-dependent β-thalassemia (NTDT): The next 5 years. Am. J. Hematol. 96(3), E57–E59 (2021).
Simonetti, S., Natalini, A., Peruzzi, G. & Di Rosa, F. A DNA/Ki67-based flow cytometry assay for cell cycle analysis of antigen-specific CD8 T cells in vaccinated mice. J. Vis. Exp. 167, 2 (2021).
Frisch, B. J. Hematopoietic stem cell cultures and assays. Methods Mol. Biol. 2230, 467–477 (2021).
Acknowledgements
We are grateful to Professor Lihong Pang for her helpful suggestions and thank doctors Fang Yang, Lingjie Deng, Dongmei Yang, Xiaoli Zhang, and Xueyu Li for their help in collecting clinical data. The views expressed in this article are the personal opinions of the authors and do not reflect the policy of the Department of Health and Human Services Funding.
Funding
This work was supported by grants from the National Natural Science Foundation of China (Nos. 81960281), Special Fund of Characteristic Innovation Team of the First Affiliated Hospital of Guangxi Medical University (NO YYZS202008), and the construction of clinical intervention protocols Guangxi key R & D program (Guike AB20159031).
Author information
Authors and Affiliations
Contributions
H.R. is a major participant in this study and the author of the paper; F.Y., L.D., D.Y., X.Z., and X.L. participated in the collection of specimens for this topic and the analysis of subject data. L.P. participated in the design of the topic and overall experimental guidance. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: The original version of this Article contained an error. In the Materials and methods section, under subheading “Participants and samples”, “Patient demographics are provided in Table 4.” now reads: “Patient demographics are provided in Table 1."
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ruan, H., Yang, F., Deng, L. et al. Human m6A-mRNA and lncRNA epitranscriptomic microarray reveal function of RNA methylation in hemoglobin H-constant spring disease. Sci Rep 11, 20478 (2021). https://doi.org/10.1038/s41598-021-99867-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-99867-9
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.