Abstract
Magnesium based niobium oxides (Mg–Nb–O) were prepared by solid-state reactions owing to understand the function of transition metal oxides as promoters/catalysts for practical application. Magnesium niobate (Mg3Nb6O11) was synthesized for the first time in nearly pure form reported in this context. MgNb2O6 and Mg4Nb2O9 were prepared in oxidizing conditions; on the contrary, Mg3Nb6O11 preferred reducing environment. Stoichiometric mixtures of the precursor materials MgO, Nb2O5 and/or metallic Nb were annealed for the syntheses which revealed the effect of temperature on phase formation, reaction kinetics and heat of reaction. The products were examined by ex-situ, in-situ X-ray diffraction (XRD) and differential scanning calorimetry (DSC). Crystallographic parameters of various binary and ternary compounds (Mg/Nb/O) formed in different calcination conditions, were extracted by Rietveld method. In-situ experiment results in single step reaction for the MgNb2O6 synthesis and the heat of formation of the solid-state reaction obtained to be minimum (93 kJ/mol). In contrast, the formation of Mg4Nb2O9 and Mg3Nb6O11 compounds towards pure phases rather complicated due to multistep reactions and corresponding heat of formation were estimated to be 140 and 190 kJ/mol. Experimental results have been discussed based on kinetic and thermodynamic constrains.
Similar content being viewed by others
Introduction
Hydrogen storage represents an important step in the development of hydrogen economy and various storage systems were reported in the literatures. Among these systems, magnesium hydride (MgH2) is an interesting material for H2 storage1,2 owing to high abundance in the lithosphere, cost-effective and less toxic properties3,4. However, higher temperature is required in H2 absorption/desorption cycles which are characterized by relatively slow reaction kinetics.
Different strategies have been proposed to overcome these problems such as ball milling and addition of catalysts, particularly, transition metal oxides (TMO), metals, alloys, etc.5. Huge interest in TMO was reported in the literatures. It has been revealed from the reviews that niobia (Nb2O5) is a competent additive even though its activity is still an unresolved issue as a promoter/additive to alter the reaction kinetics of the system6,7,8,9.
Milling of MgH2 with Nb2O5 influenced H2 absorption/desorption kinetics and Mg–Nb–O ternary compounds were formed during H2 sorption cycles10,11,12. In particular, a reactive pathway model was proposed by Friedrichs et al.13 point out the fact that the reduction of Nb2O5 into metallic Nb, followed by successive formation of magnesium based oxides (Mg–Nb–O) which facilitate H2 transport into the solid structures. Recently, the effect of the presence of Mg–Nb mixed oxide compounds (MgNb2O6, Mg4Nb2O9 and Mg3Nb6O11) on H2 absorption properties of MgH2 was investigated9,14,15.
A number of possibilities for the formation of binary and ternary compounds with various oxidation states of Nb are available in the literatures16,17,18,19,20,21,22,23,24,25. Abbattista et al.26 obtained an orthorhombic phase of MgNb2O3.6 from reduction of MgNb2O6. Marinder et al.27 also reported the preparation of Mg3Nb6O11 from precursor mixtures of MgO/NbO2 with different molar ratios of Mg:Nb. The synthesis of some ternary compounds, e.g., MgNb2O6, Mg4Nb2O9, Mg3Nb6O11 and Mg5Nb4O15 were reported elsewhere24,28. Pagola et al.24 proposed an effective method of solid-state synthetic reactions Mg–Nb oxides basically annealing commercial precursor materials. During preparation of MgNb2O6 compound from the starting materials (MgO and Nb2O5), MgNb2O6 phase of columbite structure is usually obtained including corundum-like Mg4Nb2O929. Crystallographic, microstructural and morphological features of MgNb2O6 and Mg4Nb2O9 compounds were reported elsewhere30. Moreover, they are stable phases at room temperature explored by You et al.31. A crystallographic investigation of the orthorhombic columbite-like MgNb2O6 phase was done and the fine-structural features of this compound were established from neutron diffraction investigation24. Mg4Nb2O9 shows corundum-type (α-Al2O3) structure resulting from order of Mg2+ and Nb5+ ions32,33. Nonetheless, proper selection of starting materials, calcination conditions to be optimized and reaction mechanism aiming to have pure phases not yet studied in details.
The present work aims to explore in details the synthetic routes for the preparation of three Mg–Nb–O compounds (e.g., MgNb2O6, Mg4Nb2O9 and Mg3Nb6O11) in nearly pure phases and the products were characterized by ex-situ and in-situ XRD and DSC experiments.
Methodology
Materials and methods
Commercially available MgO, Nb2O5 and Nb (Sigma-Aldrich, Germany) were employed as precursor materials for the solid-state synthesis. MgNb2O6 and Mg4Nb2O9 were prepared by annealing MgO/Nb2O5 with stoichiometric ratio in oxidizing atmosphere. A mixture of MgO/Nb2O5/Nb powders was heated in evacuated quartz ampoules for Mg3Nb6O11 preparation. The mixtures were annealed from room temperature (RT) to 473, 673, 873, 1073, 1273 and 1473 K for 24 h and heating rate was set at 10 K/min for all the cases.
Characterization
XRD analysis
Structural analysis of the as-prepared materials was carried out by ex-situ XRD diffractometer (Panalytical) with a radiation source of Cu Kα. A reaction chamber (Anton Paar XRK 900) was employed for in-situ XRD study. XRD patterns were recorded in isothermal conditions following suitable temperature step programs with a step size of 0.017° for 6 min from RT to 1173 K at 10 K/min. The experiment was carried out in a steel-made sample holder under vacuum condition and its thermal expansion was estimated to be shifted nearly 0.1° with a reference of α-quartz. In fact, the shifting of the observed peaks did not alter the lattice constants considerably. Therefore, during in-situ XRD, the surface of the powdered sample moved significantly from the goniometer centre. Continuous vacuum was introduced into the reaction chamber. The diffraction patterns were documented as a function of time and the experiments of the preparation of MgNb2O6, Mg4Nb2O9 and Mg3Nb6O11 required 10–18 h.
DSC analysis
Differential scanning calorimetry (DSC) thermograms were recorded due to the synthesis of Mg–Nb oxides from RT to 1473 K at 10 K/min using a calorimeter (Setaram) of high temperature with a flow of He and Ar. The reference (αAl2O3, ca. 0.2 g) and mixtures of precursor materials were loaded in Pt crucibles.
Crystallographic information of the powder samples built on XRD patterns were evaluated by using MAUD (Material Analysis Using Diffraction). MAUD is oriented to the studies of material science. It is a general analytical program based on diffraction/reflectivity data and mainly supports Rietveld method34.
Results and discussion
Ex-situ experiment
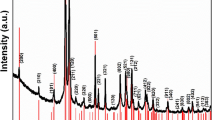
Solid-state synthetic phase evolution is a function of temperature during heating the precursor materials. XRD patterns corresponding to ex-situ measurements of the as-prepared samples were recorded at RT. The XRD patterns monitored along the process of formation of MgNb2O6, Mg4Nb2O9 and Mg3Nb6O11 are reported in the Figs. 1, 2 and 3, respectively and the phase abundance estimated by Rietveld refinement has been inserted in Tables 1, 2 and 3. XRD patterns of the samples were recorded from 10° to 90°; however, due to simplicity and more clear presentation, only the data from 20° to 50° are reported. A detailed description of phase composition of various binary and ternary oxides grown up with increasing temperatures towards solid state synthesis of pure Mg–Nb–O compounds was evaluated by Rietveld refinement with excellent fittings (R% = 1–4%).
During the synthesis of MgNb2O6 (Fig. 1 and Table 1), the binary precursor materials (MgO and Nb2O5) reacted according to the following reaction:
Detailed synthetic method of MgNb2O6 was discussed in the previous study35. In the current issue XRD patterns at RT and maximum reaction temperature 1473 K are shown (Fig. 1a,b). In fact, no solid state reactions were carried out from RT to less than 673 K followed by calcination at 673–1073 K; the evolution of columbite-like MgNb2O6 phase was observed. A nearly pure MgNb2O6 phase was obtained at 1273 K and the solid-state reaction of the desired compound was completed at 1473 K (97 wt%) excepting traces of unreacted more stable monoclinic Nb2O5 (Table 1).
In the case of Mg4Nb2O9 preparation (Fig. 2 and Table 2) the precursor oxides are basically unaffected by the treatment in the range RT-873 K (Fig. 2a,b,c,d). The MgNb2O6 phase appeared as intermediate after annealed at 1073 K (Fig. 2e). With increasing temperature up to 1273 K, diffraction patterns showed simultaneous presence of various binary and ternary compounds (Mg/Nb/O) (Fig. 2f). In this step, the fraction of MgNb2O6 decreased and a considerable amount of the corundum-type Mg4Nb2O9 was formed. At higher test temperature (1473 K), almost pure Mg4Nb2O9 phase was observed (Fig. 2g). The whole process can be described by the following stoichiometry:
However, the final formation of the mixed phase which started at 1473 K occurs on the basis of a reaction between MgO and MgNb2O6 according to the following reaction:
The phase evolution leading to Mg3Nb6O11 is more complicated than in the previous two cases owing to the presence of more reactive metallic Nb in the precursor mixture. The chemical composition remains nearly constant up to 673 K (Fig. 3a,b). Upon annealing at 873 K, niobium oxides (NbO2 and NbO) of lower oxidation states were appeared.
The formation of niobium monoxide (NbO) and dioxide (NbO2) can be explained considering that the synthesis was carried out in static vacuum in order to avoid oxidation of metallic Nb. However, in these conditions, for a semiconducting oxide like Nb2O5, oxygen depletion easily occurs and metallic Nb is thus partially oxidized by the released oxygen (Reactions 4 and 5). Similar effects were also reported for Mg–Ng–O compounds16.
Only at higher temperature (1073 K) MgNb2O6 appeared as a predominant phase (Fig. 2e). In addition, at this temperature various peaks related to Mg3Nb6O11 were observed and its phase quantity gradually increased with the temperature. With raising the synthetic temperature to 1273 K, the abundance of both MgNb2O6 and binary oxides decreased and some amount of Mg4Nb2O9 was observed together with the desired Mg3Nb6O11 phase (Fig. 3f). Then at higher temperature (1473 K), approximately a single phase of the ternary compound (92 wt%) was obtained with some contamination only by MgNb2O6 (8 wt%) (Fig. 3g).
In-situ experiment
In-situ experiments, aimed to study kinetics of the preparation of Mg–Nb–O compounds, were carried out from RT to 1173 K in isothermal conditions (Figs. 4, 5, 6) and corresponding amount of phases obtained by Rietveld analysis are reported in Tables 4, 5 and 6.
Considering that no appreciable reactions take place among the parent materials at lower temperature during in-situ experiments. After an initial pattern acquisition at RT, the solid-state synthetic temperature was increased directly to 973 K during MgNb2O6 preparation and up to 1073 K in the other two cases (Figs. 4, 5, 6 and Tables 4, 5, 6).
In-situ XRD data for MgNb2O6 preparation are shown in Fig. 4 and corresponding phase compositions are inserted in Table 4. In this case the MgNb2O6 phase appeared at 1123 K following Reaction 1 and the abundance of this phase increased with the temperature as proved by ex-situ measurements. Phase evolution with temperature (298–1173 K) are described in our previous work35.
In the case of in-situ Mg4Nb2O9 preparation (Fig. 5) and resultant phase compositions are introduced in Table 5, where only the MgNb2O6 phase was detected for the reasons reported earlier. The presence of this phase, however, confirms the compound representing a key step in the Mg4Nb2O9 phase formation (Reactions 1 and 3).
Similarly, MgNb2O6 phase was observed as an early stage during in-situ study of Mg3Nb6O11 (Fig. 6) and respective phase contents are placed in Table 6. In fact, this phase evolution is much more complicated than the other two phases as also detected in ex-situ experiment, might be owing to the addition of more reactive metallic Nb. The evolution of the various phases indicates that, even in vacuum condition, metallic Nb is easily oxidized at lower temperatures hindering the formation of Mg3Nb6O11.
Through this approach of the ternary compound synthesis, in contrast to ex-situ method, no pure phases were appeared due to temperature and kinetic restrictions. In fact, the highest temperature that can be reached by the in-situ equipment is lower than that of ex-situ preparation because of instrumental limitation. Moreover, incomplete phase transformation is determined by the reaction time inferior to the ex-situ experiments; however, the in-situ test is very important to comprehend the kinetics of the solid-state synthesis. Besides, low-valence niobium oxides (NbO and NbO2) were evidenced for all the cases during in-situ experiment focus the fact of vacuum conditions at which the reactions were carried out.
The evolution of the various Mg–Nb oxide phases during ex-situ experiments are discussed earlier and compared them to the in-situ outcomes. Since it was not possible to obtain the Mg4Nb2O9 and Mg3Nb6O11 phases with the in-situ approach; a comparison between the two approaches can be done only for MgNb2O6. Nevertheless, the following considerations can be done:
Nearly 24 wt% of the MgNb2O6 phase was obtained at 873 K and 1073–1173 K by ex-situ and in-situ tests, respectively point out that in-situ measurement required higher temperature (Fig. 7, Tables 1 and 4)35. This indicates that MgNb2O6 formation is promoted by an oxidizing ambient. Moreover, the reasons behind Mg4Nb2O9 that was not appeared during in-situ experiments can be explained as the growing of the compound started at higher temperature. This evidence also can be interpreted that the formation of Mg4Nb2O9 is composed, at least, by two steps. Both the experiments indicate that the MgNb2O6 phase represents the first step during Mg4Nb2O9 preparation (Reactions 2 and 3). This phase then evolved to the final product at higher temperature. On the contrary, Mg3Nb6O11 formation is not parallel to the others and its mechanism is still an open question. Ex-situ experiment shows that the Mg4Nb2O9 and Mg3Nb6O11 phases were simultaneously appeared at about 1073 K and at this temperature, Mg4Nb2O9 formation is favoured (15%) rather than Mg3Nb6O11 (10%). In-situ measurement clearly shows that the grown-up of MgNb2O6 and Mg4Nb2O9 compounds were hampered in vacuum conditions. Moreover, they possibly react as follows (Reaction 6) at higher temperature to form Mg3Nb6O11 as evidenced by the quantity of the reactants obtained from Rietveld analysis decreased and that of the product increased (1273–1473, Table 3):
In addition, during the formation of Mg3Nb6O11 compound, the MgNb2O6 and Mg4Nb2O9 represent the intermediate steps which evolve to the final product at higher temperature according to the following reactions (Reaction 7 and 8):
Taking into account Reactions 4 and 5, the previous mechanism also explains why oxides with low oxidation states (NbO and NbO2) are always observed during the preparation of Mg3Nb6O11.
DSC studies
The preparation of the Mg–Nb–O compounds was studied by DSC analysis (Fig. 8) monitoring the final products by XRD with the aim of measuring the heat of reaction of the synthesis during the experiment. In the case of MgNb2O6 and Mg4Nb2O9, pure phases were observed at the highest test temperatures; whereas, a yield of 70% was obtained for Mg3Nb6O11 (Figure not attached due to simplicity).
Heating the starting materials for the synthesis up to 1400 K, three curves are obtained as shown in Fig. 8. All curves are characterized by exothermic signal spreads on a very large range of temperature (~ 200 K). Two peaks for each compound can be observed. In the case of MgNb2O6, the presence of two peaks nearly at 1090 and 1180 K (Fig. 8a) suggests that the overall process for the compound formation is complicated than the simple solid-state reaction reported earlier (Reaction 1). The presence of the two steps in the synthetic reaction can be interpreted through the following arguments:
The heating method connected to the DSC measurement is quite similar to that of the in-situ experiment where it was carried out in the reducing environment. In-situ results in fact also point out that the reaction is kinetically low and the intermediate products were formed as NbO and NbO2 in the range of 1023–1173 K.
The low temperature peaks in the DSC thermograms can be explained on the basis of the formation of such intermediates which, in this particular case, further reacted at higher temperature since they were not detected by XRD of the final product. A similar behaviour was revealed from the DSC curves for Mg4Nb2O9 and Mg3Nb6O11 (Fig. 8b,c). In these cases two peaks were also observed but they were shifted at higher temperature (1090 K for the both, and 1220 K and 1205 K for the Mg4Nb2O9 Mg3Nb6O11, respectively) (Fig. 8) indicating a multi-step pathway in the ternary oxide formation.
The heat of formation (HF), in the case of MgNb2O6, was obtained to be minimum (93 kJ/mol) calculated from the DSC results. In addition, the HF of Mg4Nb2O9 and Mg3Nb6O11 compounds towards pure phases were found 140 and 190 kJ/mol, respectively. Considering that the formation reaction is fully accomplished for MgNb2O6 and Mg4Nb2O9. Moreover, it is possible to obtain the HF for the two oxides from the DSC curves (Fig. 9) which results in − 286.17 kJ/g atom and − 294.95 kJ/g atom for MgNb2O6 and the Mg4Nb2O9, respectively. The driving force (DF) of the compounds can be calculated from available database when the synthesis is performed starting from MgO and Nb2O5 (Fig. 9).
This DF values are very close to the cases being 10.41 kJ/g atom and 9.40 kJ/g atom for the MgNb2O6 and Mg4Nb2O9, respectively thus MgNb2O6 representing the more stable phase in agreement with XRD results which clearly showed that during Mg4Nb2O9 preparation, the MgNb2O6 was always obtained as intermediate. Similar calculation for MgNb2O6 was reported earlier by our group35.
Conclusions
In this work three Mg–Nb–O compounds of MgNb2O6, Mg4Nb2O9 and Mg3Nb6O11 were successfully prepared by solid-state reactions and the formation mechanism was characterized by means of XRD performed both in-situ and ex-situ. XRD analysis shows that the solid-state reactions leading to the ternary compounds are kinetically slow; therefore, high temperature with long time was needed for calcination of the precursor materials to obtain nearly pure phases. Moreover, the formation of MgNb2O6 and Mg4Nb2O9 was hampered when the synthesis was carried out in absence of oxygen. XRD also shows that MgNb2O6 represents an intermediate in the Mg4Nb2O9 and Mg3Nb6O11 formation. For the first time the heat of formation for the MgNb2O6 and Mg4Nb2O9 phases were estimated by means of DSC analysis which shows that the MgNb2O6 is thermodynamically more stable phase. The as-prepared three pure Mg–Nb–O phases, which are utilized as catalysts/additives to improve H2 sorption kinetics of MgH2 towards practical application, will be discussed in the forthcoming issues.
References
Züttel, A. Materials for hydrogen storage. Mater. Today 6, 24–33 (2003).
Chen, P. & Zhu, M. Recent progress in hydrogen storage. Mater. Today 11, 36–43 (2008).
Selvam, P., Viswanathan, B. & Swamy, C. S. Magnesium and magnesium alloy hydrides. Int. J. Hydrog. Energy 11, 169–192 (1986).
Gerard, N. & Ono, S. Hydrogen in intermetallic compounds II. Springer, 178–82 (1992).
Oelerich, W., Klassen, T. & Bormann, R. Metal oxides as catalysts for improved hydrogen sorption in nanocrystalline Mg-based materials. J. Alloys Compd. 315, 237–242 (2001).
Barkhordarian, G., Klassen, T. & Bormann, R. Fast hydrogen sorption kinetics of nanocrystalline Mg using Nb2O5 as catalyst. Scr. Mater. 49, 213–217 (2003).
Barkhordarian, G., Klassen, T. & Bormann, R. Effect of Nb2O5 content on hydrogen reaction kinetics of Mg. J. Alloys Compd. 364, 242–246 (2004).
Dolci, F., Di Chio, M., Baricco, M. & Giamello, E. Niobium pentoxide as promoter in the mixed MgH2/Nb2O5 system for hydrogen storage: A multitechnique investigation of the H2 uptake. J. Mater. Sci. 42, 7180–7185 (2007).
Dolci, F., Di Chio, M., Baricco, M. & Giamello, E. The interaction of hydrogen with oxidic promoters of hydrogen storage in magnesium hydride. Mater. Res. Bull. 44, 194–197 (2009).
Friedrich, O. et al. MgH2 with Nb2O5 as additive for hydrogen storage: Chemical, structural and kinetic behavior with heating. J. Acta Mater. 54, 105–110 (2006).
Schimmel, H. G., Huot, J., Chapon, L. C., Tichelaar, F. D. & Mulder, F. M. Hydrogen cycling of niobium and vanadium catalyzed nanostructured magnesium. J. Am. Chem. Soc. 127, 14348–14354 (2005).
Mandal, T. K., Sebastian, L., Gopalakrishnan, J., Abrams, L. & Goodenough, J. B. Hydrogen uptake by barium manganite at atmospheric pressure. Mater. Res. Bull. 39, 2257–2264 (2004).
Friedrich, O. et al. Nb2O5 “Pathway effect” on hydrogen sorption in Mg. J. Phys. Chem. B 110, 7845–7850 (2006).
Rahman, M. W., Livraghi, S., Dolci, F., Baricco, M. & Giamello, E. Hydrogen sorption properties of ternary Mg-Nb-O phases synthesized by solid-state reaction. Int. J. Hydrog. Energy 36, 7932–7936 (2011).
Rahman, M. W. et al. Effect of Mg–Nb oxides addition on hydrogen sorption in MgH2. J. Alloys Compd. 509S, S438–S443 (2011).
Bruck, E., Route, R. K., Raymakers, R. J. & Feigelson, R. S. J. Cryst. Grow. Surface stability of lithium triborate crystals grown from excess B2O3 solutions. 128, 933–937 (1993).
Hong, Y. S., Park, H. B. & Kim, S. J. Preparation of Pb(Mg1/3Nb2/3)O-3 powder using a citrate-gel derived columbite MgNb2O6 precursor and its dielectric properties. J. Eur. Ceram. Soc. 18, 613–619 (1998).
Kong, L. B., Ma, J., Huang, H. & Zhang, R. F. Crystallization of magnesium niobate from mechanochemically derived amorphous phase. J. Alloys Compd. 340, L1–L4 (2002).
Belous, A., Ovchar, O., Jancar, B. & Bezjak, J. The effect of non-stoichiometry on the microstructure and microwave dielectric properties of the columbites A2+ Nb2O6. J. Eur. Ceram. Soc. 27, 2933–2936 (2007).
Belous, A. G. et al. Synthesis and properties of columbite-structure Mg 1− x Nb 2 O 6− x. Inorg Mater. 43, 412–417 (2007).
Sreedhar, K. & Pavaskas, N. R. Synthesis of MgTiO3 and Mg4Nb2O9 using stoichiometrically excess MgO. Mater Lett. 53, 452–455 (2002).
Kim, N. K. Synthesis chemistry of MgNb2O6 and Pb(Mg1/3Nb2/3)O-3. Mater Lett. 32, 127–130 (1997).
Camargo, E. R., Kakihana, M., Longo, E. & Leite, E. R. Pyrochlore-free Pb(Mg1/3Nb2/3)O-3 prepared by a combination of the partial oxalate and the polymerized complex methods. J. Alloys Compd. 314, 140–146 (2001).
Pagola, S., Carbonio, R. E., Alonso, J. A. & Fernàndez-Dìaz, M. T. Crystal structure of MgNb2O6 columbite from neutron powder diffraction data and study of the ternary system MgO-Nb2O5-NbO, with evidence of formation of new reduced pseudobrookite Mg5-xNb4+xO15-∂ (1.14≤ x≤ 1.60) phases. J. Solid State Chem. 134, 76–84 (1997).
Fu, Z.-F., Liu, P., Chen, X.-M., Ma, J.-L. & Zhang, H.-W. Low-temperature synthesis of Mg4Nb2O9 nanopowders by high-energy ball-milling method. J. Alloys Compd. 493, 441–444 (2010).
Abbattista, F. & Rolando, P. Metaniobates MgNb2O6 , MnNb2O6 and on respectively reduction products MgNb2O3.67 and MnNb2O3.67. Anal. Chim. 61, 196 (1971).
Marinder, B. O. Mg3Nb6O11—Oxide containing isolated octahedra of niobium atoms—Structure determination and refinement from X-ray powder film data. Chem. Scr. 11, 97–101 (1977).
Rahman, M. W., Livraghi, S., Enzo, S., Giamello, E. & Baricco, M. Synthesis and characterization of ternary Mg–Nb–O compounds. In 3rd World Congress of Young Scientists on Hydrogen Energy Systems (HYSYDAYS–2009), October 7–9, 2009, Turin, Italy.
Ananta, S. Phase morphology evolution of magnesium niobate powders synthesized by solid-state reaction. Mat. Let. 58, 2781–2786 (2004).
Sun, D.C., Senz, S. & Hesse, D. Crystallography, microstructure and morphology of Mg4Nb2O9/MgO and Mg4Ta2O9/MgO interfaces formed by topotaxial solid state reactions. 26, 3181–3190 (2006).
You, Y.C., Park, H.L., Song, Y.G., Moon, H.S. & Kim, G.C. Stable phases in the MgO-Nb2O5 system at 1250-degrees-C, J. Mater. Sci. Lett. 13, 1487–1489 (1994).
Wong-Ng, W., McMurdie, H.F., Paretzkin, B., Zhang, Y., Davis, K.L., Hubbard, C.R., Dragoo, A.L. & Stewart, J.M. Standard X-ray diffraction powder patterns of ceramic phases. 2, 191–201 (1987).
Bertaut, E. F., Corliss, L., Forrat, F., Aleonard, R. & Pauthenet, R. Etude de niobates et tantalates de metaux de transition bivalents. J. Phys. Chem. Solids 21, 234–251 (1961).
Lutterotti L, Matthies S, Wenk H-R, Schulz AJ, Richardon J. J. of Apply. Phys. 81 (1997) p. 594–600. MAUD is available at http://www.ing.unitn.it/~maud.
Rahman, M. W. Preparation of magnesium diniobate by solid–state reactions and its role for hydrogen storage. J. Aust. Ceram. Soc. 55, 579–586 (2019).
Acknowledgements
The author acknowledges the technical support of Professor Marcello Baricco of Università di Torino, Italy.
Funding
This research didn't get a particular award from subsidizing organizations in general society, business, or not-revenue driven areas.
Author information
Authors and Affiliations
Contributions
The sole author Md. Wasikur Rahman conducted the entire project, particularly, synthesized, characterized and analyzed materials as well as wrote and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The author declares no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rahman, M.W. Synthetic approach of ternary magnesium niobate (Mg–Nb–O) compounds. Sci Rep 11, 16065 (2021). https://doi.org/10.1038/s41598-021-95690-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-95690-4
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.