Abstract
Along with early-onset cancers, multiple primary cancers (MPCs) are likely resulting from increased genetic susceptibility; however, the associated predisposition genes or prevalence of the pathogenic variants genes in MPC patients are often unknown. We screened 71 patients with MPC of the stomach, colorectal, and endometrium, sequencing 65 cancer predisposition genes. A subset of 19 patients with early-onset MPC of stomach and colorectum were further evaluated for variants in cancer related genes using both normal and tumor whole exome sequencing. Among 71 patients with MPCs, variants classified to be pathogenic were observed in 15 (21.1%) patients and affected Lynch Syndrome (LS) genes: MLH1 (n = 10), MSH6 (n = 2), PMS2 (n = 2), and MSH2 (n = 1). All carriers had tumors with high microsatellite instability and 13 of them (86.7%) were early-onset, consistent with LS. In 19 patients with early-onset MPCs, loss of function (LoF) variants in RECQL5 were more prevalent in non-LS MPC than in matched sporadic cancer patients (OR = 31.6, 2.73–1700.6, p = 0.001). Additionally, there were high-confidence LoF variants at FANCG and CASP8 in two patients accompanied by somatic loss of heterozygosity in tumor, respectively. The results suggest that genetic screening should be considered for synchronous cancers and metachronous MPCs of the LS tumor spectrum, particularly in early-onset. Susceptibility variants in non-LS genes for MPC patients may exist, but evidence for their role is more elusive than for LS patients.
Similar content being viewed by others
Introduction
The identification of inherited DNA variants in cancer predisposition genes is clinically important for both cancer prevention and treatment as it can reduce cancer morbidity and mortality in individuals carrying pathogenic variants1. The prevalence of pathogenic variants in cancer predisposition genes is lower than 1% of the healthy population and 5–8% in patients diagnosed with cancer2,3,4. Pathogenic variants in cancer predisposition genes are therefore rare. On the other hand, wide-spread surveillance screening may result in false positive findings or findings of uncertain significance, yielding to increased anxiety and unnecessary or ineffective clinical procedures. Thus, continued research supporting more effective and precise genetic screening guidelines is needed1.
Multiple primary cancers (MPCs), referring to two or more histologically distinct cancers diagnosed in one individual, is gradually recognized as an important medical problem with a reported frequency of 2–17% in the cancer population5,6. Along with early-onset cancer, MPC have been regarded as high-risk of heritability7 and those patients therefore represent excellent candidates for genetic screening. However, limited data is available for the prevalence and type of pathogenic variants in the cancer predisposition genes of these patients.
Gastric cancer (GC) and colorectal cancer (CRC) are the two most common cancers in Korea, with 5-year survival up to 70%8 and their combination is the most common instance of MPCs, representing ~ 2% of Korean GC or CRC cases9,10. About 15% of each of the cancer types can be characterized by elevated microsatellite instability (MSI) caused by mismatch repair (MMR) deficiency11,12,13,14,15. Together with Endometrial cancer (EC)—another cancer characterized by MSI16—GC and CRC are the main cancer types associated with Lynch Syndrome (LS), caused by pathogenic variants in MMR genes, resulting in up to an 80% lifetime risk of cancers17,18. In particular, 2–5% of CRC and EC, as well as ~ 15% of early-onset CRC are thought to be associated with LS19,20. MPCs with cancer types in the spectrum of LS therefore represent some of the strongest candidates for genetic screening, as the prevalence of LS among MPC is unknown.
Here we present the results of the genetic screening of 71 patients with MPC of the stomach, colorectal and endometrium. We evaluate the distribution and prevalence of pathogenic variants in 65 cancer predisposition genes, including LS susceptibility genes, as a function of age and clinicopathological features. We further characterize inherited coding variants focusing on DNA repair and cancer related genes in 19 early-onset MPC (eoMPC) patients, proposing candidate MPC susceptibility genes based on the analysis of loss of function (LoF) variants in candidate tumor suppressor genes, their relative prevalence and association with the mutational landscape of the associated tumors.
Results
Identification of Lynch Syndrome patients by multigene targeting panel
We investigated the prevalence of LoF variants in cancer predisposition genes using a cohort of 71 patients with MPCs. Fifty-four (76.1%) patients were diagnosed with GC and CRC (23–42.6% synchronous) while 14 diagnosed with EC and CRC (N = 13) or GC (N = 1). Three patients had 3 or more types of cancers. Thirty (42.3%) patients were diagnosed with MPC before age 55 and classified as eoMPCs. A family history of cancer was more prevalent in eoMPCs: of the 56 patients for which family history was available, twenty-four (42.9%) patients had two or more first-degree relatives affected with any type of cancer, and 15 of them were eoMPCs (Odds Ratio [OR] = 8.58, 2.21–39.59, p < 0.001). Twenty-six (36.6%) patients had one or more MSI-H tumors, a higher proportion than the one observed in single-cancers of the same type (~ 15%)11,12,13,16 (Supplemental File 1, Table S1). This suggests an important contribution of defects in MMR to MPC development. The germline DNA of the patients was sequenced using a multigene targeting panel of 65 cancer predisposition genes. A total of 15/71 (21.1%) patients were found to carry variants classified to be pathogenic or likely pathogenic (P/LP) and all the variants affected LS related genes (Table 1, Table S2 in Supplemental File 1): MLH1 (n = 10), MSH6 (n = 2), PMS2 (n = 2), and MSH2 (n = 1), a distribution comparable to the previous reports17. There was one patient with multi-locus inherited neoplasia alleles syndrome (MINAS) harboring germline pathogenic variants in both MLH1 and BRCA1 gene21. One of recurrent P/LP variants of MLH1 was c.1758dupC, a common variant in Korean LS patients22. There was a P/LP variant in MSH2 (c.942 + 3A > T) that was reported as a frequent de novo mutation and founder variant in Newfoundland23,24. in a patient diagnosed with four types of cancer (GC, CRC, EC, and lung cancer). Of the remaining 56 of patients, 42 had VUS altering 43 genes, leaving the contribution of these genes to MPC undetermined.
Characteristics of LS patients
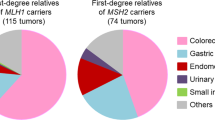
All 15 LS patients had at least one MSI-H tumor in contrast to 11/56 non-LS patients. LS patients were also more likely to have two or more first-degree relatives (9/10, OR = 17.67, 2.12–836.73, p = 0.001, Supplemental file 1, Tables S1 and S2). The association of the variants with tumor characteristics were further investigated. The loss of expression of the protein corresponding to the gene with P/LP variants was confirmed in all 19 MSI-H tumors investigated from all LS patients (Supplemental file 1, Table S3)25. The LS patients were more likely to be diagnosed with eoMPC (13/15, OR = 14.31, 2.79–144.22, p < 0.001, Fig. 1) but equally likely to present with synchronous tumors (6/15, OR = 0.89, 0.23–3.26, p > 0.999). This suggests that consistent with Lynch syndrome, LS MPC patients are diagnosed earlier. However, the variants are unlikely to influence the relative timing of the two cancer diagnosis. More importantly, these results showed that the majority of MPC patients do not have pathogenic variants known to cause LS. Even restricting to 13 patients with eoMPCs and strong family history of cancer (two or more first-degree relatives over two successive generations), cancer predisposition genes were not altered in nearly half of the patients (6/13, Table S2 in the Supplemental file 1). The likely genetic cause of MPC in these patients remains undetermined.
A scatter plot between age of first and second cancer by with/without pathogenic or likely pathogenic (P/LP) germline variants and concurrency of the tumors in each patient. There were 15 (21.1%) of patients with germline P/LP variants (red colors). Among early-onset multiple primary cancers (MPCs), 43.3% (13/30) patients had P/LP variants while only 4.9% (2/41) patients were related to P/LP variants in late-onset MPCs.
Identification of candidate cancer predisposition genes in early-onset MPC
To identify additional variants and genes underlying a possible cancer susceptibility in non-LS patients, we sequenced the whole exome of 19 eoMPC patients with gastric and colorectal cancer which is the most common combination of MPCs in Korea9,10. Seven of the patients were LS patients, as determined above, and used as controls for the cancer predisposition genes discovery process. We identified 111,842 high-confidence variants across all patients, of which 3,211 were rare (MAF < 0.01) in East-Asian population and predicted to be damaging, affecting 2,675 genes. We investigated a set of 382 genes (referred to as cancer genes) involved in DNA repair, cancer susceptibility and progression (Supplemental file 1, Table S4)26. Of those, 66 were altered by 82 variants, including 7 in the MLH1 gene of the LS patients, therefore confirming the panel sequencing results and validating the initial WES variant calling and filtering approach.
For a given candidate cancer predisposition genes, we expect that the prevalence of LoF alterations observed in eoMPC cohort would be higher than in a matching sporadic cancer cohort. Despite some LS phenotypes being reported26, TCGA gastric and colorectal cancer patients are mainly from sporadic cancers. We selected 70 of these patients with East-Asian ancestry to closely match the Korean ancestry of eoMPC cohort and reduce spurious findings resulting from population differences. In these patients, we identified a total of 87 LoF variants affecting 47/382 cancer genes. We confirmed that LoF variants in MLH1 were more prevalent in LS patients than in TCGA patients (7/7 vs 2/70 p < 0.001 Table 2, Table S5 in the Supplemental file 1). Importantly, 5/7 of the MLH1 LoF variants in LS patients were predicted to be high-confidence LoF variants, indicating that most, but not all, known pathogenic variants can be identified through this approach. RECQL5 was the only gene significantly more altered in non-LS patients than in TCGA patients (4/12 vs 1/70, OR:31.66, 2.73–1700.6, p = 0.0013, Table 2). In particular two of these patients had the same RECQL5 variant (p.R441Q which is not seen in the TCGA patients (p = 0.02, Table S5in the Supplemental file 1) The variant is rare, with a global minor allelic fraction of 3.6 × 10–4. Interestingly 89 of the 101 observed minor alleles (out of 278,974 total alleles observed) in the gnomAD dataset27 belong to the East-Asian population, suggesting a possible Korean-specific effect.
Bi-allelic alterations in corresponding tumors as a guide for cancer predisposition genes identification
According to Knudson’s two-hit model28, the wild-type allele of tumor suppressor genes is frequently lost or mutated in the tumor. Such combination of inherited deleterious variants with somatic loss—referred to as bi-allelic alteration—can help prioritize candidate cancer predisposition genes26,29. We sequenced the exome of 37 tumors from all 19 patients with eoMPC (Supplemental file 1, Table S7) and identified 35 somatic bi-allelic alterations by LoF mutations (N = 5), loss of heterozygosity (N = 27) or both (N = 3) focusing on candidate germline LoF genes.
The MLH1 gene was somatically altered in 13/15 tumors from LS patients and at least one tumor from each of the 7 LS patient. (Table 3). Importantly, 5/7 MLH1 variants in LS patients were high-confidence LoF variants. This observation confirms the validity of the investigation of bi-allelic alteration to identify cancer predisposition genes, at least for high-confidence LoF variants in known predisposition genes. In contrast, the variants in the main candidate susceptibility gene in non-LS patients, RECQL5, were not high-confidence LoF variant and the genes was not affected in any of the 6 tumors from 4 variant carriers or any of the 22 tumors from all non-LS patients. Expanding the analysis to other candidate genes affected by LoF germline variants in non-LS patients, we identified 17 genes in 10 patients also affected by bi-allelic alterations in one or more tumors. Two were altered via mutations and 15 through LOH. Among these, 2 genes were affected by high-confidence LoF germline variants: FANCG (germline splice site variant, c.307 + 1G > C) in gastric tumor of dou_004 and CASP8 (germline deletion variant, c.658_659del) in colorectal tumor of dou_010 (Table 3). Interestingly, this analysis also revealed somatic alterations of LS genes in absence of inherited LS mutation in a non-LS patient (dou_009) with both of MSI-H tumors in the stomach and colon. Indeed, somatic allelic imbalance was observed in MLH1 (stomach and colon) and PMS2 (stomach only) and the expression of both encoded proteins was lost from both tumors. Loss of one allele in the tumor would likely not be sufficient to impair MMR, raising the possibility of undetected germline LoF variants in these LS genes in this patient, perhaps through alterations not easily detected by WES30. There was one tumor (colon cancer of dou_005) with double somatic mutations31 in MMR genes (c.582-591del at MLH1 and c.493delT at PMS2, variant allele fraction was 0.186 and 0.188, respectively) and the patient harbors germline pathogenic variant at MLH1. The details of LoF germline variants and somatic alteration of the corresponding tumors were described in Table S6 (Supplementary file 1).
Discussion
As expected, the present result confirmed that individuals with MPC, particularly early onset, have a higher likelihood of LS: MMR related P/LP germline variants were observed in 21% of MPC (15/71) and in 43% of eoMPC (13/30) patients, a proportion comparable to ~ 15% of early-onset CRC20. The elevated extracolonic cancer risk following colorectal cancer in Lynch syndrome was reported (~ 5 and 40 times of gastric and endometrial cancer risk compared to general population, respectively)32, and this study supports those results. Therefore, due to the high prevalence of germline mutations individuals with MPC in the LS tumor spectrum should undergo germline testing, including when MSI status is unknown.
Because MSI-H was considered as a hallmarks of LS, universal screening of all CRC and EC has been recommended33,34. In addition, MSI screening for gastric cancers in regions where gastric cancer is highly prevalent like Korea is a way to identify individuals and families who can benefit from germline testing of LS genes, so that surveillance and risk reducing interventions can be undertaken along with cascade testing of family members. At the same time, MSI testing is valuable for other types of solid cancers for other purpose: MSI-H is a biomarker for response to immune checkpoint inhibitors that is a breakthrough for treating advanced solid cancers regardless of its origin35. As MSI-H is predictive of LS across a broad tumor spectrum36, screening of tumor MSI status in all patients with an initial cancer, especially for LS spectrum tumors and early-onset cancer, and consecutive germline testing for patients with MSI-H tumor will help refine the diagnosis, giving an opportunity to diagnose LS and avoid or detect the second cancer as early as possible.
Genetic analysis of families with high occurrence of cancer using cancer predisposition genes panels or exome sequencing has become standard to identify the underlying cancer susceptibility variants. But penetrance, interaction with environmental factors, and size of the pedigree may affect power of such linkage studies1,37,38,39. Evaluating both germline and somatic alterations is another reasonable approach to find cancer predisposition genes26,29. The present results showed that characterizing recurrent LoF genes, truncating variants and loss of heterozygosity are useful to prioritize candidate cancer predisposition genes; any combination of them unambiguously identified germline MLH1, a known cancer predisposition genes in LS patients, in multiple patients clinically diagnosed with LS. It suggests that MPC is a strong phenotype to find cancer predisposition genes, therefore this approach would be worthy to be expanded to other combinations of MPCs. However, the types of MPCs would be different by geographical regions or ancestry: region-specific environmental factors can contribute to the oncogenesis and lead to different cancer types within the LS spectrum. For instance, MPCs including GC could be a hallmark of LS in Korea but not in the United States where GC incidence is much lower. This could explain why GC risk in LS may have been underestimated17,40. Therefore, geographical and/or ancestry specific cancer epidemiology needs to be accounted for in the genetic screening guidelines.
In non-LS patients with eoMPC, however, evidence of germline susceptibility was more elusive. RECQL5 was a frequently recurrent gene and variant (p.R441Q) compared to East Asian sporadic cancer population (TCGA). Despite careful matching, some differences between the eoMPC and matched TCGA cohorts remained and could have confounded this observation. Notably, eoMPC patients were all younger than 55 and exclusively of Korean descent, while the TCGA patients included in the analysis were not selected for age and included non-Korean Asians. Importantly, the allele frequency of the RECQL5 variant was 0.4% of EAS population in gnomAD, and there was no evidence of somatic LOH in the corresponding tumors, suggesting that additional analysis and experiment are required to establish its pathogenicity. CASP8 and FANCG were high-confidence LoF germline gene with bi-allelic alteration in CRC and GC, respectively. CASP8 encodes a member of caspase family and play a role in apoptosis, and some of its polymorphism have been reported as susceptibility to various cancers including CRC41,42,43. FANCG is a gene encoding Fanconi anemia (FA) group G protein, and part of the FA DNA damage repair pathway and in which germline pathogenic variants often predispose to cancers44,45,46. Thus, the CASP8 and FANCG variants identified in these two MPC cases could underlie their disease susceptibility though additional functional studies would be necessary to demonstrate their pathogenicity47,48,49,50.
Despite evaluating all inherited coding variants in cancer genes and somatic changes in the corresponding tumors, we did not find a clear inherited predisposition that caused multiple cancers in over half of patients with eoMPC, even with strong family cancer history. Not only cancer predisposition genes, but also environmental factors such as smoking and alcohol also increase the risk of cancers51,52,53.
Our study has inherent limitations. The present results did not cover epigenetic changes affecting candidate cancer predisposition genes in tumors, and methylation of the MLH1 promoter in particular is known to be a mechanism for somatic loss of function in ~ 20% of CRC54. Furthermore, we restricted the analysis to 382 well studied cancer genes, more likely to impact cancer susceptibility, therefore leaving the possibility to miss un-expected cancer predisposition genes. The complete analysis of whole exomes to identify rare disease susceptibility variants would indeed be intractable for the cohort under study. Careful analysis of the pedigree and genetic comparison of family members, a typical standard in such genetic susceptibility studies, was not possible due to the absence of family history information in many cases and to the retrospective nature of the study. However, this study is to our knowledge the largest study of patients with MPCs, evaluated using the same multigene targeting panel, and where both germline and multiple tumors DNA were investigated in the highest risk patients.
eoMPC of colorectal, endometrial, and gastric cancer are considerably enriched for LS patients, supporting the genetic screens of related family members as well as enhanced monitoring for younger patients after their first diagnosis. Routine tumor screening of MSI for patients with initial LS related cancers and consecutive germline testing for patients with MSI-H is worthy to diagnose LS and early detection or avoid second cancer. While susceptibility variants in non-LS genes for MPC patients may exist, evidence for their role is more elusive than for LS patients and would require deeper genetic investigation and complementary functional studies.
Methods
Population
We performed a retrospective population-based study targeting patients who were treated for two or more cancers in the stomach, colorectum, or endometrium at Severance Hospital, Yonsei University College of Medicine between January 2001 and December 2016. All methods were carried out in accordance with relevant guidelines and regulations. This study was approved by Institutional Review Board (IRB) of Severance hospital of the Yonsei University Health System and by the IRB of the University of California, San Diego (4-2017-0434, 191,543). The IRB of Severance hospital of the Yonsei University Health System waived the requirement for patient informed consent as the study is retrospective by design. The patients were selected using following criteria: (1) two or more cancers were pathologically confirmed, (2) multiple tumors were histologically different, not a metastatic or recurrent tumor from one cancer, (3) normal tissues were available and histologically confirmed for sequencing. DNA was obtained from formalin fixed paraffin embedded (FFPE) normal and tumor tissues. Early-onset MPC (eoMPC) was defined by the diagnosis of a second cancer at age 55 or younger. The clinico-pathologic characteristics of the patients and tumors including age, sex, family history of cancer, MSI or MMR status of tumors were evaluated. For evaluating MSI status, two mononucleotide repeat markers (BAT25 and BAT26) and three dinucleotide repeat markers (D5S346, D2S123, and D17S250) were used by polymerase chain reaction55, and MSIsensor was used with cut-off of MSI score > 3.5 in Whole Exome Sequencing (WES) data56. To evaluate MMR protein expression, immunohistochemistry (IHC) was conducted in four MMR genes; MLH1 (ready-to-use, clone M1, Roche, Indianapolis, IN, USA), MSH2 (ready-to-use, clone G219-1129, Roche), MSH6 (1:100, clone 44, Cell Marque, Rocklin, CA, USA), and PMS2 (1:40, clone MRQ28, Cell Marque). An MMR-deficient (dMMR) tumor was defined as a tumor showing loss of expression of any of the four MMR proteins. If a tumor was classified as any one of MSI-high (MSI-H) or deficiency MMR (dMMR) it was considered as MSI-H. The concurrency of tumors was classified as synchronous tumors when the interval between tumors was less than 1 year, otherwise considered as metachronous tumors.
Germline multigene targeting panel
Germline DNA of patients with MPCs was evaluated using a customized targeted capture sequencing panel (OncoRisk, Celemics, Seoul, Korea) covering all coding sequences and intron–exon boundaries of the coding exons of known 65 cancer predisposition genes (Supplemental file 1, Table S8)57. Both structural variants and nucleotide variants were evaluated. The germline variants were classified into pathogenic, likely pathogenic, variant of uncertain significance (VUS) and reported following the guidelines of the American College of Medical Genetics and Genomics 201758. Only pathogenic or likely pathogenic (P/LP) germline variants in cancer predisposition genes were considered in the analysis. Read-depth based detection of structural variants was conducted using the ExomeDepth software. Chromosomal copy number variations (CNVs) detected by ExomeDepth were further crosschecked using our custom pipeline; this retrieved base-level depth-of-coverage for each bam file using SAMtools software and normalized the depths against those of other samples in the same batch. There were no significant pathogenic CNV detected in the studied samples.
Whole exome sequencing
Data generation
WES of normal and tumors was conducted for patients with eoMPCs in the stomach and colon. Genomic DNA was extracted from the confirmed normal and tumor tissues of FFPE. SureSelect sequencing libraries were prepared according to the manufacturer’s instructions (Agilent SureSelect All Exon V6 kit, Santa Clara, CA, USA), and HISEQ2500 sequencing system (Illumina™, San Diego, CA, USA) performed sequencing with read lengths of 2 × 100 bp. The statistics and quality metrics of normal and tumors were described in Supplemental file 1, Tables S7 and S9.
Data analysis
The reads were aligned to hg19 reference genome by Burrows-Wheeler Aligner software (BWA 0.7.17) and duplicated reads were removed by Picard (MarkDuplicates) through bcbio-nextgen (v.1.2.3)59 For germline and somatic analysis, variants were called by Genome Analysis Toolkit Haplotype joint caller (GATK v3.9) and mutect260,61, respectively. Variants were annotated by refGene using ANNOVAR62, CADD scores (CADD13 and CADDindel)63, population allelic frequency in ExAC v3.064, and membership to ClinVar65.
To discover novel cancer predisposition genes, we focused on high-confidence (TLOD > 12, FS < 10, SEQQ > 60, MQ ≥ 60, STRANDQ > 40, DP > 10), rare (minor allele fraction was < 0.01 in ExAC of Eastern Asian population), and damaging (exonic/splicing, non-synonymous variants with > 20 of CADD score) variants in 382 genes of 11 cancer-relevant pathways26, and they were defined as LoF germline variants. High-confidence of LoF variants were predicted by Loss-Of-Function Transcript Effect Estimator (LOFTEE) plugin for Ensembl VEP (v.99) that targeting stop-gained, splice site disrupting, frameshift variants66,67. Copy number alterations of tumors compared to normal was estimated using CNVkit (V0.8)68 The gene level copy number ratio was calculated as the weighted mean of all bins covered by the whole segment overlapping the gene. Genes had ≥ 3 segments of copy number changes were included, and log2 copy number < − 0.3 was defined as deletion. Somatic allelic imbalance (AI) in tumors of a given heterozygous germline variants were estimated using hapLOHseq69. Germline variants of tumors were obtained by GATK haplotype caller and filtering out variants were not observed in germline variants in normal sample. Phase of genotypes were estimated with a companion phasing utility with hapLOHseq. Event prevalence was set 0.1, and AI was defined when posterior probability of being in AI was over 90%. To assess loss of heterozygosity (LOH) of a given heterozygous germline variants in tumors as a secondary hit mechanism of inactivation of cancer predisposition genes26,28, we evaluated germline and somatic bi-allelic alteration of corresponding tumors. When there was any one of somatic damaging mutation or AI event in candidate germline cancer predisposition genes, it was considered as bi-allelic alteration.
To compare the frequency of recurrent cancer predisposition genes in patients of eoMPCs to that of the cancer genome atlas (TCGA) cohort, germline data of East-Asian patients (≥ 80% of admixture)70 with stomach cancer or colorectal cancer in TCGA cohort was used26,71. Variants were filtered for rare and damaged variants similar to LoF germline variants in MPCs cohort. For recurrent variants and genes in eoMPCs cohort, the frequency was compared between eoMPCs cohort and selected TCGA cohort.
Statistical analysis
Continuous variables were compared by two-sided Mann–Whitney test and categorical variables were compared by two-sided Fisher’s exact test. P < 0.05 was considered as statistically significant and R version 3.6.1(R Foundation for Statistical Computing, Vienna, Austria) was used for generating figures and statistical analysis.
References
Rahman, N. Realizing the promise of cancer predisposition genes. Nature 505, 302–308. https://doi.org/10.1038/nature12981 (2014).
Huang, K. L. et al. Pathogenic germline variants in 10,389 adult cancers. Cell 173, 355–370. https://doi.org/10.1016/j.cell.2018.03.039 (2018).
Evans, J. P., Powell, B. C. & Berg, J. S. Finding the rare pathogenic variants in a human genome. JAMA 317, 1904–1905. https://doi.org/10.1001/jama.2017.0432 (2017).
Mersch, J. et al. Prevalence of variant reclassification following hereditary cancer genetic testing. JAMA 320, 1266–1274. https://doi.org/10.1001/jama.2018.13152 (2018).
Cybulski, C., Nazarali, S. & Narod, S. A. Multiple primary cancers as a guide to heritability. Int J Cancer 135, 1756–1763. https://doi.org/10.1002/ijc.28988 (2014).
Vogt, A. et al. Multiple primary tumours: challenges and approaches, a review. ESMO Open 2, e000172. https://doi.org/10.1136/esmoopen-2017-000172 (2017).
Whitworth, J. et al. A clinical and genetic analysis of multiple primary cancer referrals to genetics services. Eur. J. Hum. Genet. 23, 581–587. https://doi.org/10.1038/ejhg.2014.157 (2015).
Hong, S. et al. Cancer statistics in korea: incidence, mortality, survival, and prevalence in 2017. Cancer Res. Treat. 52, 335–350. https://doi.org/10.4143/crt.2020.206 (2020).
Cho, I. et al. Risk factors for double primary malignancies and their clinical implications in patients with sporadic gastric cancer. Eur. J. Surg. Oncol. 40, 338–344. https://doi.org/10.1016/j.ejso.2013.10.027 (2014).
Yun, H. R. et al. Double primary malignancy in colorectal cancer patients–MSI is the useful marker for predicting double primary tumors. Int. J. Colorectal. Dis. 24, 369–375. https://doi.org/10.1007/s00384-008-0541-x (2009).
Cancer Genome Atlas Research. Comprehensive molecular characterization of gastric adenocarcinoma. Nature 513, 202–209. https://doi.org/10.1038/nature13480 (2014).
Cancer Genome Atlas. Comprehensive molecular characterization of human colon and rectal cancer. Nature 487, 330–337. https://doi.org/10.1038/nature11252 (2012).
Choi, Y. Y. et al. Microsatellite instability and programmed cell death-ligand 1 expression in stage II/III gastric cancer: post hoc analysis of the CLASSIC randomized controlled study. Ann. Surg. 270, 309–316. https://doi.org/10.1097/SLA.0000000000002803 (2019).
Kim, C. G. et al. Effects of microsatellite instability on recurrence patterns and outcomes in colorectal cancers. Br. J. Cancer 115, 25–33. https://doi.org/10.1038/bjc.2016.161 (2016).
Guastadisegni, C., Colafranceschi, M., Ottini, L. & Dogliotti, E. Microsatellite instability as a marker of prognosis and response to therapy: a meta-analysis of colorectal cancer survival data. Eur. J. Cancer 46, 2788–2798. https://doi.org/10.1016/j.ejca.2010.05.009 (2010).
Cancer Genome Atlas Research. Integrated genomic characterization of endometrial carcinoma. Nature 497, 67–73. https://doi.org/10.1038/nature12113 (2013).
Moller, P. et al. Cancer incidence and survival in Lynch syndrome patients receiving colonoscopic and gynaecological surveillance: first report from the prospective Lynch syndrome database. Gut 66, 464–472. https://doi.org/10.1136/gutjnl-2015-309675 (2017).
Koornstra, J. J. et al. Management of extracolonic tumours in patients with Lynch syndrome. Lancet Oncol. 10, 400–408. https://doi.org/10.1016/S1470-2045(09)70041-5 (2009).
Cohen, S. A., Pritchard, C. C. & Jarvik, G. P. Lynch syndrome: from screening to diagnosis to treatment in the era of modern molecular oncology. Ann. Rev. Genom. Hum. Genet. 20, 293–307. https://doi.org/10.1146/annurev-genom-083118-015406 (2019).
Pearlman, R. et al. Prevalence and spectrum of germline cancer susceptibility gene mutations among patients with early-onset colorectal cancer. JAMA Oncol. 3, 464–471. https://doi.org/10.1001/jamaoncol.2016.5194 (2017).
Whitworth, J. et al. Multilocus inherited neoplasia alleles syndrome: a case series and review. JAMA Oncol. 2, 373–379. https://doi.org/10.1001/jamaoncol.2015.4771 (2016).
Shin, Y. K. et al. Germline mutations in MLH1, MSH2 and MSH6 in Korean hereditary non-polyposis colorectal cancer families. Hum. Mutat 24, 351. https://doi.org/10.1002/humu.9277 (2004).
Desai, D. C. et al. Recurrent germline mutation in MSH2 arises frequently de novo. J. Med. Genet. 37, 646–652. https://doi.org/10.1136/jmg.37.9.646 (2000).
Woods, M. O. et al. High frequency of hereditary colorectal cancer in Newfoundland likely involves novel susceptibility genes. Clin. Cancer Res. 11, 6853–6861. https://doi.org/10.1158/1078-0432.CCR-05-0726 (2005).
Sarode, V. R. & Robinson, L. Screening for lynch syndrome by immunohistochemistry of mismatch repair proteins: significance of indeterminate result and correlation with mutational studies. Arch. Pathol. Lab Med. 143, 1225–1233. https://doi.org/10.5858/arpa.2018-0201-OA (2019).
Buckley, A. R., Ideker, T., Carter, H., Harismendy, O. & Schork, N. J. Exome-wide analysis of bi-allelic alterations identifies a Lynch phenotype in The Cancer Genome Atlas. Genome Med. 10, 69. https://doi.org/10.1186/s13073-018-0579-5 (2018).
Karczewski, K. J. et al. The mutational constraint spectrum quantified from variation in 141,456 humans. Nature 581, 434–443. https://doi.org/10.1038/s41586-020-2308-7 (2020).
Knudson, A. G. Jr. Mutation and cancer: statistical study of retinoblastoma. Proc. Nat. Acad. Sci. USA 68, 820–823. https://doi.org/10.1073/pnas.68.4.820 (1971).
Park, S., Supek, F. & Lehner, B. Systematic discovery of germline cancer predisposition genes through the identification of somatic second hits. Nat. Commun. 9, 2601. https://doi.org/10.1038/s41467-018-04900-7 (2018).
Rhees, J., Arnold, M. & Boland, C. R. Inversion of exons 1–7 of the MSH2 gene is a frequent cause of unexplained Lynch syndrome in one local population. Fam Cancer 13, 219–225. https://doi.org/10.1007/s10689-013-9688-x (2014).
Pearlman, R. et al. Clinical characteristics of patients with colorectal cancer with double somatic mismatch repair mutations compared with Lynch syndrome. J. Med. Genet. 56, 462–470. https://doi.org/10.1136/jmedgenet-2018-105698 (2019).
Win, A. K. et al. Risks of primary extracolonic cancers following colorectal cancer in lynch syndrome. J. Nat. Cancer Inst. 104, 1363–1372. https://doi.org/10.1093/jnci/djs351 (2012).
Gupta, S. et al. NCCN Guidelines insights: genetic/familial high-risk assessment: colorectal, Version 32017. J Natl Compr Canc Netw 15, 1465–1475. https://doi.org/10.6004/jnccn.2017.0176 (2017).
Hampel, H. et al. Screening for Lynch syndrome (hereditary nonpolyposis colorectal cancer) among endometrial cancer patients. Cancer Res. 66, 7810–7817. https://doi.org/10.1158/0008-5472.CAN-06-1114 (2006).
Le, D. T. et al. PD-1 blockade in tumors with mismatch-repair deficiency. N Engl. J. Med. 372, 2509–2520. https://doi.org/10.1056/NEJMoa1500596 (2015).
Latham, A. et al. Microsatellite instability is associated with the presence of lynch syndrome pan-cancer. J. Clin. Oncol. 37, 286–295. https://doi.org/10.1200/JCO.18.00283 (2019).
Galvan, A., Ioannidis, J. P. & Dragani, T. A. Beyond genome-wide association studies: genetic heterogeneity and individual predisposition to cancer. Trends Genet. 26, 132–141. https://doi.org/10.1016/j.tig.2009.12.008 (2010).
Pirinen, M., Donnelly, P. & Spencer, C. C. Including known covariates can reduce power to detect genetic effects in case-control studies. Nat. Genet. 44, 848–851. https://doi.org/10.1038/ng.2346 (2012).
Hindorff, L. A. et al. Potential etiologic and functional implications of genome-wide association loci for human diseases and traits. Proc. Natl. Acad. Sci. USA 106, 9362–9367. https://doi.org/10.1073/pnas.0903103106 (2009).
Chen, S. et al. Prediction of germline mutations and cancer risk in the Lynch syndrome. JAMA 296, 1479–1487. https://doi.org/10.1001/jama.296.12.1479 (2006).
Sun, T. et al. A six-nucleotide insertion-deletion polymorphism in the CASP8 promoter is associated with susceptibility to multiple cancers. Nat. Genet. 39, 605–613. https://doi.org/10.1038/ng2030 (2007).
Zhang, Y. et al. A systematic analysis of the association studies between CASP8 D302H polymorphisms and breast cancer risk. J. Genet. 96, 283–289. https://doi.org/10.1007/s12041-017-0774-y (2017).
Ying, Y., Xu, J., Qi, Y., Zhang, M. & Yang, Y. CASP8 rs3834129 (-652 6N insertion/deletion) polymorphism and colorectal cancer susceptibility: an updated meta-analysis. J. Cancer 9, 4166–4171. https://doi.org/10.7150/jca.27110 (2018).
Chen, H., Zhang, S. & Wu, Z. Fanconi anemia pathway defects in inherited and sporadic cancers. Transl. Pediatr. 3, 300–304. https://doi.org/10.3978/j.issn.2224-4336.2014.07.05 (2014).
Esteban-Jurado, C. et al. The Fanconi anemia DNA damage repair pathway in the spotlight for germline predisposition to colorectal cancer. Eur. J. Hum. Genet. 24, 1501–1505. https://doi.org/10.1038/ejhg.2016.44 (2016).
Huang, J. P. et al. FANCA D1359Y mutation in a patient with gastric polyposis and cancer susceptibility: A case report and review of literature. World J. Gastroenterol. 24, 4412–4418. https://doi.org/10.3748/wjg.v24.i38.4412 (2018).
Soung, Y. H. et al. CASPASE-8 gene is inactivated by somatic mutations in gastric carcinomas. Cancer Res. 65, 815–821 (2005).
Soung, Y. H. et al. Caspase-8 gene is frequently inactivated by the frameshift somatic mutation 1225_1226delTG in hepatocellular carcinomas. Oncogene 24, 141–147. https://doi.org/10.1038/sj.onc.1208244 (2005).
Ramanagoudr-Bhojappa, R. et al. Multiplexed CRISPR/Cas9-mediated knockout of 19 Fanconi anemia pathway genes in zebrafish revealed their roles in growth, sexual development and fertility. PLoS Genet. 14, e1007821. https://doi.org/10.1371/journal.pgen.1007821 (2018).
Pulliam-Leath, A. C. et al. Genetic disruption of both Fancc and Fancg in mice recapitulates the hematopoietic manifestations of Fanconi anemia. Blood 116, 2915–2920. https://doi.org/10.1182/blood-2009-08-240747 (2010).
Hashibe, M. et al. Contribution of tobacco and alcohol to the high rates of squamous cell carcinoma of the supraglottis and glottis in Central Europe. Am. J. Epidemiol. 165, 814–820. https://doi.org/10.1093/aje/kwk066 (2007).
Gandini, S. et al. Tobacco smoking and cancer: a meta-analysis. Int. J. Cancer 122, 155–164. https://doi.org/10.1002/ijc.23033 (2008).
Services, U. S. D. o. H. a. H. 14th report on carcinogens. https://ntp.niehs.nih.gov/whatwestudy/assessments/cancer/roc/index.html (2016).
Li, X. et al. MLH1 promoter methylation frequency in colorectal cancer patients and related clinicopathological and molecular features. PLoS ONE 8, e59064. https://doi.org/10.1371/journal.pone.0059064 (2013).
Umar, A. et al. Revised Bethesda Guidelines for hereditary nonpolyposis colorectal cancer (Lynch syndrome) and microsatellite instability. J. Natl. Cancer Inst. 96, 261–268. https://doi.org/10.1093/jnci/djh034 (2004).
Niu, B. et al. MSIsensor: microsatellite instability detection using paired tumor-normal sequence data. Bioinformatics 30, 1015–1016. https://doi.org/10.1093/bioinformatics/btt755 (2014).
Eoh, K. J. et al. Detection of germline mutations in patients with epithelial ovarian cancer using multi-gene panels: beyond BRCA1/2. Cancer Res. Treat. 50, 917–925. https://doi.org/10.4143/crt.2017.220 (2018).
Li, M. M. et al. Standards and guidelines for the interpretation and reporting of sequence variants in cancer: a joint consensus recommendation of the association for molecular pathology, American society of clinical oncology, and college of American pathologists. J. Mol. Diagn. 19, 4–23. https://doi.org/10.1016/j.jmoldx.2016.10.002 (2017).
BCBIo-nextgen v1.2.3. https://doi.org/10.5281/zenodo.3743344.
VanderAuwera, G. A. et al. From FastQ data to high confidence variant calls: the Genome Analysis Toolkit best practices pipeline. Curr. Protoc. Bioinf. 43, 111011–111033. https://doi.org/10.1002/0471250953.bi1110s43 (2013).
DePristo, M. A. et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat. Genet. 43, 491–498. https://doi.org/10.1038/ng.806 (2011).
Wang, K., Li, M. & Hakonarson, H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 38, e164. https://doi.org/10.1093/nar/gkq603 (2010).
Rentzsch, P., Witten, D., Cooper, G. M., Shendure, J. & Kircher, M. CADD: predicting the deleteriousness of variants throughout the human genome. Nucleic Acids Res. 47, D886–D894. https://doi.org/10.1093/nar/gky1016 (2019).
Karczewski, K. J. et al. The ExAC browser: displaying reference data information from over 60 000 exomes. Nucleic Acids Res. 45, D840–D845. https://doi.org/10.1093/nar/gkw971 (2017).
Landrum, M. J. et al. ClinVar: public archive of relationships among sequence variation and human phenotype. Nucleic Acids Res. 42, D980-985. https://doi.org/10.1093/nar/gkt1113 (2014).
McLaren, W. et al. The ensembl variant effect predictor. Genome Biol. 17, 122. https://doi.org/10.1186/s13059-016-0974-4 (2016).
MacArthur, D. G. et al. A systematic survey of loss-of-function variants in human protein-coding genes. Science 335, 823–828. https://doi.org/10.1126/science.1215040 (2012).
Talevich, E., Shain, A. H., Botton, T. & Bastian, B. C. CNVkit: genome-wide copy number detection and visualization from targeted DNA sequencing. PLoS Comput. Biol. 12, e1004873. https://doi.org/10.1371/journal.pcbi.1004873 (2016).
Vattathil, S. & Scheet, P. Haplotype-based profiling of subtle allelic imbalance with SNP arrays. Genome. Res. 23, 152–158. https://doi.org/10.1101/gr.141374.112 (2013).
Harismendy, O., Kim, J., Xu, X. & Ohno-Machado, L. Evaluating and sharing global genetic ancestry in biomedical datasets. J. Am. Med. Inf. Assoc. 26, 457–461. https://doi.org/10.1093/jamia/ocy194 (2019).
Buckley, A. R. et al. Pan-cancer analysis reveals technical artifacts in TCGA germline variant calls. BMC Genom. 18, 458. https://doi.org/10.1186/s12864-017-3770-y (2017).
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government. (2017R1D1A1B03032553) We would like to thank to Severance Research Initiative project from Yonsei University College of Medicine to support this study.
Author information
Authors and Affiliations
Contributions
Y.Y.C., J.H.C., and O.H. designed and supervised the research. X.X., D.N., Y.Y.C., J.E.L. generated sequencing data and Y.Y.C. performed the statistical analysis, prepared the figures and tables, and drafted the manuscript. Y.Y.C., J.H.C., and O.H. designed the experiments. S.S., J.E.L. conducted experiment. S.L., J.S.P., J.J., H.K., S.H.N. screened patients and collected specimen. Y.Y.C., L.M. and O.H. wrote the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Choi, Y.Y., Shin, SJ., Lee, J.E. et al. Prevalence of cancer susceptibility variants in patients with multiple Lynch syndrome related cancers. Sci Rep 11, 14807 (2021). https://doi.org/10.1038/s41598-021-94292-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-94292-4
This article is cited by
-
Clinicopathologic and genomic characteristics of mucinous gastric adenocarcinoma
Gastric Cancer (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.