Abstract
Recent evidence suggests that cellular perturbations play an important role in the pathogenesis of cardiovascular diseases. Therefore, we analyzed the association between the levels of urinary metabolites and arterial stiffness. Our cross-sectional study included 330 Korean men and women. The brachial-ankle pulse wave velocity was measured as a marker of arterial stiffness. Urinary metabolites were evaluated using a high-performance liquid chromatograph-mass spectrometer. The brachial-ankle pulse wave velocity was found to be positively correlated with l-lactate, citrate, isocitrate, succinate, malate, hydroxymethylglutarate, α-ketoisovalerate, α-keto-β-methylvalerate, methylmalonate, and formiminoglutamate among men. Whereas, among women, the brachial-ankle pulse wave velocity was positively correlated with cis-aconitate, isocitrate, hydroxymethylglutarate, and formiminoglutamate. In the multivariable regression models adjusted for conventional cardiovascular risk factors, three metabolite concentrations (urine isocitrate, hydroxymethylglutarate, and formiminoglutamate) were independently and positively associated with brachial-ankle pulse wave velocity. Increased urine isocitrate, hydroxymethylglutarate, and formiminoglutamate concentrations were associated with brachial-ankle pulse wave velocity and independent of conventional cardiovascular risk factors. Our findings suggest that metabolic disturbances in cells may be related to arterial stiffness.
Similar content being viewed by others
Introduction
Cardiovascular diseases (CVD) remain a major cause of mortality worldwide, which are mainly associated with atherosclerosis1. Recent studies revealed that arterial stiffness, as measured by pulse wave velocity (PAW), is associated with a powerful biomarker that indicates an increase in the likelihood of a future clinical event, disease recurrence, or progression of CVD2,3,4.
CVD has a complex etiology, with multiple risk factors and mechanisms contributing to its development. Hypertension, tobacco use, and high levels of cholesterol are significant risk factors for increased plaque burden and CVD5,6. In cells, various mechanisms and aberrations such as metabolic abnormalities, energy deficit, deregulation of autophagy, endoplasmic reticulum stress, and activation of apoptosis contribute to CVD pathogenesis7.
Detecting metabolites in the urine is one of the metabolic profiling technologies. The liquid chromatography-mass spectrometry (LC–MS) is used for the qualitative and quantitative determination of urine metabolites with very high sensitivity and specificity8,9. LC–MS is a combination of high-performance liquid chromatography (HPLC) and mass spectrometry (MS), which is widely used in pharmaceutical, chemical, and food applications 8,9,10,11. Many studies have reported that the levels of urine metabolites are significantly different in various diseases such as asthma11, autism spectrum disorder12, gestational diabetes mellitus13, jaundice14, prostate cancer15, purine and pyrimidine disorders16, and catecholamine-producing tumors17. One study revealed that plasma metabolites appear strongly correlated with PWV in women18. Among the urinary metabolites, the tricarboxylic acid cycle (TCA cycle) metabolites, also known as the citric acid cycle metabolites, were considered as byproducts of cellular metabolism important for the biosynthesis of nucleotides, lipids, and proteins19,20,21. Several studies revealed that urine metabolites are also associated with mitochondrial dysfunction22,23,24 and congenital mitochondrial diseases in children25.
Considering that cellular perturbations are related to CVD7,26,27,28, as a biomarker of cellular metabolism, urine metabolites may also be associated with CVD. However, the relationship of urine metabolomics analysis with clinical markers of CVD, such as brachial-ankle pulse wave velocity (baPWV), has not been comprehensively evaluated. Therefore, this study aimed to investigate the metabolomic signature according to baPWV to identify the novel metabolites associated with PWV and understand the molecular mechanisms underlying arterial stiffness.
Results
Characteristics of the study participants
The baseline characteristics of the participants are presented in Table 1. The mean age was 57.8 years. Among 330 subjects, 52.7% were men. The mean baPWV was 14.3 ± 3.0 m/s. Precisely, 63.9% of the subjects had significant physical activity and 19.7% had a metabolic syndrome.
Association of urine metabolites and baPWV
The correlation of baPWV with urine metabolites was evaluated (Table 2). Among men, the levels of l-lactate, citrate, isocitrate, succinate, malate, hydroxymethylglutarate, α-ketoisovalerate, α-keto-β-methylvalerate, methylmalonate, and formiminoglutamate were positively associated with baPWV. Among women, the levels of cis-aconitate, isocitrate, hydroxymethylglutarate, and formiminoglutamate were positively associated with baPWV.
The distribution of the levels of key metabolites with baPWV
We selected isocitrate, hydroxymethylglutarate, and formiminoglutamate, which were significantly associated with baPWV. The distribution of the levels of three urine metabolites (isocitrate, hydroxymethylglutarate, and formiminoglutamate) according to baPWV is shown in Fig. 1. Figure 1 shows the positive correlation of isocitrate, hydroxymethylglutarate and formiminoglutamate with baPWV.
The Scatter plots of the levels of key metabolites with baPWV. Each concentration of the metabolites (isocitrate (a), hydroxymethylglutarate (b) and formiminoglutamate (c)) is logarithmically transformed and standardized. Men are shown as triangles, and women are displayed as circles. Solid lines are fit for all subjects; dense dotted lines are fit for men, and sparse dotted lines are fit for women. r indicates Pearson correlation coefficients. BaPWV brachial-ankle pulse wave velocity, PWV, pulse wave velocity.
Standardized regression coefficients of the levels of key metabolites for baPWV
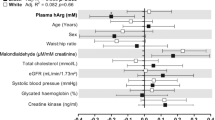
The relationship between baPWV and the levels of urine metabolites was assessed after adjusting for potential confounders (Fig. 2). In the crude model (model 1), positive relationships of baPWV with the levels of isocitrate, hydroxymethylglutarate, and formiminoglutamate were observed (P = 0.001, P < 0.001, and P < 0.001, respectively). After the adjustment for all the confounders (age, sex, mean blood pressure (BP), heart rate, total cholesterol, high-density lipoprotein (HDL) cholesterol, alcohol history, physical activity, smoking history, and medication history of hypertension, diabetes, and dyslipidemia), the positive relationship between baPWV and the three metabolites (isocitrate, hydroxymethylglutarate, and formiminoglutamate) levels was significant (P = 0.001, P = 0.029, and P < 0.001, respectively) (model 5). In the subjects excluding those with hypertension, diabetes, or statin use, and alcohol and smoking history (N = 151), the levels of isocitrate, hydroxymethylglutarate, and formiminoglutamate were significantly related to baPWV (P < 0.001, P = 0.005, and P = 0.003, respectively) (model 6).
Standardized regression coefficients of the levels of key metabolites for baPWV. The concentrations of the key metabolites (isocitrate (a), hydroxymethylglutarate (b) and formiminoglutamate (c)) are logarithmically transformed and inserted in the regression models. Model 1 constitutes crude models. Model 2 additionally includes the variables of age and sex. Model 3 additionally includes variables of mean BP and heart rate. Model 4 additionally includes variables of total cholesterol, HDL cholesterol, BMI. Model 5 additionally includes the following variables: medication history of hypertension, diabetes, and dyslipidemia, habits of smoking, significant alcohol consumption, and physical activity. In Model 6, the subjects with history of medications, smoking, and significant alcohol consumption were excluded. The covariates in Model 6 were same as those included in Model 4. Error bars show standard error of the means. BaPWV brachial-ankle pulse wave velocity, BP blood pressure, HDL high-density lipoprotein, BMI body mass index.
Discussion
In this cross-sectional study of 330 men and women, higher levels of urine isocitrate, hydroxymethylglutarate, and formiminoglutamate were significantly associated with high baPWV after adjusting for conventional cardiovascular (CV) risk factors.
Previous studies have demonstrated that cellular mechanisms, including intracellular hyperglycemia, increasing fatty acid flux and oxidation, and mitochondrial dysfunction, play a role in coronary atherogenesis and cardiomyopathy7,29,30,31.
One of the cellular mechanisms, higher fatty acid flux and oxidation, is related to various heart diseases30,31. During cardiac ischemia, elevated rates of fatty acid oxidation result in the inhibition of glucose oxidation by inhibiting the activity of pyruvate dehydrogenase leading to increased production of l-lactate31. As in previous reports, higher lactate levels were related to cardiac problems such as heart failure, endocardial damage, and cardiac ischemia 32,33. In this study, l-lactate levels were positively correlated with baPWV in men, before adjusting for confounders. However, we did not find any significant correlation of l-lactate levels with baPWV after adjusting for confounders. This discrepancy may have arisen from our small sample size and non-homogeneous study population. Further research is needed to investigate the relationship of l-lactate and arterial stiffness in a large population.
Among the cellular perturbations, mitochondrial dysfunction also plays a central role in the development of CVD7,26,29,30,34. Mitochondria contribute in oxygen sensing, cellular signaling, cell stress regulation, and energy production. The mitochondrial respiratory chain is the major pathway of energy production. Mitochondria are very sensitive to nutrient and oxygen supply and can adapt to a changing environment. When this adaptation is impaired, it leads to a progressive decline of mitochondrial function, which is associated with abnormalities in the respiratory chain and ATP synthesis, increased oxidative stress, and activation of signaling proteins35. Reactive oxygen species (ROS) overproduction then leads to the oxidation of lipids and proteins and promotes atherogenesis by inducing endothelial dysfunction, vessel inflammation, and accumulation of oxidized low-density lipoprotein (LDL)28. The diagnosis of mitochondrial dysfunction relies on a combination of clinical presentation, measurement of metabolites, and analysis of respiratory chain function22,36. Alban et al. showed the relationship between mitochondrial respiratory chain activities and urine metabolites in 75 cases24. Another previous study indicated that mitochondrial dysfunction is accompanied by the excretion of citric acid cycle intermediates22. Elevated levels of one of the citric acid cycle intermediates in urine, isocitrate, may reflect mitochondrial nicotinamide adenine dinucleotide phosphate (NADP) + -isocitrate dehydrogenase inactivity. Mitochondrial NADP+-isocitrate dehydrogenase catalyzes the oxidative decarboxylation of isocitrate, producing alpha-ketoglutarate, and regulates cardiomyocyte energy and redox status. Some studies suggest that mitochondrial NADP+-isocitrate dehydrogenase dysfunction contributes to the etiology of cardiomyopathy37,38, cancer39, and cell damage after kidney ischemia–reperfusion injury40. In our study, we demonstrated that urine isocitrate levels were significantly associated with baPWV and was still significantly correlated with baPWV after adjusting for confounders.
Regarding citric acid cycle intermediates, high levels of hydroxymethylglutarate may reflect the inadequate endogenous synthesis of CoQ1041,42. Some studies have demonstrated that CoQ10 might play a role in the prevention of heart ailments, inhibition of LDL oxidation, and progression of atherosclerosis43. Consistent with previous studies, our results reveal the relationship between baPWV and urine hydroxymethylglutarate; after the adjustment of confounders, urine hydroxylmethylglutarate levels were still significantly associated with baPWV.
Succinate is a metabolic intermediate of the TCA cycle within cells, like hydroxymethylglutarate44. In mitochondria, succinate is produced as an intermediate metabolite formed from the conversion of succinyl-CoA and is oxidized by succinate dehydrogenase (SDH) to form fumarate. Meanwhile, succinate is also produced from succinic semialdehyde via the γ-aminobutyric acid (GABA) shunt44. Generally, succinate is considered an intracellular metabolite, but succinate also has been shown to accumulate in the extracellular tissue environments related to the conditions of stress and inflammation. For instance, in microbial fermentation, succinate is also formed by the reversal of partial TCA cycle reactions45,46,47. In this study, urine hydroxymethylglutarate levels were significantly associated with baPWV, but the same was not true for urine succinate. Although hydroxymethylglutarate and succinate are in close connection in the TCA cycle, the mechanisms for succinate release are diverse and unclear, which resulted in the different results.
Formiminoglutamic acid is an intermediate metabolite in the degradative conversion of histidine to glutamic acid. An increase in urinary formiminoglutamic acid levels may be seen in patients with folic-acid deficiency, vitamin B12 deficiency, and liver disease48,49. Some studies showed that folate deficiency is associated with heart disease50 and CVD51,52. Folate supplementation delays the development of atherosclerotic lesion by modulating monocyte chemotactic protein-1 (MCP-1) and vascular endothelial growth factor (VEGF) DNA methylation levels53. In our study, urinary formiminoglutamate levels were positively correlated with higher baPWV, independent of confounders.
Our study has several limitations. First, our study was cross-sectional; thus, we could not demonstrate a causative relationship between urine metabolites and baPWV. Second, as noted earlier, we had a small sample, and our population was considerably heterogeneous; the subjects studied had various underlying diseases and a wide difference in age. Third, we lacked sufficient disease history of the subjects which might be related to arterial stiffness. Finally, we did not consider the day by day variability in urine measurement and did not reflect the long-term data. Additional long-term studies about the metabolic status can help overcome this limitation.
In conclusion, our study reveals that higher concentrations of urine isocitrate, hydroxymethylglutarate, and formiminoglutamate were positively correlated with higher baPWV, independent of conventional CV risk factors. Our findings show that the changes in these metabolites were associated with arterial stiffness. Further studies are required to reveal the causal relationship between urine metabolites and baPWV. This may serve as a new useful clinical biomarker for predicting elevated arterial stiffness and may help understand the molecular mechanisms underlying arterial stiffness.
Methods
Study population
The participants enrolled in this cross-sectional study included adults who visited the Chaum Life Center, CHA University, Seoul, Republic of Korea. Among all adults who attended the periodic medical check-up from November 2016 to December 2018, 927 participants agreed to participate in the study and had undergone urine metabolomic analyses. Subjects were required not to take supplements, including vitamin and herbs, for at least 1 week. We excluded 591 subjects who had not undergone measurements of baPWV. Subjects with renal disease, malignant disease, thyroid disease, collagen diseases, or infections, and acute disease, abnormal liver function, and a history of stroke, angina, or myocardial infarction were also excluded. Finally, 174 men and 156 women were enrolled in our study. This study was approved by the institutional review board of CHA Bundang Medical Center. The participants provided informed consent prior to enrollment. All the procedures were carried out in accordance with the relevant guidelines.
Medical history, metabolic syndrome and lifestyle habits
The medical history, medication, and lifestyle habits of the subjects were collected. The presence of metabolic syndrome (MetS) was defined by the National Cholesterol Education Program Adult Treatment Panel III criteria54. The cutoff values for central obesity were applied in accordance with a well-validated previous Korean study55. The MetS was defined by the presence of three or more of the following components: (1) waist circumference ≥ 90 cm for man and ≥ 85 cm for women, (2) systolic BP ≥ 130 mmHg or diastolic BP ≥ 85 mmHg or antihypertensive medication use, (3) HDL cholestrol < 1.04 mmol/L for men and 1.29 mmol/L for women, (4) triglyceride > 1.69 mmol/L, and (5) elevated fasting blood glucose ≥ 6.11 mmol/L or taking hypoglycemic agents.
Patients were categorized as non-smokers or current smokers based on their smoking habits. Significant alcohol consumption was defined as > 21 standard drinks/week in men and > 14 standard drinks/week in women over a 2-year period56. Significant physical activity was defined as ≥ 3 exercise/week.
Anthropometric measurements
Height and weight were measured in centimeters and kilograms, respectively, using standardized protocols while the subjects were dressed in light clothing and had their shoes off. Body mass index (BMI) was calculated from height and weight. BP was measured after resting for 10 min in a sitting position using an automatic sphygmomanometer (TM-2655P, A&D Company, Tokyo, Japan) with an appropriate cuff size. The mean BP calculated using a formula in which the diastolic BP is doubled and added to the systolic BP and the composite sum was divided by 357.
Biochemical measurements
Blood samples were collected and subsequently analyzed at a central certified laboratory at the CHA Gangnam Medical Center. Blood samples were collected from the antecubital vein early in the morning after an 8-h overnight fast. Fasting plasma glucose, high-density lipoprotein (HDL) cholesterol, and triglyceride levels were measured using a chemistry autoanalyzer (Hitachi 7600-110, Tokyo, Japan). Glomerular filtration rate (GFR) determined using the Modification of Diet in Renal Disease method58.
Measurements of baPWV
BaPWV was measured using a non-invasive vascular screening device (VP-1000 plus, Omron Healthcare, CA, USA). Subjects were examined in the supine position for at least 5 min, with elbows and ankles fastened to the blood pressure cuff. Electrocardiographic electrodes sheet and telepathy were attached on both wrists and placed at the second intercostal space at the left margin of the sternum. These values were measured after individuals had rested for at least 5 min. We adopted the average value of the left and right baPWV.
Measurements of urine metabolites
After 8-h fasting, the urine samples were collected from each subject and promptly placed in a freezer. Urine samples were analyzed at the Eone Laboratory, Inc. (Yeonsu-gu, Incheon, Republic of Korea). Fasting samples are usually used to explore how systemic metabolism differs between populations with different dietary habits59,60. The creatinine concentrations of the urine samples were determined before the analysis, and urinary concentrations of metabolites were normalized with urine creatinine to minimize the variability in urine concentrations.
The urine samples (300 μL) of the subjects were transferred into the autosampler vial. The samples were prepared according to the standardized protocol and injected into the HPLC–tandem mass spectrometry (HPLC–MS/MS) system. The HPLC–MS/MS analyses were conducted with AB Sciex Triple Quad 4500 MD (Framingham, MA, USA) with an electrospray ionization interface. The quantification was performed using an Organic Acids Urine LC–MS/MS analysis kit (ZIVAK, Kocaeli, Turkey). Injection volumes were 20.0 μL, and the flow rate throughout the analysis was 0.25 mL/min. The selected reaction monitoring transitions and the related optimized declustering potential, collision energy, and collision cell exit potential for the different analytes were determined according to the manufacturer’s manual. For all metabolites, a standard curve of known concentrations consisting of three concentrations was built. Quantification of metabolites in samples was based on these standard curves and carried out automatically with the MS controller MultiQuant MD 3.0.2.
Statistical analysis
SPSS version 25.0 (IBM, Armonk, NY, USA) was used to perform all data analyses. All continuous variables are reported as means ± standard deviations (SDs) or median (interquartile range). The categorical variables are expressed as number (percentage). We converse normalized the metabolite data, as the metabolite concentrations were not normally distributed. To identify the metabolites that associate with PWV, Spearman correlation analysis of the metabolite levels with baPWV was performed.
The metabolites that were significantly related to baPWV in both men and women were considered as key metabolites. We drew scatter plots to show the association between key metabolites and baPWV. Fit lines were also drawn in the plots.
To avoid the effects of the confounding factors, associations of metabolites with urine metabolites were tested using covariate-adjusted multivariable regression to examine the relationship between urine metabolites and baPWV with the adjustment of confounding factors. Model 1 means a crude model. First, demographic factors, age, and sex were included in model 2. Then, mean BP and heart rate were additionally included in Model 3 as strongly influencing factors for PWV. Total cholesterol, HDL cholesterol levels, and BMI, which are conventional CV risk factors, were additionally included in model 4. As factors that may potentially affect PWV, medication history of hypertension, diabetes mellitus, and dyslipidemia, smoking and alcohol history, and physical activity were additionally included in model 5. To minimize the effects of the medications for hypertension, diabetes mellitus, and dyslipidemia and smoking and alcohol history, the regression models were analyzed again in the subjects without a medication history of hypertension, diabetes mellitus, or statin use, and alcohol and smoking history (model 6). For all analyses, a P-value < 0.05 was considered statistically significant.
References
Benjamin, E. J. et al. Heart disease and stroke statistics-2019 update: A report from the American Heart Association. Circulation 139, e56–e528. https://doi.org/10.1161/CIR.0000000000000659 (2019).
Wilkinson, I. B., Maki-Petaja, K. M. & Mitchell, G. F. Uses of arterial stiffness in clinical practice. Arterioscler. Thromb. Vasc. Biol. 40, 1063–1067. https://doi.org/10.1161/ATVBAHA.120.313130 (2020).
Kim, H. L. & Kim, S. H. Pulse wave velocity in atherosclerosis. Front. Cardiovasc. Med. 6, 41. https://doi.org/10.3389/fcvm.2019.00041 (2019).
Ohkuma, T. et al. Brachial-ankle pulse wave velocity and the risk prediction of cardiovascular disease: An individual participant data meta-analysis. Hypertension 69, 1045–1052. https://doi.org/10.1161/HYPERTENSIONAHA.117.09097 (2017).
Dahlof, B. Cardiovascular disease risk factors: Epidemiology and risk assessment. Am. J. Cardiol. 105, 3A-9A. https://doi.org/10.1016/j.amjcard.2009.10.007 (2010).
Yusuf, S. et al. Modifiable risk factors, cardiovascular disease, and mortality in 155,722 individuals from 21 high-income, middle-income, and low-income countries (PURE): A prospective cohort study. Lancet 395, 795–808. https://doi.org/10.1016/S0140-6736(19)32008-2 (2020).
Chistiakov, D. A., Shkurat, T. P., Melnichenko, A. A., Grechko, A. V. & Orekhov, A. N. The role of mitochondrial dysfunction in cardiovascular disease: A brief review. Ann. Med. 50, 121–127. https://doi.org/10.1080/07853890.2017.1417631 (2018).
Rodriguez-Morato, J., Pozo, O. J. & Marcos, J. Targeting human urinary metabolome by LC-MS/MS: A review. Bioanalysis 10, 489–516. https://doi.org/10.4155/bio-2017-0285 (2018).
Grebe, S. K. & Singh, R. J. LC-MS/MS in the clinical laboratory—Where to from here? Clin. Biochem. Rev. 32, 5–31 (2011).
Pratima, N. A. & Gadikar, R. Liquid chromatography-mass spectrometry and its applications: A brief review. Arch. Org. Inorg. Chem. Sci. 1, 26–34 (2018).
Pitt, J. J. Principles and applications of liquid chromatography-mass spectrometry in clinical biochemistry. Clin. Biochem. Rev. 30, 19–34 (2009).
Chen, Q., Qiao, Y., Xu, X. J., You, X. & Tao, Y. Urine organic acids as potential biomarkers for autism-spectrum disorder in Chinese children. Front. Cell Neurosci. 13, 150. https://doi.org/10.3389/fncel.2019.00150 (2019).
Leitner, M. et al. Combined metabolomic analysis of plasma and urine reveals AHBA, tryptophan and serotonin metabolism as potential risk factors in gestational diabetes mellitus (GDM). Front. Mol. Biosci. 4, 84. https://doi.org/10.3389/fmolb.2017.00084 (2017).
Wang, X. et al. Urine metabolomics analysis for biomarker discovery and detection of jaundice syndrome in patients with liver disease. Mol. Cell Proteomics 11, 370–380. https://doi.org/10.1074/mcp.M111.016006 (2012).
Klupczynska, A. et al. A study of low-molecular-weight organic acid urinary profiles in prostate cancer by a new liquid chromatography-tandem mass spectrometry method. J. Pharm. Biomed. Anal. 159, 229–236. https://doi.org/10.1016/j.jpba.2018.06.059 (2018).
Monostori, P. et al. Extended diagnosis of purine and pyrimidine disorders from urine: LC MS/MS assay development and clinical validation. PLoS ONE 14, e0212458. https://doi.org/10.1371/journal.pone.0212458 (2019).
Eisenhofer, G., Peitzsch, M. & McWhinney, B. C. Impact of LC-MS/MS on the laboratory diagnosis of catecholamine-producing tumors. TrAC, Trends Anal. Chem. 84, 106–116 (2016).
Menni, C. et al. Metabolomic study of carotid-femoral pulse-wave velocity in women. J. Hypertens. 33, 791–796. https://doi.org/10.1097/HJH.0000000000000467 (2015).
Martinez-Reyes, I. & Chandel, N. S. Mitochondrial TCA cycle metabolites control physiology and disease. Nat. Commun. 11, 102. https://doi.org/10.1038/s41467-019-13668-3 (2020).
Hollywood, K., Brison, D. R. & Goodacre, R. Metabolomics: Current technologies and future trends. Proteomics 6, 4716–4723. https://doi.org/10.1002/pmic.200600106 (2006).
Pan, D., Lindau, C., Lagies, S., Wiedemann, N. & Kammerer, B. Metabolic profiling of isolated mitochondria and cytoplasm reveals compartment-specific metabolic responses. Metabolomics 14, 59. https://doi.org/10.1007/s11306-018-1352-x (2018).
Hancock, M. R. Mitochondrial dysfunction and the role of the non-specialist laboratory. Ann. Clin. Biochem. 39, 456–463. https://doi.org/10.1258/000456302320314467 (2002).
Sharma, K. et al. Metabolomics reveals signature of mitochondrial dysfunction in diabetic kidney disease. J. Am. Soc. Nephrol. 24, 1901–1912. https://doi.org/10.1681/ASN.2013020126 (2013).
Alban, C., Fatale, E., Joulani, A., Ilin, P. & Saada, A. The relationship between mitochondrial respiratory chain activities in muscle and metabolites in plasma and urine: A retrospective study. J. Clin. Med. https://doi.org/10.3390/jcm6030031 (2017).
Shatla, H. M. et al. Role of plasma amino acids and urinary organic acids in diagnosis of mitochondrial diseases in children. Pediatr. Neurol. 51, 820–825. https://doi.org/10.1016/j.pediatrneurol.2014.08.009 (2014).
Dominic, E. A. et al. Mitochondrial cytopathies and cardiovascular disease. Heart 100, 611–618. https://doi.org/10.1136/heartjnl-2013-304657 (2014).
Tsutsui, H., Kinugawa, S. & Matsushima, S. Mitochondrial oxidative stress and dysfunction in myocardial remodelling. Cardiovasc. Res. 81, 449–456. https://doi.org/10.1093/cvr/cvn280 (2009).
Victor, V. M., Apostolova, N., Herance, R., Hernandez-Mijares, A. & Rocha, M. Oxidative stress and mitochondrial dysfunction in atherosclerosis: Mitochondria-targeted antioxidants as potential therapy. Curr. Med. Chem. 16, 4654–4667. https://doi.org/10.2174/092986709789878265 (2009).
Shah, M. S. & Brownlee, M. Molecular and cellular mechanisms of cardiovascular disorders in diabetes. Circ. Res. 118, 1808–1829. https://doi.org/10.1161/CIRCRESAHA.116.306923 (2016).
Fillmore, N., Mori, J. & Lopaschuk, G. D. Mitochondrial fatty acid oxidation alterations in heart failure, ischaemic heart disease and diabetic cardiomyopathy. Br. J. Pharmacol. 171, 2080–2090. https://doi.org/10.1111/bph.12475 (2014).
Liu, Q., Docherty, J. C., Rendell, J. C., Clanachan, A. S. & Lopaschuk, G. D. High levels of fatty acids delay the recovery of intracellular pH and cardiac efficiency in post-ischemic hearts by inhibiting glucose oxidation. J. Am. Coll. Cardiol. 39, 718–725. https://doi.org/10.1016/s0735-1097(01)01803-4 (2002).
Matsushita, K. et al. The association of plasma lactate with incident cardiovascular outcomes: The ARIC Study. Am. J. Epidemiol. 178, 401–409. https://doi.org/10.1093/aje/kwt002 (2013).
Carter, G. & Gavin, J. B. Endocardial damage induced by lactate, lowered pH and lactic acid in non-ischemic beating hearts. Pathology 21, 125–130. https://doi.org/10.3109/00313028909059548 (1989).
Murphy, E. et al. Mitochondrial function, biology, and role in disease: A scientific statement from the American Heart Association. Circ. Res. 118, 1960–1991. https://doi.org/10.1161/RES.0000000000000104 (2016).
Jazwinski, S. M. The retrograde response: When mitochondrial quality control is not enough. Biochim. Biophys. Acta 400–409, 2013. https://doi.org/10.1016/j.bbamcr.2012.02.010 (1833).
Walker, U. A., Collins, S. & Byrne, E. Respiratory chain encephalomyopathies: A diagnostic classification. Eur. Neurol. 36, 260–267. https://doi.org/10.1159/000117269 (1996).
Benderdour, M. et al. Decreased cardiac mitochondrial NADP+-isocitrate dehydrogenase activity and expression: A marker of oxidative stress in hypertrophy development. Am. J. Physiol. Heart Circ. Physiol. 287, H2122–H2131. https://doi.org/10.1152/ajpheart.00378.2004 (2004).
Benderdour, M., Charron, G., DeBlois, D., Comte, B. & Des-Rosiers, C. Cardiac mitochondrial NADP+-isocitrate dehydrogenase is inactivated through 4-hydroxynonenal adduct formation: An event that precedes hypertrophy development. J. Biol. Chem. 278, 45154–45159. https://doi.org/10.1074/jbc.M306285200 (2003).
Reitman, Z. J. & Yan, H. Isocitrate dehydrogenase 1 and 2 mutations in cancer: Alterations at a crossroads of cellular metabolism. J. Natl. Cancer Inst. 102, 932–941. https://doi.org/10.1093/jnci/djq187 (2010).
Han, S. J. et al. Mitochondrial NADP(+)-dependent isocitrate dehydrogenase deficiency exacerbates mitochondrial and cell damage after kidney ischemia-reperfusion injury. J. Am. Soc. Nephrol. 28, 1200–1215. https://doi.org/10.1681/ASN.2016030349 (2017).
Folkers, K. et al. Lovastatin decreases coenzyme Q levels in humans. Proc. Natl. Acad. Sci. U.S.A. 87, 8931–8934. https://doi.org/10.1073/pnas.87.22.8931 (1990).
Enna, S. J. & Bylund, D. B. xPharm: The Comprehensive Pharmacology Reference 1–10 (Elsevier, 2007).
Kumar, A., Kaur, H., Devi, P. & Mohan, V. Role of coenzyme Q10 (CoQ10) in cardiac disease, hypertension and Meniere-like syndrome. Pharmacol. Ther. 124, 259–268. https://doi.org/10.1016/j.pharmthera.2009.07.003 (2009).
Connors, J., Dawe, N. & Van Limbergen, J. The role of succinate in the regulation of intestinal inflammation. Nutrients 11, 25. https://doi.org/10.3390/nu11010025 (2018).
He, W. et al. Citric acid cycle intermediates as ligands for orphan G-protein-coupled receptors. Nature 429, 188–193. https://doi.org/10.1038/nature02488 (2004).
Kushnir, M. M., Komaromy-Hiller, G., Shushan, B., Urry, F. M. & Roberts, W. L. Analysis of dicarboxylic acids by tandem mass spectrometry. High-throughput quantitative measurement of methylmalonic acid in serum, plasma, and urine. Clin. Chem. 47, 1993–2002 (2001).
Wust, J. Presumptive diagnosis of anaerobic bacteremia by gas-liquid chromatography of blood cultures. J. Clin. Microbiol. 6, 586–590 (1977).
Rose, D. P. Value of detection of formiminoglutamic acid in urine for the diagnosis of malabsorption states. Br. Med. J. 1, 1031–1034. https://doi.org/10.1136/bmj.1.5441.1031 (1965).
Luhby, A. L., Cooperman, J. M. & Teller, D. N. Urinary excretion of formiminoglutamic acid: Application in diagnosis of clinical folic acid deficiency. Am. J. Clin. Nutr. 7, 397–406. https://doi.org/10.1093/ajcn/7.4.397 (1959).
Rook, G. D., Lopez, R., Shimizu, N. & Cooperman, J. M. Folic acid deficiency in infants and children with heart disease. Br. Heart J. 35, 87–92. https://doi.org/10.1136/hrt.35.1.87 (1973).
Ward, M. Homocysteine, folate, and cardiovascular disease. Int. J. Vitam Nutr. Res. 71, 173–178. https://doi.org/10.1024/0300-9831.71.3.173 (2001).
Verhaar, M. C., Stroes, E. & Rabelink, T. J. Folates and cardiovascular disease. Arterioscler. Thromb. Vasc. Biol. 22, 6–13. https://doi.org/10.1161/hq0102.102190 (2002).
Cui, S. et al. Folic acid supplementation delays atherosclerotic lesion development by modulating MCP1 and VEGF DNA methylation levels in vivo and in vitro. Int. J. Mol. Sci. 18, 990. https://doi.org/10.3390/ijms18050990 (2017).
National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 106, 3143–3421 (2002).
Lee, S. Y. et al. Appropriate waist circumference cutoff points for central obesity in Korean adults. Diabetes Res. Clin. Pract. 75, 72–80. https://doi.org/10.1016/j.diabres.2006.04.013 (2007).
Chalasani, N. et al. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases. Hepatology 67, 328–357. https://doi.org/10.1002/hep.29367 (2018).
Pocock, G. & Richards, C. D. The Human Body: An Introduction for the Biomedical and Health Sciences (Oxford University Press, 2009).
Levey, A. S. et al. A more accurate method to estimate glomerular filtration rate from serum creatinine: A new prediction equation. Modification of diet in renal disease study group. Ann. Intern. Med. 130, 461–470. https://doi.org/10.7326/0003-4819-130-6-199903160-00002 (1999).
Smith, L. et al. Important considerations for sample collection in metabolomics studies with a special focus on applications to liver functions. Metabolites 10, 104. https://doi.org/10.3390/metabo10030104 (2020).
Ivey, K. L. et al. Identifying the metabolomic fingerprint of high and low flavonoid consumers. J. Nutr. Sci. 6, e34. https://doi.org/10.1017/jns.2017.27 (2017).
Acknowledgements
I would also like to thank Soo-Kyung Chun for her assistance in collecting the data.
Author information
Authors and Affiliations
Contributions
M.K. supervised the project. Y.K. and J.H. analyzed the data and wrote the manuscript draft. Y.K. provided constructive recommendations on this project and proofread the article. All the authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Haam, JH., Kim, YS., Cho, DY. et al. Elevated levels of urine isocitrate, hydroxymethylglutarate, and formiminoglutamate are associated with arterial stiffness in Korean adults. Sci Rep 11, 10180 (2021). https://doi.org/10.1038/s41598-021-89639-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-89639-w
This article is cited by
-
Update on the Use of Pulse Wave Velocity to Measure Age-Related Vascular Changes
Current Hypertension Reports (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.