Abstract
Inertial measurement units (IMUs) are increasingly popular and may be usable in clinical routine to assess gait. However, assessing their intra-session reliability is crucial and has not been tested with foot-worn sensors in healthy participants. The aim of this study was to assess the intra-session reliability of foot-worn IMUs for measuring gait parameters in healthy adults. Twenty healthy participants were enrolled in the study and performed the 10-m walk test in single- and dual-task ('carrying a full cup of water') conditions, three trials per condition. IMUs were used to assess spatiotemporal gait parameters, gait symmetry parameters (symmetry index (SI) and symmetry ratio (SR)), and dual task effects parameters. The relative and the absolute reliability were calculated for each gait parameter. Results showed that spatiotemporal gait parameters measured with foot-worn inertial sensors were reliable; symmetry gait parameters relative reliability was low, and SR showed better absolute reliability than SI; dual task effects were poorly reliable, and taking the mean of the second and the third trials was the most reliable. Foot-worn IMUs are reliable to assess spatiotemporal and symmetry ratio gait parameters but symmetry index and DTE gait parameters reliabilities were low and need to be interpreted with cautious by clinicians and researchers.
Similar content being viewed by others
Introduction
Locomotion is an acquired fundamental human motor activity1. Motor command allows muscle contraction control and subsequent joint movements, permitted by the integration of multiple sensory information continuously2. Optical motion analysis systems are considered as the gold-standard to assess gait parameters3. These parameters allow the understanding of the process of human locomotion3. However, their clinical use is poor and “gait testing” is still considered as “research” in the United states4. Indeed, these systems are expensive, they further require experienced individuals for data collection and analysis and are difficult to transport between care units4. On the contrary, clinicians are looking for easy to use, transportable and affordable gait analysis systems. Inertial measurement units (IMUs) containing 3-axis accelerometers fully meet these specific needs, making them increasingly popular and usable in clinical routine, to diagnose, determine severity of disease, select appropriate treatment and predict prognosis5. IMUs can be placed for instance on trunk, sternum, shank or feet6. Foot-worn IMUs allow quantification of symmetry between left and right foot7 (i.e. to assess differences in the contribution of each limb to propulsion and control during walking8,9) and permit to differentiate the phase of stance in load, foot flat and push ratio10. However, to allow in clinical practice the expanded use of these sensors, testing their reliability is crucial. When a measurement is repeated by the same participant, the reliability refers to the reproducibility of this measurement11. More-precisely, the intra-session reliability refers to the reliability of different trials performed during the same session of time11,12. The reliability should always be reported by authors to better describe the sources of imprecision in clinical trials results11.
A very recent meta-analysis questioning validity and reliability of wearable inertial sensors in healthy adults during walking3 have reported seven published studies that have used foot-worn sensors to assess spatiotemporal gait parameters10,13,14,15,16,17,18, among which two of them (28.6%) used Physilog IMUs10,17. Concurrent validity of the Physilog foot-worn inertial sensors and associated gait analysis package for the spatiotemporal gait analysis has previously been validated as compared to optical motion capture system in healthy adults17,19 but also in pathological populations (i.e., children with cerebral palsy20, patients with Parkinson disease21 or after a stroke22). However, to the best of our knowledge, no study has assessed the intra-session reliability of spatiotemporal gait parameters or the number of trial required to obtain reliable gait parameters. Since the concurrent validity between Physilog foot-worn inertial sensors and associated gait analysis package and optical motion capture systems has already been established17,19, the present study was designed to solely focus on intra-session reliability of Physilog foot-worn inertial sensors for measuring gait parameters in healthy adults. Besides, the meta-analysis of Kobsar et al. focused on the assessment of spatiotemporal gait parameters measured in single-task condition3. However, the assessment of gait in dual-task condition is recommended for ecological validity to assess safety risk and function progression23. Indeed, the ability to maintain balance while simultaneously performing a cognitive task is essential for daily living and has been implicated as a risk factor of falls in older adults24,25. Dual task is defined as “the concurrent performance of two tasks that can be performed independently and have distinct and separate goals”23. The dual task effect can further be calculated to quantify costs of multitasking23,26. Interestingly, when comparing daily-living and in-lab gait pattern in older adults, Hillel et al. in 2019 have recently reported that gait parameters “obtained during daily-living were closer in value to the dual task values measured in the laboratory setting” (27, page 1). Furthermore, gait characteristics in dual task allowed better classification of patients with and without cognitive impairment than single task28.
Besides, previous studies that have used foot-worn inertial sensors from Physilog to assess spatiotemporal gait parameters during walking at self-selected comfortable speed revealed a high heterogeneity regarding the walking protocols used (e.g. 5 m walking test17,19 versus 50 m walking test10). The 10-m walk test is a clinical test timing how long it takes a patient to walk 10 meters29. It was validated in a wide range of conditions and is generalizable to clinical practice even if clear and standardized instructions have to be given to allow accurate data during walking and appropriate clinical decisions29. A rolling start and finish was considered as easier and more convenient for patient with neurological conditions29. Besides, a self-selected or comfortable speed should be used as less random measurement errors than walking at maximum speed were found30. In its original form, the 10-m walk test was based on the measurement of the walking speed (m/s) only, calculated from the time to walk 10 m. Interestingly, the use of IMUs during the performance of this clinical test now allows to further compute relevant data such as spatiotemporal gait parameters and their symmetry.
Along these lines, the aim of the present study was to assess the intra-session reliability of foot-worn Physilog IMUs for measuring gait parameters under single-task and dual-task walking in healthy adult during the 10-m walk test. More precisely, the objectives are 1) to evaluate the reliability, standard error of measurement (SEM) and minimal detectable change (MDC) values of spatiotemporal gait parameters and symmetry gait parameters obtained, (2) to evaluate the reliability, SEM and MDC values of dual task effects obtained from gait parameters, and (3) to determine the number of trials required to ensure reliable gait assessment.
Methods
Participants
All participants signed informed consent and approved to participate in Folomi study as healthy control participants31. The study was approved by local ethic committee (CPP Ile De France 1, RCB: 2017-A03468-45, date of agreement: July 17th, 2018, Last version: V6.0, June 17th, 2020), is registered in Clinical trials (NCT03761212) and was performed in accordance with relevant guidelines and regulations.
In line with previous studies on reliability on healthy10,17,19 and pathological gait20,22,32,33, twenty healthy participants (10 first men and 10 first women of healthy participants of the Folomi study34) were included in the present study.
Participants should be aged between 18 to 65 years, able to walk 180 m without technical help, with a public health insurance (French social security). The non-inclusion criteria were: (1) Musculo-skeletal, cardio-respiratory or neurologic disease that could affect gait, (2) Hip or knee arthroplasty done, (3) Not able to speak French, (4) Desire of pregnancy in the following 18 months and (5) Adults protected by laws (Article L1121-5)31. Demographic and anthropometric characteristics of study participants (age, height, leg height, body mass, body mass index) were obtained from the participants. Leg length (in meters) was measured from the anterior superior iliac spine to the medial malleolus35.
Experimental protocol
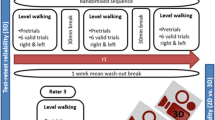
A 10-m Walk test (10MWT) was performed on a 14 m walkway for a rolling start (2 m) and finish (2 m) of the 10 m walk29, at comfortable walking speed36. This walking task was performed in single- and dual-task conditions (3 trials per condition)31.
In the dual-task condition, participants had to carry a full cup of water in their dominant hand with the following instruction: “perform both tasks as well as possible”26,37. The examiner noted whether there was any spillage of water26. This manual task has been shown to significantly alter spatiotemporal gait parameters during walking in healthy adults with significant reduction of gait speed, stride length and cadence38.
Gait assessment
Two wireless inertial sensor systems (Physilog5, 200 Hz, BioAGM, Gait Up, CH), fixed on the dorsal part of the participant’s feet using Velcro straps (behind the base of the fifth metatarsal22), were used to collect gait data (Supplementary Fig. S1). Gait assessments were performed by the same examiner.
Spatiotemporal gait parameters (Table 1) were calculated with the Gait Analysis Software provided by Gait Up (CH)39. The two first and last steps were removed from the analysis40,41 and at least 16 steps were included in the analysis. The mean of left and right gait parameters were calculated and used for calculation of mean gait parameters:
-
Speed (m s−1): Mean walking stride velocity of forward walking
-
Cadence (step/minute): Number of steps in a minute
-
Stride length (m): Distance between two consecutive footprints on the ground, from the heel of a foot to the heel of the same foot, one cycle after
-
Swing (%): Portion of the cycle during which the foot is in the air and does not touch the ground
-
Stance (%): Portion of the cycle during which part of the foot touches the ground
-
Double support (%): Portion of the cycle where both feet touch the ground
-
Load (%): Portion of the stance between the heel strike and the foot being flat on the ground
-
Foot Flat (%): Portion of the stance where the foot is fully flat on the ground
-
Push (%): Portion of the stance between the foot being flat on the ground and the toe leaving the ground at take off
Speed, cadence and stride length were normalized to have non dimensional values using the formula described by Hof42 and Pinzone et al.35 :
\(Normalized speed \left( {speed N} \right) = \frac{Speed}{{\sqrt {gl_{0} } }}\) with g the acceleration of gravity (= 9.81 m/s2) and l0 leg length; \(Normalized cadence \left( {cadence N} \right) = Cadence*\sqrt {\frac{{l_{0} }}{g}}\) with g the acceleration of gravity and l0 leg length; \(Normalized stride length \left( {stride length N} \right) = \frac{stride length}{{l_{0} }}\) with l0 leg length.
Gait symmetry measures
To assess the contribution of each limb to propulsion and control during walking8,9, symmetry gait parameters can be used9. Among these parameters, the symmetry index (SI)9 and the symmetry ratio (SR)43 are the more commonly used. The SI, which is the most commonly used and cited in published studies on gait symmetry9,43,44, was calculated. SI was calculated with the following formula for each spatiotemporal gait parameter (except double support)9:
with XL the value of each parameter of left foot and XR the value of each parameter of right foot.
A value of SI of 0 indicates full symmetry, while SI ≥ 100% indicates its asymmetry.
The symmetry ratio (SR), described as easier to interpret, is recommended on the basis of potential clinical utility in patients after stroke which often display asymmetry between left and right leg43. SR was calculated with the following formula for each spatiotemporal gait parameter (except double support)43:
with XL the value of each parameter of left foot and XR the value of each parameter of right foot.
A SR of 1 indicates full symmetry, while a SR of 2.0 on gait speed indicates that the right foot is twice faster than left foot.
Dual task effects parameters
Dual task effects (DTE) were calculated to assess the influence of the addition of a secondary attention demanding task23,26. DTE can be calculated as a relative measure of change (DTE) or in percentage (DTE%)26. DTE, as the relative measure of change in performance 45, was calculated from mean parameters as26:
\(DTE = {\text{single task performance}} - {\text{dual task performance }}\).
DTE has the same unit as the gait parameter that is used in the calculation. A negative value of DTE for gait speed, cadence, stride length, swing and foot flat is associated to better performance in dual task; while a negative value for stance, double support, load and push is associated to worse performance in dual task26. DTE can also be calculated as a percentage (DTE%), being unit-less, and permitting comparison between parameters26:
\(DTE \% = { }\frac{{{\text{single task performance}} - {\text{dual task performance}}}}{{\text{single task performance}}}X100\).
Same interpretation as DTE can be done regarding positive and negative values.
Statistical analysis
Spatiotemporal gait parameters, gait symmetry measures, and dual task effects, were normally distributed (Shapiro–Wilk normality test). A repeated measures analysis of variance (RM-ANOVA) with the number of trials used as within subject factor was performed on gait parameters, ICC and SEM values46. When significant effect of the number of trials was found, a Tukey post-hoc test was used to compare differences between trials46.
Intraclass correlation coefficients (ICC) were used to compute the relative reliability, and standard error of measurement (SEM) and minimal detectable change (MDC) were used to compute the absolute reliability across the trials 1–2–3 in each condition (single- and dual-task).
The relative reliability was evaluated by the calculation of a 2-way fixed ICC2,1 (for absolute agreement)47. ICC values inferior to 0 were considered as poor reliability, between 0.01 to 0.2 as slight, between 0.21–0.4 as fair, between 0.41–0.6 as moderate, 0.61–0.8 as substantial and 0.8–1.00 as almost perfect48. SEM, expressed in the same unit as gait parameters, corresponds to the absolute measure of the variability of the errors of measurements. SEM informs on the precision of gait parameters of individual examinees49. SEM was calculated with the following formula50:
with SD the standard deviation of the parameters from all patients and ICC the relative reliability.
MDC is the minimum value for which a difference can be considered as “real”, and was generated with the formula15:
with SEM the Standard Error of Measurement.
SEM% and MDC% were also calculated as a percentage of mean for each parameter. SEM% values were classified as ‘low’ (SEM% ≤ 10%) or ‘high’ (SEM% > 10%)51. MDC% values were classified as ‘low’ (MDC% ≤ 20%), ‘acceptable’ (20% < MDC% < 40%) and ‘high’ (MDC% ≥ 40%)51 with a MDC% < 10% considered as excellent and a MDC% < 40% as acceptable52. These two classifications were based on previous publications32,51,53 as no clear criteria for interpretation of SEM% and MDC% are available.
Limits of agreements (LOA) and Bland and Altman plots of the differences between trials against their mean, were used to assess the magnitude of disagreement between trials. A difference between trials outside the LOA can be considered as real change (Fig. 2 and Supplementary Fig. S2)53.
Ethics approval and consent to participate
The study was approved by local ethic committee (CPP Ile De France 1, RCB: 2017-A03468-45, date of agreement: July 17th, 2018, Last version: V6.0, June 17th, 2020). The study is registered on ClinicalTrials.gov, with the following ID: NCT03761212 and follow the SPIRIT checklist. Written informed consent were obtained from all participants by a physiotherapist or a doctor. Any modification to the initial protocol will be presented to the local ethic committee and has to be accepted before application and registered in ClinicalTrials.gov.
Results
Healthy adults included in the present study were 44.9 (11.7) years old. They were 174.7 (8.1) meters height and 69.3 (11.6) kg weight.
Intra-session relative and absolute reliability of gait parameters
Table 1 reports means and standard deviations of gait parameters obtained for each trial and for the three trials, in the single-task and the dual-task condition, and for dual task effects (DTE) and DTE% parameters. p values obtained from RM-ANOVA for differences between trials are also reported. Results showed that no significant difference was found between trials in all gait parameters in single- and dual-task conditions and for DTE and DTE% parameters (Table 1).
Tables 2 and 3 report relative reliability with intraclass correlation coefficients (ICC) and limits of agreement (LOA) values for spatiotemporal gait parameters obtained in the single-task and the dual-task condition, respectively.
On the one hand, results showed that almost all ICC of spatiotemporal gait parameters were above 0.81 and considered as almost perfect relative reliability in single- and dual-task conditions49, except for cadence, normalized cadence, swing, and stance with ICC above 0.61 and considered as substantial reliability. On the other hand, symmetry index (SI) and symmetry ratio (SR) gait parameters showed lower ICC values with ICC ranging from 0 to 0.25 for speed and cadence, and from 0.15 to 0.88 for slength, swing, stance, LDr, FFr and PUr.
Figure 1 present relative reliability with ICC values for spatiotemporal and symmetry gait parameters in the single-task and the dual-task conditions.
Intraclass correlation coefficients calculated with the means of trial 1–2, 1–3, 2–3 and 1–2-3 in single-task (a) and dual-task (b) conditions; Dotted color lines correspond to relative reliability thresholds with: slight (black: 0.00 < ICC < 0.2), fair (red: 0.21 < ICC < 0.4), moderate (orange: 0.41 < ICC < 0.6), substantial (yellow: 0.61 < ICC < 0.8), and almost perfect (green: 0.81 < ICC < 1.0) reliability 49. Abbreviations: LDr = Load Ratio, FFr = Foot Flat ratio, PUr = Push ratio, nprm = normalized, SI = symmetry index, SR = symmetry ratio, slength = stride length
Table 4 (single task condition) and Table 5 (dual task condition) report absolute reliability with standard error of measurement (SEM) and minimal detectable change (MDC) in the same unit as the parameter and in percentage, calculated between each trial (T1-T2, T1-T3, T2-T3 and T1-T2-T3). The results showed that SEM percentages were comprised between 1.05 and 6.58% in the single-task condition for spatiotemporal and normalized spatiotemporal gait parameters. For symmetry ratio, SEM percentage ranged from 1.66 to 27.22%, while symmetry index ranged from 42.76 to 193.73%. In the dual-task condition, comparable values for SEM and MDC were found.
The comparisons of means of ICC between trials showed that slightly higher ICC and lower SEM and MDC were obtained when pooling gait parameters from the second and the third trials, compared to the first and second trials, to the first and third trials, or to the three trials in the single-task and the dual-task conditions.
Bland and Altman plots for speed, cadence, and stride length in single task and dual task conditions can be found in Fig. 2.
Intra-session relative and absolute reliability of dual-task effects on gait parameters
Table 6 presents relative reliability with ICC and LOA values on gait parameters for DTE and DTE%. Table 7 shows absolute reliability of DTE with SEM and minimal detectable change (MDC) calculated between each trial (T1-T2, T1-T3, T2-T3 and T1-T2-T3) for DTE and DTE% respectively. The results showed that DTE (0.00 < ICC < 0.82, 61.05 < SEM% < 3666.49) and DTE% (0.00 < ICC < 0.82, 68.03 < SEM% < 14,994.32) reliabilities were low. Relative reliability of DTE % expressed with ICC was comprised between 0.61 and 1.00 for speed, stride length and double support, between 0.21 to 0.41 for cadence, load ratio and foot flat ratio (0.21 < ICC < 0.41), and between 0.00 and 0.21 for swing, stance and push ratio.
Bland and Altman plots for speed, cadence, and stride length DTE and DTE% can be found in Supplementary Fig. S2.
Discussion
The aim of the present study was to assess the reliability of Physilog5 foot-worn IMUs to assess gait in healthy adults during the 10-m walk test in single and dual task conditions. The objectives were 1) to evaluate the reliability, standard error of measurement (SEM) and minimal detectable change (MDC) values of spatiotemporal gait parameters and symmetry gait parameters obtained, (2) to evaluate the reliability, SEM and MDC values of dual task effects obtained from gait parameters, and (3) to determine the number of trials required to ensure reliable gait assessment.
Relative reliability (ICC) was considered as substantial to perfect for spatiotemporal gait parameters in both single- and dual-task conditions. Speed and stride length were the most reliable parameters in line with previous studies on healthy participants3. In a study using also Physilog IMUs with patients after stroke, comparable results on relative reliability (0.639 < ICC0.999) were found22. Absolute reliability (SEM and MDC) is not always reported in reliability studies3 and SEM was reported by only one study using Physilog sensors in healthy participants for local dynamic stability parameter54. Absolute reliability on stroke patients was lower (i.e. higher SEM% = 3.4 < SEM% < 14.8) but could be explained by the typical pathologic gait associated with stroke and hemiparesis that could lead to more variability (illustrated with higher SEM on paretic leg)22. In a study on healthy adults (n = 39, mean age: 23.8 ± 6.2) assessing gait with foot-worn IMUs15, MDC in single task was comparable to the results of the present study with 0.12 m.s−1 for gait speed (between 0.13 and 0.16 m.s−1 in the present study), 0.11 for stride length (between 0.07 and 0.09 m in the present study). However, lower MDC were found for cadence (2.7215 vs between 7.58 and 9.52 steps/minute in the present study), and for stance and swing percentage (1.4915 vs between 1.75 and 3.29% in the present study). These differences could be explained by the algorithm used to assess gait parameters as one used Mobility Lab (APDM, Inc., Portland, OR) while the present study used Gait Analysis Software (V5.3.0) from Gait Up (CH). MDC is used to evaluate the difference to be considered “real” than measurement error. It is important to note that MDC values reported in the present study can be used for an individual subject to assess change in performance before and after intervention15. However, MDC used to determine meaningful improvement in a group of participants has to take into account the size of the group (n) and is calculated as: \(MDC_{{group}} = MDC_{{individual}} \div \sqrt n\).
Foot-worn IMUs (Physilog5, BioAGM, Gait Up, CH) were thus reliable to assess spatiotemporal gait parameters during walking at comfortable speed, regardless of the single or dual-task condition in healthy adults. This was not the case for symmetry gait parameters which showed less reliability than spatiotemporal gait parameters. We believe that this result is of importance considering the popularity and the relevance of symmetry gait parameters in clinical setting55. Indeed, while under healthy conditions and unconstrained walking conditions, gait pattern is generally symmetric (i.e. the left and the right lower limbs behave similarly)56, increased levels of gait asymmetry are typically observed in a variety of clinical populations such as elderly fallers57, individuals with lower-limb amputations58, knee osteoarthritis59, hip arthroplasties60, or after knee anterior cruciate ligament reconstruction61, people with multiple sclerosis62, persons with diabetes and peripheral neuropathy63, patients with Lewy body disease64, with Parkinson disease65,66, or after a stroke44,67. For instance, Fling et al. 2018 recently reported that people with Parkinson disease exhibited significant increased spatial (e.g., step length) and temporal (e.g., step time) asymmetries of the lower extremities during gait than age-matched healthy control participants66. Recent results of Wei et al. (2017) further revealed that gait asymmetry is an important factor for the prediction of falls in stroke patients68. Also of note is that chronic gait asymmetry could also lead to increased risk of lower-limb overuse injuries and articular joint degeneration due to the increased weight bearing and propulsion demands placed on one lower limb during walking, in both pathological69,70,71,72 and healthy populations73. Along these lines, gait asymmetry can be used as (i) a metric to assess pathology status, patients’ functional recovery or disease progression, (ii) a target for interventions aiming to improve gait performance, and (iii) outcome to evaluate and compare different gait rehabilitation programs interventions43,55,74,75,76,77. Interestingly, various rehabilitation programs have been shown to improve gait to a more symmetric gait pattern in individuals with lower-limb amputations (see for review78), stroke survivors (see for review79) or patients with Parkinson disease80,81.
In the present study, two formulae widely employed in clinical practice, namely symmetry ratio and symmetry index, were used to quantify gait symmetry9,43,82. Relative and absolute reliabilities of symmetry index of spatiotemporal gait parameters were low, as indicated by low values of ICC, high values of SEM% and high values of MDC%. Accordingly, following existing recommendations22,51,52, we would not recommend using SI of spatiotemporal gait parameters for the experimental conditions used in the present study at least. Interestingly, the symmetry ratio (SR) of spatiotemporal gait parameters showed better relative reliability that SI, as indicated by higher values of ICC (except for speed and stride length) and better absolute reliability, as indicated by acceptable SEM% and MDC% (SEM% and MDC% < 20%). This result is of particular interest since SR is easier to interpret by clinicians than SI43. Indeed, a SR of 2 indicates that the right foot was twice faster than left foot43, while SI gives a percentage of asymmetry that is more difficult to interpret9. All in all, SR of spatiotemporal gait parameters should be preferred to SI by clinicians and researchers to assess gait symmetry.
Dual task effects are often used to assess the cost of performing another task while walking and permits comparisons between studies with various secondary task26,45. However, the reliability of DTE is not commonly assessed in gait reliability studies but has to be tested to allow the use of this indicator in gait studies24. In our study, DTE% showed slightly better reliability with higher ICC for almost all gait parameters. Relative reliability of DTE % was considered as substantial to almost perfect for speed, stride length and double support (0.61 < ICC < 1.00), fair to moderate for cadence, load ratio and foot flat ratio (0.21 < ICC < 0.41), and slight to fair for swing, stance and push ratio (0.00 < ICC < 0.21). SEM percentage were high for all DTE parameters (68.03 < SEM% < 14,994.32%). These results are in line with previous studies reporting DTE reliability (0.19 < ICC < 0.5524; 0.002 < ICC < 0.88283) in participants with (n = 23, mean age: 80.6 ± 6.4) and without (n = 27, mean age: 76.1 ± 5.6) cognitive impairment disorders using a counting secondary task. One explanation of lower reliability of DTE is that measurement errors of each single task and dual task conditions tends to inflation of the overall measurement error when combined to obtain the DTE83. Besides, we asked participants not to prioritize one task26,37 and some may have chosen a posture-first strategy84, while others may have chosen a posture-second strategy85, leading to variability of gait performance that could have been amplified with DTE calculation24. Besides, the reliability of gait performance in dual task conditions can be dependent of the complexity of the dual-task used as a concurrent task to gait86,87, of the level of cognitive state of participants88 or of the educational level for verbal fluency tasks89. Thus, researchers or clinicians should be aware of the poor reliability of DTE. Even if DTE% of spatiotemporal gait parameters showed slightly better reliability than DTE of spatiotemporal gait parameters, their relative and absolute reliabilities were low (low ICC, and high SEM% and MDC%). Following existing recommendations22,51,52, we would not recommend using DTE and DTE% of spatiotemporal gait parameters for the experimental conditions used in the present study at least.
In the present study, even if the reliability of spatiotemporal gait parameters was substantial to perfect in both the single-task and the dual-task conditions, we observed slightly better ICC (i.e. higher), SEM and MDC (i.e. lower) when the reliability was calculate from the second and the third trials for most of gait parameters. While numerous studies calculated gait parameters using the average of three trials in older adults37,90 or pathological populations91, the number of trials to ensure reliable measurements using intra-session reliability has been less investigated92,93. Our results showed that the first trial appears less reliable and are in line with those reported in previous studies on healthy93 and pathological populations92. Thus, the mean of at least two trials should be used for better reliability of spatiotemporal gait parameters and the first trial could be used, like other studies, as a warming trial94.
A first limitation of the study is the lack of formal cognitive assessment of the participants considering the presumable impact of individual cognitive function on gait performance8895,96,97,98,99. Growing evidence suggests that the reliance on cognitive control processes during walking can be increased when performing a more complex locomotor task96, or when concurrently executing a dual-task (see for reviews88,98,99). Furthermore, a recent systematic review concluded that individuals with cognitive impairment can demonstrate decrease of gait performance98. What is more, previous studies have reported that motor slowing preceded cognitive decline in healthy older adults, suggesting that a decrease of gait performance could be a predictor of cognitive decline (see for recent reviews100,101). Another limitation of the present study is that reliability was assessed during a single experimental session, which by definition did not permit the calculation of the inter-session reliability. Inter-session reliability refers to the extent of agreement between measurements of a measure in sessions performed at different moments (usually at 1 h or 1 week interval)102. It is used to describe error magnitude between two sessions of time to further study disease evolution or the impact of treatments102,103. Furthermore, reliability was tested in healthy middle-aged adults. To allow the use of these sensors in clinical practice, the reliability has to be further tested in elderly and pathological populations.
To conclude, foot-worn IMUs are reliable to assess spatiotemporal and symmetry ratio gait parameters in healthy participants in both single- and dual-task conditions. Reliability of symmetry index and DTE gait parameters was low and we do not recommend the use of these parameters for the experimental conditions used in the present study. Future studies should examine the number of strides necessary to ensure better reliability of these index to allow their use in clinical practice and research104. Future studies should also focus on the assessment of reliability of 10-m walk test in pathological populations and should open up perspectives with other clinical tests (e.g. 6MWT or Timed Up and Go test) that could capture different features of gait and mobility in daily life.
Data availability
The data of the present manuscript can be available on demand to the corresponding author.
References
Bouisset, S. & Maton, B. Muscles, Posture et Mouvement: Bases et Applications de la Méthode Électromyographique (Hermann, Paris, 1996).
Abu-Faraj, Z., Harris, G., Smith, P. & Hassani, S. Human Gait and Clinical Movement Analysis. in 1–34 (2015). https://doi.org/10.1002/047134608X.W6606.pub2.
Kobsar, D. et al. Validity and reliability of wearable inertial sensors in healthy adult walking: A systematic review and meta-analysis. J. Neuroeng. Rehabil. 17, 62 (2020).
Simon, S. R. Quantification of human motion: Gait analysis-benefits and limitations to its application to clinical problems. J. Biomech. 37, 1869–1880 (2004).
Baker, R. Gait analysis methods in rehabilitation. J. NeuroEngin. Rehabil. 3, 4 (2006).
Iosa, M., Picerno, P., Paolucci, S. & Morone, G. Wearable inertial sensors for human movement analysis. Expert Rev. Med. Devices 13, 641–659 (2016).
Sabatini, A. M., Martelloni, C., Scapellato, S. & Cavallo, F. Assessment of walking features from foot inertial sensing. IEEE Trans. Biomed. Eng. 52, 486–494 (2005).
Sadeghi, H. Local or global asymmetry in gait of people without impairments. Gait Posture 17, 197–204 (2003).
Błażkiewicz, M., Wiszomirska, I. & Wit, A. Comparison of four methods of calculating the symmetry of spatial-temporal parameters of gait. Acta Bioeng. Biomech. 16, 29–35 (2014).
Mariani, B., Rouhani, H., Crevoisier, X. & Aminian, K. Quantitative estimation of foot-flat and stance phase of gait using foot-worn inertial sensors. Gait Posture 37, 229–234 (2013).
Lachin, J. M. The role of measurement reliability in clinical trials. Clin. Trials Lond. Engl. 1, 553–566 (2004).
Wedege, P., Steffen, K., Strøm, V. & Opheim, A. I. Reliability of three-dimensional kinematic gait data in adults with spinal cord injury. J. Rehabil. Assist. Technol. Eng. 4, 2055668317729992 (2017).
Hamacher, D., Hamacher, D., Taylor, W. R., Singh, N. B. & Schega, L. Towards clinical application: Repetitive sensor position re-calibration for improved reliability of gait parameters. Gait Posture 39, 1146–1148 (2014).
Sijobert, B. et al. Implementation and validation of a stride length estimation algorithm, using a single basic inertial sensor on healthy subjects and patients suffering from Parkinson’s disease. Health (N. Y.) 7, 704–714 (2015).
Washabaugh, E. P., Kalyanaraman, T., Adamczyk, P. G., Claflin, E. S. & Krishnan, C. Validity and repeatability of inertial measurement units for measuring gait parameters. Gait Posture 55, 87–93 (2017).
Kitagawa, N. & Ogihara, N. Estimation of foot trajectory during human walking by a wearable inertial measurement unit mounted to the foot. Gait Posture 45, 110–114 (2016).
Mariani, B., Rochat, S., Büla, C. J. & Aminian, K. Heel and toe clearance estimation for gait analysis using wireless inertial sensors. IEEE Trans. Biomed. Eng. 59, 3162–3168 (2012).
Benoussaad, M., Sijobert, B., Mombaur, K. & Azevedo Coste, C. Robust foot clearance estimation based on the integration of foot-mounted IMU acceleration data. Sensors 16, 12 (2015).
Mariani, B. et al. 3D gait assessment in young and elderly subjects using foot-worn inertial sensors. J. Biomech. 43, 2999–3006 (2010).
Brégou Bourgeois, A., Mariani, B., Aminian, K., Zambelli, P. Y. & Newman, C. J. Spatio-temporal gait analysis in children with cerebral palsy using, foot-worn inertial sensors. Gait Posture 39, 436–442 (2014).
Mariani, B., Jiménez, M. C., Vingerhoets, F. J. G. & Aminian, K. On-shoe wearable sensors for gait and turning assessment of patients with Parkinson’s disease. IEEE Trans. Biomed. Eng. 60, 155–158 (2013).
Lefeber, N., Degelaen, M., Truyers, C., Safin, I. & Beckwee, D. Validity and reproducibility of inertial physilog sensors for spatiotemporal gait analysis in patients with stroke. IEEE Trans. Neural Syst. Rehabil. Eng. Publ. IEEE Eng. Med. Biol. Soc. 27, 1865–1874 (2019).
McIsaac, T. L., Lamberg, E. M. & Muratori, L. M. Building a Framework for a Dual Task Taxonomy. BioMed Research International https://www.hindawi.com/journals/bmri/2015/591475/ (2015).
Yang, L., Liao, L. R., Lam, F. M. H., He, C. Q. & Pang, M. Y. C. Psychometric properties of dual-task balance assessments for older adults: A systematic review. Maturitas 80, 359–369 (2015).
Beauchet, O. et al. Stops walking when talking: A predictor of falls in older adults?. Eur. J. Neurol. 16, 786–795 (2009).
Yang, L., He, C. & Pang, M. Y. C. Reliability and validity of dual-task mobility assessments in people with chronic stroke. PLoS ONE 11(1), e0147833 (2016).
Hillel, I. et al. Is every-day walking in older adults more analogous to dual-task walking or to usual walking? Elucidating the gaps between gait performance in the lab and during 24/7 monitoring. Eur. Rev. Aging Phys. Act. 16, 6 (2019).
Kikkert, L. H. J. et al. Gait characteristics and their discriminative power in geriatric patients with and without cognitive impairment. J. Neuroeng. Rehabil. 14, 84 (2017).
Tyson, S. & Connell, L. The psychometric properties and clinical utility of measures of walking and mobility in neurological conditions: A systematic review. Clin. Rehabil. 23, 1018–1033 (2009).
Stephens, J. M. & Goldie, P. A. Walking speed on parquetry and carpet after stroke: Effect of surface and retest reliability. Clin. Rehabil. 13, 171–181 (1999).
Soulard, J. et al. Gait as predictor of physical function in axial spondyloarthritis: The prospective longitudinal FOLOMI (Function, Locomotion, Measurement, Inflammation) study protocol. Rheumatol. Int. https://doi.org/10.1007/s00296-019-04396-4 (2019).
Wüest, S., Massé, F., Aminian, K., Gonzenbach, R. & de Bruin, E. D. Reliability and validity of the inertial sensor-based Timed ‘Up and Go’ test in individuals affected by stroke. J. Rehabil. Res. Dev. 53, 599–610 (2016).
Gerber, C. N., Carcreff, L., Paraschiv-Ionescu, A., Armand, S. & Newman, C. J. Reliability of single-day walking performance and physical activity measures using inertial sensors in children with cerebral palsy. Ann. Phys. Rehabil. Med. https://doi.org/10.1016/j.rehab.2019.02.003 (2019).
Rudwaleit, M. et al. Defining active sacroiliitis on magnetic resonance imaging (MRI) for classification of axial spondyloarthritis: A consensual approach by the ASAS/OMERACT MRI group. Ann. Rheum. Dis. 68, 1520–1527 (2009).
Pinzone, O., Schwartz, M. H. & Baker, R. Comprehensive non-dimensional normalization of gait data. Gait Posture 44, 68–73 (2016).
Graham, J. E., Ostir, G. V., Fisher, S. R. & Ottenbacher, K. J. Assessing walking speed in clinical research: A systematic review. J. Eval. Clin. Pract. 14, 552–562 (2008).
Beauchet, O. et al. Guidelines for assessment of gait and reference values for spatiotemporal gait parameters in older adults: The biomathics and Canadian gait consortiums initiative. Front. Hum. Neurosci. 11, 353 (2017).
Kwon, Y., Kwon, J. W. & Cho, I. H. The difference of gait characteristic according to the variety of dual tasks in young healthy adults. Work Read. Mass 63, 33–38 (2019).
Dadashi, F. et al. Gait and foot clearance parameters obtained using shoe-worn inertial sensors in a large-population sample of older adults. Sensors 14, 443–457 (2014).
Truong, P. H., Lee, J., Kwon, A.-R. & Jeong, G.-M. Stride counting in human walking and walking distance estimation using insole sensors. Sensors 16, 823 (2016).
Anwary, A. R., Yu, H. & Vassallo, M. An automatic gait feature extraction method for identifying gait asymmetry using wearable sensors. Sensors 18, 676 (2018).
Hof, A. Scaling gait data to body size. Gait Posture 4, 222–223 (1996).
Patterson, K. K., Gage, W. H., Brooks, D., Black, S. E. & McIlroy, W. E. Evaluation of gait symmetry after stroke: A comparison of current methods and recommendations for standardization. Gait Posture 31, 241–246 (2010).
Lauziere, S., Betschart, M., Aissaoui, R. & Nadeau, S. Understanding Spatial and Temporal Gait Asymmetries in Individuals Post Stroke. Int. J. Phys. Med. Rehabil. 2, 1–11 (2014).
Kelly, V. E., Janke, A. A. & Shumway-Cook, A. Effects of instructed focus and task difficulty on concurrent walking and cognitive task performance in healthy young adults. Exp. Brain Res. Exp. Hirnforsch. Exp. Cerebrale 207, 65–67 (2010).
Lee, S. & Lee, D. K. What is the proper way to apply the multiple comparison test?. Korean J. Anesthesiol. 71, 353–360 (2018).
Koo, T. K. & Li, M. Y. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J. Chiropr. Med. 15, 155–163 (2016).
Landis, J. R. & Koch, G. G. The measurement of observer agreement for categorical data. Biometrics 33, 159–174 (1977).
Harvill, L. M. Standard error of measurement. Educ. Meas. Issues Pract. 10, 33–41 (1991).
Walton, D. M. et al. Reliability, standard error, and minimum detectable change of clinical pressure pain threshold testing in people with and without acute neck pain. J. Orthop. Sports Phys. Ther. 41, 644–650 (2011).
Bourke, A. K., Scotland, A., Lipsmeier, F., Gossens, C. & Lindemann, M. Gait characteristics harvested during a smartphone-based self-administered 2-minute walk test in people with multiple sclerosis: Test–retest reliability and minimum detectable change. Sensors 20, 5906 (2020).
Huang, S.-L., Hsieh, C.-L., Wu, R.-M. & Lu, W.-S. Test–retest reliability and minimal detectable change of the beck depression inventory and the Taiwan geriatric depression scale in patients with Parkinson’s disease. PLoS ONE 12(9), e0184823 (2017).
Bland, J. M. & Altman, D. G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet Lond. Engl. 1, 307–310 (1986).
Reynard, F. & Terrier, P. Local dynamic stability of treadmill walking: Intrasession and week-to-week repeatability. J. Biomech. 47, 74–80 (2014).
Cabral, S. Gait Symmetry Measures and Their Relevance to Gait Retraining. In Handbook of Human Motion 429–447 (Springer International Publishing, 2018). https://doi.org/10.1007/978-3-319-14418-4_201.
Kodesh, E., Kafri, M., Dar, G. & Dickstein, R. Walking speed, unilateral leg loading, and step symmetry in young adults. Gait Posture 35, 66–69 (2012).
Yogev, G., Plotnik, M., Peretz, C., Giladi, N. & Hausdorff, J. M. Gait asymmetry in patients with Parkinson’s disease and elderly fallers: When does the bilateral coordination of gait require attention?. Exp. Brain Res. 177, 336–346 (2007).
Mattes, S. J., Martin, P. E. & Royer, T. D. Walking symmetry and energy cost in persons with unilateral transtibial amputations: Matching prosthetic and intact limb inertial properties. Arch. Phys. Med. Rehabil. 81, 561–568 (2000).
Mills, K., Hettinga, B. A., Pohl, M. B. & Ferber, R. Between-limb kinematic asymmetry during gait in unilateral and bilateral mild to moderate knee osteoarthritis. Arch. Phys. Med. Rehabil. 94, 2241–2247 (2013).
Tsai, T.-Y. et al. Asymmetric hip kinematics during gait in patients with unilateral total hip arthroplasty: In vivo 3-dimensional motion analysis. J. Biomech. 48, 555–559 (2015).
Winiarski, S. & Czamara, A. Evaluation of gait kinematics and symmetry during the first two stages of physiotherapy after anterior cruciate ligament reconstruction. Acta Bioeng. Biomech. 14, 91–100 (2012).
Rudroff, T. & Proessl, F. Effects of muscle function and limb loading asymmetries on gait and balance in people with multiple sclerosis. Front. Physiol. 9, 531 (2018).
Kernozek, T. W., Greany, J. F. & Heizler, C. Plantar loading asymmetry in American Indians with diabetes and peripheral neuropathy, with diabetes only, and without diabetes. J. Am. Podiatr. Med. Assoc. 103, 106–112 (2013).
Mc Ardle, R., Galna, B., Donaghy, P., Thomas, A. & Rochester, L. Do Alzheimer’s and Lewy body disease have discrete pathological signatures of gait?. Alzheimers Dement. J. Alzheimers Assoc. 15, 1367–1377 (2019).
Orcioli-Silva, D. et al. Double obstacles increase gait asymmetry during obstacle crossing in people with Parkinson’s disease and healthy older adults: A pilot study. Sci. Rep. 10, 2272 (2020).
Fling, B. W., Curtze, C. & Horak, F. B. Gait asymmetry in people with Parkinson’s disease is linked to reduced integrity of callosal sensorimotor regions. Front. Neurol. 9, 215 (2018).
Oken, O. & Yavuzer, G. Spatio-temporal and kinematic asymmetry ratio in subgroups of patients with stroke. Eur. J. Phys. Rehabil. Med. 44, 127–132 (2008).
Wei, T.-S., Liu, P.-T., Chang, L.-W. & Liu, S.-Y. Gait asymmetry, ankle spasticity, and depression as independent predictors of falls in ambulatory stroke patients. PLoS ONE 12, e0177136 (2017).
Alharbi, S. Gait asymmetry and the risk of knee osteoarthritis in post-stroke individuals. (University of Salford, 2019).
Patterson, K. K. et al. Gait asymmetry in community-ambulating stroke survivors. Arch. Phys. Med. Rehabil. 89, 304–310 (2008).
Lloyd, C. H., Stanhope, S. J., Davis, I. S. & Royer, T. D. Strength asymmetry and osteoarthritis risk factors in unilateral trans-tibial, amputee gait. Gait Posture 32, 296–300 (2010).
Adamczyk, P. G. & Kuo, A. D. Mechanisms of gait asymmetry due to push-off deficiency in unilateral amputees. IEEE Trans. Neural Syst. Rehabil. Eng. Publ. IEEE Eng. Med. Biol. Soc. 23, 776–785 (2015).
Heil, J., Loffing, F. & Büsch, D. The influence of exercise-induced fatigue on inter-limb asymmetries: A systematic review. Sports Med. Open 6, 39 (2020).
Jung, J., Choi, W. & Lee, S. Immediate augmented real-time forefoot weight bearing using visual feedback improves gait symmetry in chronic stroke. Technol. Health Care Off. J. Eur. Soc. Eng. Med. 28, 733–741 (2020).
Rozanski, G. M., Wong, J. S., Inness, E. L., Patterson, K. K. & Mansfield, A. Longitudinal change in spatiotemporal gait symmetry after discharge from inpatient stroke rehabilitation. Disabil. Rehabil. 42, 705–711 (2020).
Drużbicki, M. et al. Changes in gait symmetry after training on a treadmill with biofeedback in chronic stroke patients: A 6-month follow-up from a randomized controlled trial. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 22, 4859–4868 (2016).
Plotnik, M., Wagner, J. M., Adusumilli, G., Gottlieb, A. & Naismith, R. T. Gait asymmetry, and bilateral coordination of gait during a six-minute walk test in persons with multiple sclerosis. Sci. Rep. 10, 12382 (2020).
Highsmith, M. J. et al. Gait Training interventions for lower extremity amputees: A systematic literature review. Technol. Innov. 18, 99–113 (2016).
Alingh, J. F., Groen, B. E., Van Asseldonk, E. H. F., Geurts, A. C. H. & Weerdesteyn, V. Effectiveness of rehabilitation interventions to improve paretic propulsion in individuals with stroke—a systematic review. Clin. Biomech. Bristol Avon 71, 176–188 (2020).
Frazzitta, G., Pezzoli, G., Bertotti, G. & Maestri, R. Asymmetry and freezing of gait in parkinsonian patients. J. Neurol. 260, 71–76 (2013).
Ricciardi, L. et al. Working on asymmetry in Parkinson’s disease: Randomized, controlled pilot study. Neurol. Sci. Off. J. Ital. Neurol. Soc. Ital. Soc. Clin. Neurophysiol. 36, 1337–1343 (2015).
Viteckova, S. et al. Gait symmetry measures: A review of current and prospective methods. Biomed. Signal Process. Control 42, 89–100 (2018).
Venema, D. M., Hansen, H., High, R., Goetsch, T. & Siu, K.-C. Minimal detectable change in dual-task cost for older adults with and without cognitive impairment. J. Geriatr. Phys. Ther. https://doi.org/10.1519/JPT.0000000000000194 (2018).
Shumway-Cook, A., Woollacott, M., Kerns, K. A. & Baldwin, M. The effects of two types of cognitive tasks on postural stability in older adults with and without a history of falls. J. Gerontol. A. Biol. Sci. Med. Sci. 52, M232-240 (1997).
Bloem, B. R., Grimbergen, Y. A. M., van Dijk, J. G. & Munneke, M. The, “posture second” strategy: A review of wrong priorities in Parkinson’s disease. J. Neurol. Sci. 248, 196–204 (2006).
Smith, E., Cusack, T., Cunningham, C. & Blake, C. The influence of a cognitive dual task on the gait parameters of healthy older adults: A systematic review and meta-analysis. J. Aging Phys. Act. 25, 671–686 (2017).
Hunter, S. W., Divine, A., Frengopoulos, C. & Montero Odasso, M. A framework for secondary cognitive and motor tasks in dual-task gait testing in people with mild cognitive impairment. BMC Geriatr. 18, 202 (2018).
Al-Yahya, E. et al. Cognitive motor interference while walking: A systematic review and meta-analysis. Neurosci. Biobehav. Rev. 35, 715–728 (2011).
Santos Nogueira, D., Azevedo Reis, E. & Vieira, A. Verbal fluency tasks: Effects of age, gender, and education. Folia Phoniatr. Logop. Off. Organ Int. Assoc. Logop. Phoniatr. IALP 68, 124–133 (2016).
Brown, K. C. et al. Gait speed and variability for usual pace and pedestrian crossing conditions in older adults using the GAITRite walkway. Gerontol. Geriatr. Med. https://doi.org/10.1177/2333721415618858 (2015).
Jonathan, A. et al. Gait speed and 1-year mortality following cardiac surgery: A landmark analysis from the society of thoracic surgeons adult cardiac surgery database. J. Am. Heart Assoc. 7, e010139 (2018).
Fransen, M., Crosbie, J. & Edmonds, J. Reliability of gait measurements in people with osteoarthritis of the knee. Phys. Ther. 77, 944–953 (1997).
Brach, J. S., Perera, S., Studenski, S. & Newman, A. B. The reliability and validity of measures of gait variability in community-dwelling older adults. Arch. Phys. Med. Rehabil. 89, 2293–2296 (2008).
Byun, S., Han, J. W., Kim, T. H. & Kim, K. W. Test-retest reliability and concurrent validity of a single tri-axial accelerometer-based gait analysis in older adults with normal cognition. PLoS ONE 11, e0158956 (2016).
Beurskens, R., Steinberg, F., Antoniewicz, F., Wolff, W. & Granacher, U. Neural correlates of dual-task walking: Effects of cognitive versus motor interference in young adults. Neural Plast. 2016, 8032180 (2016).
Stöckel, T. & Mau-Moeller, A. Cognitive control processes associated with successful gait performance in dual-task walking in healthy young adults. Psychol. Res. 84, 1766–1776 (2020).
Holtzer, R., Epstein, N., Mahoney, J. R., Izzetoglu, M. & Blumen, H. M. Neuroimaging of mobility in aging: A targeted review. J. Gerontol. A. Biol. Sci. Med. Sci. 69, 1375–1388 (2014).
Yang, Q. et al. Gait change in dual task as a behavioral marker to detect mild cognitive impairment in elderly persons: A systematic review and meta-analysis. Arch. Phys. Med. Rehabil. 101, 1813–1821 (2020).
Yogev-Seligmann, G., Hausdorff, J. M. & Giladi, N. The role of executive function and attention in gait. Mov. Disord. Off. J. Mov. Disord. Soc. 23, 329–342; quiz 472 (2008).
Kikkert, L. H. J., Vuillerme, N., van Campen, J. P., Hortobágyi, T. & Lamoth, C. J. Walking ability to predict future cognitive decline in old adults: A scoping review. Ageing Res. Rev. 27, 1–14 (2016).
Quan, M. et al. Walking pace and the risk of cognitive decline and dementia in elderly populations: A meta-analysis of prospective cohort studies. J. Gerontol. A. Biol. Sci. Med. Sci. 72, 266–270 (2017).
Shrout, P. E. & Fleiss, J. L. Intraclass correlations: Uses in assessing rater reliability. Psychol. Bull. 86, 420–428 (1979).
Kluge, F. et al. Towards mobile gait analysis: Concurrent validity and test-retest reliability of an inertial measurement system for the assessment of spatio-temporal gait parameters. Sensors 17, 1522 (2017).
Hollman, J. H. et al. Number of strides required for reliable measurements of pace, rhythm and variability parameters of gait during normal and dual task walking in older individuals. Gait Posture 32, 23–28 (2010).
Acknowledgements
The authors would like to thank all the participants of the study and the Folomi team for conceiving and designing the Folomi study (J. Soulard, N. Vuillerme, P. Gaudin, L. Grange, A. Baillet, J-L Cracowski, R. Juvin, J. Vaillant).
Funding
This study is part of the Ph.D. thesis of the first author Julie Soulard (Univ. Grenoble Alpes, AGEIS, France & Grenoble Alps University Hospital, Grenoble, France). This work is supported by the French Ministry of Health and Solidarity with the “Nursing and Paramedical Hospital Research Program” year 2016 (PHRIP-16–0528) and by the French National Research Agency in the framework of the "Investissements d’avenir” program (ANR-10-AIRT-05 and ANR-15-IDEX-02). The sponsors had no involvement in the design of the study, the collection, analysis and interpretation of data, and in writing the manuscript. This work further forms part of a broader translational and interdisciplinary research program, GaitAlps (NV).
Author information
Authors and Affiliations
Contributions
This work is part of the PhD thesis of the first author J.S, co-supervised by J.V and N.V. J.S, J.V and N.V contributed to conception and design of this study. J.S performed data collection. J.S and R.B organized the database and performed the statistical analysis. All authors interpreted the data. J.S wrote the first draft of the manuscript. All authors contributed to manuscript revision, read and approved the submitted version of the manuscript. Each author has approved the submitted version. They have agreed both to be personally accountable for the author’s own contributions and to ensure that questions related to the accuracy or integrity of any part of the work, even ones in which the author was not personally involved, are appropriately investigated, resolved, and the resolution documented in the literature.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Soulard, J., Vaillant, J., Balaguier, R. et al. Spatio-temporal gait parameters obtained from foot-worn inertial sensors are reliable in healthy adults in single- and dual-task conditions. Sci Rep 11, 10229 (2021). https://doi.org/10.1038/s41598-021-88794-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-88794-4
This article is cited by
-
Can transcranial direct current stimulation combined with interactive computerized cognitive training boost cognition and gait performance in older adults with mild cognitive impairment? a randomized controlled trial
Journal of NeuroEngineering and Rehabilitation (2024)
-
Assessing falls in the elderly population using G-STRIDE foot-mounted inertial sensor
Scientific Reports (2023)
-
Validity and reliability of the Apple Health app on iPhone for measuring gait parameters in children, adults, and seniors
Scientific Reports (2023)
-
DUO-GAIT: A gait dataset for walking under dual-task and fatigue conditions with inertial measurement units
Scientific Data (2023)
-
Comprehensive validation of a wearable foot sensor system for estimating spatiotemporal gait parameters by simultaneous three-dimensional optical motion analysis
BMC Sports Science, Medicine and Rehabilitation (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.