Abstract
To investigate the role of quantitative ultrasound (QUS) radiomics to predict treatment response in patients with head and neck squamous cell carcinoma (HNSCC) treated with radical radiotherapy (RT). Five spectral parameters, 20 texture, and 80 texture-derivative features were extracted from the index lymph node before treatment. Response was assessed initially at 3 months with complete responders labelled as early responders (ER). Patients with residual disease were followed to classify them as either late responders (LR) or patients with persistent/progressive disease (PD). Machine learning classifiers with leave-one-out cross-validation was used for the development of a binary response-prediction radiomics model. A total of 59 patients were included in the study (22 ER, 29 LR, and 8 PD). A support vector machine (SVM) classifier led to the best performance with accuracy and area under curve (AUC) of 92% and 0.91, responsively to define the response at 3 months (ER vs. LR/PD). The 2-year recurrence-free survival for predicted-ER, LR, PD using an SVM-model was 91%, 78%, and 27%, respectively (p < 0.01). Pretreatment QUS-radiomics using texture derivatives in HNSCC can predict the response to RT with an accuracy of more than 90% with a strong influence on the survival.
Clinical trial registration: clinicaltrials.gov.in identifier NCT03908684.
Similar content being viewed by others
Introduction
Radiomics is an emerging field in oncology involving advanced computational imaging analysis and typically involves the application of artificial intelligence for meaningful interpretation of data1. Tumour images are now recognized as more than greyscale images, and computer vision can unfold information that can be linked with underlying genotypic and phenotypic features2. Imaging forms an integral role in oncology in diagnosis, disease staging, treatment planning, the assessment of treatment response, and tumour surveillance. Standard imaging modalities involve morphological-based techniques like ultrasonography (US), computed tomography (CT), magnetic resonance imaging (MRI) or functional imaging like positron emission tomography (PET) or functional MRI (fMRI). The use of radiomics to serve as potential noninvasive biomarkers in risk stratification, in prediction and monitoring of treatment response has generated interest to develop strategies towards precision oncology1,3.
Head and neck malignancies accounted for 890,000 new cases worldwide (seventh most common globally) and 450,000 deaths in 20184. Radical radiotherapy (RT) with or without concurrent chemotherapy forms the primary treatment for a majority of patients with head and neck squamous cell carcinomas (HNSCC) arising from the oropharynx, hypopharynx, or larynx leading to excellent organ preservation5. Locally advanced cancer is present in approximately 40–60% of patients during presentation, which involves advanced primary disease and/or regional lymph nodes (LN)5,6. In the past decades, technological advances in RT with intensity-modulated radiotherapy (IMRT) and image-guided radiotherapy (IGRT) have led to a reduction of radiation toxicities like xerostomia7. The survival outcome of patients with locally advanced HNSCC remains compromised despite such advancements. As a result, there is increasing interest regarding the use of response-guided adaptive RT, with several ongoing trials investigating dose-escalation strategies for patients deemed to be non-responders8,9.
As imaging has a crucial role in the management of HNSCC, radiomic analysis has been undertaken for different imaging modalities like PET, CT, and MRI10,11,12. Study endpoints have been variable with some studies investigating molecular characteristics, while others have linked radiomic features with survival outcomes with encouraging results. Quantitative ultrasound (QUS) uses data typically not interpreted by clinical B-mode US devices. QUS uses raw radiofrequency (RF) data, which can provide information related to tissue microstructure elastic properties and underlying biology13. Tumour treatment response can be detected using QUS much earlier than conventional imaging due to ongoing changes in elastic properties associated with cell death14,15. Radiomic analysis of QUS has proven to be useful in the determination of response to neoadjuvant chemotherapy in breast cancer16,17,18,19.
In previous studies, QUS obtained before and during treatment was shown to be promising in predicting treatment response for head-neck malignancies20,21. In a cohort of 32 patients, pre-treatment QUS could predict the response at 3 months with an accuracy of 88%20. In a subsequent study including 36 patients, the classifier performance was improved when QUS features were obtained during radiotherapy as early as after 1 week of treatment compared to pr-treatment21. In the current study, the number of patients has been increased to 59, and includes a homogeneous group of HNSCC (excluding other tumour histologies included in the previous study such as nasopharynx, parotid, and others). Also, the study methodology has significant development incorporating third-order imaging features (texture derivatives), using more advanced machine learning classifiers, and final correlation with clinical outcomes. In addition, the endpoint of final response beyond 3 months in partial responders was analyzed in the present study. The imaging features used in the study were spectral parameters, texture of spectral parameters (QUS-Tex1), and second-order texture analysis of QUS-Tex1 features (QUS-Tex1-Tex2). Five spectral parameters were used-mid-band fit (MBF), spectral slope (SS), spectral intercept (SI), average scatterer diameter (ASD), and average acoustic concentration (AAC). The machine learning classifiers included Fischer’s linear discriminant analysis (FLD), k-nearest neighbours (KNN), and support vector machines-radial basis function (SVM-RBF). Four textural features of contrast (CON), correlation (COR), energy (ENE), and homogeneity (HOM) were analyzed. To the best of our knowledge, this is the first study of QUS-radiomics using higher-order imaging features to predict different groups of radiation responses, which were linked to clinical outcomes.
Results
Clinical characteristics
A total of 59 patients with node-positive HNSCC were included in the current analysis. The different patient, disease, and treatment-related features have been summarized in Table 1. None of the clinical features were significantly distributed between the three response groups (ER/LR/PD). In the entire group, the median age was 61 years (range, 39 to 80 years), with 12 patients being 70 years or above. The median size of the lymph nodes was 3.1 cm (range 1.3 to 7 cm), without any significant difference between the three response groups. The most common primary site was oropharynx in 42, followed by carcinoma unknown primary in 8, larynx in 6, and hypopharynx in 3 patients. The human papillomavirus (HPV) p16 immunostaining was available in 44 patients, with positive staining in 38 (86%). Concurrent chemotherapy was used in 49, cetuximab (without chemotherapy) in 2, and RT alone in 8 patients. All patients included in the current study completed the scheduled course of RT.
Clinical outcomes
During the first response evaluation at 3 months, 22 patients had a complete response and were designated as early responder (ER), as described earlier. For the remaining 37 patients, 29 were designated as late responder (LR) as they had complete disease resolution with a median of 6 months from RT completion (range 4 to 10 months), whereas 8 patients had persistent/progressive disease (PD) involving treated target disease (primary and nodal target volumes). Median follow up for all the patients was 32 months (range 5 to 64 months). At follow up for the current study, 18 patients had recurrent disease (local-1, nodal-5, distant-9, local-nodal-1, local-nodal-distant-2). The 2-year and 5-year recurrence-free survival (RFS) for all patients was 72% and 68%, respectively. The 2-year and 5-year overall survival (OS) for the entire cohort was 86% and 61%, respectively. Both the RFS and OS were significantly different between the three response groups (Table 1). The 2-year RFS for the ER, LR, and PD was 96%, 74%, and 13%, respectively (p < 0.01).
Feature analysis
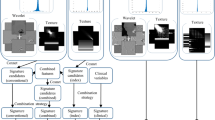
Representative B-mode images and the corresponding representative QUS and QUS-texture maps are presented in Fig. 1 for three patients, one each from the ER, LR, and PD groups. Obvious intratumoral heterogeneity was evident from the spectral parametric maps and their texture maps. Figure 1 demonstrates typical hypoechogenicity for the tumour and apparent heterogeneity in quantitative ultrasound parameters.
Representative ultrasound B-mode (uppermost row), QUS spectral parametric maps of AAC and MBF, and the corresponding texture images from one patient in each of the three response groups-early responder (a), late responder (b), and persistent disease (c). QUS parametric images include the largest involved lymph node (central region bounded by closed dotted white curve). The colour bar ranges are 0 to 150 dB/cm3 for AAC, − 20 to 20 dB for MBF and arbitrary unit for the texture features. The scale bar represents 1 cm.
Table 2 indicates the features that exhibited statistically significant differences between the different response groups. Three months after RT completion, 12 features were found to be significantly different between the complete response (ER) and partial/non-responder (LR/PD) groups. These included two spectral parameters (MBF, SI), one QUS-Tex1 feature (MBF-CON), and 9 QUS-Tex1-Tex2 features. Between the LR and PD groups, a different set of 12 features had different distributions. One spectral feature (ASD), three QUS-Tex1 features (AAC-CON, ASD-CON, and AAC-HOM), and 8 QUS-Tex1-Tex2 features were found to have different values between these two response groups. The scatter plots of all the 105 features between the binary response groups have been shown in Supplementary Fig. S1.
Classification performances
Table 3 presents the classifier performance using the three classifiers for the two binary response groups. The SVM-RBF model demonstrated the best results in differentiating the ER from the LR/PD group with a sensitivity, specificity, accuracy, and AUC of 86%, 95%, 92%, and 0.91, respectively. The three features selected for building the classifier model were QUS-Tex1-Tex2 parameters (MBF-HOM-CON, MBF-ENE-CON, ASD-HOM-ENE). For the SVM model, the accuracy was improved by 3% with parameter tuning. To study the influence of texture-derivatives on the classifier performance, the analyses were carried out in a step-wise manner, once using spectral and QUS-Tex1 features only and then finally, with all the 105 features incorporating QUS-Tex1-Tex2 features. The inclusion of texture-derivatives led to a significant improvement of the classifier performances. Without the texture-derivatives, the AUC was 0.70, 0.72, 0.51 for FLD, KNN, and SVM-RBF, which was improved to 0.75, 0.80, and 0.91 when all the QUS-Tex1-Tex2 features were included. The ROC plots using the three classifiers for the two different endpoints (with and without texture-derivatives) are shown in Fig. 2. The pictorial representation of the hyperplane plot using the SVM-RBF model to classify the two response groups at 3 months has been presented in Fig. 3.
ROC plots of predictive models for different endpoints developed using spectral and texture features alone (upper row) and those developed using all the spectral, texture, and texture-derivate features (lower row). The endpoints considered are 3-month complete responder versus partial responder/non-responder (a,c), late responder versus persistent/progressive disease (b,d). Three standard classification algorithms that include FLD, KNN, and SVM-RBF were evaluated as indicated in the inset legend. The classification models that include texture-derivate features (lower row) achieved higher AUC values in general compared to those developed without texture-derivate features.
In order to differentiate the LR from PD, the SVM-RBF performed better than the other two models with sensitivity, specificity, accuracy, and AUC of 97%, 88%, 95%, and 0.97, respectively. The two selected features in the model were SS and SS-HOM-CON. Similar to the previous endpoint, the inclusion of texture derivatives improved the AUC for the SVM-RBF model from 0.92 to 0.97.
Finally, each patient was assigned to one of the three predicted response groups using the SVM-RBF model. The 2-year RFS for the predicted ER, predicted LR, and predicted PD groups were 91%, 78%, and 27%, respectively (p < 0.01). Figure 4 shows the comparison of the RFS survival plots generated using the actual response groups and the radiomics-predicted response groups.
Discussion
Head and neck malignancies represent a diverse group of malignancies arising commonly from the epithelial lining of the upper aerodigestive tracts and associated glandular structures5. Outcomes in HNSCC are strongly influenced by clinical-pathological factors like the site of primary disease, disease stage, performance status of the patient, and causative factors like tobacco abuse or HPV. For patients with locally-advanced HNSCC or node-positive disease, the prognosis is poor, with an estimated 5-year survival rate being approximately 50% or less5. Also, with the presence of vital anatomical and physiological structures in the head and neck region, in patients cured of their disease, long-term treatment-related late toxicities can have significant adverse effects on quality of life22,23. There is an unmet need for the development of reliable biomarkers, which can help in better risk-stratification so that existing treatment strategies can be tailored accordingly. The introduction of computational techniques and artificial intelligence in imaging and medicine has led to the development of the field of radiomics, with standard imaging modalities producing new potential noninvasive biomarkers. In the current study, the role of ultrasound before radiation treatment was investigated in predicting final clinical response to therapy, using higher-order imaging features.
In the past few years, there had been an increasing number of studies investigating the role of radiomics in HNSCC, primarily involving CT, PET, or MRI10,11,12. The majority of the radiomic studies have investigated endpoints like molecular characteristics (HPV status) and clinical outcomes like local control, distant metastasis, and overall survival. Although favourable results have been obtained from most of the studies, there has been variance with regards to image processing, feature selection strategies, machine learning algorithms, and validation approaches24. A recent multi-institutional has indicated the utility of pre-operative contrast-enhanced CT-based radiomics in predicting histopathological extranodal extension25. In a study involving 128 patients with head and neck malignancies, Bogowicz et al. had shown radiomics analysis of CT features from the primary tumour and lymph nodes to correlate with loco-regional control26. Similarly, in a study involving 300 patients with HPV-related oropharyngeal cancers, Kwan et al. associated distant metastasis with radiomic features obtained from radiation planning CT images27. Vallières et al. had demonstrated that radiomic analysis of pretreatment PET and CT images could be used in a risk assessment for loco-regional recurrence and distant metastasis in a study of 300 patients with head neck malignancies undergoing radical intent RT28. Fewer imaging studies have investigated response to therapy as a study endpoint in head neck malignancies. Liu et al. performed texture analysis of T1, T2, and diffusion MR sequences in 53 patients with NPC treated with chemoradiation, which was able to predict the treatment response with an accuracy of approximately 90%29. Similarly, Wang et al. used pretreatment MRI in 120 patients with NPC to predict response to induction chemotherapy30.
Ultrasonography is frequently used in the management of HNSCC, more commonly indicated in the characterization of suspicious neck nodes, as well as to guide biopsy or cytology procedures for histopathological confirmation31,32. B-mode US is more popularly used in clinical scenarios. The use of QUS involves technically identical ultrasound devices, but with raw RF data collected and analyzed quantitatively. QUS detects the elastic properties of the tissue microstructure and is influenced by parameters like cell size, shape, and organization33. Microcellular elastic properties have been demonstrated to be different across different grades of tumours, which have distinct biological behaviour34. In tissue characterization, QUS techniques have been proven to be extremely sensitive in detecting ongoing changes with treatment-induced cell death since it leads to changes in nuclear and cell structures properties with cell fragmentation, pyknosis, and the formation of apoptotic bodies, ultimately changing scatterer elastic properties14,15. Clinical studies investigating QUS-radiomics indicated this methodology to be useful in predicting response to neoadjuvant chemotherapy (NAC) in patients with locally advanced breast cancer (LABC) before starting treatment16. Similarly, response changes with NAC can be detected as early during the first week of treatment, correlating with the final response obtained from histopathological examination months after the treatment is started17. This methodology is being evaluated in a randomized clinical trial of QUS-radiomics guided adaptive chemotherapy, where patients predicted to have inadequate response can have modifications made to their NAC (clinicaltrials.gov identifier NCT04050228). The application of QUS imaging for head and neck malignancies is a relatively new technique, with initial results from 32 patients showing a predictive accuracy of 88% using spectral and texture features (18). Compared to other imaging modalities, QUS-based radiomics has the advantage of rapid scan acquisition that can be performed with a portable device, with excellent patient compliance. A QUS-based approach is expected to provide more biologically-linked information compared to other imaging like CT or MRI as the imaging information can be tuned to pick up details from a smaller foci of cells and is sensitive to approximately 10–20 cell diameters using a mean frequency of 6 MHz) (24).
In the current study, using an SVM-based model, the accuracy of classification between complete responders versus non-responders/partial responders at 3 months and late responders vs persistent/progressive disease patients was 92% and 95%, respectively. There were 12 features at each time having a differential distribution between the two binary groups. At the 3 month time, most of the features that were different between the two response groups were related to the MBF parameter. Mid-band fit denotes the value of linear fit at the central frequency and is influenced by scatterer size, shape, and organization (24). The 3-feature set used in the classification model used texture-derivatives of one ASD parameter and 2 MBF parameters. ASD is dependant on cell size, suggesting a differential structural organization between the two response groups, which impacted radiation response. Between the LR and PD response groups, the values were seen to be different for many of the AAC parameters, which accounts for the density of scatterers, their organization, and their elastic properties. The 2-feature SVM-model used SS and SS-texture-derivative features, with SS representing information related to cell shape and size. It is essential to note the majority of features selected in model development were texture-derivatives suggesting intratumoral heterogeneity to have an important implication on response to radiation and clinical outcomes. Also, the inclusion of higher-order texture features led to an improvement in classifier performance, indicating a better representation of spatial heterogeneity with third-order imaging analysis.
Response to radiation had a strong influence on clinical outcomes (both RFS and OS). The patients with an early complete response within the first 3 months are likely to harbour disease sensitive to RT and had the best outcomes. Patients who exhibited a delayed response demonstrated an intermediate prognosis. As expected, patients with residual or progressive disease in an irradiated volume indicated radioresistant disease and showed the poorest RFS and OS amongst the patient groups. The radiomics model developed here can be utilized as a tool for predicting clinical radiosensitivity, with links to ultimate survival outcomes. The development of treatment-escalation strategies has been explored elsewhere using response-guided adaptive radiotherapy to improve outcomes in patients with poor outcomes8,35. Similarly, de-escalation strategies can be considered in patients with an expected better prognosis in order to avoid treatment-related toxicities. Recent studies exploring treatment de-escalation with the replacement of concurrent cisplatin with cetuximab in HPV-positive oropharyngeal cancer resulted in negative results36,37. These results explored a generalized approach using HPV as a marker of de-intensification of chemotherapy but turned out to be ineffective. The use of a QUS-based radiomics model can help in predicting clinical radiosensitivity aiding in better risk-stratification on an individual patient basis and guiding towards precision medicine.
The current analysis involved a relatively small number of patients. The study here is currently being continued, and with further expansion, it is expected to lead to the generation of more robust and reproducible imaging biomarker models. Thus study included binary endpoints at two different times due to a small number of samples. With the inclusion of more patients, advanced classification algorithms like deep learning strategies and more robust validation strategies can be undertaken. In addition, given the technical challenges of imaging primary head and neck tumours (deep location, tissue interfaces), in the patients here, the neck node was scanned. Given the high correlation of response of both the primary tumour and index neck node following treatment, using nodal imaging features alone suggests the clinical utility of the QUS-radiomics methods evaluated in the work here.
Methods and materials
Patient selection
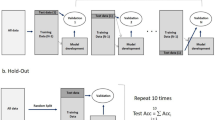
The prospective study was conducted based on good clinical practice according to the declarations of Helsinki and institutional research ethics board approval (Sunnybrook Health Sciences Center, Canada) and registered with clinicaltrials.gov (identifier NCT03908684, registered on 09/04/2019). Patients with a diagnosis of biopsy-proven HNSCC with metastatic LN, the latter amenable to ultrasound imaging from a primary disease involving oropharynx, hypopharynx, larynx, or carcinoma of an unknown primary (CUP) treated with radical RT were eligible for this study. Patients with poor performance status (Eastern Co-operative Oncology Group > 1), prior treatment for HNSCC, previous history of RT to the head and neck region, severe medical or psychiatric comorbidities with a life expectancy of < 6 months or unreliable for follow up were considered as exclusion criteria. Informed consent was obtained from all patients accrued in this study. Patients with nasopharyngeal cancer (NPC), CUP suspected from NPC (Epstein-Barr virus positivity or histological suspicion), oral cavity primary or no follow up after treatment completion for response assessment were excluded from the current analysis. The study methodology has been represented in Fig. 5.
Treatment protocols and response evaluation
Study participation had no influence on patient treatment or follow up. All patients were treated using a uniform radiation dose regimen of 70 Gy/33–35 fractions over 6–7 weeks to the high-risk volume using IMRT and IGRT techniques according to standard institutional practice. The use of concurrent systemic therapy was at the discretion of the responsible medical oncologist. The first response to treatment was evaluated at approximately 3 months following completion of RT with clinical examination, endoscopy, and morphological imaging with CT/MRI supplemented with PET-CT. Patients with complete resolution of the primary tumour and index lymph node < 1 cm at the end of 3 months were considered as “Early Responders” (ER). Patients with a residual nodal size of < 1 cm with strong radiological suspicion of residual disease or histological confirmation were excluded from the ER category. Patients with partial response or non-responders at 3 months were further followed up with clinical examinations, additional imaging, histopathological examination, or surgical intervention. Patients who experienced complete resolution of disease beyond 3 months were labelled as “Late Responders” (LR). Other patients were labelled as “persistent or progressive disease” (PD) involving the primary or index LN. Further follow up was done every 3–6 months in the initial 2 years and thereafter every 6–12 months as indicated. Study accrual was carried out between January 2015 and October 2019, with the final analysis done in June 2020.
Image acquisition
Ultrasound RF data were collected from the index LN most amenable to imaging using a Sonix RP ultrasound imaging system (Ultrasonix, Vancouver, Canada) or an equivalent OEM system from Elekta Ltd (Montreal, Canada) before starting RT. Imaging was performed by a research sonographer with experience in head & neck ultrasound imaging. The system used a linear array transducer (Elekta: 4DL14-5/39, Sonix RP: L14-5/60) with a center frequency of 6.5 MHz with a bandwidth of 3–8 MHz. A 40 MHz sampling frequency was used for digitization. Data were acquired along 512 scan lines, spanning a 6 cm lateral field-of-view and the focal depth of 1.75 cm (Sonix RP) and 2.5 cm (Elekta). Earlier work has indicated that with appropriate data normalization using standardized techniques, QUS features are consistent between different clinical ultrasound devices38. The index LN was manually contoured on US B-mode images and designated as the region of interest (ROI). For each patient, typically 3–5 slices were selected from the entire ROI at regular intervals. Given the technical challenges in volumetric acquisition and analysis of conventional probe-based ultrasound, the slices were selected to represent different areas of the lymph node. Quantitative ultrasound spectroscopic analysis, associated texture analysis, and texture-derivate analyses were performed on the selected regions from the target LN as described below.
Quantitative ultrasound spectral features
Spectral analysis was performed using the RF data associated with the segmented area of the index LN. A sliding window analysis was used with a 2-mm by 2-mm kernel to create parametric images for each QUS spectral parameter. A 94% overlap was used between adjacent sliding windows in both axial and lateral directions. Prior to spectral analysis, a Hanning gating function was applied on individual RF scan lines within the window. The power spectrum was generated using a Fast Fourier Transform (FFT) method. An average power spectrum was obtained from RF signals within the window. The power spectrum was normalized using a reference phantom technique using 5–30 µm glass beads embedded in a homogeneous medium of oil droplets embedded in gelatin39,40. For the phantom, the measured attenuation coefficient and speed of sound were 0.8 dB/cm/MHz and 1540 m/s, respectively (the University of Wisconsin, Department of Medical Physics, Madison, WI, USA). Attenuation compensation was applied to account for the attenuation from the intervening tissue layers (intervening tissue and tumour) considering 1 dB/cm/MHz for the overlying breast tissues39,41. The attenuation coefficient estimate (ACE) for the tumour was determined using a spectral difference method. This considered the rate of change in the log spectral power magnitude for the frequency bandwidth with depth (over the tumour region) relative to the reference phantom39,40. Five spectral parameters were determined from the power spectrum-mid-band fit (MBF), spectral slope (SS), spectral intercept (SI), average scatterer diameter (ASD), and average acoustic concentration (AAC). Further details and the biological correlations of individual spectral parameters have been described in previous publications33,42. The values of the different spectral features were obtained from the individual windows, and the average weighted values from all the slices were used as first-order imaging features.
Texture parameters
Colour-coded quantitative ultrasound-based parametric maps were generated based on the values of individual spectral parameters from each of the corresponding windows. A grey level co-occurrence matrix (GLCM) method was used to determine texture features to quantify intra-tumoural heterogeneity43. The GLCM method analyses the spatial relationship between neighbouring pixels at different angular directions. The grey level intensities of each of the parametric images were linearly scaled into 16 discrete values. The GLCM matrices were created from each QUS-parametric map at inter-pixel distances: 1, 2, 3, 4, 5 pixels and at four angular directions: 0°, 45°, 90°, and 135°. Four textural features of contrast (CON), correlation (COR), energy (ENE), and homogeneity (HOM) were extracted and subsequently averaged over distances and angular directions to generate second-order imaging features (QUS-Tex1). Therefore, 5 spectral features led to the generation of 20 QUS-Tex1 features.
The third-order imaging features were texture-derivatives (QUS-Tex1-Tex2). A previous study involving breast cancer had demonstrated an improvement in the classifier performances with the inclusion of higher-order imaging features in the form of texture derivatives44. Texture-derivate analysis was done by creating intermediate texture-encoded maps using sliding window analysis with a 15-pixel by 15-pixel window with each pixel in these maps representing quantification of local textures within the concerned window. A second pass GLCM analysis was performed on the texture maps, resulting in 80 QUS-Tex1-Tex2 features.
The weighted averaged measures of the features were used for building models for predicting response. A total set of 105 QUS-radiomic features (5 spectral, 20 QUS-Tex1, 80 QUS-Tex1-Tex2) were acquired before starting RT. Imaging was obtained nominally within 24 h in advance of the start of treatment, although an interval of 7 days was allowed according to the study protocol.
Statistical analysis
Binary endpoints were used for constructing the radiomics model due to a relatively smaller number of patients in our study, and multiple classifier outputs will need a larger sample size for robust classification. The first endpoint was the response at 3 months following completion of RT-complete responders vs. partial/non-responders (i.e. ER vs LR + PD). The other classification endpoint was LR vs PD.
The distribution of categorical variables (clinical features) between the three response groups was studied using the Pearson chi-square test and Fisher’s exact test as appropriate. For the feature values, a Shapiro–Wilk test was performed to test the normality of distribution. For comparison between the binary response groups, an unpaired t-test was used for normally distributed data, while a Mann–Whitney U-Test was used for non-parametric data. The survival analysis was performed using the Kaplan–Meier product-limit method, with the date of starting RT considered as baseline. A comparison of the different factors on survival was conducted using a log-rank test. A p-value of < 0.05 was used as a threshold of statistical significance.
Machine learning classifiers
Three standard computational algorithms were used for the development of a radiomics model: Fischer’s linear discriminant analysis (FLD), k-nearest neighbours (KNN), and support vector machines-radial basis function (SVM-RBF). The FLD is a linear classification algorithm exploring multi-dimensional feature space that maximizes the ratio between-class to within-class variance. The KNN is an instance-based classification algorithm studying class association of a test point in the feature space. It is based on the spatial distribution of most of the points neighbouring the test point and the distance between those points to the test point. The classifier was tested using k-values of 1 to 5 nearest neighbours to find the one leading to the most optimal classification. The SVM-RBF algorithm used a nonlinear classification that maximizes the margin between the two specified classes. The input data was mapped into a higher-dimensional space using kernel functions where the data are supposed to have better distribution, which then applied a hyperplane optimally separating the two classes in this higher-dimensional feature space. A Gaussian radial basis function (RBF) as the kernel function was used in our study. The model parameters (soft margin parameter C and the free parameter γ) were optimized using a grid search method.
A forward sequential-feature-selection (SFS) technique was used for classification using predetermined endpoints. To avoid the overfitting of the predictive model, the number of features for the final classification was limited to 3 for ER vs. LR/PD and 2 features for LR vs. PD45. The classifier performance was trained based on F1-Score (the harmonic average of precision and sensitivity). A leave-one-out (LOO) cross-validation technique was used for validation and testing the efficacy of the classifier performances, which involves training the classification model with all observations except one which was used to test the developed model. Receiver operating characteristics (ROC) analysis was performed utilizing sensitivity, specificity, accuracy, and area under curve (AUC). The segmentation, feature extraction, texture analysis, and machine learning classification were done using MATLAB 2019b (MathWorks Inc., USA). Survival analysis was performed using IBM SPSS version 21 (IBM corporation).
Conclusions
The use of a pretreatment texture-derivative based QUS-radiomics model was able to predict the final response to radiation with excellent accuracy (more than 90%). Clinical radiosensitivity had a strong influence on the survival outcomes, and a support vector machine classifier could accurately identify patients at higher risk of disease recurrence based on QUS-predictive factors.
Data availability
Data will be shared upon request to the corresponding author and the institutional ethics committee according to the policy of Sunnybrook Health Sciences Centre, Toronto.
References
Lambin, P. et al. Radiomics: The bridge between medical imaging and personalized medicine. Nat. Rev. Clin. Oncol. 14, 749–762 (2017).
Gillies, R. J., Kinahan, P. E. & Hricak, H. Radiomics: Images are more than pictures, they are data. Radiology 278, 563–577 (2015).
Aerts, H. J. W. L. The potential of radiomic-based phenotyping in precision medicine: A review. JAMA Oncol. 2, 1636–1642 (2016).
Bray, F. et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 68, 394–424 (2018).
Chow, L. Q. M. Head and neck cancer. N. Engl. J. Med. 382, 60–72 (2020).
Marur, S. & Forastiere, A. A. Head and neck squamous cell carcinoma: Update on epidemiology, diagnosis, and treatment. Mayo Clin. Proc. 91, 386–396 (2016).
Nutting, C. M. et al. Parotid-sparing intensity modulated versus conventional radiotherapy in head and neck cancer (PARSPORT): A phase 3 multicentre randomised controlled trial. Lancet Oncol. 12, 127–136 (2011).
Berwouts, D. et al. Long-term outcome of 18F-fluorodeoxyglucose-positron emission tomography-guided dose painting for head and neck cancer: Matched case-control study. Head Neck 39, 2264–2275 (2017).
Morgan, H. E. & Sher, D. J. Adaptive radiotherapy for head and neck cancer. Cancers Head Neck 5, 1 (2020).
Wong, A. J., Kanwar, A., Mohamed, A. S. & Fuller, C. D. Radiomics in head and neck cancer: From exploration to application. Transl. Cancer Res. 5, 371–382 (2016).
Giraud, P. et al. Radiomics and machine learning for radiotherapy in head and neck cancers. Front. Oncol. 9, 174 (2019).
Haider, S. P., Burtness, B., Yarbrough, W. G. & Payabvash, S. Applications of radiomics in precision diagnosis, prognostication and treatment planning of head and neck squamous cell carcinomas. Cancers Head Neck 5, 6 (2020).
Oelze, M. L. & Mamou, J. Review of quantitative ultrasound: Envelope statistics and backscatter coefficient imaging and contributions to diagnostic ultrasound. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 63, 336–351 (2016).
Czarnota, G. J. et al. Ultrasound imaging of apoptosis: High-resolution non-invasive monitoring of programmed cell death in vitro, in situ and in vivo. Br. J. Cancer 81, 520–527 (1999).
Kolios, M. C., Czarnota, G. J., Lee, M., Hunt, J. W. & Sherar, M. D. Ultrasonic spectral parameter characterization of apoptosis. Ultrasound Med. Biol. 28, 589–597 (2002).
Tadayyon, H. et al. A priori prediction of neoadjuvant chemotherapy response and survival in breast cancer patients using quantitative ultrasound. Sci. Rep. 7, 45733 (2017).
Sannachi, L. et al. Response monitoring of breast cancer patients receiving neoadjuvant chemotherapy using quantitative ultrasound, texture, and molecular features. PLoS ONE 13, e0189634 (2018).
DiCenzo, D. et al. Quantitative ultrasound radiomics in predicting response to neoadjuvant chemotherapy in patients with locally advanced breast cancer: Results from multi-institutional study. Cancer Med. 9, 5798–5806 (2020).
Quiaoit, K. et al. Quantitative ultrasound radiomics for therapy response monitoring in patients with locally advanced breast cancer: Multi-institutional study results. PLoS ONE 15, e0236182 (2020).
Tran, W. T. et al. Predictive quantitative ultrasound radiomic markers associated with treatment response in head and neck cancer. Future Sci. OA 6, 433 (2019).
Tran, W. T. et al. Quantitative ultrasound delta-radiomics during radiotherapy for monitoring treatment responses in head and neck malignancies. Future Sci. OA https://doi.org/10.2144/fsoa-2020-0073 (2020).
Bentzen, S. M., Rosenthal, D. I., Weymuller, E. A. & Trotti, A. Increasing toxicity in nonoperative head and neck cancer treatment: Investigations and interventions. Int. J. Radiat. Oncol. Biol. Phys. 69, S79–S82 (2007).
Ringash, J. Quality of life in head and neck cancer: Where we are, and where we are going. Int. J. Radiat. Oncol. Biol. Phys. 97, 662–666 (2017).
Guha, A. et al. Radiomic analysis for response assessment in advanced head and neck cancers, a distant dream or an inevitable reality? A systematic review of the current level of evidence. Br. J. Radiol. 93, 20190496 (2020).
Kann, B. H. et al. Multi-institutional validation of deep learning for pretreatment identification of extranodal extension in head and neck squamous cell carcinoma. JCO 38, 1304–1311 (2019).
Bogowicz, M., Tanadini-Lang, S., Guckenberger, M. & Riesterer, O. Combined CT radiomics of primary tumor and metastatic lymph nodes improves prediction of loco-regional control in head and neck cancer. Sci. Rep. 9, 15198 (2019).
Kwan, J. Y. Y. et al. Radiomic biomarkers to refine risk models for distant metastasis in HPV-related oropharyngeal carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 102, 1107–1116 (2018).
Vallières, M. et al. Radiomics strategies for risk assessment of tumour failure in head-and-neck cancer. Sci. Rep. 7, 10117 (2017).
Liu, J. et al. Use of texture analysis based on contrast-enhanced MRI to predict treatment response to chemoradiotherapy in nasopharyngeal carcinoma. J. Magn. Reson. Imaging 44, 445–455 (2016).
Wang, G. et al. Pretreatment MR imaging radiomics signatures for response prediction to induction chemotherapy in patients with nasopharyngeal carcinoma. Eur. J. Radiol. 98, 100–106 (2018).
Ahuja, A. T. & Ying, M. Sonographic evaluation of cervical lymph nodes. Am. J. Roentgenol. 184, 1691–1699 (2005).
Lin, C.-M. et al. The application of ultrasound in detecting lymph nodal recurrence in the treated neck of head and neck cancer patients. Sci. Rep. https://doi.org/10.1038/s41598-017-04039-3 (2017).
Sannachi, L. et al. Non-invasive evaluation of breast cancer response to chemotherapy using quantitative ultrasonic backscatter parameters. Med. Image Anal. 20, 224–236 (2015).
Tadayyon, H., Sadeghi-Naini, A. & Czarnota, G. J. Noninvasive characterization of locally advanced breast cancer using textural analysis of quantitative ultrasound parametric images. Transl. Oncol. 7, 759–767 (2014).
Bentzen, S. M. & Gregoire, V. Molecular imaging-based dose painting: A novel paradigm for radiation therapy prescription. Semin. Radiat. Oncol. 21, 101–110 (2011).
Gillison, M. L. et al. Radiotherapy plus cetuximab or cisplatin in human papillomavirus-positive oropharyngeal cancer (NRG Oncology RTOG 1016): A randomised, multicentre, non-inferiority trial. The Lancet 393, 40–50 (2019).
Mehanna, H. et al. Radiotherapy plus cisplatin or cetuximab in low-risk human papillomavirus-positive oropharyngeal cancer (De-ESCALaTE HPV): An open-label randomised controlled phase 3 trial. The Lancet 393, 51–60 (2019).
Sannachi, L. et al. Quantitative ultrasound monitoring of breast tumour response to neoadjuvant chemotherapy: Comparison of results among clinical scanners. Ultrasound Med. Biol. 46, 1142–1157 (2020).
Labyed, Y. & Bigelow, T. A. Estimating the total ultrasound attenuation along the propagation path by using a reference phantom. J. Acoust. Soc. Am. 128, 3232–3238 (2010).
Labyed, Y., Bigelow, T. A. & McFarlin, B. L. Estimate of the attenuation coefficient using a clinical array transducer for the detection of cervical ripening in human pregnancy. Ultrasonics 51, 34–39 (2011).
Duric, N. et al. Development of ultrasound tomography for breast imaging: Technical assessment. Med. Phys. 32, 1375–1386 (2005).
Sannachi, L. et al. Breast cancer treatment response monitoring using quantitative ultrasound and texture analysis: Comparative analysis of analytical models. Transl. Oncol. 12, 1271–1281 (2019).
Haralick, R. M., Shanmugam, K. & Dinstein, I. Textural features for image classification. IEEE Trans. Syst. Man Cybern. 3, 610–621 (1973).
Dasgupta, A. et al. Quantitative ultrasound radiomics using texture derivatives in prediction of treatment response to neo-adjuvant chemotherapy for locally advanced breast cancer. Oncotarget 11, 3782–3792 (2020).
Park, S. H. & Han, K. Methodologic guide for evaluating clinical performance and effect of artificial intelligence technology for medical diagnosis and prediction. Radiology 286, 800–809 (2018).
Acknowledgements
We would like to thank all the patients for their participation in the study. Our sincere gratitude to the physicians and other health care staff for their support in patient care. We express our regard to the Terry Fox Foundation for funding support.
Funding
Terry Fox Foundation Program Project Grant from the Hecht Foundation (Grant Number 1083). The funding agencies had no role in the study design, study methodology, study results, or in the preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Study concept: L.O., A.D. and G.J.C.; Acquisition of data, interpretation of data, writing-original draft and revision, writing-final approval: All authors; Project administration, funding acquisition: G.J.C.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Osapoetra, L.O., Dasgupta, A., DiCenzo, D. et al. Assessment of clinical radiosensitivity in patients with head-neck squamous cell carcinoma from pre-treatment quantitative ultrasound radiomics. Sci Rep 11, 6117 (2021). https://doi.org/10.1038/s41598-021-85221-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-85221-6
This article is cited by
-
Advances in personalized radiotherapy
BMC Cancer (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.