Abstract
Autologous reconstruction using abdominal flaps remains the most popular method for breast reconstruction worldwide. We aimed to evaluate a prediction model using machine-learning methods and to determine which factors increase abdominal flap donor site complications with logistic regression. We evaluated the predictive ability of different machine learning packages, reviewing a cohort of breast reconstruction patients who underwent abdominal flaps. We analyzed 13 treatment variables for effects on the abdominal donor site complication rates. To overcome data imbalances, random over sampling example (ROSE) method was used. Data were divided into training and testing sets. Prediction accuracy, sensitivity, specificity, and predictive power (AUC) were measured by applying neuralnet, nnet, and RSNNS machine learning packages. A total of 568 patients were analyzed. The supervised learning package that performed the most effective prediction was neuralnet. Factors that significantly affected donor-related complication was size of the fascial defect, history of diabetes, muscle sparing type, and presence or absence of adjuvant chemotherapy. The risk cutoff value for fascial defect was 37.5 cm2. High-risk group complication rates analyzed by statistical method were significant compared to the low-risk group (26% vs 1.7%). These results may help surgeons to achieve better surgical outcomes and reduce postoperative burden.
Similar content being viewed by others
Introduction
Ever since Hartrampf first introduced breast reconstruction using transverse abdominal flaps to the world in 19821, abdominal flap breast reconstruction including transverse rectus abdominis muscle (TRAM) flap and deep inferior epigastric artery perforator (DIEP) flap has become a mainstay technique among autologous breast reconstruction surgeons, and has become one of the most popular methods worldwide2,3. However, as with all autologous reconstruction methods, morbidity after abdominal flap surgery cannot be avoided. One of the most troublesome complications related to the donor site would be postoperative weakness of the abdominal wall and rectus muscles caused by damage to the deep fascia and the rectus muscles themselves during flap harvesting4,5,6,7. These complications greatly affect the patient's quality of life after surgery8.
Techniques for elevation of the abdominal flap have evolved to reduce morbidity of the donor site as much as possible. Examples include adjustments to the classic TRAM flap, muscle sparing TRAM flap, and DIEP flap5. Each technique still maintains unique risks however, such as in the case of DIEP elevated with a single perforator, where the risk of fat necrosis is high when a large flap volume is harvested9,10. In attempts to mediate this risk, surgeons try to harvest more perforator vessels, but this in turn leads to a larger fascial defect. There have been numerous studies regarding the risks of abdominal complication and defect closure in abdominal flap surgeries that state conflicting conclusions4,5,6,7,11,12.
In addition, since complication rates of the donor part after operation are not very high, and typical symptoms, such as abdominal bulging or hernia diagnosed mainly through subjective complaints of patients, a large patient cohort study with follow-up through unified standards and methods is difficult to establish. Also, even if such a study were possible, the data may be severely imbalanced due to the low prevalence rates of individual complication types. It would thus, be difficult to evaluate multiple factors regarding flap elevation and the wound closure process at the donor site.
Machine learning can augment existing statistical processing methods to provide a more accurate and efficient prediction through data obtained in real life, including various clinical situations13. Machine learning is a subfield of artificial intelligence in computer science that has evolved from the study of pattern recognition and computer learning theory. It is a technology that studies and builds a system that learns based on empirical data, performs predictions, and consistently improves on its own performance by modifying its algorithms. Rather than executing strictly defined static program instructions, machine learning algorithms build internal models to derive predictions or decisions based on input data. In the process of establishing a machine learning prediction model in creating the algorithm, it is important to sample a wide variance of data within a range that does not harm overall data integrity14,15,16,17. Using machine learning, we aimed to evaluate factors responsible for donor-related complication, and to evaluate the odds ratio between high versus low risk cohorts to provide more accurate and detailed information for patients in the process of planning, performing, and following up cases of TRAM flap surgeries.
Methods
Patients
A retrospective cohort study was conducted for patients who underwent muscle-sparing type TRAM and DIEP (muscle sparing type 3) flaps for immediate or delayed unilateral breast reconstruction at Seoul National University Bundang Hospital from 2006 through 2019. All study participants provided informed consent and the study design was approved by the institutional review board.
Data collection
A comprehensive review was conducted on electronic medical records, clinical data warehouse systems of our institution, and radiographic imaging studies. A total of 13 factors related to muscle sparing TRAM flap elevation and donor closure were investigated including: age, body mass index, smoking status, prediagnosed diabetes mellitus or hypertension, history of open surgery, history of abdominal laparoscopy or endoscopic surgery, preoperative chemotherapy, elevated flap weight, muscle sparing type, size of fascia defect after flap elevation, and closure pattern of fascia during donor closure (primary closure, nonabsorbable mesh assisted closure, acellular dermal matrix assisted closure). If the patient complained of abdominal bulging or hernia during the outpatient follow-up process, a complication was present, regardless of the severity of symptoms or corrective surgery.
Statistical methods
For this study, the R program version 3.6.3 (The R Foundation for Statistical Computing, Vienna, Austria) was used. Installed and used packages were: mlbench, moonBook, random over sampling example (ROSE), rpart, neuralnet, Metrics, pROC, ROCR, nnet, devtools, and RSNNS. Logistic regression was performed to investigate the effect of each factor on donor-related complications. Risk factors affecting abdominal complication were investigated among 15 variables by logistic regression, and such selection was determined according to statistical significance. Missing values were excluded. Statistical significance was set at p < 0.05. Goodness of fit was evaluated through the Hosmer–Lemeshow test. AUC (area under curve) was measured through the ROC (receiver operating characteristic) curve.
ROSE: sMOTE oversampling
As the imbalance of clinical data in present study was severe due to an extremely low complication rate, ROSE oversampling was performed using a method previously published17. In cases of applying machine learning to real world data, there are several ways to solve for class imbalances, defined as an overly large discrepancy weighted towards one class. There are methods such as class weight, which gives heavier cost to minority classes; down-sampling, which subtracts randomly from the majority class; and up-sampling, which randomly replicates minority classes. The ROSE package used by the authors is a synthetic minority sampling technique that interpolates from minority classes while down sampling the majority class18.
Neuralnet
The neuralnet package has very flexible functions for training feed-forward neural networks19. To approximate the functional relationship, this package can theoretically process covariates and response variables as well as any number of hidden layers and hidden neurons. Although computational cost can increase exponentially with complexity, there is also an option to stop the iteration at any point. The maximum number of iteration steps can be defined by the user and is reached before the algorithm converges. In addition, the package provides functions to visualize results, and generally includes several functions to facilitate the use of neural networks. Total of six neurons were utilized repeatedly, with resilient backpropagation with backtracking. Error function was sum of squared errors and logistic function with threshold of 0.08 was applied. Stepmax was set at 1e6, as the result yielded better results than 1e5.
NNET
The nnet package is also a widely used neural network package because both categorical data and continuous variables can be classified simply without any additional work20. Like neuralnet, it works using a single thread and is a relatively simple package by recent standards without any significant GPU requirements. Furthermore, although the basic functional process itself is simple, adding more hidden layers does not significantly lead to increased additional computational cost or GPU usage frequency. One hidden layer was applied, with two different set of bias progression. The number of total hidden nodes (size) was set at 6, repeating maxit was set at 500, decay weight parameter was applied in order to prevent overfitting of the predictive results. Different subsets were recategorized in order to yield final sensitivity and specificity, by creating total of four different subsets.
RSNNS: neural networks using the stuttgart neural network simulator (SNNS)
SNNS is a library, containing many standard implementations of neural networks. It provides several different neural network and visualization functions, including multi-layer perceptrons (MLP) capabilities21. The RSNNS package wraps SNNS functionality to make it available from within the R program. Using the RSNNS low-level interface, the algorithmic functionality and flexibility of SNNS can be accessed. Furthermore, the package contains a convenient high-level interface, so that the most common neural network topologies and learning algorithms integrate seamlessly into the R program. Total of 500 train data and 68 test data was utilized, additionally categorized into input and output datasets in order to perform multi-layer perceptron function. Total learning number (maxit) was set to 1000 in order to achieve more predictive power.
Risk cutoff point
By separating factors identified as a direct cause of donor-related complication risk, the optimal reference point representing the risk of complication risk was searched through the ROC curve through the Youden index method, and data was classified above and below the cutoff point22. Following the classification, the risk assessment was conducted separately by dividing the high-risk group and the low-risk group.
Results
A total of 568 patients who underwent muscle sparing TRAM operation for breast reconstruction were enrolled in this study. Patient demographics and variables were summarized in Table 1. Mean follow up period was 68 months. Among the patients, a total of 37 (6.5%) patients complained of abdominal bulging and confirmed with physical examination by the surgeon who operated on, of which 12 (2.1%) were radiographically diagnosed abdominal hernia, and 7 (1.2%) required hernia repair surgery.
Among the different treatment variables analyzed through multivariable logistic regression, the factors that significantly affected occurrences of abdominal complication were: large fascial defect size (OR 1.62), muscle sparing type 0 (OR 1.35), diabetes mellitus (OR 1.71), and adjuvant chemotherapy (OR 2.1).
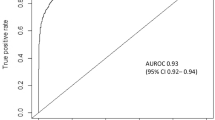
After ROSE oversampling, the AUC obtained through the test run was 0.89 (Fig. 1). Training and testing sets were divided in an 80:20 ratio, with each training set pre-processed through sMOTE oversampling. The prediction score, sensitivity, specificity, and AUC as measured by the neuralnet, nnet, and RSNNS packages were summarized in Table 2. Among the three packages, the prediction accuracy of the neuralnet package was the best at 81%, and the model accuracy was also the highest (Fig. 2). The prediction rate was the best in the neuralnet prediction function, when the hidden layers were set to 3 layers (Fig. 3) and the reverse propaganda algorithm was used.
(Left) ROC (receiver operating characteristics) curve of neuralnet prediction package. AUC was 0.79. (Right) ROC (receiver operating characteristics) curve of nnet prediction package. AUC was 0.75. R program version 3.6.3 (The R Foundation for Statistical Computing, Vienna, Austria) was used in creating diagram.
Plot diagram of highest predictive neuralnet function (left) and the result (right). There were six hidden neurons each involved in three hidden layers, resilient backpropagation with backtracking algorithm, sum of squared errors for error calculation, and logistic function for smoothing the result. Maximum steps were set at 106. R program version 3.6.3 (The R Foundation for Statistical Computing, Vienna, Austria) was used in creating diagram.
The cutoff point for maximal sensitivity and specificity regarding complication risk for fascial defect size was 37.5 cm2 (Fig. 4). The predicted probability of abdominal complication between the high- and low-risk group was calculated using the highest prediction function of neuralnet package. The high-risk group was designated as: having a fascial defect size of 37.5 cm2 or more, having chemotherapy, have history of diabetes mellitus, and performing muscle sparing type 0 procedure. The probability of abdominal complication risk for these patients was 26%. In contrast the low-risk group had: a fascial defect size less than 37.5 cm2, no adjuvant chemotherapy, without diabetes, and muscle sparing type 3 surgery, with a probability of 1.7% for abdominal complications. Additionally, different probabilities of abdominal complications in each situation based on risk factors were categorized (Fig. 5).
Discussion
This study is the first, to our knowledge, to implement a prediction model utilizing machine learning methods for the risk assessment of complications in breast reconstruction with abdominal free flap. The authors applied an efficient prediction model using a neural network technique. Though complications with abdominal free flaps are rare and studies on them are relatively easy to neglect, once it occurs, these donor-related complications give patients and surgeons significant clinical burden23,24,25. In this study, we aimed to present accurate information for more effective risk assessment by patients and doctors.
Breast reconstruction with abdominal flap has evolved from initial methods of harvesting whole muscle, to muscle sparing and DIEA perforator-based flap. Efforts have been made to minimize the amount of tissue harvested from the donor site to reduce postoperative functional deficit and to reduce donor site related complications. Donor-related morbidity is known to be related to the amount of tissue that is harvested from the abdomen during flap elevation5. However, even in muscle sparing or DIEP flap elevation procedures, too many perforators can be harvested, leading to abdominal fascia and rectus muscle disruptions, especially in cases when a larger flap volume is needed or when bilateral breast reconstruction is performed. Harvesting perforators from both medial and lateral rows to reduce fat necrosis was shown to lead to more fascial defect10. Techniques using single perforator or single row perforator have their own drawbacks as it was shown that the frequency of fat necrosis was high with long-term flap volume decrease after these surgeries10,26,27.
This study revealed that prediction accuracy can be efficiently maintained (AUC 0.89) by sMOTE oversampling using the ROSE package of the R program, overcoming severe data imbalances due to low complication rates. Also, although the patient cohort size that was used in this study was not comparably large, the integrity of data after oversampling was high enough to establish neural network models. Among the three neural network packages utilized, neuralnet package outperformed the other two in every category including prediction scores, AUC of ROC curve, sensitivity, and specificity. An impressive prediction accuracy could be obtained (81%) through neuralnet function when setting three different hidden layers and binary classification output. Lastly, use of parallelized capabilities of multi-layer perception was assumed to fail to overperform compared to other two packages. The RSNNS package was the weakest in performance and adding more hidden layers in the settings did not result in better prediction scores (Fig. 5).
The lesson to be learned from this study is that important factors that significantly increase risk for donor-related complications in the muscle sparing tram flap were: defect size at flap elevation, adjuvant chemotherapy, history of diabetes mellitus, and muscle sparing technique type. Notably, the statistical cutoff point for defect size that significantly increases the complication risk is 37.5 cm2. Of course, it is better to minimize defects in all surgeries, but when performing surgery in the real-world practice, the position of the lateral and medial row perforators is different for each patient and for each race. Further, the width of the rectus muscle may differ, and the defect size is dependent on the volume of the flap being elevated. This data provides information that can be used as an effective reference for accurately discriminating the risk before and after surgery up to the follow-up period for each patient.
RSNNS (R Using Stuttgart Neural Network Simulator) performed lower than classical machine learning packages, neuralnet and NNET. The main strength of RSNNS package is that it has various neural network and visualization function with unlimited ability to increase perceptron layers in utilizing variable datasets (Fig. 6). However, in multi-layered perceptron, hidden layer between input and output layer can not define the standard value of hidden layer output, without backpropagation that was utilized in both neuralnet and NNET packages. The feedforward neural network is assumed to underperform under prefabricated data with ROSE oversampling. However, different training sets with various implementation details should be utilized in order to maintain the utmost method in processing oversampled data, such as abdominal complication after autologous breast reconstruction.
Plot diagram of RSNNS function, with five hidden neurons (left) and nine hidden neurons (right). Different options were tested with RSNNS package, however failed to give satisfying prediction values. R program version 3.6.3 (The R Foundation for Statistical Computing, Vienna, Austria) was used in creating diagram.
In addition, this study was the first in this field to create a most accurate deep learning prediction model, which was analyzed and validated by using imbalanced data, corrected through sMOTE oversampling, and utilizing three representative packages among neural network machine learning algorithms. Due to the nature of machine learning prediction models, the results may vary depending on the quality and quantity of the data, but by defining the method and package settings used to obtain the most efficient and high AUC when using a prediction model with limited and imbalanced data like in the reconstructive surgical field, these models can be used and replicated effectively for risk assessment of patient outcomes in free flap operation as well as other reconstructive surgical procedures.
The authors note that this study had several limitations encountered. First, it was difficult to specifically define abdominal donor-related complication as there are no exact diagnostic criteria or standards, and among patients with symptoms, not all patients underwent further imaging workup or had hernia repair operation during their variable follow up time span. The most important diagnostic finding to confirm presence of complication was bulging of the surgical site when the patient was in the upright position. Since the patient's symptomatic complaint and the physical examination of the surgeon were the most accurate diagnostic measure, we used these to determine occurrence of donor-related complications for this retrospective review. However, in all cases where the patient complained of having abdominal hernia or bulging, the responsible surgeon always performed physical examination to confirm it, and majority of the cases were radiographically confirmed with abdominal CT or sonography. In addition, the cases that were primarily diagnosed by the surgeon during follow-up period without primary complaints from the patients were also included.
In addition, the possibility that there are additional risk factors, other than the 15 factors investigated in this study, cannot be excluded. According to previous studies, factors other than those analyzed in this study may be responsible for abdominal donor-related complication inducing: harvested perforator vessel number and denervation of intercostal nerve4,5,6,7,11,12. There were, likewise, several other factors that could have been considered, such as: how long the surgery took, whether there was a flap-related complication after surgery, the extent of blood loss during surgery, and the duration of hospitalization. To facilitate efficient operation of the risk prediction model through supervised learning however, we chose a method of organizing the data by narrowing it down to several more likely factors. In addition, in the process of simplifying data, clustering and normalization of data was performed and data mining was unified for various factors as a result, such as: more specific types/sizes of mesh used for fascia closure and data on the manufacturer or the size of acellular dermal matrix that was used for fascia closure. Although this data was clustered, it is impossible to completely rule out the possibility that each subfactor could be independently responsible for various consequences in the patient's recovery process after surgery. For these limitations, it is necessary to analyze individual factors more effectively through further studies on risk assessment.
We admit that there are inherent limitations to this type of research, and that various efforts have been involved to find effective triggers through many previous studies. Many studies on how to select key variables that affect final prediction outcome in order to execute machine learning studies have been conducted using existing classical statistical methods, not ML packages, or by simply referring to previous references17,28,29. However, our study acknowledged such limitations and focused on providing more efficient information by applying the advanced data processing model to patients and physicians in the treatment process before and after the surgery. Logistic multivariate regression in fact is also a kind of statistical prediction model. It was used to screen and select which factor is responsible in affecting the final outcome. If every clinical variable could have been objectively investigated and analyzed accurately and completely, it would have been a perfect thesis. However, the technical difficulties regarding data processing remains, and the authors, to our knowledge, chose the most efficient way in processing the clinical data regarding such a complicated, multi-factorial, overfitted and imbalanced breast reconstruction outcome data.
Conclusion
To create a more accurate and efficient deep learning-based prediction model of donor-related complications in autologous breast reconstruction, we overcame various limitations inherent to unequal data, and validated the predictive accuracy between three representative neural network packages. This method of risk assessment for rare, but troublesome complications through a machine learning approach, performed by the authors for the first time, can potentially be replicated and used for studying complications in other procedures. Using this model, defect size at flap elevation, adjuvant chemotherapy, and muscle sparing technique type were found to be factors that significantly increase risks for post-operative complications. These results may help surgeons to achieve better surgical outcomes and reduce postoperative burden (Supplementary information S1 and S2).
References
Hartrampf, C. R., Scheflan, M. & Black, P. W. Breast reconstruction with a transverse abdominal island flap. Plast. Reconstr. Surg. 69(2), 216–225 (1982).
Song, W. J. et al. Current status of and trends in post-mastectomy breast reconstruction in Korea. Arch. Plast. Surg. 47(2), 118–125 (2020).
Lemaine, V. et al. Autologous breast reconstruction versus implant-based reconstruction: How do long-term costs and health care use compare?. Plast. Reconstr. Surg. 145(2), 303–311 (2020).
Chang, E. I. et al. Comprehensive analysis of donor-site morbidity in abdominally based free flap breast reconstruction. Plast. Reconstr. Surg. 132(6), 1383–1391 (2013).
Egeberg, A., Rasmussen, M. K. & Sørensen, J. A. Comparing the donor-site morbidity using DIEP, SIEA or MS-TRAM flaps for breast reconstructive surgery: A meta-analysis. J. Plast. Reconstr. Aesth. Surg. 65(11), 1474–1480 (2012).
Paik, J. M. et al. Donor site morbidity following DIEP flap for breast reconstruction in Asian patients: Is it different?. Microsurgery 35(8), 596–602 (2015).
Schaverien, M. V., Perks, A. & McCulley, S. Comparison of outcomes and donor-site morbidity in unilateral free TRAM versus DIEP flap breast reconstruction. J. Plast. Reconstr. Aesth. Surg. 60(11), 1219–1224 (2007).
Ochoa, O. et al. Prospective longitudinal patient-reported satisfaction and health-related quality of life following DIEP flap breast reconstruction: Relationship with body mass index. Plast. Reconstr. Surg. 143(6), 1589–1600 (2019).
Lie, K. H., Barker, A. S. & Ashton, M. W. A classification system for partial and complete DIEP flap necrosis based on a review of 17,096 DIEP flaps in 693 articles including analysis of 152 total flap failures. Plast. Reconstr. Surg. 132(6), 1401–1408 (2013).
Mulvey, C. L. et al. Increased flap weight and decreased perforator number predict fat necrosis in DIEP breast reconstruction. Plast. Reconstr. Surg. Glob. Open. 1(2), 1–7 (2013).
Salgarello, M., Tambasco, D. & Farallo, E. DIEP flap donor site versus elective abdominoplasty short-term complication rates: A meta-analysis. Aesth. Plast. Surg. 36(2), 363–369 (2012).
Ferguson, R. E. Jr. & Pu, L. L. Repair of the abdominal donor-site fascial defect with small intestinal submucosa (Surgisis) after TRAM flap breast reconstruction. Ann. Plast. Surg. 58(1), 95–98 (2007).
Alpaydin, E. Introduction to machine learning. 2020: MIT press.
Kanevsky, J. et al. Big data and machine learning in plastic surgery: A new frontier in surgical innovation. Plast. Reconstr. Surg. 137(5), 890e–897e (2016).
Knoops, P. G. et al. A machine learning framework for automated diagnosis and computer-assisted planning in plastic and reconstructive surgery. Sci. Rep. 9(1), 13597. https://doi.org/10.1038/s41598-019-49506-1 (2019).
Mantelakis, A. & Khajuria, A. The applications of machine learning in plastic and reconstructive surgery: Protocol of a systematic review. Syst. Rev. 49(1), 44. https://doi.org/10.1186/s13643-020-01304-x (2020).
O’Neill, A. C. et al. Development and evaluation of a machine learning prediction model for flap failure in microvascular breast reconstruction. Ann. Surg. Oncol. 27(9), 3466–3475 (2020).
Lunardon, N., Menardi, G. & Torelli, N. ROSE: A Package for Binary Imbalanced Learning. R Journal. 6(1), 79–89 (2014).
Günther, F. & Fritsch, S. neuralnet: Training of Neural Networks. R J. 2(1), 30–38 (2010).
Ripley, B., Venables, W. & Ripley, M. B. Package ‘nnet’. R package version. 7, 3–12 (2016).
Bergmeir, C., & Benítez, J. M. Neural networks in R using the Stuttgart neural network simulator: RSNNS. J. Stat. Softw. 46(7). https://www.jstatsoft.org/article/view/v046i07 (2012).
Fluss, R., Faraggi, D. & Reiser, B. Estimation of the Youden Index and its associated cutoff point. Biom J. 47(4), 458–472 (2005).
Damen, T. H. et al. Improving outcomes in microsurgical breast reconstruction: lessons learnt from 406 consecutive DIEP/TRAM flaps performed by a single surgeon. J. Plast. Reconstr. Aesth. Surg. 66(8), 1032–1038 (2013).
Kind, G. M., Rademaker, A. W. & Mustoe, T. A. Abdominal-wall recovery following TRAM flap: a functional outcome study. Plast. Reconstr. Surg. 99(2), 417–428 (1997).
Nahabedian, M. Y., Tsangaris, T. & Momen, B. Breast reconstruction with the DIEP flap or the muscle-sparing (MS-2) free TRAM flap: is there a difference?. Plast. Reconstr. Surg. 115(2), 436–444 (2005).
Grover, R. et al. The impact of perforator number on deep inferior epigastric perforator flap breast reconstruction. Arch. Plas. Surg. 41(1), 63–70 (2014).
Uda, H. et al. Comparison of abdominal wall morbidity between medial and lateral row-based deep inferior epigastric perforator flap. J. Plast. Reconstr. Aesth. Surg. 68(11), 1550–1555 (2015).
Jo, C. et al. Transfusion after total knee arthroplasty can be predicted using the machine learning algorithm. Arthroscopy 28(6), 1757–1764 (2020).
Panesar, S. et al. Machine learning versus logistic regression methods for 2-year mortality prognostication in a small, heterogeneous glioma database. World Neurosurg. X. 2:100012
Acknowledgements
This study was approved by institutional review board of Seoul National University (IRB number B2007-624-101) that follows the tenets set by the declaration of Helsinki. Patient data was anonymized to protect confidentiality. This work was supported by Grant no. 09-2020-002 from the Seoul National University Bundang Hospital Research Fund. The authors wish to thank Prof. Edward Choi of Graduate School of Artificial Intelligence, Korean Advanced Institute of Science and Technology for his helpful comments and discussions.
Author information
Authors and Affiliations
Contributions
Y.M.: study design, data collection, statistical analysis, interpretation, and initial manuscript writing. S.J.: data collection and bibliography review. C.H.: data collection, critical review, manuscript revision. E.K., E.K., H.S.: data collection and critical review. E.Y.: data collection, interpretation, and critical review. J.J.H.: study design, statistical analysis, interpretation, manuscript revision. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Myung, Y., Jeon, S., Heo, C. et al. Validating machine learning approaches for prediction of donor related complication in microsurgical breast reconstruction: a retrospective cohort study. Sci Rep 11, 5615 (2021). https://doi.org/10.1038/s41598-021-85155-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-85155-z
This article is cited by
-
The usefulness of artificial intelligence in breast reconstruction: a systematic review
Breast Cancer (2024)
-
Feasibility of anomaly score detected with deep learning in irradiated breast cancer patients with reconstruction
npj Digital Medicine (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.