Abstract
The identification of factors predisposing to severe COVID-19 in young adults remains partially characterized. Low birth weight (LBW) alters cardiovascular and lung development and predisposes to adult disease. We hypothesized that LBW is a risk factor for severe COVID-19 in non-elderly subjects. We analyzed a prospective cohort of 397 patients (18–70 years) with laboratory-confirmed SARS-CoV-2 infection attended in a tertiary hospital, where 15% required admission to Intensive Care Unit (ICU). Perinatal and current potentially predictive variables were obtained from all patients and LBW was defined as birth weight ≤ 2.500 g. Age (adjusted OR (aOR) 1.04 [1–1.07], P = 0.012), male sex (aOR 3.39 [1.72–6.67], P < 0.001), hypertension (aOR 3.37 [1.69–6.72], P = 0.001), and LBW (aOR 3.61 [1.55–8.43], P = 0.003) independently predicted admission to ICU. The area under the receiver-operating characteristics curve (AUC) of this model was 0.79 [95% CI, 0.74–0.85], with positive and negative predictive values of 29.1% and 97.6% respectively. Results were reproduced in an independent cohort, from a web-based survey in 1822 subjects who self-reported laboratory-positive SARS-CoV-2 infection, where 46 patients (2.5%) needed ICU admission (AUC 0.74 [95% CI 0.68–0.81]). LBW seems to be an independent risk factor for severe COVID-19 in non-elderly adults and might improve the performance of risk stratification algorithms.
Similar content being viewed by others
Introduction
COVID-19 is a mild or asymptomatic condition in the majority of patients, but in up to 1–2% it may result in severe disease and death1,2. Older age, male sex and coexisting conditions are the main risk factors described so far for severe COVID-19 disease3,4,5,6,7,8. However, a small proportion of young and apparently healthy adults may eventually require critical care. There is a need for comprehensive models that identify factors associated to the risk of severe forms of COVID-199.
The association between low birth weight (LBW) and adult health has long been recognized10,11,12. LBW, defined as ≤ 2500 g13,14, can result from fetal growth restriction, prematurity or both13. Fetal growth restriction has been associated with increased cardiovascular mortality10,15, lower lung functional capacity16,17,18 and increased respiratory morbidity18,19 in adulthood. Likewise, prematurity has been described as a risk factor for suboptimal cardiovascular20 and lung21 development and a greater predisposition to heart failure22 and lung disease23 later in life. For studies in adults, birth weight is an accessible and robust surrogate for fetal growth restriction and preterm births, and a strong predictor of short and long-term morbidity24.
From the above observations, we hypothesized that LBW could increase the risk of developing severe illness in non-elderly adults with COVID-19. To test this hypothesis, we designed a prospective study in confirmed COVID-19 patients (18–70 years) admitted to our institution, a public, tertiary, referral, university hospital in Spain (development dataset) and validated the model in an independent cohort of self-reported laboratory-confirmed COVID-19 subjects recruited through a web-based survey (validation dataset).
Results
Development dataset
Figure 1 (left panel) presents the CONSORT diagram of the testing cohort. Out of 516 potentially eligible patients (with laboratory-confirmed SARS-CoV-2 infection by real time polymerase chain reaction (RT-PCR) of nasopharyngeal swab samples) during the study period, the development cohort included 397 patients with available perinatal information (77%). Based on clinical assessment of severity, 98 patients (24.7%) followed home hospitalization care and 299 (75.3%) were hospitalized, 60 of whom (20%) were eventually taken care of in the Intensive Care Unit (ICU) (25 (42%) required mechanical ventilation and none died). Table 1 displays the characteristics of the 337 non-critically ill patients (home hospitalization (n = 98) or in hospital (n = 239)) with those treated in the ICU (n = 60). The latter were older, more frequently males, had a higher body mass index (BMI) and a higher prevalence of hypertension. Of note, they were also born with LBW (18.3 vs. 9.5%, p = 0.041) and suffered fetal growth restriction (25 vs. 14.8%, p = 0.043) more often. Individuals born with LBW had a higher probability for ICU admission as compared with those with normal birth weight (Fig. 2).
Table 2 displays crude and adjusted Odds Ratio (aOR) for ICU admission. In the multivariate model, age (aOR 1.04 [1–1.07], P = 0.012), male sex (aOR 3.3 [1.72–6.67], P < 0.001), hypertension (aOR 3.4 [1.69–6.72], P = 0.001), and LBW (aOR 3.61 [1.55–8.43], P = 0.003) remained independent predictors of ICU. As shown in Fig. 3 (left), the area under the receiver-operating characteristics curve (AUC) for predicting ICU admission was 0.79 (95% CI, 0.74–0.85). The model had a sensitivity of 91.7%, specificity of 60.2%, positive predictive value (PPV) of 29.1% and negative predictive value (NPV) of 97.6% (Table 3).
Validation dataset
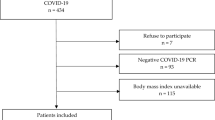
Figure 1 (right panel) presents the CONSORT diagram of the validation dataset. We received 9320 responses of subjects aged 18 to 70 years who referred symptoms suggestive of COVID-19. Among them, 1822 self-reported COVID-19 confirmed by RT-PCR. A total of 1215 of them (67%) reported mild symptoms and did not require hospital admission whereas 607 (33%) were hospitalized of whom 46 (8%) patients required ICU admission (30 of them (65% of ICU patients) were mechanically ventilated, and one male patient (2% of ICU patients) died at the age 68 years as reported later by her daughter.
Table 4 shows the characteristics of the 1776 non-critically ill patients (treated at home (n = 1215) or in hospital (n = 561)) with those treated in the ICU (n = 46). Like we observed in the developing cohort, ICU patients in the validation dataset were older, more frequently males, had a higher BMI and a higher prevalence of hypertension. Importantly, again, they were born with LBW (19.6 vs. 7.3%, p = 0.006) and suffered fetal growth restriction (26.1 vs. 12.4%, p = 0.010) more often. In this validation dataset, the prevalence of prematurity was also higher in ICU patients (23.9 vs. 10.9%, p = 0.011).
The model obtained in development dataset was applied to the validation dataset, obtaining an AUC of 0.74 (95% CI 0.68–0.81) (Fig. 2, right panel), with a sensitivity of 73.9%, specificity of 67.3%, PPV of 5.5% and NPV of 99% (Table 3).
Discussion
This study provides evidence that recording birth weight might improve the prognostic stratification of COVID-19 in non-elder patients. Most young patients present mild forms of COVID-19, but a small proportion might require admission to ICU for severe complications3,4,5,6,7,8, which is clearly associated to non-obvious predisposing factors. Early interventions in COVID-19 have demonstrated to reduce mortality4,6. Consequently, the identification of predisposing factors –particularly in a priori non high-risk subjects- might allow early therapeutic measures eventually preventing serious evolution to serious illness. In this study we evaluated an innovative approach by studying early life risk factors not usually taken into account in current clinical practices. Birth weight is one of the most universally recorded information for any given individual and self-recalled birth weight has demonstrated to be a reliable information, particularly in subjects born after the 1960s41. If confirmed in future studies that LBW identifies high risk for complicated COVID-19, this should be included in initial assessment of non-elder infected subjects and would offer opportunities for early interventions to prevent complications.
Previous studies
Despite the large number of studies on prognostic factors for severe COVID-193,4,5,6,7,8,25,26,27,28,29,30,31,32,33,34,35,36, to our knowledge no previous study has investigated the predictive role of early life events as a risk factor for severe COVID-19 in adulthood. Results confirmed our working hypothesis, which was aligned with a long-standing research line in this field11,12,18,19,37. Besides, results confirmed previous studies showing a strong predictive value for severe COVID-19 of older age, male sex and coexisting conditions such as hypertension3,4,5,6,7,8,25,26,27,28,29,30,31. The fact that we studied non-elderly adults (≤ 70 years) may have limited the identification of significant associations with other reported coexisting conditions such as chronic lung disease2,6,7,8,27, diabetes2,3,4,5,7,8,25,26,30,32, obesity8,31,33 or cancer3,34. Current or previous smoking status35 and chronic treatment with ACE inhibitors36 were not associated with COVID-19 severity in our dataset. We acknowledge that other unstudied confounders may have interfered our results.
Interpretation of novel findings
Our results suggest that LBW is an independent risk factor for severe COVID-19 in adulthood. This finding is consistent with previous epidemiological and experimental studies supporting the developmental origin of adult diseases. Adverse in utero environment induces permanent changes in the structure, function and metabolism of the developing fetal organs. Most developmental changes of early life persist in the long term which leads to a greater risk of disease in adulthood10,11. It is suggested that fetal adaptation to perinatal events represents a ‘first hit’ leading to latent susceptibility, which combined with a ‘second hit’ later in life could increase the risk for adult diseases10,11. This notion has been consistently demonstrated in experimental research38, but evidence in humans is limited. The COVID-19 pandemic represents a unique opportunity to study the response of a significant number of individuals born LBW to a specific and well-defined stressor.
LBW has been consistently associated with increased adult cardiovascular mortality, hypertension, metabolic syndrome, diabetes and lung morbidity10,11,12,15,16,17,18,19,20,21. LBW can be a result of being born too small—fetal growth restriction- and/or too early—prematurity. Fetal growth restriction is caused by placental insufficiency leading to a sustained reduction in fetal oxygen and nutrient supply11. This triggers an adaptive fetal response including cardiovascular remodeling12,39, increased blood pressure12, altered lipoprotein profile, lost of nephrons, and disturbed pulmonary alveolarization and vascular growth11. In prematurity, key developmental stages have to take place ex utero in non-physiological conditions40 leading to cardiovascular hypertrophy and impaired lung development, insulin sensitivity and bone density20,21,22,40.
Strengths and limitations
This study has some strengths and limitations that merit comment. Among the strengths, we prospectively collected information spanning the full COVID-19 clinical spectrum, from mild to hospitalized and ICU patients. Likewise, we validated our observations in an independent dataset. Finally, we included only non-elderly subjects (< 70 years) to avoid the potential confounding effect of age-related comorbidities. The study sample size was too small to assess the predictive value of LBW across age ranges. We acknowledge that the evidence here presented should be validated in another prospective hospital cohort. We opted for an online survey to shorten validation time. We acknowledge also a potential selection bias since there were virtually no deaths in our study population. Firstly, mortality rate for COVID-19 was very low in our hospital (8%, 194/2425) with most cases occurring in subjects > 70 years-old. Secondly, we tried to contact all COVID-19 patients identified in the EMRs, but a few very severe cases were directly intubated and died preventing the interview for the study. In addition, we acknowledge the potential inaccuracy of the perinatal data obtained by interview or online survey. However, self-reported birth weight has demonstrated to be a good surrogate of adverse in utero environment, particularly in non-elderly subjects41. Although the prevalence of LBW in our study populations lie within the range reported by recent international and national estimates42, we acknowledge that a selection bias cannot be fully discarded. Finally, we acknowledge that future studies might unveil non-identified confounders not considered in the design of this study that might have affected the present results.
Conclusions
Our data suggest that low birth weight increases the risk of severe COVID-19 in non-elderly adults. This new information further supports the importance of early life events in adult diseases. If confirmed in future studies, LBW should be considered in frisk stratification algorithms for COVID-19.
Methods
Development dataset
Study design, population and ethics
Prospective observational cohort that included non-elderly adults (aged 18 to 70 years) consecutively attended at Hospital Clínic of Barcelona from March 25 to April 25, 2020 with laboratory-confirmed SARS-CoV-2 infection by real time polymerase chain reaction (RT-PCR) of nasopharyngeal swab samples. Sample size was determined by the time window of opportunity of the study. Criteria for hospital admission (COVID-19 pneumonia) and therapeutic management while in hospital followed the in-house protocols. The primary outcome of the study was admission to the ICU, which was determined by the attending physician on a patient by patient basis following standard clinical assessment criteria43. Follow-up time was censored on May 25, 2020 so that each patient had at least 30 days of observation. The study was approved by the Ethical Committee of Hospital Clínic (HCB/2020/0353) and written informed consent was obtained from all patients. All research was performed in accordance with Regulation EU 2016/679 of the European Parliament and of the Council of April 27, 2016 on the protection of individuals.
Data collection
Cases were identified by daily review of hospital attendance logs in electronic medical records. Likewise, demographics, smoking exposure, coexisting conditions, treatment received during the last two weeks before hospitalization, need for ICU admission, complications or death during the clinical course, and therapeutic management interventions were obtained by reviewing electronic medical records. On the other hand, perinatal (birth weight and gestational age at delivery) and childhood (“asthma” or other respiratory disease in childhood) data were obtained by a face-to-face or telephone interview. Birth weight centiles were calculated adjusted by gender and gestational age at birth according to local standards44. LBW was defined as birth weight equal or below 2500 g14. Fetal growth restriction was defined as a birth weight below the 10th centile for gestational age45 and preterm delivery as born before 37 weeks of gestation46.
Validation dataset
Study design, setting and population
To validate externally the performance of the prognostic algorithm created by the development cohort, we collected independent data from self-selected volunteers who declared laboratory-confirmed SARS-CoV-2 infection through an anonymous multilingual (Spanish, Catalan, Italian, English and French) online survey (Limey Survey GmbH, Germany). The survey was disseminated via email and social media, and it was open and free for all subjects who self-reported to have laboratory confirmed COVID-19. Demographic information, coexisting conditions, perinatal and childhood data, COVID-19 related symptoms and need for hospitalization or admission to ICU were collected (voluntary sampling) using an anonymous web-based cross-sectional survey from April 1 to May 31, 2020.
Statistical analysis
Results are presented as counts (percentage) or mean (SD) as appropriate. Our aim was to determine whether being born LBW increases susceptibility to severe COVID-19 in non-elderly adults. Our main outcome measure was admission to ICU for COVID-19. Our main exposure measure was LBW defined as birth weight ≤ 2500 g. Other exposure measures were age, sex, body mass index, smoking status, presence of comorbidities (hypertension, cardiovascular disease, diabetes mellitus, obesity, chronic lung disease and malignancy), fetal growth restriction (defined as birth weight below 10th centile), prematurity (defined as delivery occurring before 37 weeks) and childhood lung disease. Variables with p < 0.05 on univariate analyses were entered in the multivariate logistic regression analysis to determine independent risk factors for ICU admission. A forward stepwise selection algorithm was applied to select the final model in the development dataset. Odds ratio and 95% confidence interval (95% CI) were calculated. Hosmer and Lemeshow test were used to assess the goodness of fit of the final model47. Analysis of the Receiver Operating Curve (ROC) was used to evaluate the predictive performance of the model in the development datasets and the optimal cut-off was computed using Youden criteria48. The model determined in the development dataset was used to predict ICU admission in the validation dataset and we report the statistical parameters for development and validation. All p-values are two-sided and considered statistically significant if < 0.05. Data were analysed with SPSS v26 and R software version 3.6.2 (R project for statistical computing, Vienna, Austria).
References
Guan, W. J. et al. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 328, 1708–1720 (2020).
Yang, X. et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study. Lancet Respir. Med. 8, 475–481 (2020).
Guan, W.J., Liang, W.H., Zhao, Y., Liang, H., Chen, Z., Li, Y. Comorbidity and its impact on 1590 patients with COVID-19 in China: A nationwide analysis (Internet). Eur. Respir. J. (2020). http://www.embase.com/search/results?subaction=viewrecord&from=export&id=L631357733%0A. https://doi.org/10.1183/13993003.00547-2020
Wang, D. et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA J. Am. Med. Assoc. 323, 1061–1069 (2020).
Shi, Y. et al. Host susceptibility to severe COVID-19 and establishment of a host risk score: Findings of 487 cases outside Wuhan. Crit. Care. 24, 2–5 (2020).
Cummings, M.J., Baldwin, M.R., Abrams, D., et al. Epidemiology, clinical course, and outcomes of critically ill adults with COVID-19 in New York City: A prospective cohort study. medRxiv (internet). 6736:2020.04.15.20067157 (2020).
Chidambaram, V. et al. Factors associated with disease severity and mortality among patients with COVID-19: A systematic review and meta-analysis. PLoS ONE 15(11), e0241541. https://doi.org/10.1371/journal.pone.0241541 (2020) ((eCollection202)).
Preliminary estimates of the prevalence of selected underlying health conditions among patients with coronavirus disease 2019—United States, February 12–March 28, 2020. MMWR Morb. Mortal. Wkly. Rep. 69, 382–386 (2020).
Schnake-Mahl, A., Carty, M. G., Sierra, G. & Toyin-Ajayi, M. P. Identifying patients with increased risk of severe COVID-19 complications: Building an actionable rules-based model for care teams. N. Engl. J. Med. https://doi.org/10.1056/CAT.20.0116 (2020).
Barker, D. The fetal and infant origins of adult disease. BMJ 301, 1111 (1990).
Crispi, F., Miranda, J. & Gratacós, E. Long-term cardiovascular consequences of fetal growth restriction: Biology, clinical implications, and opportunities for prevention of adult disease. Am. J. Obstet. Gynecol. 218, S869–S879 (2018).
Crispi, F. et al. Fetal growth restriction results in remodeled and less efficient hearts in children. Circulation 121, 2427–2436 (2010).
Kramer, M. S. Determinants of low birth weight: Methodological assessment and meta-analysis. Bull. World Health Organ. 65, 663–737 (1987).
Schieve, L. A. et al. Low and very low birth weight in infants conceived with use of assisted reproductive technology. N. Engl. J. Med. 346, 731–737 (2002).
Leon, D. A. et al. Reduced fetal growth rate and increased risk of death from ischaemic heart disease: Cohort study of 15 000 Swedish men and women born 1915–29. BMJ 317, 241–245 (1998).
den Dekker, H. T., Jaddoe, V., Reiss, I. K., de Jongste, J. C. & Duijts, L. Fetal and infant growth patterns and risk of lower lung function and asthma the generation R study. Am. J. Respir. Crit. Care Med. 197, 183–192 (2018).
Pike, K., Jane Pillow, J. & Lucas, J. S. Long term respiratory consequences of intrauterine growth restriction. Semin. Fetal Neonatal Med. 17, 92–98 (2012).
Agusti, A. & Faner, R. Lung function trajectories in health and disease. Lancet Respir. Med. 4, 358–364 (2019).
Agustí, A. & Hogg, J. C. Update on the pathogenesis of chronic obstructive pulmonary disease. N. Engl. J. Med. 381, 1248–1256 (2019).
Telles, F. et al. Changes in the preterm heart from birth to young adulthood: A meta-analysis. Pediatrics 146, e20200146 (2020).
Kotecha, S. J. et al. Effect of preterm birth on later FEV1: A systematic review and meta-analysis. Thorax 68, 760–766 (2013).
Carr, H., Cnattingius, S., Granath, F., Ludvigsson, J. F. & Edstedt Bonamy, A. K. Preterm birth and risk of heart failure up to early adulthood. J. Am. Coll. Cardiol. 69, 2634–2642 (2017).
Martinez, F. D. Early-life origins of chronic obstructive pulmonary disease. N. Engl. J. Med. 375, 871–878 (2016).
Hughes, M. M., Black, R. E. & Katz, J. 2500-g low birth weight cutoff: History and implications for future research and policy. Matern. Child Health J. 21, 283–289 (2017).
Huang, C. et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395, 497–506 (2020).
Izurieta, H. S. et al. Natural history of COVID-19: Risk factors for hospitalizations and deaths among >26 million U.S. Medicare beneficiaries. J. Infect. Dis. https://doi.org/10.1093/infdis/jiaa767 (2020).
Goodman, K. E. et al. Impact of sex and metabolic comorbidities on COVID-19 mortality risk across age groups: 66,646 inpatients across 613 US hospitals. Clin. Infect. Dis. https://doi.org/10.1093/cid/ciaa1787 (2020).
Working group for the surveillance and control of COVID-19 in Spain, et al. The first wave of the COVID-19 pandemic in Spain: Characterisation of cases and risk factors for severe outcomes, as at 27 April 2020. Euro Surveill. (2020).
Chen, N. et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 395, 507–513 (2020).
Bae, S., Kim, S. R., Kim, M. N., Shim, W. J. & Park, S. M. Impact of cardiovascular disease and risk factors on fatal outcomes in patients with COVID-19 according to age: A systematic review and meta-analysis. Heart https://doi.org/10.1136/heartjnl-2020-317901 (2020).
Pricehaywood, E. G., Burton, J., Fort, D. & Seoane, L. Hospitalization and mortality among black patients and white patients with COVID-19. N. Engl. J. Med. https://doi.org/10.1056/NEJMsa2011686 (2020).
Guo, W. et al. Diabetes is a risk factor for the progression and prognosis of COVID-19. Diabetes Metab. Res. Rev. 1, 1–9 (2020).
Chu, Y., Yang, J., Shi, J., Zhang, P. & Wang, X. Obesity is associated with increased severity of disease in COVID-19 pneumonia: A systematic review and meta-analysis. Eur. J. Med. Res. 25, 64. https://doi.org/10.1186/s40001-020-00464-9 (2020).
Sng, C. C. T. et al. Cancer history and systemic anti-cancer therapy independently predict COVID-19 mortality: A UK tertiary hospital experience. Front. Oncol. 10, 595804. https://doi.org/10.3389/fonc.2020.595804 (2020).
Vardavas, C. I. & Nikitara, K. COVID-19 and smoking: A systematic review of the evidence. Tob. Induc. Dis. 18, 20 (2020).
Reynolds, H. R. et al. Renin-angiotensin-aldosterone system inhibitors and risk of covid-19. N. Engl. J. Med. 382, 2441–2448 (2020).
Lange, P. et al. Lung-function trajectories leading to chronic obstructive pulmonary disease. N. Engl. J. Med. 373, 111–122 (2015).
Rueda-Clausen, C. F., Morton, J. S., Lopaschuk, G. D. & Davidge, S. T. Long-term effects of intrauterine growth restriction on cardiac metabolism and susceptibility to ischaemia/reperfusion. Cardiovasc. Res. 90, 285–294 (2011).
Rodríguez-López, M. et al. Descriptive analysis of different phenotypes of cardiac remodeling in fetal growth restriction. Ultrasound Obstet. Gynecol. 50, 207–214 (2017).
Luu, T. M., Katz, S. L., Leeson, P., Thebaud, B. & Nuyt, A. M. Preterm birth: Risk factor for early-onset chronic diseases. CMAJ 188, 736–740 (2016).
Nilsen, T. S., Kutschke, J., Brandt, I. & Harris, J. R. Validity of self-reported birthweight: Results from a Norwegian twin sample. Twin Res. Hum. Genet. 20, 406–413 (2017).
Blencowe, H. et al. National, regional, and worldwide estimates of low birthweight in 2015, with trends from 2000: A systematic analysis. Lancet Glob. Health. 7, e849–e860. https://doi.org/10.1016/S2214-109X(18)30565-5 (2019).
Nates, J. L. et al. ICU admission, discharge, and triage guidelines: A framework to enhance clinical operations, development of institutional policies, and further research. Crit. Care Med. 44, 1553–1602 (2016).
Figueras, F. et al. Customized birthweight standards for a Spanish population. Eur. J. Obstet. Gynecol. Reprod. Biol. 136, 20–24 (2008).
American College of Obstetricians and Gynecologists. Fetal growth restriction. ACOG Practice Bulletin No. 204. Obs. Gynecol. 133, e97–109 (2019).
American College of Obstetricians and Gynecologists. Definition of term pregnancy. ACOG Practice Bulletin No. 579. Obs. Gynecol. 122, 1139–1140 (2013).
Hosmer, D. W. & Lemeshow, S. Applied Logistic Regression (Wiley, New York, 2000).
Youden, W. J. Index for rating diagnostic tests. Cancer 3, 32–35 (1950).
Acknowledgments
Authors thank all participants in the study for their willingness to contribute to medical research as well as all the health workers who fought the COVID-19 pandemia.
Funding
This research was partially supported by “La Caixa” Foundation and Hospital Clínic, Barcelona, Spain. The funding source had no involvement in study design, in the collection, analysis, and interpretation of data; in the writing of the report; nor in the decision to submit the paper for publication.
Author information
Authors and Affiliations
Contributions
Fa.C. and Fr.C. postulated the hypothesis. Fa.C., Fr.C., E.G. and A.A. designed the study and wrote the study protocol. O.S., JR.B., I.B., R.F., A.T., M.L. and K.V. contributed to the study protocol design. Fa.C., Fr.C., M.L., M.C. and M.T. recruited the patients and performed the data collection. F.G. designed the web-based survey. R.B., Fa.C., Fr.C. and M.L. performed the statistical analysis. Fa.C., Fr.C., E.G. and A.A. analysed and interpreted the results. Fa.C., Fr.C., E.G. and A.A. wrote the main manuscript text. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Crispi, F., Crovetto, F., Larroya, M. et al. Low birth weight as a potential risk factor for severe COVID-19 in adults. Sci Rep 11, 2909 (2021). https://doi.org/10.1038/s41598-021-82389-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-82389-9
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.