Abstract
Pannexin 3 (Panx3), a member of the gap junction pannexin family is required for the development of hard tissues including bone, cartilage and teeth. However, the role of Panx3 in skin development remains unclear. Here, we demonstrate that Panx3 regulates skin development by modulating the transcription factor, Epiprofin (Epfn). Panx3−/− mice have impaired skin development and delayed hair follicle regeneration. Loss of Panx3 in knockout mice and suppression by shRNA both elicited a reduction of Epfn expression in the epidermis. In cell culture, Panx3 overexpression promoted HaCaT cell differentiation, cell cycle exit and enhanced Epfn expression. Epfn−/− mice and inhibition of Epfn by siRNA showed no obvious differences of Panx3 expression. Furthermore, Panx3 promotes Akt/NFAT signaling pathway in keratinocyte differentiation by both Panx3 ATP releasing channel and ER Ca2+ channel functions. Our results reveal that Panx3 has a key role factor for the skin development by regulating Epfn.
Similar content being viewed by others
Introduction
The skin is the largest organ of the human body, acting as the first line of defense from external factors with protective, immunologic, metabolism, thermoregulatory, and sensory functions1. Skin integrity and its normal function depend on the ability of keratinocytes to form the permeability barrier of the stratum corneum2. The epidermis contains five distinct layers: 1) basal, 2) spinosum, 3) granulosum, 4) lucidum and 5) corneum layers. Layers one through four are formed through the sequential differentiation of keratinocytes. The maturation process of keratinocytes has an integral relationship with a gradual amplification in calcium from basal layer of stem cells and proliferation progenitor cells to the outermost cornified layer containing terminally differentiated keratinocytes3. Calcium plays a well-known critical role in barrier function repair and skin homeostasis by regulating keratinocytes proliferation and differentiation4.
Maintenance of homeostasis in the skin requires direct intracellular and cell-extracellular communication5. Gap junction proteins play an important role in cell communications through their function as channels, allowing the exchange of various molecules, such as ions, metabolites, second messengers, and morphogens between adjoining cells6,7. There are two main families of gap junction proteins in vertebrates, connexins (Cxs) and pannexins (Panxs)8 with the latter being comprised of three family members, Panx1, 2 and 39. Panx1 is ubiquitously expressed in most tissues, while Panx2 appears to be more restricted to the central nervous system10. Panx3 is highly expressed in osteoblasts, synovial fibroblasts, bones, cartilage, teeth and skin9,11,12 and is found to cell differentiation and cell cycle exit of preosteoblast and chondrocyte13,14,15. Loss of Panx3 in mice lead to severe skeletal abnormalities16,17. Panx3 acts as a channel having different functions depending on its localization in the cell. At the plasma membrane, Panx3 releases intracellular ATP into the extracellular space, to activate PI3K/Akt pathway. This phosphorylation of Akt initiates endoplasmic reticulum (ER)-associated Panx3 to increase intracellular Ca2+ levels, which upregulates the calmodulin/calcineurin/NFAT signaling pathway to promote differentiation11,14,18. Panx3 deficiency delays wound healing in the skin by reducing epithelial–mesenchymal transition, keratinocytes migration, and collagen remodeling19. However, the functions of Panx3 in skin tissue development remain poorly understood.
Epiprofin (Epfn), also known as Sp6, is a zinc finger transcription factor that functions to promote both the proliferation and differentiation of keratinocytes20. Epfn−/− mice display defects in tooth morphology, supernumerary tooth formation, digit fusion in the limbs, skin abnormalities and hairlessness20,21,22,23,24. In cell culture, low Epfn expression promotes proliferation of keratinocyte progenitors, whereas high Epfn expression promotes cell cycle exit and induces differentiation through the activation of Notch120.
In this study, we show that Panx3 mediates keratinocyte differentiation by regulating Epfn. We find a thin epidermis in Panx3−/− mice and reduced Epfn expression in skin. In HaCaT cells, Panx3 overexpression increases Epfn expression and keratinocyte differentiation, while shRNA ablation of Panx3 decreases the presence of Epfn. In contrast, Epfn deletion does not interfere with Panx3 expression in skin and keratinocyte differentiation, suggesting that Panx3 is an upstream regulatory molecule of Epfn during skin development. Further, Panx3 promotes keratinocyte differentiation via Epfn signaling through the activation of Akt/NFAT signaling by its ATP releasing channel and ER Ca2+ channel functions.
Results
Panx3−/− mice show a thin epidermis and delayed hair follicle regeneration
To elucidate the role of Panx3 in the skin, we first compared histological cross-sections of the skin in Panx3+/− and Panx3−/− mice at multiple post-natal ages (Fig. 1A; Supplementary Fig. 1). The skin phenotype between Panx3+/+ and Panx3+/− mice were comparable and was used as a control (Supplementary Fig. 2). On postnatal day four (P4), Panx3−/− mice showed less pigmentation (Supplementary Fig. 1), a thinner epidermis and dermis than Panx3+/− mice (Fig. 1B). The number of hair follicles in Panx3−/− skin was lower than that of Panx3+/− skin (Fig. 1A,C). Further examination revealed Panx3−/− mice showed no obvious difference in the first hair cycle and no premature hair follicle anagen cessation, hair loss, or hair thinning commonly associated with severe malnutrition (Fig. 1A, P4 and P10). However, in the second hair cycle from five to eight weeks old, Panx3−/− mice demonstrated a delayed hair follicle cycle with a prolonged time in the telogen (quiescent) phase (Fig. 1A, P20 and P38; Supplementary Fig. 1).
Panx3 regulates skin structure and hair follicle cycle. (A) Representative histology images (H&E staining) of Panx3+/− and Panx3−/− mice skin on different postnatal days. (B) Higher magnification H&E stained images of Panx3+/− and Panx3−/− mice skin on P4. Ep: epidermis; D: dermis. (C) Measurements of epidermis and dermis thickness and hair follicle numbers in Panx3+/− and Panx3−/− mice on P4. *p < 0.01. Error bars represent the mean ± SD; N = 3.
Panx3−/− mice show the inhibition of keratinocyte differentiation
We next investigated the localization of Panx3 within the epidermis. In Panx3+/− mice, Panx3 was expressed throughout the epidermis from the basal layer to the granular layer, as well as the corneal layer in the skin at adult stage (Fig. 2A, a, left panel in upper lane). Panx3 immunostaining was effectively eliminated when the anti-Panx3 antibody was mixed with its cognate peptide (Fig. 2A, a, left panel in middle lane). Cx43, which is ubiquitously expressed in skin25, was not abolished in Panx3 −/− mice (Fig. 2A, a and b, middle panel in bottom lane).
Panx3 deficiency inhibits keratinocyte differentiation. (A) Immunostaining of skin of 6 weeks old Panx3+/− (top and middle) and Panx3−/− (bottom) mice for to Panx3 (red) and Cx43 (green) in the presence (top and middle) or absence (bottom) of Panx3 cognate peptide (middle) or control peptide (S-peptide) (top) (a). Dotted lines indicate borders between epidermis and dermis. (B) Immunostaining of skin of Panx3+/− and Panx3−/− mice on P4 and P38 with antibody to Notch1(red). The nuclei were counterstained with DAPI (blue in merged) (a). Relative fluorescence intensity comparison of images were analyzed (M&M) (A,b and B,b). *p < 0.01. Error bars represent the mean ± SD; N = 3.
Since Panx3−/− mice showed an inability to regulate keratinocyte differentiation, we next observed the expression of keratinocyte differentiation marker, Notch1, at P4 and P38. Unlike Panx3+/− mice, we found no evidence of Notch1 expression in the epidermis layer at either P4 and P38 of Panx3−/− mice, (Fig. 2B, a and b). These results suggest that Panx3 regulates keratinocyte differentiation.
Panx3 promotes HaCaT cell differentiation and inhibits cell proliferation
We next analyzed Panx3 function in keratinocyte differentiation using the human immortalized keratinocyte cell line, HaCaT cells. HaCaT cells normally are maintained in calcium-free keratinocyte media (KM) and can be induced to differentiate through the addition of high Ca2+ concentration in the media (KM + high Ca2+). We also used an overexpression model (pCMV6-Panx3) and shPanx3 to further establish the influence of Panx3 on keratinocyte differentiation (Supplementary Fig. 3). Panx3 overexpression increased the expression of the terminal keratinocyte differentiation marker Filaggrin and Notch1, while showing decreased Ki67, a marker for cell proliferation (Fig. 3A, a and b). Moreover, quantitative RT-PCR (qPCR) analysis showed the differentiation markers, K1, and K10 were increased in Panx3 overexpressed cells, while immature markers, K5, and K14 were decreased (Fig. 3B). In addition, Panx3 overexpression inhibited HaCaT cells proliferation (Fig. 4A), while promoting cell cycle arrest at G0/G1 phase (Fig. 4B; Supplementary Fig. 4). Together, these results indicate that Panx3 promotes keratinocyte differentiation and inhibits cell proliferation.
Panx3 regulates HaCaT cell differentiation. (A) Immunostaining of Panx3 (green), Filaggrin (red), and Notch1 (purple), Ki67 (yellow) in pCMV6-mock or pCMV6-Panx3 transfected HaCaT cells cultured in normal condition media (a). Relative fluorescence intensity comparison of images and % of Ki67 positive cells were analyzed (b). *p < 0.01. Error bars represent the mean ± SD; N = 3. The nuclei were counterstained with DAPI (blue in merged). (B) qPCR for K1, K10, K5, and K14 in pCMV6-mock or pCMV6-Panx3 transfected HaCaT cells. Results represent the mean ± SD; N = 3. *p < 0.01.
Panx3 promotes cell cycle exit in HaCaT cell proliferation. (A) pCMV6-mock or pCMV6-Panx3 were transiently transfected HaCaT cells were cultured in normal condition media for the indicated days. *p < 0.01. Error bars represent the mean ± SD; N = 3. (B) FACS analysis of cell cycle on pCMV6-mock or pCMV6-Panx3 transiently transfected HaCaT cells cultured in normal condition media for 3 days. The cells were stained with propidium iodide, and cell cycle stages were measured by FACS analysis. The panels represent distribution of cells (%) in the G0/G1, S, and G2/M phases. **p < 0.05, *p < 0.01. Error bars represent the mean ± SD; N = 3.
Panx3 regulates Epfn in keratinocyte differentiation
Keratinocyte development is notably modulated by a zinc finger transcription factor, Epiprofin (Epfn)20. Low Epfn expression promotes HaCaT cell proliferation. High Epfn expression shifts the cell fate from proliferation to differentiation by activating the Notch signaling pathway20. Thus, we hypothesed that Epfn could be involved in Panx3 mediated keratinocyte differentiation. We first compared Epfn expression in skin between Panx3+/− and Panx3−/− mice by qPCR. Epfn expression was significantly decreased in Panx3−/− mice (Fig. 5A). We next observed Epfn expression and localization in skin of both Panx3+/− and Panx3−/− mice by immunohistochemistry. Panx3+/− mice revealed that Epfn was expressed in hair follicles including inner- and outer- root sheath cells at P4 as well as in both the epidermis and within hair follicle keratinocytes at P38. In contrast, Epfn was significantly reduced in Panx3−/− mice at both ages (Fig. 5B, a and b). In addtion, our HaCaT model demonstrated a similar high expression of Epfn in Panx3 overexpressing cells, while Epfn was reduced in shPanx3 cells (Fig. 5C, a and b). During differentiation condition (KM + high Ca2+), both Panx3 and Epfn demonstrated a similar expression pattern in control HaCaT cells compared with those in normal media (KM) (Fig. 5C, a and b). In KM + high Ca2+ condition, proliferation marker, K14 was reduced, while differentiation markers, Filaggrin, K10 and Panx3 were increased (Supplementary Fig. 5A). Further, shPanx3 reduced Panx3 and differentiation marker, Notch1 expression in KM + Ca2+ condition (Supplementary Fig. 5B). These results indicate that Panx3 regulates Epfn expression during keratinocyte differentiation.
Panx3 regulates Epfn expression during keratinocyte differentiation. (A) qPCR for Panx3 and Epfn using total RNA prepared from skin of 6 weeks old Panx3+/− and Panx3−/− mice. Results represent the mean ± SD; N = 3. *p < 0.01. (B) Immunostaining of Epfn (red) in Panx3+/− and Panx3−/− mice skin of P4 and P38. The nuclei were counterstained with DAPI (blue in merged) (a). Relative fluorescence intensity comparison of images were analyzed (b). *p < 0.01. Error bars represent the mean ± SD; N = 3. (C) Immunostaining for Panx3 (green) and Epfn (red) in pCMV6-Panx3 or shPanx3 transiently transfected HaCaT cells cultured in normal medium (KM) and differentiation medium containing 1.4 mM Ca2+ (KM + high Ca2+). The nuclei were counterstained with DAPI (blue in merged) (a). Relative fluorescence intensity comparison of images were analyzed (b). *p < 0.01. Error bars represent the mean ± SD; N = 3.
Epfn deficiency doesn’t interfere with Panx3 expression
We further asked whether Epfn modulates Panx3 expression in keratinocyte differentiation. Q-PCR with skin of Epfn+/+ or Epfn−/− mice showed that Panx3 expression was comparable (Fig. 6A). Moreover, Panx3 expression was comparable in HaCaT cell with si-Control or si-Epfn by immunostaining (Fig. 6B, a and b). These results suggest that Epfn is not required for Panx3 expression and is likely downstream of its cellular function. For further confirmation that Epfn is a downstream molecule of Panx3 during keratinocyte differentiation, we analyzed Notch1, a downstream target of both Epfn20 and Panx3 (Figs. 2B and 3A) by si-Epfn and Epfn overexpression (pcDNA3.1-Epfn)20,23 into Panx3 overexpressed and shPanx3 transfected HaCaT cells, respectively (Fig. 6C,D). si-Epfn reduced Notch1 expression in Panx3 overexpressed cells, which normally upregulated Notch1 expression (Fig. 6C, a and b), while Epfn overexpression increased Notch 1 in shPanx3 transfected cells, which downregulated Notch1 (Fig. 6D, a and b). Thoes results indicate that Epfn is downstream of Panx3 during keratinocyte differentiation.
Epfn deficiency does not interfere with Panx3 expression. (A) qPCR for Panx3 and Epfn using total RNA prepared from skin of 6 weeks old Epfn+/+ and Epfn−/− mice. Results represent the mean ± SD; N = 3. *p < 0.01. NS, not significant. (B) Immunostaining of Panx3 (green) and Epfn (red) in si-Control or si-Epfn transfected HaCaT cells cultured in normal medium. The nuclei were counterstained with DAPI (blue in merged) (a). Relative fluorescence intensity comparison of images were analyzed (b). (C) Western blots of Epfn and Notch1 in Panx3 overexpressed HaCaT cells transfected with or without si-Epfn (a). Quantification of the ratios of Epfn/α-Tubulin (upper panel) and Notch1/α-Tubulin (lower panel). (D) Western blots of Epfn and Notch1 in shPanx3 transfected HaCaT cells with or without pcDNA3.1-Epfn (a). Quantification of the ratios of Epfn/α-Tubulin (upper panel) and Notch1/α-Tubulin (lower panel). *p < 0.01. NS, not significant.
Panx3 regulates HaCaT differentiation via Akt/NFAT signaling
We next asked which signaling pathways are involved in Panx3-mediated keratinocyte differentiation through Epfn. Previously, we reported that Panx3 functions as ATP releasing channel and ER Ca2+ channel in HaCaT cell19. Thus, we hypothesized that Panx3 mediated ATP and Ca2+ signaling could be involved. To answer this question, we analyzed the activation of Akt and NFAT in HaCaT cells by Western blotting (Fig. 7). Panx3 overexpression promoted phosphorylation of Akt and the level of dephosphorylated NFATc1 (active form) (Fig. 7A,B). We also found Epfn and Notch1 were also increased by Panx3 overexpression. To analyze how Panx3 channel functions are involved, we first applied Panx3 antibody to block Panx3 ATP at the cell membrane11,14. Application of Panx3 antibody inhibited both Akt and NFATc1 activation in Panx3 overexpressed cells and therefore decreased Epfn and Notch1 expression. To test the 2nd function of Panx3 as an ER Ca2+ channel mediated by ATP/PI3K/Akt signals14,19, a mutation was introduced to Panx3 at serine 68 (Ser68Ala) . to impair Panx3 ER Ca2+ release18. Ser68Ala inhibited the activation of NFATc1 and both Epfn and Notch1 expression (Fig. 7A, a, b and c). These results suggest that Panx3 as an ER Ca2+ channel strongly regulates Epfn expression through NFATc1/Ca2+ signaling for keratinocyte differentiation. Further, since Panx3 ER Ca2+ channel is activated by Akt signaling14,18,19, we investigated Epfn expression and NFATc1 activation with Akt dominant negative vector (Akt-DN) to confirm whether Panx3 ER Ca2+ channel function is critical for Epfn expression. Akt-DN inhibited NFATc1 activation and decreased Epfn and Notch1 expression (Fig. 7B, a, b and c). These results indicate that Panx3 regulates Epfn expression through Panx3 ER Ca2+ channel function during keratinocyte differentiation.
Panx3 ER Ca2+ channel regulates Epfn via Akt/NFAT signaling pathway. HaCaT cells were cultured for 3 days in normal media. Western blotting was performed with antibodies to Panx3, Epfn, Notch1, phospho-Akt (P-Akt), Akt, phospho-NFATc1 (P-NFATc1), NFATc1, and α-Tubulin. The cropped blots were used in the figures. Full blot is shown in the Supplementary Information (Full Original Blots-I–IV). (B) Quantification of the ratios of Epfn/α-Tubulin (upper panel of A,b and B,b) and Notch1/α-Tubulin (lower panel of A,b and B,b), P-Akt/Akt (upper panel of A,c and B,c) and P-NFATc1/NFATc1 (lower panel of A,c and b,c). **p < 0.05, *p < 0.01. Error bars represent the mean ± SD; N = 3.
Discussion
Here we demonstrated that Panx3 regulates skin development by promoting keratinocyte differentiation via the transcription factor, Epfn. At the tissue level, Panx3−/− mice revealed abnormal skin including less pigmentation, a thinner epidermis and dermis as well as a decrease in hair follicles. At the cellular level, Panx3−/− mice demonstrate decreased Notch1 expression during skin development. In an overexpression cell model, Panx3 promoted keratinocyte cell cycle arrest and led to cell differentiation. Further, Panx3 overexpression promoted Epfn expression during keratinocyte differentiation, while Panx3−/− mice and shPanx3 showed decreased expression. However, Epfn is not crucial to regulate Panx3 expression. Panx3 promotes the activation of NFAT signaling via Panx3 ER Ca2+ channel to Epfn expression to enhance Notch1 for keratinocyte differentiation (Fig. 8).
Panx3 signaling pathway in keratinocyte differentiation. Schematic representation of Panx3 signaling pathway in keratinocyte differentiation. 1) ATP releasing Panx3 channel activates Akt signaling in autocrine or paracrine manner. 2) Panx3 ER Ca2+ channel opened by Akt signaling promotes calmodulin (CaM)-NFAT signaling by raising up intracellular Ca2+ levels. 3) Dephosphorylated NFATc1 (active form NFATc1) enters the nucleus and binds to the promoter regions of Epfn. Epfn then promotes Nocth1 expression.
Panx3 is expressed in the epidermis, especially in areas containing actively differentiating keratinocytes in human and murine skin samples19,26,27, but the exact mechanisms of how Panx3 regulates skin development has not yet been fully explored. The in vivo and in vitro data presented here demonstrate that Panx3 expression is required to promote keratinocyte differentiation during skin development. Although we did not observe serious skin defects in Panx3−/− mice, the diminished expression of the differentiation marker Notch1 in the skin demonstrates the crucial role of Panx3 in regulating the activities of keratinocytes. The epidermis is composed of five functionally different layers of keratinocytes at different stages of differentiation3. From bottom to the upper most are the stratum basal, spinosum, granulosum, lucidum and corneum layers. The deep basal layer contains proliferative epidermal stem cells from which the superficial layers are derived and differentiate. Within these epidermis layers, calcium forms a steep gradient with the highest concentration at the stratum granulosum28. This is an indication that keratinocytes require low-level endogenous Ca2+ to maintain a basic bioactivity for proliferation and high-level Ca2+ for differentiation. Ca2+, acting as an important second messenger, is a key regulator of the proliferation, differentiation and morphology in keratinocytes29,30. A critical level of cytosolic calcium, achieved either by the influx of extracellular calcium or by intercellular communication via cell–cell contacts, will trigger mechanisms required for initiation of keratinocyte differentiation31. Panx3 functions as plasma membrane channel and gap junction which allow the transfer of extracellular small molecules, including Ca2+ between cell-extracellular spaces and other cells, respectively. In addition, Panx3 also possesses a unique function as ER Ca2+ channel, which can be stimulated to release endogenous Ca2+ into cytosol11,12,14. Our results confirm that Panx3 has a tight relationship with keratinocyte proliferation and differentiation. In osteoblast differentiation, ATP release through Panx3 activates PI3K/Akt pathway to promote Panx3 ER Ca2+ release in an autocrine and paracrine manner. As an ER Ca2+ channel, Panx3 is phosphorylated at the Serine 68 residue by Akt signaling, which raises intracellular Ca2+ levels and activates other Ca2+-dependent signaling pathways including NFAT18,19. Here we find that during keratinocyte differentiation, Panx3 functions first as an ATP releasing channel, then as an ER Ca2+ channel19 and promotes the activation of Akt and NFAT,respectively, similar to osteoblasts. Additonally, we found a change in the cellular morphological characteristics in Panx3 overexpressd HaCaT cells in both KM and KM + high Ca2+ (Figs. 3A and 5C). Intracellular Ca2+ can regulate cell morphology and the cornification process of epidermal keratinocytes32,33. Panx3 mediated intracellular Ca2+ signaling pathway also plays a role for cell morphology, although we need further analysis to elucidate the mechanism. Previous study showes that Panx3 overexpression reduced cell proliferation as well as it maintains epidermal architecture in the rat epidermal keratinocytes (REKs) with the epidermal organotypic culture system26. Our data was consistent with the previous report about cell proliferation. However, our study can not address more detail Panx3 functions for epidermal architecture with organotypic culture in this article.
During the wound healing process, Panx3−/− mice show delayed epithelialization by inhibiting Wnt and BMP signaling pathways19. Wnt/β-catenin signaling promotes keratinocyte proliferation in the hair follicles and in the interfollicular epidermis34. Panx3 expression inhibits Wnt/β-catenin signaling in osteoprogenitor proliferation13, whereas its overexpression inhibits HaCaT cell proliferation and promotes cell cycle arrest. Although biological evidence of Panx3 for keratinocyte proliferation is similar to that in osteoprogenitor cells, Panx3 function to Wnt/β-catenin signaling in keratinocyte, especially in the case of wound healing is not same. BMP2 promotes Panx3 expression during osteoblast differentiation14. As well as Wnt/β-catenin signaling, Panx3 function to BMP signaling in keratinocyte may has differences from that in other cell types. In addition to cell type difference, it is expected that the mechanism in normal keratinocyte differentiation may differ from in the case of wound healing process and requires future experimentation to answer those questions.
Typically, hair follicles regenerate in cyclic bouts consisting of growth (Anagen phase), regression (Catagen phase), and rest (Telogen phase) phases that collectively result in the production of a mature, fully grown hair shaft that extends over the surface of the skin35. Hair follicle stem cells in the epithelial niche provide the necessary number and type of specialized cells that are required to construct de novo hair36, while the signals are conveyed to the mesenchymal niche to induce dermal papilla growth37. The switch between active and quiescent states of hair follicles is a balance between Wnt signal and BMP signal that emanate from the dermal papilla38. Calcium channels activity bulge stem cells of the hair follicles cells also contribute to the quiescence-growth cycle39. The delayed hair follicle cycling of Panx3−/− mice indicates that Panx3 has a potential role to regulate the activation of epithelial stem cells in hair follicles. We found that hair follicle stem cells were maintained in the quiescent stage and delayed hair follicle regeneration in Panx3−/− mice (data not shown). According to our data, Panx3 may regulate the activation of hair follicle stem cells, but not their maintenance. Adult Panx3−/− mice exhibited impaired de novo production of hair shafts and all temporary hair cell lineages, owing to a prolonged quiescent phase of the first hair cycle.
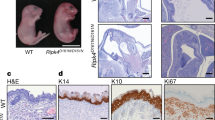
Epfn was identified by differential hybridization using mRNA isolated from embryonic mouse molars40. Epfn and Sp6 are different transcripts generated by alternative promoters from the same gene, but code for the same protein41. In contrast to Sp6 expressed ubiquitously in all adult tissues42, Epfn is only expressed during mouse embryogenesis in a tissue-specific manner in teeth, skin, hair follicle and limb buds. Besides severe defects in tooth morphology, Epfn−/− mice also display skin abnormalities and hairlessness20. It is well documented that Epfn is a key cell cycle regulator of keratinocytes: low levels of Epfn increased the proliferation of HaCaT, while high levels of Epfn promoted cell cycle exit and differentiation20. Panx3 overexpression promoted Epfn expression, while Panx3−/− mice and shPanx3 reduced it. However, Epfn−/− mice and siRNA knockdown of Epfn did not demonstrate a change Panx3 expression. These results indicate that Panx3 is an upstream regulator of Epfn involved in keratinocyte differentiation. Unlike the severe skin abnormality of Epfn−/− mice, however, Panx3 −/− mice do not show obvious skin abnormalities and hairlessness, only displaying a delayed hair cycle and hair regeneration in the early stage of hair growth, suggesting a possible compensation from the other gap junction proteins. A previous study shows that connexin 43 (Cx43) and Panx1 express higher in Panx3−/− mice skin than those in Panx3 +/− mice skin19. However, in our study, Cx43, Panx1 and Panx2 expression were not changed in Panx3−/− mice compared with Panx3+/− mice (Fig. 2A; Supplementary Fig. 6). Thus, other gap junction proteins in skin may help to maintain the metabolism of Panx3−/− skin43,44.
Cx43, is ubiquitously expressed in skin and contributes to keratinocyte differentiation45. Cx43 mutations directly result in oculodentodigital dysplasia (ODDD) showing the particularly strong abnormalities in the eyes, teeth, and fingers46 and rarely demonstrate skin abnormalities. Because Cx43-null mice die at birth due to cardiac malformation, analysis of skin disorders in Cx43-null mice is difficult47. According to the studies with Cx43 conditional KO mice and mutant mice, Cx43 function is compensated by other Cxs family members such as Cx26 and Cx30 in keratinocyte differentiation48,49,50. Likewise for osteoblast differentiation, Cx43 expression is regulated by Panx3 via transcription factor, osterix. Cx43 expression is decreased by Panx3−/− mice in bone. On the other hand, Cx43 expression was comparable in skin of Panx3−/− mice. Thus, there are tissue dependent roles between Panx3 and Cx43.
Panx1, a member of the pannexin family, is ubiquitously expressed, especially in the central nervous system51. Panx1 is highly expressed in neonatal but not in aged mouse skin and regulates skin development by providing proper skin architecture. Panx1 is required for keratinocyte migration, dermal fibroblast proliferation, and keratinocyte differentiation at early stage, particularly52. Panx3 reduces cell proliferation of keratinocytes in the organotypic epidermis26. Panx1−/− mice show reduced dermal area, but increased hypodermal thickness in dorsal skin. Furthermore, Panx3 expression is increased in skin of Panx1−/− mice. Thus, the functional differences between Panx1 and Panx3 may compensate for each other, especially at late stages of skin development. In Panx1 and Panx3 double knock-out mouse (dKO), it was shown that neonatal dKO mice have reduced dorsal skin thickness, similar to Panx1−/ mice phenotype43. According to previous reports and our current results, the compensation mechanism between Panx1 and Panx3 may be due to an age dependency.
Panx3 channels are involved in ATP/Ca2+ signaling pathway during osteoblast differentiation in multiple ways11,14. First, extracellular ATP released through Panx3 channel activates PI3K-Akt signaling pathway. This in turn leads to the phosphorylation or ER-associated Panx3 to release ER Ca2+ store to elevate intracellular Ca2+ and activate the calmodulin (CaM)-NFAT pathway. Panx3 mediated NFAT activity promotes osterix (also known as Sp7) transcription to differentiate mature osteoblast16. Panx3 also can activate NFAT through its function as an ATP releasing channel as well as its role as an ER Ca2+ channel during keratinocyte differentiation19. NFAT is essential for maintaining hair follicle stem cell quiescence53. and regulates primary keratinocyte cells differentiation in vitro by physical association with Sp1/Sp354. We found Panx3 regulates Epfn/Sp6 expression. Epfn is also upregulated by Ca2+. Thus, Ca2+ signaling plays a role for Epfn regulation. Actually, Epfn promoter has NFAT binding sequence (data not shown). Thus, Panx3 mediated NFAT signaling can regulate Epfn expression. Besides, Epfn may combine with NFAT directly or indirectly to differentiate cells. Epfn is also reported to promote Notch1 by binding to the Notch1 promoter region20. We found that Panx3 ER Ca2+ channel strongly regulates Epfn expression. However, it is still uncertain how Panx3 ATP releasing function and gap junction channel regulate keratinocyte differentiation. Blocking of Panx3 ER Ca2+ channel function with the Panx3 mutation construct, Ser68Ala, a small amount of Epfn expression still could be observed, but Notch1 expression remained low (Fig. 7A). These results suggest that other Panx3 functions may also be involved. Panx3 acting as an ATP releasing channel reduces intracellular ATP, resulting in a decrease in AMP-activated protein kinase (AMPK) activity and cAMP/PKA signaling in odontoblasts, chondrocytes and osteoblasts12,13,15. AMPK regulates Notch1 stability55, while PKA is also known to regulate Notch1 during astrocytic differentiation56. Thus, it is possible that ATP release through Panx3 can contribute to keratinocyte differentiation. Glycosylated Panx3 can form a membrane channel, but not gap junction44,57,58. However, it has also been reported that Panxs can form gap junction channel with the loss of glycosylation59. Thus, un-glycosylated Panx3 gap junction channel may contribute keratinocyte differentiaiton. Our results suggest that Panx3 regulates keratinocyte differentiation via NFAT/Ca2+ signaling activated by Panx3’s function as an ER Ca2+ channel following enhancement of Epfn/Notch1 signaling pathways.
In summary, our data have provided new evidence in keratinocyte differentiation mechanism that Panx3 is an upstream regulator of Epfn via Akt/NFAT signaling pathways by its channel functions.
Materials and methods
Animals
Pannexin 3 homozygous (Panx3−/−), heterozygous mice (Panx3+/−) mice16 and Epiprofin homozygous mice (Epfn−/−)40were generated as described before. Mice were housed under controlled conditions (i.e., temperature, 22 ± 2 °C, relative humidity 65 ± 15%, and 12 h light/dark cycle). All mice had access to a commercial diet and filtered water ad libitum. The animal protocol approved by the NIDCR Animal Care and Use Committee was used for maintaining and handling mice (protocol number ASP15-775). All animals were housed in an animal facility approved by the American Association for the Accreditation of Laboratory Animal Care. For analyzing hair follicle cycling, pair of sibling Panx3+/− and Panx3−/− mice on postborn day 4 (P4), 10 (P10), 20 (P20), 25 (P25) and 38 (P38) were anesthetized with isoflurane. The fur on the dorsal skin was removed with an electric shaver followed by applying VEET gel cream to remove the hair shafts. All experimental procedures in the manuscript were approved by the Animal Care and Use Committee of the National Institute of Dental and Craniofacial Research (protocol no.15-775) and Tohoku University (protocol no. 2017DnA-035), and performed in accordance with relevant guidelines and regulations.
Cell culture
HaCaT cells were obtained from Silvio Gutkind (UCSD). HaCaT cells were cultured in Keratinocyte-SFM (Gibco, Gaithersburg, MD, USA) with EGF and BPE supplements. For transfection, exponentially growing HaCaT cells were transfected with Panx3 expression vector (pCMV-6 Panx3) (5 μg per 3 × 104 cells) (Origene, Rockville, USA. SC305706) or shRNA plasmid for Panx3 (shPanx3) (Origene, Rockville, USA. TG302692) and their negative controls pCMV6-mock and shControl vector by using a Lipofectamine 3000 Reagent kit (ThermoFisher, Walkersville, USA.) followed by the manufacturer’s protocol19. For inhibition of Epfn, HaCaT cells were transfected with siRNA for Epfn (si-Epfn) (Origene, Rockville, USA. SR312895) and their negative control (si-Control) by using a transfection reagent designed for RNAi duplex SiTran1.0 (Origene, Rockville, USA. TT300001) followed by manufacturer’s protocol.
For the cell proliferation assay, HaCaT cells were prepared in 96-well plates at a density of 5 × 103 cells per well for culture. After 12 h, the medium was removed and a Cell Counting Kit (CCK)-8 (Dojindo, Japan) was used according to manufacturer’s instruction to determine the cell numbers based upon reaction product optical density (OD) value. To achieve this, 100 μl of fresh medium containing 10 μl of CCK-8 was added into each well, and the cells were cultured for an additional 1 h, during which time the CCK-8 reagent reacted with cellular metabolic products. The absorbance of the reaction products was then measured with a spectrophotometer at a wavelength of 450 nm using an enzyme-linked immunoadsorbent assay microplate reader (Tecan, USA).
For the cell differentiation assay, HaCaT cells were cultured in normal condition keratinocyte media (KM) with 1.4 mM calcium chloride (Quality Biological. INC. USA) at 37 °C under an atmosphere of 95% air and 5% CO2 for three days to induce differentiation.
Histological analysis
Tissue samples were retrieved at various time points and prepared for histological and immunohistochemical examination. The specimens were fixed overnight in 4% (v/v) Paraformaldehyde Phosphate Buffer Solution (Wako, Japan), embedded in paraffin and then cross-sectioned longitudinally into 5-μm sections using a Leica microtome for Gill’s 3 hematoxylin and aqueous eosin Y solution (H&E) to visualize the overall tissue morphology. All samples were analyzed using an upright microscope, and images were acquired using ScanScope camera (Leica Biosystems)19.
Immunostaining
Tissue sections were deparaffinized in xylene and rehydrated in descending ethanol solution sections. The samples were then immersed in 0.3% H2O2 to terminate peroxidase activity. After being immersed with antigen retrieval reagent (Dako. S1699) and heated with Decloaking Chamber (Biocare Medical) to improve antigen exposure, sections were applied with 0.1% sodium borohydride (Sigma) at room temperature for 30 min to reduce autofluorescence. Then, sections were blocked with PowerBlock (Biogenex. HK083) for 30 min at room temperature and incubated in a primary antibody solution diluted with antigen diluted reagent with concentration of 1:250, for overnight at 4 °C. Primary antibodies were detected by Alexa Fluor 488 (Invitrogen)-, Cy-3-, or Cy-5 (Jackson ImmunoResearch Laboratories)-conjugated secondary antibody of 1:250 with DAPI (Sigma) 1:1000. Finally, the slides were mounted using a mounting medium (Thermo scientific. TA-030-FM). Five samples from each time point were analyzed manually19.
HaCaT cells (1 × 104) were seeded in a 35-mm glass bottom dish and transfected with pCMV6-mock, pCMV6-Panx3, shControl, and shPanx3 vectors, or si-Control, or si-Epfn and cultured in KM for 3 days. The cells were then fixed in 4% Paraformaldehyde Phosphate Buffer Solution (Wako, Japan) + 5% sucrose (MP Biomedicals, France) for 20 min at room temperature followed by 0.1% Triton X-100 (Sigma) for penetration for 5 min and blocked by Power Block (Biogenex) for 30 min. Primary antibodies were applied at 4 °C overnight and were detected by Alexa Fluor 488 (Invitrogen)-, Cy-3-, or Cy-5 (Jackson ImmunoResearch Laboratories)-conjugated secondary antibody of 1:250 with DAPI (Sigma) 1:1000. Immunostaining imaging was performed on a laser scanning A1R MP confocal microscope (Nikon) equipped with plan apo 20 × (NA = 0.75), plan fluor 40 × (NA = 1.30), and plan apo 60 × (NA = 1.4) objectives. Rabbit anti-Panx3 antibody, inhibitory Panx3, and scrambled peptides15, and Rabbit anti-Epfn antibody23 have been described previously. Mouse anti-connexin 43 (Thermofisher. 138300), Rabbit anti-Notch1 (Cell Signaling Technology. 3608s), Rabbit anti-Ki67 (abcam, ab833Rabbit anti-Filaggrin (Biorbyt. Orb101353) antibodies were used.
ImageJ software (version 2.0.0-rc-69/1.52p, https://imagej.net; provided in the public domain by the National Institutes of Health, Bethesda, MD, USA) is used to analize the quatification of fluorescence intesity. Open figures in ImageJ and turn the RGB figure to RGB stack (Image > Type > RGB stack). Select the stack to work on. Open Threshold (Image > Adjust > Threshold), adjust the threshold to the area of target signals. Use the same setting to evaluate the other figures in the same assay. The fluorescence intensity or the ratio of fluorescence intensity of Panx3+/− to Panx3−/− is evaluated.
For calculating the percentage of Ki67 positive cells, four views of each cell condition were randomly selected. The total number of nuclei and the nuclei with positive Ki67 signals were counted with ImageJ.
RT-PCR
Total RNA from cells or skin tissues was extracted using the RNeasy Mini Kit and RNase-Free DNase set (Qiagen, USA) according to the manufacturer’s instruction. The RNA was reverse transcribed using the iScript (Bio-Rad.1708840). Total RNA (0.5 μg) was used for reverse transcription to generate cDNA, which was used as a template for PCRs with gene-specific primers. Primers were designed with Mac Vector software (version 17, https://macvector.com/index.html) (Table 1). Real-time PCR amplification was performed with iQ SYBR Green Supermix (Bio-Rad. 64047529) and a C1000 thermocycler (Bio-Rad). Real-time PCR was performed for 45 cycles at 95 °C for 20 s, 60 °C for 20 s, and 72 °C for 20 s. Ribosomal protein S29 was used to normalize gene expression in the samples. All reactions were run in triplicate19.
Flow cytometry analysis
HaCaT cells (1 × 106) were seeded in a 10-cm dish and transfected with pCMV6-mock and pCMV6-Panx3 vectors and cultured in KM for three days. The cells were then collected by centrifugation at 120 × g for five min, and fixed with cold 70% ethanol for 24 h followed by washing twice with PBS and staining with PI/RNAse solution (Thermofisher) for 15 min in the dark. The DNA content was analyzed with CellQuest software (http://www.icms.qmul.ac.uk/flowcytometry/flowcytometry/guides/CellQuest%20user%20guide.pdf) on FACSCalibur Station (Becton Dickinson).
Western blot analysis
HaCaT cells were lysed in Mammalian Cell Lysis Buffer (Abcam. Ab179835) with a halt protease inhibitor cocktail (Thermo Scientific. Rockford IL. USA 78437) on ice. Cells lysis was ultrasonic on ice, done five times with a rotation of 15 s at 1 Hz action and a 15-s stop. Protein concentrations were determined by the BCA kit (Thermofisher. 23225). Ten μg of protein from each group were resolved on sodium dodecyl sulfate (SDS)-polyacrylamide gels (Thermofisher. NP0342) and then transferred onto polyvinyl difluoride membranes (Thermofisher. IB401002). Primary antibodies, Rabbit anti-Epfn23, Goat anti-Panx3 (Santa Cruz. sc-51386), Mouse anti-phospho-Akt (Cell Signaling. 4051s), Rabbit anti-Akt antibody (Cell Signaling. 9272), Rabbit anti-phospho-NFAT antibody (Thermofisher. PA5-64484), Mouse anti-NFAT (BD Biosciences. 556602), Mouse anti-Notch1 antibody (Thermofisher. MA5-11961) were used to detect the corresponding proteins. Western blots were developed using SuperSignal West Dura Extended Duration Substrate (Thermo Scientific) and then photographed at Amersham Imager 600 (GE)19.
Statistical analyses
Results are expressed as the mean ± SD. Statistical analysis was performed by the Mann–Whitney U test to compare two groups. For more than two groups, data were analyzed by Kruskal–Wallis test. GraphPad Prism 7 software (https://www.graphpad.com/scientific-software/prism/) was used, in which differences between groups were considered significant when p < 0.05 (5%).
References
Fenner, J. & Clark, R. A. F. In Skin Tissue Engineering and Regenerative Medicine (eds Albanna, M. Z. & Holmes Iv, J. H.) 1–17 (Academic Press, 2016).
Berardesca, E. In Wound Healing and Skin Physiology (eds Altmeyer, P., Hoffmann, K., el Gammal, S., & Hutchinson, J.) 505–511 (Springer, Berlin Heidelberg).
Bikle, D. D., Xie, Z. & Tu, C. L. Calcium regulation of keratinocyte differentiation. Expert Rev. Endocrinol. Metab. 7, 461–472. https://doi.org/10.1586/eem.12.34 (2012).
Denda, M., Fuziwara, S. & Inoue, K. Influx of calcium and chloride ions into epidermal keratinocytes regulates exocytosis of epidermal lamellar bodies and skin permeability barrier homeostasis. J. Investig. Dermatol. 121, 362–367. https://doi.org/10.1046/j.1523-1747.2003.12367.x (2003).
Desai, R. A., Gao, L., Raghavan, S., Liu, W. F. & Chen, C. S. Cell polarity triggered by cell-cell adhesion via E-cadherin. J. Cell Sci. 122, 905–911. https://doi.org/10.1242/jcs.028183 (2009).
Plotkin, L. I., Davis, H. M., Cisterna, B. A. & Saez, J. C. Connexins and pannexins in bone and skeletal muscle. Curr. Osteoporos Rep. 15, 326–334. https://doi.org/10.1007/s11914-017-0374-z (2017).
Esseltine, J. L. & Laird, D. W. Next-generation connexin and pannexin cell biology. Trends Cell Biol. 26, 944–955. https://doi.org/10.1016/j.tcb.2016.06.003 (2016).
Beyer, E. C. & Berthoud, V. M. Gap junction gene and protein families: connexins, innexins, and pannexins. Biochim. Biophys. Acta Biomembr. 1860, 5–8. https://doi.org/10.1016/j.bbamem.2017.05.016 (2018).
Baranova, A. et al. The mammalian pannexin family is homologous to the invertebrate innexin gap junction proteins. Genomics 83, 706–716. https://doi.org/10.1016/j.ygeno.2003.09.025 (2004).
Makarenkova, H. P., Shah, S. B. & Shestopalov, V. I. The two faces of pannexins: new roles in inflammation and repair. J. Inflamm. Res. 11, 273–288. https://doi.org/10.2147/JIR.S128401 (2018).
Ishikawa, M. & Yamada, Y. The role of pannexin 3 in bone biology. J. Dent. Res. 96, 372–379. https://doi.org/10.1177/0022034516678203 (2017).
Iwamoto, T. et al. Pannexin 3 regulates proliferation and differentiation of odontoblasts via its hemichannel activities. PLoS ONE 12, e0177557. https://doi.org/10.1371/journal.pone.0177557 (2017).
Ishikawa, M., Iwamoto, T., Fukumoto, S. & Yamada, Y. Pannexin 3 inhibits proliferation of osteoprogenitor cells by regulating Wnt and p21 signaling. J. Biol. Chem. 289, 2839–2851. https://doi.org/10.1074/jbc.M113.523241 (2014).
Ishikawa, M. et al. Pannexin 3 functions as an ER Ca(2+) channel, hemichannel, and gap junction to promote osteoblast differentiation. J. Cell Biol. 193, 1257–1274. https://doi.org/10.1083/jcb.201101050 (2011).
Iwamoto, T. et al. Pannexin 3 regulates intracellular ATP/cAMP levels and promotes chondrocyte differentiation. J. Biol. Chem. 285, 18948–18958. https://doi.org/10.1074/jbc.M110.127027 (2010).
Ishikawa, M. et al. Pannexin 3 and connexin 43 modulate skeletal development through their distinct functions and expression patterns. J. Cell Sci. 129, 1018–1030. https://doi.org/10.1242/jcs.176883 (2016).
Oh, S. K. et al. Pannexin 3 is required for normal progression of skeletal development in vertebrates. FASEB J. 29, 4473–4484. https://doi.org/10.1096/fj.15-273722 (2015).
Ishikawa, M. et al. Pannexin 3 ER Ca(2+) channel gating is regulated by phosphorylation at the Serine 68 residue in osteoblast differentiation. Sci. Rep. 9, 18759. https://doi.org/10.1038/s41598-019-55371-9 (2019).
Zhang, P. et al. Pannexin-3 deficiency delays skin wound healing in mice due to defects in channel functionality. J. Invest. Dermatol. 139, 909–918. https://doi.org/10.1016/j.jid.2018.08.033 (2019).
Nakamura, T. et al. Epiprofin orchestrates epidermal keratinocyte proliferation and differentiation. J. Cell Sci. 127, 5261–5272. https://doi.org/10.1242/jcs.156778 (2014).
Hertveldt, V. et al. The development of several organs and appendages is impaired in mice lacking sp6. Dev. Dyn. 237, 883–892. https://doi.org/10.1002/dvdy.21355 (2008).
Ibarretxe, G. et al. Epiprofin/Sp6 regulates Wnt-BMP signaling and the establishment of cellular junctions during the bell stage of tooth development. Cell Tissue Res. 350, 95–107. https://doi.org/10.1007/s00441-012-1459-8 (2012).
Nakamura, T. et al. Transcription factor epiprofin is essential for tooth morphogenesis by regulating epithelial cell fate and tooth number. J. Biol. Chem. 283, 4825–4833. https://doi.org/10.1074/jbc.M708388200 (2008).
Nakamura, T. et al. Epiprofin regulates enamel formation and tooth morphogenesis by controlling epithelial–mesenchymal interactions during tooth development. J. Bone Miner. Res. 32, 601–610. https://doi.org/10.1002/jbmr.3024 (2017).
Kretz, M., Maass, K. & Willecke, K. Expression and function of connexins in the epidermis, analyzed with transgenic mouse mutants. Eur. J. Cell Biol. 83, 647–654. https://doi.org/10.1078/0171-9335-00422 (2004).
Celetti, S. J. et al. Implications of pannexin 1 and pannexin 3 for keratinocyte differentiation. J. Cell Sci. 123, 1363–1372. https://doi.org/10.1242/jcs.056093 (2010).
Cowan, K. N., Langlois, S., Penuela, S., Cowan, B. J. & Laird, D. W. Pannexin1 and Pannexin3 exhibit distinct localization patterns in human skin appendages and are regulated during keratinocyte differentiation and carcinogenesis. Cell Commun. Adhes. 19, 45–53. https://doi.org/10.3109/15419061.2012.712575 (2012).
Menon, G. K., Grayson, S. & Elias, P. M. Ionic calcium reservoirs in mammalian epidermis: ultrastructural localization by ion-capture cytochemistry. J. Invest. Dermatol. 84, 508–512. https://doi.org/10.1111/1523-1747.ep12273485 (1985).
Tu, C. L. & Bikle, D. D. Role of the calcium-sensing receptor in calcium regulation of epidermal differentiation and function. Best Pract. Res. Clin. Endocrinol. Metab. 27, 415–427. https://doi.org/10.1016/j.beem.2013.03.002 (2013).
Maurer, J. K., Gibbons, B. A. & Bruce, R. D. Morphometric assessment of thymic size variation in laboratory rabbits. Toxicol. Pathol. 18, 407–411. https://doi.org/10.1177/019262339001800307 (1990).
Pillai, S., Bikle, D. D., Mancianti, M. L., Cline, P. & Hincenbergs, M. Calcium regulation of growth and differentiation of normal human keratinocytes: modulation of differentiation competence by stages of growth and extracellular calcium. J. Cell Physiol. 143, 294–302. https://doi.org/10.1002/jcp.1041430213 (1990).
Li, F. et al. Dynamics and mechanisms of intracellular calcium waves elicited by tandem bubble-induced jetting flow. Proc. Natl. Acad. Sci. USA 115, E353–E362. https://doi.org/10.1073/pnas.1713905115 (2018).
Murata, T. et al. Transient elevation of cytoplasmic calcium ion concentration at a single cell level precedes morphological changes of epidermal keratinocytes during cornification. Sci. Rep. 8, 6610. https://doi.org/10.1038/s41598-018-24899-7 (2018).
Huelsken, J., Vogel, R., Erdmann, B., Cotsarelis, G. & Birchmeier, W. beta-Catenin controls hair follicle morphogenesis and stem cell differentiation in the skin. Cell 105, 533–545. https://doi.org/10.1016/s0092-8674(01)00336-1 (2001).
Muller-Rover, S. et al. A comprehensive guide for the accurate classification of murine hair follicles in distinct hair cycle stages. J. Invest. Dermatol. 117, 3–15. https://doi.org/10.1046/j.0022-202x.2001.01377.x (2001).
Rompolas, P. & Greco, V. Stem cell dynamics in the hair follicle niche. Semin. Cell Dev. Biol. 25–26, 34–42. https://doi.org/10.1016/j.semcdb.2013.12.005 (2014).
Plikus, M. V. et al. Self-organizing and stochastic behaviors during the regeneration of hair stem cells. Science 332, 586–589. https://doi.org/10.1126/science.1201647 (2011).
Plikus, M. V. et al. Cyclic dermal BMP signalling regulates stem cell activation during hair regeneration. Nature 451, 340–344. https://doi.org/10.1038/nature06457 (2008).
Yucel, G. et al. State-dependent signaling by Cav1.2 regulates hair follicle stem cell function. Genes Dev. 27, 1217–1222. https://doi.org/10.1101/gad.216556.113 (2013).
Nakamura, T. et al. The Kruppel-like factor epiprofin is expressed by epithelium of developing teeth, hair follicles, and limb buds and promotes cell proliferation. J. Biol. Chem. 279, 626–634. https://doi.org/10.1074/jbc.M307502200 (2004).
Hertveldt, V. et al. The Sp6 locus uses several promoters and generates sense and antisense transcripts. Biochimie 89, 1381–1387. https://doi.org/10.1016/j.biochi.2007.05.011 (2007).
Scohy, S. et al. Identification of KLF13 and KLF14 (SP6), novel members of the SP/XKLF transcription factor family. Genomics 70, 93–101. https://doi.org/10.1006/geno.2000.6362 (2000).
Abitbol, J. M. et al. Double deletion of Panx1 and Panx3 affects skin and bone but not hearing. J. Mol. Med. (Berl.) 97, 723–736. https://doi.org/10.1007/s00109-019-01779-9 (2019).
Penuela, S. et al. Pannexin 1 and pannexin 3 are glycoproteins that exhibit many distinct characteristics from the connexin family of gap junction proteins. J. Cell Sci. 120, 3772–3783. https://doi.org/10.1242/jcs.009514 (2007).
Zhang, X. F. & Cui, X. Connexin 43: key roles in the skin. Biomed. Rep. 6, 605–611. https://doi.org/10.3892/br.2017.903 (2017).
Paznekas, W. A. et al. Connexin 43 (GJA1) mutations cause the pleiotropic phenotype of oculodentodigital dysplasia. Am. J. Hum. Genet. 72, 408–418. https://doi.org/10.1086/346090 (2003).
Reaume, A. G. et al. Cardiac malformation in neonatal mice lacking connexin43. Science 267, 1831–1834. https://doi.org/10.1126/science.7892609 (1995).
Langlois, S. et al. Connexin levels regulate keratinocyte differentiation in the epidermis. J. Biol. Chem. 282, 30171–30180. https://doi.org/10.1074/jbc.M703623200 (2007).
Kretz, M. et al. Altered connexin expression and wound healing in the epidermis of connexin-deficient mice. J. Cell Sci. 116, 3443–3452. https://doi.org/10.1242/jcs.00638 (2003).
Flenniken, A. M. et al. A Gja1 missense mutation in a mouse model of oculodentodigital dysplasia. Development 132, 4375–4386. https://doi.org/10.1242/dev.02011 (2005).
Bruzzone, R., Hormuzdi, S. G., Barbe, M. T., Herb, A. & Monyer, H. Pannexins, a family of gap junction proteins expressed in brain. Proc. Natl. Acad. Sci. USA 100, 13644–13649. https://doi.org/10.1073/pnas.2233464100 (2003).
Penuela, S. et al. Panx1 regulates cellular properties of keratinocytes and dermal fibroblasts in skin development and wound healing. J. Invest. Dermatol. 134, 2026–2035. https://doi.org/10.1038/jid.2014.86 (2014).
Horsley, V., Aliprantis, A. O., Polak, L., Glimcher, L. H. & Fuchs, E. NFATc1 balances quiescence and proliferation of skin stem cells. Cell 132, 299–310. https://doi.org/10.1016/j.cell.2007.11.047 (2008).
Santini, M. P., Talora, C., Seki, T., Bolgan, L. & Dotto, G. P. Cross talk among calcineurin, Sp1/Sp3, and NFAT in control of p21(WAF1/CIP1) expression in keratinocyte differentiation. Proc. Natl. Acad. Sci. USA 98, 9575–9580. https://doi.org/10.1073/pnas.161299698 (2001).
Li, H., Lee, J., He, C., Zou, M. H. & Xie, Z. Suppression of the mTORC1/STAT3/Notch1 pathway by activated AMPK prevents hepatic insulin resistance induced by excess amino acids. Am. J. Physiol. Endocrinol. Metab. 306, E197-209. https://doi.org/10.1152/ajpendo.00202.2013 (2014).
Angulo-Rojo, C., Manning-Cela, R., Aguirre, A., Ortega, A. & Lopez-Bayghen, E. Involvement of the Notch pathway in terminal astrocytic differentiation: role of PKA. ASN Neuro 5, e00130. https://doi.org/10.1042/AN20130023 (2013).
Penuela, S., Bhalla, R., Nag, K. & Laird, D. W. Glycosylation regulates pannexin intermixing and cellular localization. Mol. Biol. Cell 20, 4313–4323. https://doi.org/10.1091/mbc.E09-01-0067 (2009).
Boyce, A. K. J., Epp, A. L., Nagarajan, A. & Swayne, L. A. Transcriptional and post-translational regulation of pannexins. Biochim. Biophys. Acta Biomembr. 1860, 72–82. https://doi.org/10.1016/j.bbamem.2017.03.004 (2018).
Ruan, Z., Orozco, I. J., Du, J. & Lu, W. Structures of human pannexin 1 reveal ion pathways and mechanism of gating. Nature https://doi.org/10.1038/s41586-020-2357-y (2020).
Acknowledgements
We thank Kenneth Yamada for critical suggestions. This work was supported in part by the Intramural Research Program of the National Institute of Dental and Craniofacial Research, USA [Grant Number DE000483-24, -25, and-26 to Y.Y.]; Grants-in-Aid from the Ministry of Education, Science, and Culture of Japan [Grant Number 16H05514 to M.I.]; Grants-in-Aid for Scientific Research-KAKENHI-Challenging Research (Exploratory) [Grant Number 19K22698 to M.I.and 18K19634 to T.N.]; Grants-in-Aid for Scientific Research-KAKENHI-Fostering Joint International Research [Grant Number 16KK0196 to M.I.]; the National Nature Science Foundation of China [NSFC81500811 to B.H.]. M.I. was supported in part by a Research Fellowship from the Japan Society for the Promotion of Science for Young Scientists.
Author information
Authors and Affiliations
Contributions
P.Z, M.I, and Y.Y conceptualized the experiments and wrote the manuscript. P.Z and M.I performed the experiments and the data analysis, with contributions from A.D, T.N, and B.H. M.I generated Panx3−/− mice. M.I, and Y.Y supervised the work. All authors gave final approval for submission. This paper is dedicated to the memory of Yoshihiko Yamada.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, P., Ishikawa, M., Doyle, A. et al. Pannexin 3 regulates skin development via Epiprofin. Sci Rep 11, 1779 (2021). https://doi.org/10.1038/s41598-021-81074-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-81074-1
This article is cited by
-
Pannexin 3 deletion reduces fat accumulation and inflammation in a sex-specific manner
International Journal of Obesity (2022)
-
Pannexin 3 channels in health and disease
Purinergic Signalling (2021)
-
Mechanisms of ATP release in pain: role of pannexin and connexin channels
Purinergic Signalling (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.