Abstract
In this paper, we investigate an norovirus (NoV) epidemic model with stochastic perturbation and the new definition of a nonlocal fractal–fractional derivative in the Atangana–Baleanu–Caputo (ABC) sense. First we present some basic properties including equilibria and the basic reproduction number of the model. Further, we analyze that the proposed stochastic system has a unique global positive solution. Next, the sufficient conditions of the extinction and the existence of a stationary probability measure for the disease are established. Furthermore, the fractal–fractional dynamics of the proposed model under Atangana–Baleanu–Caputo (ABC) derivative of fractional order “\({p}\)” and fractal dimension “\({q}\)” have also been addressed. Besides, coupling the non-linear functional analysis with fixed point theory, the qualitative analysis of the proposed model has been performed. The numerical simulations are carried out to demonstrate the analytical results. It is believed that this study will comprehensively strengthen the theoretical basis for comprehending the dynamics of the worldwide contagious diseases.
Similar content being viewed by others
Introduction
The NoV is disseminated by numerous factors that can promptly surge the transmission and henceforth the subsequent disease. The virus is known to be relatively sporadic, with more than half of the infections occurring during the cold season1. Seasonal fluctuation is attributed to ecological factors as well as demographic behaviour. Norovirus, for instance, is more easily feasting in cooler temperatures and might be aided by greater rainfall2,3. Other factors in the population may influence the intensity of norovirus epidemics. The virus infects people of all ages, however it is most common in kids below the age of five4. According to epidemiological studies, that the very earliest norovirus disease arises in childhood. Serious problems and fatality are often more common among the aging and handicapped, according to5,6. As a result, while disease is self-limiting in healthy people, the consequences for certain high-risk groups can be devastating. Such populations have also been observed to have much longer shedding intervals, which could lengthen an epidemic7,8,9,10.
Scholars from different fields have been working to prevent or minimize the rate of infection in certain groups. Mathematicians too have worked on the evaluation of relevant nonlinear dynamics of problems connected to infection, such as epidemics (see, for example11,12,13). Since 1990, mathematicians and biologists have been working hard to learn more about how epidemics travel and how to prevent them from growing in the community. In the area of preventing diseases, mathematicians are also playing an important contribution by employing mathematical modeling approaches as well as optimal solutions14,15,16,17,18,19,20. Infection modelling and study have become increasingly popular in current years as a means of better comprehending the mechanisms of developing epidemics. The approaches of mathematical modeling and optimality may then be employed to come up with a decent control strategy or technique for managing infectious diseases. Most academics have highlighted the stability and optimality for viral models with a non-linear incident operator using deterministic model techniques21,22. To come up with an effective control strategy or treatment for infectious disease, mathematical modelling in various dynamical systems, such as fractional and stochastic modelling, might be applied.
The integer order calculus of differentiation and integration has been developed to rational or complex numbers in modern calculus, illustrating the predicament among two integer numbers as in. The fractional differential equation is also valuable for gaining both analytical and numerical solutions for a multitude of problems23,24. Because establishing a precise solution is tough, numerous researchers look at FDEs for optimising and estimated solutions using pre-existing methodologies. They used Modified Euler approaches, Taylor’s series approach, Adams Bash-Forth techniques, predictor-corrector strategy, and other integral transforms, as well as wave-lets methods, to cope of the problem numerically. Recently, Atangana25 introduced a new nonlocal operator with the combination of fractional order and fractal dimension known as fractal–fractional (FF) differential and integral operators. The FF operator has been applied as an effective tool in describing various phenomena in many areas of science and epidemiology to explore the complex real word problems that could not be modeled with classical and fractional differential and integral operators with single order.
Numerous random order derivatives with an extra degree of freedom of choice have been explored using the fractional-order (FO) and integer-order mathematical model. When compared to deterministic models, it covers the entire spectrum to every compartment and a more accurate outcome than the natural order model also FO model, and stochastic differential equations could provide a high degree of stability. Because the findings of each investigation in a stochastic process varies from the others, we must run the models multiple times and look for patterns in the expected outcomes. Several authors have studied the stability analysis of different epidemic schemes including a non-linear incidence mapping for stochastic models26,27,28.
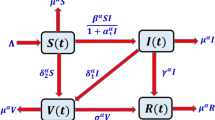
The five stochastic differential equations are used to proposed a stochastic epidemic mathematical system for NoV. The entire population is split in five partitions, each of which represents a sub-population: susceptible (H), vaccinated (V), asymptomatic or exposed (U), symptomatic or infected (A), and recovered (C), i.e., \(H(t)+V(t)+U(t)+A(t)+C(t))=N(t)\) The equations describing the model are
Here \(W_1(t), W_2(t), W_3(t), W_4(t), W_5(t)\) are independently standard Brownian motions, and \(\zeta _{1}, \zeta _{2}, \zeta _{3}, \zeta _{4}, \zeta _{5}\) are the intensities of standard Gaussain white noises, correspondingly. The parameters description given in Table 2. Here, vaccinated people also become infected via contact with symptomatic people. Note that, \(0< \tau < 1\) means \(\tau = 1\) perfect vaccine, while \(\tau = 0\) represents a vaccine that offers no protection at all.
The rest of the article layout has been established; In “Deterministic state stability” section, we determined the deterministic stability of system (1). We prove the global positive solution for system (1) in “The existence and uniqueness of positive solution” section. We employed a stochastic threshold strategy to decrease the epidemic in “Extinction of the disease” section. And in “Stationary distribution and ergodicity” section we evaluate a stationary distribution of the stochastic model. In “A fractal–fractional NoV model with Mittag–Leffler kernel” section, we analyze the proposed model through Fractal–fractional Atangana–Baleanu operator. In “Parameter estimation” section, the application of the parameter estimation is presented for the proposed model. In “Numerical simulations” section, we give simulation results to support our theoretical results. In the “Conclusion” section, we are give some conclusions.
Deterministic state stability
The deterministic form of system (1) is governed by the following set of equations
with initial conditions
The model equilibria provide useful information regarding the model trajectory over time. The model (2) posses two type of equilibrium points, the disease-free (DFE) and endemic equilibrium (EE) points.
Disease-free equilibrium
The disease-free equilibrium \((G_0)\) can be obtained by equating the right side of equations in system (2) to zero as follows:
Lemma 1
The DFE \(G_0\) is locally and globally asymptotically stable if \(R_0^D < 1\), and unstable if \(R_0^D > 1\).
Proof
The Proof of the Lemma is simple so we omit it here.
Using the next-generation matrix technique, we compute the basic reproductive ratio \(R_0^D\) using only the two equations corresponding to compartments U and A classes from system (2).
The basic reproductive ratio, defined as the spectral radius of the matrix \(\mathbf {F V}^{-1}\), is obtained as
Endemic equilibrium
The system (2) is also have EE equilibrium point which denoted by \(G_1=(H_1,V_1,U_1.A_1,C_1)\), in same way we can find \(G_1\), where
where
Lemma 2
The EE \(G_1\) is locally and globally asymptotically stable if \(R_0^D < 1\) and unstable if \(R_0^D >1\).
Proof
The Proof of the Lemma is simple so we omit it here. \(\square \)
The existence and uniqueness of positive solution
In order to addresses the existence and uniqueness of the stochastic model (1), we present the following theorem.
Theorem 1
The solution of the constructed stochastic epidemiological system (1) (H(t), V(t), U(t), A(t), C(t)) is unique for \(t\ge 0\) with initial condition \((H(0), V(0), U(0), A(0), C(0))\in {\mathbb {C}}^{5}_{+}\). Additionally, the solution will almost certainly remain in \({\mathbb {C}}^{5}_{+}\) with the unit probability, that is, \((H(0), V(0), U(0), A(0), C(0))\in {\mathbb {C}}^{5}_{+}~\forall ~t\ge 0\) almost surely (a.s).
Proof
The coefficients used in equations for the initial value of the state variables \((H(t), V(t), U(t), A(t), C(t))\in {\mathbb {C}}^{5}_{+}\) are continuous and locally lipschitz. As a result, there should be a local unique solution of the system
(H(t), V(t), U(t), A(t), C(t)) throughout \(t\in [0,\tau _e)\). The citations22,26 provide a detailed examination of the explosion duration \(\tau _e\). To demonstrate the solution’s global character, we must establish that \(\tau _e=\infty \) a.s. Suppose we have a comparatively big nonnegative number \(k_0\) such that each of the state’s starting conditions are contained within the range \([\frac{1}{k_0},k_0]\). Let the final time be specified as \(k\ge k_0\) to every positive integer.
We will use \(\inf \phi =\infty \) throughout this article, whereas \(\phi \) stands for the null set. The concept of \(\tau _k\) forces us to state that it rises as k reaches \(\infty \). Replacing \(\tau _\infty =\lim _{k\rightarrow \infty }\) for \(\tau _e\ge \tau _\infty \) a.s. After demonstrating that \(\tau _\infty =\infty \) a.s., we will assert that \(\tau _e=\infty \), and therefore (H(t), V(t), U(t), A(t), C(t)) will be found in \({\mathbb {C}}^{5}_{+}\) a.s. \(\forall t\ge 0\). Hence, proving that \(\tau _e=\infty \) a.s. suffices. Instead, two positive constants values \(\epsilon \) from (0, 1) and T must exist, such that
As a result, the integer \(k_1\ge k_0\) exists in the following form
After that, we’ll look at how to interpret a \(C^{2}\)-function \(H:{\mathbb {C}}^5_{+}\rightarrow {\mathbb {C}}_{+}\) in such a way that
It is to be noted that the H is a nonnegative function, and it can be verified from the fact that \(0\le y-\text {logy}-1,~\forall ~0<y\). Assume that \(k_0\le K\) and \(0<T\) are arbitrary. Upon applying \(At{\hat{o}}'s\) formula to Eq. (6) gives us
In Eq. (7), \(LH:{\mathbb {C}}^5_{+}\rightarrow {\mathbb {C}}_{+}\) is defined by the following equation
Thus,
Setting \(\Omega _k=\{\tau _k\le T\}\) for \(k\ge k_1\) and by Eq. (5), \(P(\Omega _k)\ge \epsilon \). Note that for each \(\omega \) from \(\Omega _k\) there must exist one or more than one \(H(\tau _k,\omega )\), \(V(\tau _k,\omega )\), \(U(\tau _k,\omega )\), \(A(\tau _k,\omega )\) and \(C(\tau _k,\omega )\) which equals
\(\frac{1}{k}\) or k. As a result \(H(H(\tau _k),V(\tau _k),U(\tau _k),A(\tau _k),C(\tau _k))\) is no less then \(\frac{1}{k}-1+\text {log}k\) or \(k-1-\text {log}k\). Therefore,
By using Eqs. (5) and (9), we can write
The indicator function of \(\Omega \) is represented as \(1_{\Omega (\omega )}\). As we get closer to \(\infty \), the contradiction \(\infty >H\big (H(0),V(0),U(0),A(0),C(0))\big )+MT=\infty \) emerges, indicating that \(\tau _\infty =\infty \) a.s. \(\square \)
Extinction of the disease
This section focuses on the criteria for disease’s extinction in system (1). Prior to prove the major findings, let’s look at an important lemmas.
Let
Lemma 3
13,14 (Strong Law of Large Number) If \(M=\{M\}_{t\ge 0}\) is now a continuous and real-valued local martingale that vanishes at \(t=0\),
Lemma 4
For arbitrary given starting value \((H(0), V(0), U(0),A(0), C(0)) \in {\mathbb {C}}_+^5,\) the solution (H(t), V(t), U(t), A(t), C(t)) for the system 2 has the upcoming properties:
Furthermore, when \(d>\frac{1}{2}(\zeta _1^2\vee \zeta _2^2\vee \zeta _3^2\vee \zeta _4^2\vee \zeta _5^2)\) holds, then
Proof
We exclude Lemma 4 proof because it is same in Lemma 4.1 in28. \(\square \)
Defined a parameter
Theorem 2
If \( R_0<1 \) and \( d >\frac{\zeta _1^2\vee \zeta _2^2\vee \zeta _3^2\vee \zeta _4^2\vee \zeta _5^2}{2}, \) then the root of system (1) fulfilling, so given as:
Proof
Describe a differentiable function \(G_0\) as
Considering Ito’s formula along with using system (1), we get
On the both sides of (19), we have integrating with limits from 0 to t and dividing with t we have the following
We’ve used Lemma 3 to get the following
Which demonstrates
It is simple to deduce below from the fourth equation of system (1).
Furthermore, on both hand sides of the first equation in system (1), integrating from 0 to t and dividing with t obtains
and considering (22), and Lemma 3, it then follows that
Similarly, we also can get
The proof for Theorem 2 is finished. \(\square \)
Stationary distribution and ergodicity
When it comes to stochastic systems, there are no endemic equilibria. As a result, the stability analysis could be utilised to investigate the disease’s persistence. As a consequence, one should focus on the existence and uniqueness theory for the stationary distribution, which, in some ways, will help with disease persistence. We shall use Hasminskii’s renowned finding29 for this task.
Let X(t) be a regular Markov process (time-homogeneous) in \(C^n_+\) for which the dynamics is as below:
The diffusion matrix is of the form
Lemma 5
22,26 The stationary distribution of the process X(t) is unique. If there exists a bounded domain having a regular boundary such that \(U,{\bar{U}}\in C^d\) \({\bar{U}}\) closure \({\bar{U}}\in C^d,\) with below properties
-
1.
The lowest eigenvalue for A(t) is bounded away form the origin for the open domain U along with its neighbourhood.
-
2.
for \(x \in C^d U,\), the mean time \(\tau \) (with which a path originating from x reaches the set U) is bounded, and for all compact subset \(K\subset C^n\), \(Sup_{x\in k} U^x\tau < \infty \). When f(.) is an integrable function having measure \(\pi \), thus
for each \(x \in C^d.\)
Describe a parameter
Theorem 3
The system (1) solution (H(t), V(t), U(t), A(t), C(t)) is ergodic, and having a unique stationary distribution. Since \(R^s_0 > 1\), \(\pi (.)\) is used.
Proof
To check the condition (2) of the Lemma 5, we should define a non-negative \(C^2-\)function \( V:C^5_+ \rightarrow C_+.\) For which we need to define
here the positive constants \(c_1, c_2\) and \(c_3\) must be calculated afterwards. We get the following results by utilising Itô’s formula and the suggested system (1).
Therefore, we have
The above implies that
Let
Namely
Consequently
In addition, we obtain
here \(c_4 > 0\) is a constant that will be decided afterward. It’s useful to illustrate that.
here \(U_k = (\frac{1}{k}, k) \times ( \frac{1}{k}, k) \times (\frac{1}{k}, k)\). The upcoming step is to show that \(V_2(H,V,U,A,C)\) has one and only one minimum value \(V_2(H_0,V_0,U_0,A_0,C_0).\) \(\square \)
The partial derivative of \(V_2(H,V,U,A,C) \) with respect to H, V, U, A, C is as follow
It’s not difficult to establish that \(V_2\) has a unique stagnation point.
Moreover, the Hessian matrix of \(V_2(H,V,U,A,C)\) at (H(0), V(0), U(0), A(0), C(0)) is
The Hessian matrix is evidently positive definite. As an outcome, \(V_2(H,V,U,A,C)\) has a least value of V2(H, V, U, A, C)
\(V_2(H(0),V(0),U(0),A(0),C(0))\). As per Eq. (30) and according to the continuity of \(V_2(H,V,U,A,C)\), we can say that \(V_2(H,V,U,A,C)\) has just one least value \(V_2(H(0),V(0),U(0),A(0),C(0))\) contained in \({\mathbb {C}}^5_+\).
Following that, we’ll define a non-negative \(C^2-\)function \(V : {\mathbb {C}}^5_+ \rightarrow {\mathbb {C}}_{+}\) as follows
Considering \(Ito's\) calculation and the proposed system, we reach at
as a consequence of which the preceding assumption can be formed:
where
The subsequent step is to establish the set
where \(\epsilon _i>0\) is a negligibly minor constant to be found afterwards for \((i = 1,2)\). We’ll split the entire \({\mathbb {C}}_{+}^{5} \backslash D\) into the preceding regions for clarity’s reason.
Now we’ll show that \(LV(H,V,U,A,C) < 0 \) on \({\mathbb {C}}_{+}^{5} \backslash D\), which is the similar as presenting it on the ten regions specified earlier.
Case 1. If \((H,V,U,A,C) \in D_1\), so by Eq. (34), it gives
Choosing \(\epsilon _1 > 0,\) yields \({\mathcal {L}}V<0\) for each \((H,V,U,A,C)\in D_1.\)
Case 2. If \((H,V,U,A,C)\in D_2\), then from Eq. (34), we can obtain
Let \( \epsilon _1 > 0\), so we can get \( {\mathcal {L}}V<0 \) for any \( (H,V,U,A,C)\in D_2. \)
Case 3. If \((H,V,U,A,C)\in D_3\), then from Eq. (34), we obtain
By taking small \(\epsilon _1,\epsilon _2 > 0,\) so, as \({\mathcal {L}}V<0\) for each \((H,V,U,A,C)\in D_3.\)
Case 4. \(If (H,V,U,A,C)\in D_4,\) from Eq. (34), we obtain
If we pick a small enough \(\epsilon _1 > 0\), then we get \({\mathcal {L}}V<0\) for every \((H,V,U,A,C)\in D_4.\).
Case 5. If \((H,V,U,A,C) \in D_5,\) from Eq. (34), we obtain
We take quite small \(\epsilon _2 > 0\), so now we can get \({\mathcal {L}}V<0\) for any \((H,V,U,A,C)\in D_5.\)
Case 6. If \((H,V,U,A,C)\in D_6,\) from Eq. (34), we obtain
We can choose sufficiently small \(\epsilon _2, \epsilon _1 > 0\), so now we can get \({\mathcal {L}}V<0\) for any \((H,V,U,A,C)\in D_6.\)
Case 7. If \((H,V,U,A,C)\in D_7,\) from Eq. (34), we obtain
By selecting smallest value of \(\epsilon _2 > 0\), so we can get \({\mathcal {L}}V<0\) for any \((H,V,U,A,C)\in D_7.\)
Case 8. If \((H,V,U,A,C)\in D_8,\) from Eq. (34), we obtain
Let we take the smallest value of \(\epsilon _2 > 0\), so we can get \({\mathcal {L}}V<0\) for any \((H,V,U,A,C)\in D_8.\)
Case 9. If \((H,V,U,A,C)\in D_9,\) from Eq. (34), we obtain
Now if \(\epsilon _1 > 0\), so now we can find \({\mathcal {L}}V<0\) for every \((H,V,U,A,C)\in D_9.\)
Case 10. If \((H,V,U,A,C)\in D_10,\) from Eq. (34), we obtain
For the smallest velue of \(\epsilon _2 > 0\), so we can obtain \({\mathcal {L}}V<0\) for any arbitrary \((H,V,U,A,C)\in D_10.\)
As an outcome, we establish that a constant \( W > 0 \) is such that which assures
Hence
Assume that \((H(0),V(0), U(0), A(0), C(0)) = (x_1, x_2, x_3,x_4,x_5) = x \in {\mathbb {C}}_{+}^{5} \backslash D\), and \(\tau ^x \) is that time for which a path start from x reach to the set D,
One can get the following by taking integration of the both hand sides of the inequality (35) from zero to \( \tau ^{(n)}(t)\), considering expectation, and utilizing Dynkin’s calculation.
As V(x) is a non-negative, therefore
We have \(P\{\tau _e = \infty \} = 1\) as a result of the proof of Theorem 3. Conversely, the system (1) can be defined as regular. As a result, if we take \(t \rightarrow \infty \) and \(n \rightarrow \infty \), we almost certainly obtain \(\tau {(n)}(t) \rightarrow \tau ^x \) almost certainly.
As a result, using Fatou’s lemma, we arrive at
Clearly, \(sup_{x\in K} {\mathbb {U}}\tau ^x < \infty ,\) here K is a compact subset from \({\mathbb {C}}_{+}^{5}\). It confirms Lemma 5 condition 2.
Additionally, the diffusion matrix of the system (1) is
Choosing \(M = \min _{(H,V,U,A,C)\in {\overline{D}}\in C^5_+}\{\zeta ^2_1H^2,\zeta ^2_2 V^2,\zeta ^2_3U^2, \zeta ^2_4A^2, \zeta ^2_5C^2\},\) we obtain
where \(\rho = (\rho _1, \rho _2, \rho _3,\rho _4,\rho _5) \in {\mathbb {C}}_{+}^{5}.\) This implies that Lemma 5 condition 1 is likewise true. Following to the preceding analysis, Lemma 5 indicates that the system (1) is ergodic and has just one stationary distribution.
A fractal–fractional NoV model with Mittag–Leffler kernel
Fractional calculus (FC) has gained much interest from the researcher and scientists, because of its uses in different fields of real-world problems than that of integer order. FC has taken advantages and popularity of modelling with memory effects30,31. Inspired from the work of Atangana25,31,32,33, in the field of FC, we convert the proposed norovirus model to investigate its parameter with the available data in system (1). The current section, deals with the approach of Atangana–Baleanu fractal–fractional (FF) derivative operator having fractional order \({p}\) and fractal dimension \({q}\). We consider the presented model (2) to fractal–fractional order in sense of \(\mathcal {ABC}\) operator. It is due to the fractional order derivatives have extra degree of freedom and some other characteristics of heredity, memory, and description of the past as well as present and future. This operator has non-singular kernel and is non-local operator. The NoV model can be shown through the following fractal–fractional differential system:
Further, the system (36) can be write in the ABC fractal–fractional into the following way
where \(\mathcal {K}_i, i=1,2,3,4,5\). In view of Eq. (37)
along with
where
Taking the first equation of (37) and by using the anti-derivative of fractal dimension and fractional order in sense of, we have
by letting \(t=t_{\flat +1}\) for \(\flat =0,1,2 \ldots ,\)
The approximate function be \(\mathcal {K}_1\) on the interval \([{t}_\varsigma ,{t}_{\varsigma +1}]\) through the interpolation polynomial as follows
which implies that
Calculating \({\mathbf {I}}_{\varsigma -1, {p}}\) and \({\mathbf {I}}_{\varsigma , {p}}\) we get
and
put \(t_\varsigma =\varsigma \Delta ,\) we get
and
substituting the values of (41) and (42) in (40), we obtain
And similarly for the other classes V, U, A and C we may find the same scheme as
Parameter estimation
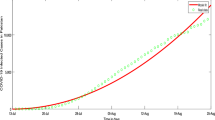
In reference34, they study the data about a NoV infectious diarrhea incident reported in a middle school in a city. To guarantee the correctness and effectiveness of the methods, the data of a 2007 NoV outbreak in a middle school in one city is used as the real data to solve the inverse problem of the parameter estimation. For the daily reports and the data set in the Eq. (48), and the related relative error is used in the goodness of fit.
where \(A_i\) is respectively the reported total number of infected, and \({\hat{A}}_i\) is the simulated total number of infected. The simulated cumulative number of infected are calculated by summing the individuals transit from the infected compartment to the recovered compartment for each day. Figure 1 shows the fit of model to the real data. Estimated values of parameters are shown in the Table 1.
Quantity of the NoV infections compare with the numerical results of integer-order model (2).
Numerical simulations
In this section, we will illustrate our analytical results by some examples with the help of numerical simulations firstly. We use the first order stochastic iterative techniques of the fourth order Range kutta method to estimate the system solution (1). We adopt stochastic methodologies to simulate the double stochastic integrals because the system is driven by five independent noises \(dW_i(t)\) for \(i=1,2,3,4,5\). These integrals are approximated utilising the pseudo periodicity of the brownian bridge and their similarity to the deterministic Fourier series approach in our numerical method. For this, we use fourth-order Range kutta stochastic iterative techniques to accomplish the resulting discretization-transformation of the model (1),
Here \( \xi _{k, i}(k=1,2,3,4)\) are four free Gaussian general variables with N(0, 1) and \(\Delta t>0\) time-increment.
The values of the parameters listed in Table 2.
Numerical simulations for stochastic stability
Now, we will use numerical simulation to investigate the numerical approximation and biological feasibility of the system (1). As a result, we used the parameters and noise intensities value from Table 2. The initial value of the Individuals susceptible H(t), vaccinated V(0), asymptomatic U(0), symptomatic A(t), and recovered R(t) are presented in Table 2 for \(t\in [0{-}150]\).
Taking into account white noises and parameter values from Table 2 (Set A), which ensure the conditions of Theorem 2, therefore, the infected population exponentially tends to zero with probability 1. The epidemic free equilibrium point shows global asymptotic stability in the related deterministic approach. As seen in Fig. 2, the disease can die.
We use parameter value from Table 2 (Set B) for the stochastic system (1) and compute \( R^s_0>1\), which ensure the condition for the NoV persistent. As shown in Fig. 3, the infection of system (1) will reside in the average, confirming the conclusions of Theorem 3. Observe 9000 attempts at \(t=500\), then compute the average value. Theorem 3 suggests that the system (1) has an ergodic stationary distribution, as shown in Fig. 4.
Example 1
(Stochastic disease-free dynamical behavior) The parameter values are taken from Table 2 (Set A). As a result, we obtain the reproduction number \({\tilde{R}}^s_0<1\), and the root of the model (1) may be satisfied by Theorem 2.
and
As a result, the pandemic will disappear from the population, as seen in Fig. 2 demonstrates that the numerical-simulation validates our strategy.
Example 2
(Stochastic endemic dynamical behavior) We get the parameter values from Table 2 (Set B). We show that \(R^s_0 >1\), and that the illness will lie or stabilised according to Theorem 3, and we illustrate our findings in Fig. 3. Theorem 3 states that the system (1) has just one stationary distribution, as shown in Fig. 3.
Simulations of (H(t), V(t), U(t), A(t), C(t)), for the stochastic models (1) with its corresponding deterministic version.
Simulations of (H(t), V(t), U(t), A(t), C(t)) for the stochastic models (1) with its corresponding deterministic version.
The probability distribution histogram of (H(t), V(t), U(t), A(t), C(t)) for the stochastic model (1).
Numerical simulation for fractal–fractional system
In this section, we applied the novel numerical approach obtained above in (43)–(47) to simulate the proposed FF NoV model (36) having different arbitrary orders “\({p}\)” and various dimensions “\({q}\)” respectively given in Figs. 5 and 6, the initial and parameter value taken from Table 2 (Set C). We simulate the ABC fractal–fractional NoV model (36) when both the fractional order “\({p}\)” and fractal order “\({q}\)” are different. Figure 5a represents the susceptible class H(t) at different fractional orders and fractal dimensions. The class declines with the passage of time as the virus enters the society will transfer to the other classes of the system. This class converges quickly at low order and slowly at high orders. Figure 5b shows the vaccinated class V(t) growing at the beginning and then became stable at different fractal–fractional orders. As the disease control, the vaccination also controls and goes to their equilibrium point. Figure 5c is the representation of asymptotic or exposed class U(t) showing decrease in its behavior quickly with the passage of time like the behavior of susceptible class at different fractal–fractional orders. In Fig. 5 done can see the dynamical behavior of symptomatic or infectious class which also shows declines as controlled by vaccination and converging to their equilibrium point or became vanishes. Figure 5e shows the class of recovered population from the said epidemic at different fractional orders and fractal dimensions for the independent variable t. The cases of recovery increases at the beginning as more people have been vaccinated and the become stable. In Fig. 6 we simulate the ABC fractal–fractional NoV model (36) when both the fractional order “\({p}\)” and fractal order “\({q}\)” varies equally. From these figures, we note that changing both “\({p}\)” and “\({q}\)” at the same time an epidemic model provides interesting and biologically more feasible results because the infected population is vanishing more significantly as compared to the fractional systems. Thus, from these graphical results, we conclude that utilizing this new idea of FF operator one can observe more accurate results and provide deeper understanding not only for an epidemic model but also to the real world problem arising in science and engineering.
Simulations of the fractional-fractal NoV model in ABC case of system (36) when \({p}=(01, 0.85, 0.75, 0.65)\) and \({q}=(01, 01, 01, 01, 01)\).
Simulations of the fractional-fractal NoV model in ABC case of system (36) when \({p}={q}=(01, 0.85, 0.75, 0.65)\).
Conclusion
In this paper, we propose an epidemic model for NoV transmission in considering environmental noise and fractal–fractional with vaccination effects. Based on the proposed model, there exist a unique time-global solution for any given positive initial value. The thresholds governing the extinction and propagation of the epidemic sickness are determined. The sufficient requirements for extinction of the disease and existence of ergodic stationary distribution of the stochastic system are then obtained from Theorems 2 and 3 utilizing Hasminskii theory and Lyapunov analysis methods. Finally, we applied the concept of FF calculus in the ABC sense to obtain the proposed model. In addition, numerical simulation are given to describe the solution behaviour of our theoretical result. We find that if the noise intensity is large, then the disease will go to extinction. The graphical simulations reveal that the fractal–fractional concept provides better and biologically more reliable results than the classical fractional and ordinary derivatives. In the future, the new approach of modeling known as fractal–fractional operator can be used confidently to study the dynamics of various infectious diseases including the novel COVID-19.
Data availability
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
References
Ahmed, S. M., Lopman, B. A. & Levy, K. A systematic review and meta-analysis of the global seasonality of norovirus. PLoS ONE 8, 75922 (2013).
Marshall, J. A. & Bruggink, L. D. The dynamics of norovirus outbreak epidemics: Recent insights. Int. J. Environ. Res. Public Health 8, 1141–1149 (2011).
Rohayem, J. Norovirus seasonality and the potential impact of climate change. Clin. Microbiol. Infect. 18, 524–527 (2009).
Carmona-Vicente, N. et al. Norovirus infections and seroprevalence of genotype GII. 4-specific antibodiesin a Spanish population. J. Med. Virol. 8, 675–682 (2015).
Din, A. & Li, Y. Stochastic optimal control for norovirus transmission dynamics by contaminated food and water. Chin. Phys. Bhttps://doi.org/10.1088/1674-1056/ac2f32 (2021).
Honma, S. et al. Epidemiological study of prevalence of genogroup II human calicivirus (Mexico virus) infections in Japan and Southeast Asia as determined by enzyme-linked immunosorbent assays. J. Clin. Microbiol. 36, 2481–2484 (1998).
Simmons, K. et al. Duration of immunity to norovirus gastroenteritis. Emerg. Infect. Dis. 13, 1260–1267 (2013).
Hall, A. J. et al. Norovirus disease in the United States. Emerg. Infect. Dis. 19, 1198–1205 (2013).
Lai, C.-C. et al. A norovirus outbreak in a nursing home: norovirus shedding time associated with age. J. Clin. Virol. 56, 96–101 (2013).
Murata, T. et al. Prolonged norovirus shedding in infants 6 months of age with gastroenteritis. Pediatr. Infect. Dis. J. 26, 46–49 (2007).
Peter, O. J., Qureshi, S., Yusuf, A., Al-Shomrani, M. & Abioye Idowu, A. A new mathematical model of COVID-19 using real data from Pakistan. Results Phys. 24, 104098 (2021).
Ariffin, M. R. K. et al. Mathematical epidemiologic and simulation modelling of first wave COVID-19 in Malaysia. Sci. Rep. 11, 1–10 (2021).
Akgül, A. A novel method for a fractional derivative with non-local and non-singular kernel. Chaos Solitons Fractals 114, 478–482 (2018).
Geng, C., Paganetti, H. & Grassberger, C. Prediction of treatment response for combined chemo-and radiation therapy for non-small cell lung cancer patients using a bio-mathematical model. Sci. Rep. 17, 1–12 (2017).
Qureshi, S. Fox H-functions as exact solutions for Caputo type mass spring damper system under Sumudu transform. J. Appl. Math. Comput. Mech. 20, 83–89 (2021).
Boucenna, D., Baleanu, D., Makhlouf, A. B. & Nagy, A. M. Analysis and numerical solution of the generalized proportional fractional Cauchy problem. Appl. Numer. Math. 167, 173–186 (2021).
Chong, K. C. et al. Mathematical modelling of the impact of treating latent tuberculosis infection in the elderly in a city with intermediate tuberculosis burden. Sci. Rep. 9, 1–11 (2019).
Qureshi, S., Chang, M. M. & Shaikh, A. A. Analysis of series RL and RC circuits with time-invariant source using truncated M, Atangana beta and conformable derivatives. J. Ocean Eng. Sci. 6, 217–227 (2021).
Din, A. & Li, Y. Stationary distribution extinction and optimal control for the stochastic hepatitis B epidemic model with partial immunity. Phys. Scr. 96, 074005 (2021).
Li, Y. & Shah, M. A. The complex dynamics of Hepatitis B infected individuals with optimal control. J. Syst. Sci. Compl. 2021(4), 1–23 (2021).
Nana-Kyere, S. et al. Hepatitis B optimal control model with vertical transmission. Appl. Math. 7, 5–13 (2017).
Ji, C. & Jiang, D. Threshold behaviour of a stochastic SIR model. Appl. Math. Model. 38, 5067–79 (2014).
Qureshi, S., Yusuf, A. & Aziz, S. Fractional numerical dynamics for the logistic population growth model under conformable Caputo: a case study with real observations. Phys. Scr. 96, 114002 (2021).
Karatas Akgül, E., Akgül, A. & Baleanu, D. Laplace transform method for economic models with constant proportional Caputo derivative. Fractal Fract. 20, 30 (2020).
Atangana, A. Fractal–fractional differentiation and integration: connecting fractal calculus and fractional calculus to predict complex system. Chaos Solitons Fractals 102, 396–406 (2017).
Din, A., Li, Y. & Yusuf, A. Delayed hepatitis B epidemic model with stochastic analysis. Chaos Solitons Fractals 146, 110839 (2021).
Din, A. & Li, Y. Lévy noise impact on a stochastic hepatitis B epidemic model under real statistical data and its fractal–fractional Atangana–Baleanu order model. Phys. Scr. 96, 124008 (2021).
Zhang, X.-B., Wang, X.-D. & Huo, H.-F. Extinction and stationary distribution of a stochastic SIRS epidemic model with standard incidence rate and partial immunity. Physica A 531, 121548 (2019).
Khasminskii, R. Z. Stochastic Stability of Differential Equations (Book). Alphen aan den Rijn, Netherlands, Sijthoff and Noordhoff (Monographs and Textbooks on Mechanics of Solids and Fluids). Mechanics: Analysis. 7, (1980).
Atangana, A. & Araz, S. İ. New concept in calculus: piecewise differential and integral operators. Chaos Solitons Fractals 145, 110638 (2021).
Atangana, A. & Baleanu, D. New fractional derivatives with non-local and non-singular kernel: theory and application to heat transfer model. Thermal Sci. 20, 763–769 (2016).
Atangana, A. & Qureshi, S. Modeling attractors of chaotic dynamical systems with fractal-fractional operators. Chaos Solitons Fractals 103, 320–337 (2019).
Baleanu, D. & Fernandez, A. On some new properties of fractional derivatives with Mittag–Leffler kernel. Commun. Nonlinear Sci. Numer. Simul. 159, 444–462 (2018).
Is there a population awareness of microbes: the dynamics of Norovirus transmission. CTModelling (2018). https://mp.weixin.qq.com/s/gMdemZE1Vl1r4DHuDqPoQw.
Acknowledgements
This work was supported by the National Natural Science Foundation of P. R. China (Nos. 11901114, 62002068), and Guangzhou Science and technology innovation general project (No. 201904010010), Young innovative talents project of Guangdong Provincial Department of Education (No. 2017KQNCX081), Natural Science Foundation of Guangdong Province (Grant No. 2017A030310598), Finance and Accounting Innovation Research Team under Guangdong, Hong Kong and Macau Greater Bay Area Capital Market (No. 2020WCXTD009).
Author information
Authors and Affiliations
Contributions
T.C.: Conceptualization, data curation, validation, formal analysis, writing—original draft. P.L.: Supervision, Project administration, Funding acquisition, Visualization, review editing. A.D.: Visualization, Writing, Methodology, Software, review editing. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cui, T., Liu, P. & Din, A. Fractal–fractional and stochastic analysis of norovirus transmission epidemic model with vaccination effects. Sci Rep 11, 24360 (2021). https://doi.org/10.1038/s41598-021-03732-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-03732-8
This article is cited by
-
Comprehensive analysis of a stochastic wireless sensor network motivated by Black-Karasinski process
Scientific Reports (2024)
-
Theoretical and numerical analysis of COVID-19 pandemic model with non-local and non-singular kernels
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.