Abstract
Fibromyalgia (FM) is a stress-related chronic pain disorder with common cognitive complaints. This study characterized cognitive dysfunction in patients with FM and explored whether these changes are linked to altered cortisol levels. Consecutive 44 patients with FM and 48 healthy controls were enrolled for the assessments of subjective and objective cognitive functions and diurnal levels of salivary cortisol (sampled at awakening, 30 min after awakening, 3 pm, and bedtime). All measurements were compared between the groups and evaluated for clinical correlation. The FM group had more subjective cognitive complaints and performed poorer in objective cognitive testing in memory (delayed recall in Chinese Version Verbal Learning Test and Taylor Complex Figure Test), language (Boston Naming Test), and executive domains (Wisconsin Card Sorting Test) after adjustments for education. The diurnal cortisol levels of patients with FM tended to be lower, especially at 30 min after awakening and bedtime. Moreover, moderate positive correlations existed between the Chinese Version Verbal Learning Test, Boston Naming Test and the morning cortisol levels within the FM group. We suggested the altered cognitive function in FM may be linked to stress maladaptation. Future studies are warranted to elucidate whether stress management improves cognitive performance in patients with FM.
Similar content being viewed by others
Introduction
Fibromyalgia (FM) is a chronic pain disorder characterized by widespread body pain and several associated symptoms such as fatigue, cognitive problems, unrefreshed sleep, and depression1. Although a complex interaction between genetic disposition and environmental factors may cause FM, its pathophysiology remains undetermined2. Many patients with FM identified stress or a stressful event during their lifetime as a pivotal trigger for their chronic pain. In fact, patients with FM reported a higher proportion of early life adversities such as physical and sexual abuse3; moreover, post-traumatic stress syndrome (PTSD) is a well-identified comorbidity of FM4. The disabling pain per se and complex comorbidities in FM (psychiatric and several others including migraine, irritable bowel syndrome, bladder hyperactivity, and restless leg syndrome) tremendously affect the quality of life of patients5. Therefore, FM may be regarded as a stress disorder2, and stress may be a window into a thorough understanding of this elusive disorder.
The most studied physiological system of stress is the hypothalamic–pituitary–adrenal axis and its downstream product cortisol6. A lower cortisol level has been reported in many stress-related disorders such as PTSD7 and chronic fatigue syndrome (CFS). In FM, however, the basal cortisol level and their diurnal change tended to be lower but inconsistent across studies8,9,10, as discussed in a recent meta-analysis11. These inconsistent results of cortisol levels in FM might be because of different methods of measurement and the lack of a gold standard11. Similar to FM, an animal study12 demonstrated chronic stress induced a hypoactive pattern of cortisol through enhanced negative feedback13, whereas childhood maltreatment (chronic stress) was associated with higher cortisol levels in patients with FM in the afternoon14. Based on the association between hypocortisolism and fatigue experience in CFS, the altered cortisol levels in FM might be considered a maladaptive response to stress15. However, some researchers argue that this is a protective mechanism because a low cortisol level may reduce allostatic load or enhance the body’s defense system against chronic inflammation16.
Cognitive problems, a core symptom of FM, were reported in more than 76% of patients with FM2, majorly involving memory, executive, and language domains17. In fact, FM is the only pain syndrome that incorporates cognitive changes into its diagnostic criteria. Cognitive changes are also pivotal for the assessment of functional disability in FM, such as that using the revised Fibromyalgia Impact Questionnaire (FIQR)18. In a recent review, decreased mental reserve due to pain, fatigue, and depression has been proposed to cause the cognitive symptoms of FM17. Nevertheless, few studies have explored the role of stress in relation to cognitive changes in FM. In animal studies, chronic stress can lead to structural and functional changes in some neural correlates of pain perception or pain modulation, notably in the hippocampus, which may concomitantly affect cognitive functions2,19. The hippocampus is vulnerable to stress owing to the high expression of glucocorticoid receptors20. In human studies, hypercortisolism is hypothesized to be crucial in the pathogenesis of Alzheimer’s disease21, whereas the connection between lower cortisol levels and cognitive dysfunction has been observed in Addison’s disease22, PTSD23, and the normal population24. Although either a low or high cortisol level beyond the physiological range seems detrimental to cognitive function, how cortisol affects cognitive function in FM remains to be investigated.
This study hypothesized that cortisol levels are altered in patients with FM, and this alteration is related to cognitive performance in FM. Accordingly, in the present study, we used comprehensive subjective and objective cognitive assessments to determine whether patients with FM display deficits in different cognitive domains and explored whether these cognitive deficits were associated with the diurnal salivary cortisol levels.
Methods
Participants and procedure
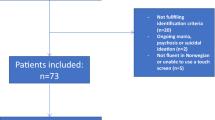
Consecutive patients with FM aged 20–60 years were enrolled from the Neurological Institute of Taipei Veterans General Hospital in Taiwan. All patients fulfilled the modified 2010 American College of Rheumatology (ACR) Fibromyalgia Diagnostic Criteria1; however, those with any autoimmune rheumatic disease were excluded. Healthy volunteers who did not have past or family histories of FM and who had not experienced any significant pain during the past year were recruited as controls. All participants denied having any history of systemic or major neuropsychiatric disease and exhibited normal results of physical and neurological examinations and brain magnetic resonance imaging (MRI). Participants who were receiving any medication or hormone therapy on a daily basis were excluded. Shift workers and patients who experienced severe dry mouth that may interfere with diurnal cortisol measurement or saliva sampling were also excluded. The institutional review board of Taipei Veterans General Hospital approved the study protocol, and each participant provided written informed consent. All methods were carried out in accordance with relevant guidelines and regulations (Declaration of Helsinki).
At the first visit, all patients with FM and healthy controls completed a questionnaire on the extent of body pain (Widespread Pain Index, [WPI]; range 0–19)1, body pain intensity (0–10 on a numerical rating scale), duration (in years) of body pain, and number and severity of the centralized symptoms of chronic pain (Symptom Severity Scale [SSS]; range 0–12), including fatigue, unrefreshing sleep, cognitive symptoms, headache, lower abdominal pain or cramps, and depression1. The FIQR was administered to all participants to assess their FM-related functional disability18. To evaluate depression severity, the Beck Depression Inventory version 1 (BDI-I) was administered to all participants25. Each participant also completed a manual tender point survey. Trained research assistants palpated on the 18 specific anatomical positions defined by the 1990 ACR FM classification at a pressure of 4.0 kg/m2 as measured using a dolorimeter26. Each participant reported the level of tenderness (0: none; 1: mild; 2: moderate; and 3: severe) at each position. The total tender points (TTP; range 0–18) and total tenderness score (TTS = sum of the tenderness level in the 18 positions; range 0–54) of each participant were determined. After completion of the standardized clinical and questionnaire assessments, all participants were scheduled for another visit to undergo cognitive assessment (see 2.2 and 2.3) and were instructed to collect saliva samples at home for diurnal cortisol measurement on the previous day of visit (see 2.4).
Subjective cognitive complaint questionnaire
We designed a Chinese questionnaire to characterize subjective cognitive complaints by asking Yes/No questions about difficulties faced by patients in various daily activities within the past 2 years. This questionnaire comprised 12 questions to identify subjective complaints in 4 different cognitive domains. The total score of this questionnaire was the sum of “Yes” to the 12 questions and therefore ranged from 0 to 12. The English version of this questionnaire is provided as Supplementary Table S1. In addition, we used the severity rating of cognitive symptoms in the SSS as a measure of the global function for subjective cognitive complaints.
Objective cognitive assessments
The objective cognitive assessments were performed by trained psychologists and research assistants, and the results were reviewed and interpreted by psychologists and neurologists. The entire cognitive testing battery included the following tests, which could be categorized into five functional domains:
-
1.
Global function: Mini–Mental State Examination27.
-
2.
Memory: Chinese Version Verbal Learning Test (CVVLT)—total correct and 10 min recall (CVVLT–10 M)28, Wechsler Memory Scale—logic memory test (WMS–LM), part 1 and 229, and Taylor Complex Figure Test (TY-CFT)—delayed recall parts30.
-
3.
Visuospatial: TY-CFT—copy30.
-
4.
Language: the modified 30-item Boston Naming Test (BNT)31 and Semantic Verbal Fluency Test32.
-
5.
Executive: Forward and Backward Digit Span Test33, a modification of the Trail-Making Test, part A and B (TMT-A or -B)34 and Wisconsin Card Sorting Test—64-card version—total number correct (WCST−TC), perseverative response (WCST–PR), and categories completed35.
Salivary cortisol level measurement
One day prior to the aforementioned cognitive assessments, all participants were instructed to follow the protocol modified from the work of McLean, et al.36 to collect their saliva in sample tubes at home at four time points: awakening, 30 min after awakening, 3 pm, and bedtime. For saliva sampling at awakening, participants were asked not to brush their teeth, eat, or drink before sampling. All collected saliva samples were brought to the hospital the next day and preserved in our laboratory refrigerator at − 20 °C until further analysis using commercial enzyme-linked immunosorbent assay kits, AssayMax ELISA Kits (Assaypro, St. Charles, MO, USA). The cortisol awakening response (CAR) was calculated as the difference in cortisol levels between awakening and 30 min after awakening37.
Statistical analysis
Because of the small sample size, Mann–Whitney U tests and chi-square tests were used to examine intergroup differences in demographic and clinical profiles, subjective cognitive complaints, and diurnal cortisol levels. For objective cognitive testing, we used stratification (education years ≤ 12 vs. > 12) to adjust education in both FM and control groups. After stratification for education, fewer participants (both FM and controls) were in the lower education group. Stratification was also applied on the results of subjective cognitive complaints questionnaire to compare the subjective and objective cognitive deficits. Correlation analysis was used to explore the association among demographic and clinical profiles, items of the subjective cognitive complaint questionnaire, objective cognitive testing, and diurnal cortisol levels. For the effect size, φ was for the between group comparisons of the subjective cognitive complaints questionnaire by chi-square test, Cohen’s d for the continuous variables such as total score of the subjective cognitive complaints questionnaire and diurnal cortisol levels, and Hedges’ g for the objective cognitive tests after stratification because of smaller sample size. SPSS version 22 (IBM Inc., Armonk, NY, USA) was used to perform the aforementioned statistical analyses, and a P value of < 0.05 was considered statistically significant. The effect size was small, medium and large if 0.1, 0.3, 0.5 for φ and 0.2, 0.5, 0.8 for Cohen’s d38 and Hedges’ g39, respectively.
Results
Demographics and clinical profile
In total, 44 patients with FM and 48 healthy controls were enrolled in this cross-sectional study. Age and sex did not differ between the FM and control groups, whereas the years of education were fewer in the FM group than in the control group (P = 0.002; Table 1). As expected, the FM group demonstrated higher TTP and TTS on palpation and reported higher scores on the WPI. Moreover, compared with controls, a higher proportion of patients with FM reported fatigue, cognitive symptoms, unrefreshed sleep, headache, and depression on the SSS; the total score on the SSS was also higher in the FM group. The scores on the BDI-I and FIQR were also higher in the FM group than in the control group (Table 1).
Subjective cognitive complaint questionnaire
Compared with the control group, the FM group reported more subjective cognitive complaints, mostly with medium effect sizes for each item (Table 2). In the FM group, 90%, 60%, 33%, and 16% of patients reported at least one symptom in executive, memory, language, and visuospatial domains, respectively. The total score on the subjective cognitive complaint questionnaire was worse in the FM group with a large effect size (FM: 4.1 ± 2.6; controls: 1.3 ± 2.2, P < 0.001, d = 1.171). The total score was highly correlated with the severity of cognitive symptoms in the SSS (0–3) in all participants (FM and controls combined, r = 0.537, P < 0.001) and controls (r = 0.478, P = 0.001) but not in the FM group (r = 0.117, P = 0.485). The total score in the FM group was moderately correlated with functional disability (FIQR, r = 0.335, P = 0.040). After stratification, patients with FM still reported more subjective cognitive complaints than the controls regardless the education level (Supplementary Table S2 and S3).
Objective cognitive function testing
The raw scores of the objective cognitive assessments of both groups were listed as Table 3. In the lower education subgroup analysis, objective cognitive performance did not differ between FM and control subjects (Table 4). In the higher education subgroup analysis, the results in the FM group performed poorer in memory (CVVLT−10 M, 7.9 ± 1.5 vs. 8.6 ± 0.6, P = 0.027, g = 0.641; TY−CFT delayed recall, 33.3 ± 2.0 vs. 34.0 ± 1.9, P = 0.041, g = 0.351), language (BNT: 28.9 ± 1.0 vs. 29.5 ± 1.0, P = 0.002, g = 0.584), and executive domains (WCST−TC: 42.1 ± 9.9 vs. 47.2 ± 8.2, P = 0.026, g = 0.026; WCST−PR: 11.8 ± 7.7 vs. 8.0 ± 5.0, P = 0.018, g = 0.593). The effect sizes were medium with the largest one in the memory domain. Furthermore, several correlations existed between the items of subjective cognitive complaints and objective cognitive testing either in the FM group (Supplementary Table S4) or in all participants (FM and controls combined; Supplementary Table S5). Notably, correlations were observed in language and executive domains but not in the memory and visuospatial domains in the FM group. Also, no correlation was observed between the total score on the subjective cognitive complaint questionnaire and objective cognitive performance.
Diurnal cortisol levels and clinical correlation
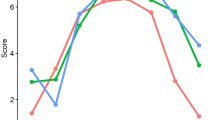
Diurnal cortisol levels in the FM group tended to be lower than those in the control group (Fig. 1). In particular, the cortisol levels were lower in the FM group at 30 min after awakening with a large effect size (FM: 0.209 ± 0.145, controls: 0.386 ± 0.243 pg/mL, P = 0.001, d = 0.890) and at bedtime with a medium effect size (FM: 0.002 ± 0.018, controls: 0.024 ± 0.024 pg/mL, P = 0.021, d = 0.568) but not at awakening (FM: 0.154 ± 0.117, controls: 0.212 ± 0.175 pg/mL, P = 0.148, d = 0.392) and 3 pm (FM: 0.067 ± 0.067, controls: 0.081 ± 0.080 pg/mL, P = 0.503, d = 0.190). The CAR was also lower in the FM group with a medium effect size (FM: 0.056 ± 0.138, controls: 0.161 ± 0.185 pg/mL, P = 0.011, d = 0.646). No correlation was observed between the diurnal cortisol levels and demographics, subjective cognitive complaints, and most clinical profiles (WPI, SSS, and BDI-I) in the FM group. However, the TTP was mild to moderately correlated with the cortisol levels at awakening (r = − 0.253, P = 0.029) and 30 min after awakening (r = − 0.341, P = 0.003). Correlations between the diurnal cortisol levels and objective cognitive performance were noted in several cognitive domains, such as memory, visuospatial, language and executive functions in the FM group (Supplementary Table S6). In contrast, the correlations were fewer and only observed in the executive domain when all participants (both FM and controls) were included in the correlation analysis (Supplementary Table S7). Among the cognitive tests showing differences between FM and controls, moderate positive correlations existed between the CVVLT−10 M and the cortisol level at 30 min after awakening (r = 0.385, P = 0.014) and between the BNT and the awakening cortisol level within the FM group (r = 0.367, P = 0.020). The CAR was not correlated with any objective cognitive tests.
Depression (the BDI-I score) was not correlated with subjective memory complaints or most of the items of the objective cognitive tests in the FM (Supplementary Table S4), and the aforementioned differences between the groups remained the same after adjustment for the BDI-I score.
Discussion
In this study, we characterized changes in the cognitive function of patients with FM and investigated their relationship with the clinical profile of FM and diurnal cortisol levels. Patients with FM had subjective cognitive complaints regarding executive, language, and memory domains. In objective cognitive testing, patients with FM demonstrated deficits mainly in memory, followed by executive, and language functions. In addition, patients with FM displayed a lower diurnal pattern of cortisol levels, particularly at 30 min after awakening with a large effect size and at bedtime with a moderate effect size. Moderate positive correlations existed between the CVVLT−10 M and the cortisol level at 30 min after awakening and between the BNT and awakening cortisol level within the FM group.
Concerning subjective cognitive complaints, our study demonstrated that patients with FM faced difficulties in memory, executive, and language domains. In agreement with our findings, subjective complaints in memory40,41, executive42, and language domains43 have been reported in patients with FM. A recent review summarizing 52 studies on cognitive function in FM also concluded that memory, attention (executive), and word-finding difficulty (language and semantic memory) are the principal subjective and objective cognitive deficits in these patients17. Consistent with this review, the present study also demonstrated deficits in memory (delayed recall in CVVLT and TY−CFT), executive (WCST) and language domains (BNT) in patients with FM.
Previous neuroimaging studies have provided a biological basis for cognitive dysfunction in patients with FM. Specifically, poor performance on WCST in our patients with FM may represent disrupted problem-solving, an ineffective hypothesis-testing strategy35, increased distractibility, or rule detection deficit44. A pertinent MRI study in FM revealed trophic changes in the anterior cingulated cortex, a brain area related to executive function45. The episodic memory deficits in delayed recall of CVVLT and TY−CFT were supported by another MRI study demonstrating decreased bilateral hippocampal volumes in FM46. On the other hand, though the BNT is commonly categorized as a language test, the worse BNT in our patients with FM may reflect poor access to semantic memory47. Reilly, et al.48 demonstrated the utility of the BNT to assess semantic deficits in patients with Alzheimer’s disease and semantic dementia, and several studies also have demonstrated semantic access deficits in patients with FM49. Moreover, the semantic access also relies on the function of the hippocampus50. Sawrie, et al.51 demonstrated that the performance on the BNT is associated with the hippocampus in patients with intractable temporal lobe epilepsy on quantitative 1H magnetic resonance spectroscopy. Thus, both episodic and semantic memory deficits may be linked to hippocampal dysfunction.
In addition to neuroimaging, an association between subjective cognitive complaints and FIQR was demonstrated in the present study and the study conducted by Gelonch, et al.42. As reported by Williams, et al.43, this association may imply that the subjective cognitive dysfunction in patients with FM may additionally result from pain severity, fatigue, mood, sleep, and other insidious clinical factors that interactively affect the overall perception of functional disability. Given the close relationship between these factors and stress, we assumed that stress is an omnibus factor that directly or indirectly affects the cognitive function of patients with FM2. In agreement, our data demonstrated that stress, as assessed by salivary cortisol levels, was associated with TTP in all participants and with the performance of patients with FM on the CVVLT–10 M and BNT.
The relationship between the altered cortisol levels and cognitive functions has been observed in several diseases. Studies on PTSD52, CFS17, Cushing syndrome53, and Addison’s disease22 have reported deficits mostly in memory, executive function, and attention, in line with our results. However, few studies have investigated the relationship between cortisol and cognition in specific functional domains in patients with FM. Sephton, et al.54 demonstrated that the mean diurnal levels of salivary cortisol were positively correlated with the performance on the visual reproduction task. Barcelo-Martinez, et al.55 demonstrated a negative correlation of the diurnal difference (the cortisol level at 8 am to 9 am—cortisol level at 4 pm to 5 pm) in serum cortisol levels with verbal memory tested using the Rey Auditory Verbal Learning Test and cognitive flexibility tested using the WCST, suggesting that a more fluctuated diurnal cortisol level may impair the cognitive function. However, in the study by Barcelo-Martinez et al., the cortisol level in the FM group was comparable to but tended to be higher than that in the control group, different from our patient group with lower cortisol levels. This difference might be because of the difference in the sampling method. Note that the stress of venepuncture in the study by Barcelo-Martinez et al. may have also biased the subsequent data on that day56.
Our study demonstrated that hypocortisolism is associated with cognitive change. The correlations between WMS–LM, TMT-B and diurnal cortisol levels may imply that attenuated diurnal cortisol levels account for the cognitive deficits. Especially, in the two cognitive tests that showed groups differences, moderate positive correlations were noted between the BNT and the awakening cortisol level, and between the CVVLT–10 M and the cortisol level at 30 min after awakening. Therefore, we suggested a generally inadequate diurnal cortisol level may be harmful to cognitive function in patients with FM. However, not all correlations between cortisol and objective cognitive tests in the FM group showed consistent results. For example, the digital backward showed a negative correlation with the awakening cortisol level. Further studies are needed to clarify the inconsistent cortisol effect upon different cognitive tests.
Although no direct evidence supports the link between altered cortisol levels and both episodic and semantic memory deficits, some indirect evidence might suggest hippocampus as a possible neural substrate for this link. The hippocampus is a limbic structure that is closely related to stress processing20 and both episodic and semantic memory function50, as tested by the CVVLT and BNT in the present study. Earlier studies in patients with PTSD reported a correlation between hippocampal atrophy and the accumulative effect of chronic stress (disease duration)57 and also a correlation between hippocampal atrophy and verbal memory deficits58. Taken together, we propose that the memory deficit in patients with FM may be associated with hippocampal dysfunction secondary to chronic stress.
Intriguingly, not all functional domains involved in subjective cognitive complaints from patients with FM demonstrated objective deficits on cognitive function testing. The subjective cognitive complaints were only correlated with the objective cognitive performances in the language and executive domains but not in the memory and visuospatial domains. In the lower education subgroup analysis, the FM group had more subjective cognitive complaints than the controls but comparable objective cognitive performances in memory and executive domains. In fact, such a “mismatch” in cognitive function has been observed in CFS17, a common comorbidity of FM, and other clinical pain conditions such as migraine59. Biologically, the mismatch may be explicable by cognitive compensation amid objective function testing. In functional MRI studies, subjective illness experience and objective performance revealed separate neural substrates60. Another study revealed that patients with FM recruited additional neural networks to achieve the same performance on the Go/NO-go test61, and the resultant mental overloading and exhaustion may aggravate subjective cognitive impairment. Psychologically, several potential factors may lead to greater subjective experience of cognitive deficits (compared with objective cognitive testing). In addition to the aforementioned perceived functional disability and mental exhaustion related to brain processing overloading, memory perfectionism, heightened self-monitoring17, and altered interoceptive awareness62 may contribute to subjective cognitive dysfunction. Studies using personality inventories have been attempting to establish a “pain personality,” including the previous conversion model and the newer vicious cycle model, which refers to the role of problematic stress coping because of the trait of harm avoidance and low self-directedness63.
This study has some limitations. First, women comprised 91% of our FM group, and the generalizability of our data is thus limited. Second, the manual tender point survey was not objective, and interpretation of result may be biased by the location of the survey sites, patient and examiner positioning, order of examination, and pressure application technique64. However, our purpose was to confirm that these patients with FM also fulfilled the 1990 ACR criteria and provided an additional assessment of tenderness following the work of Hsiao, et al.65. Third, our subjective cognitive complaint questionnaire has not been validated. In fact, validated tools for evaluating subjective cognitive decline are still under development66. We attempted to use a comprehensive questionnaire as an exploratory tool to measure subjective cognitive complaints, correlations to the cognitive problems of SSS, and objective cognitive performance (Supplementary Tables S4 and S5) in our study, which may support the validity of our subjective cognitive complaint questionnaire. Fourth, we did not assess the effort required for performing objective cognitive tests. Because body pain may affect motivation, Lockhart and Satya-Murti67 emphasized the role of inadequate effort, especially in a clinical scenario involving patients with FM, where the base rate of inadequate effort is up to 35%. However, Pidal-Miranda, et al.41 suggested that this effect might be small in clinical studies because these study volunteers usually have higher motivation and less compensation-seeking tendencies. Future studies may incorporate performance-validating tests68 to explore effort as a significant contributing factor for subjective or objective cognitive functions in FM. Fifth, the cortisol level of participants was tested only for 1 day, and testing for more days would provide more reliable results, considering day-to-day differences in levels69. Finally, our exploratory cross-sectional study demonstrated the relation between stress and cognitive change; a longitudinal study is warranted to confirm the causality.
Conclusion
Our study demonstrated that patients with FM experienced both subjective and objective cognitive changes in memory, executive and language functions. In objective cognitive testing, both episodic and semantic memory functions in patients with FM were correlated with decreased salivary cortisol levels. The link between cognitive change and cortisol suggests stress maladaptation may play some role in the cognitive dysfunction associated with FM. Additional studies must elucidate whether stress management improves cognitive performance in patients with FM.
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- FM:
-
Fibromyalgia
- CFS:
-
Chronic fatigue syndrome
- PTSD:
-
Post-traumatic stress syndrome
- ACR:
-
American College of Rheumatology
- TTP:
-
Total tender points
- TTS:
-
Total tender point score
- WPI:
-
Widespread pain index
- SSS:
-
Symptom severe score
- BDI-I:
-
Beck’s depression inventory version 1
- FIQR:
-
Revised fibromyalgia impact questionnaire
- CVVLT:
-
Chinese Version Verbal Learning Test
- CVVLT–10 M:
-
Chinese Version Verbal Learning Test—10 min recall
- WMS–LM:
-
Wechsler Memory Scale—logic memory
- TY-CFT:
-
Taylor complex figure test
- BNT:
-
The modified 30-item boston naming test
- TMT-A or -B:
-
A modification of the trail-making test, part A or B
- WCST:
-
Wisconsin card sorting test
- WCST−TC:
-
Wisconsin card sorting test—total correct
- WCST−PR:
-
Wisconsin card sorting test—perseverative response
References
Wolfe, F. et al. Fibromyalgia criteria and severity scales for clinical and epidemiological studies: A modification of the ACR preliminary diagnostic criteria for fibromyalgia. J. Rheumatol. 38, 1113–1122. https://doi.org/10.3899/jrheum.100594 (2011).
Borchers, A. T. & Gershwin, M. E. Fibromyalgia: A critical and comprehensive review. Clin. Rev. Allergy Immunol. 49, 100–151. https://doi.org/10.1007/s12016-015-8509-4 (2015).
Hauser, W., Kosseva, M., Uceyler, N., Klose, P. & Sommer, C. Emotional, physical, and sexual abuse in fibromyalgia syndrome: A systematic review with meta-analysis. Arthritis Care Res. (Hoboken) 63, 808–820. https://doi.org/10.1002/acr.20328 (2011).
Roy-Byrne, P., Smith, W. R., Goldberg, J., Afari, N. & Buchwald, D. Post-traumatic stress disorder among patients with chronic pain and chronic fatigue. Psychol. Med. 34, 363–368. https://doi.org/10.1017/s0033291703008894 (2004).
Yunus, M. B. Central sensitivity syndromes: A new paradigm and group nosology for fibromyalgia and overlapping conditions, and the related issue of disease versus illness. Semin. Arthritis Rheum. 37, 339–352. https://doi.org/10.1016/j.semarthrit.2007.09.003 (2008).
McEwen, B. S. The neurobiology of stress: From serendipity to clinical relevance. Brain Res. 886, 172–189. https://doi.org/10.1016/S0006-8993(00)02950-4 (2000).
Heim, C., Ehlert, U. & Hellhammer, D. H. The potential role of hypocortisolism in the pathophysiology of stress-related bodily disorders. Psychoneuroendocrinology 25, 1–35. https://doi.org/10.1016/S0306-4530(99)00035-9 (2000).
Griep, E. N. et al. Function of the hypothalamic-pituitary-adrenal axis in patients with fibromyalgia and low back pain. J. Rheumatol. 25, 1374–1381 (1998).
McCain, G. A. & Tilbe, K. S. Diurnal hormone variation in fibromyalgia syndrome: A comparison with rheumatoid arthritis. J. Rheumatol. Suppl. 19, 154–157 (1989).
Klerman, E. B., Goldenberg, D. L., Brown, E. N., Maliszewski, A. M. & Adler, G. K. Circadian rhythms of women with fibromyalgia. J. Clin. Endocrinol. Metab. 86, 1034–1039. https://doi.org/10.1210/jcem.86.3.7293 (2001).
Tak, L. M. et al. Meta-analysis and meta-regression of hypothalamic-pituitary-adrenal axis activity in functional somatic disorders. Biol. Psychol. 87, 183–194. https://doi.org/10.1016/j.biopsycho.2011.02.002 (2011).
Romano, G. F., Tomassi, S., Russell, A., Mondelli, V. & Pariante, C. M. Fibromyalgia and chronic fatigue: The underlying biology and related theoretical issues. Adv. Psychosom. Med. 34, 61–77. https://doi.org/10.1159/000369085 (2015).
Houshyar, H., Galigniana, M. D., Pratt, W. B. & Woods, J. H. Differential responsivity of the hypothalamic-pituitary-adrenal axis to glucocorticoid negative-feedback and corticotropin releasing hormone in rats undergoing morphine withdrawal: Possible mechanisms involved in facilitated and attenuated stress responses. J. Neuroendocrinol. 13, 875–886. https://doi.org/10.1046/j.1365-2826.2001.00714.x (2001).
Nicolson, N. A., Davis, M. C., Kruszewski, D. & Zautra, A. J. Childhood maltreatment and diurnal cortisol patterns in women with chronic pain. Psychosom. Med. 72, 471–480. https://doi.org/10.1097/PSY.0b013e3181d9a104 (2010).
Powell, D. J., Liossi, C., Moss-Morris, R. & Schlotz, W. Unstimulated cortisol secretory activity in everyday life and its relationship with fatigue and chronic fatigue syndrome: A systematic review and subset meta-analysis. Psychoneuroendocrinology 38, 2405–2422. https://doi.org/10.1016/j.psyneuen.2013.07.004 (2013).
Fries, E., Hesse, J., Hellhammer, J. & Hellhammer, D. H. A new view on hypocortisolism. Psychoneuroendocrinology 30, 1010–1016. https://doi.org/10.1016/j.psyneuen.2005.04.006 (2005).
Teodoro, T., Edwards, M. J. & Isaacs, J. D. A unifying theory for cognitive abnormalities in functional neurological disorders, fibromyalgia and chronic fatigue syndrome: Systematic review. J. Neurol. Neurosurg. Psychiatry 89, 1308–1319. https://doi.org/10.1136/jnnp-2017-317823 (2018).
Bennett, R. M. et al. The revised fibromyalgia impact questionnaire (FIQR): Validation and psychometric properties. Arthritis Res. Ther. 11, R120. https://doi.org/10.1186/ar2783 (2009).
McEwen, B. S. Protective and damaging effects of stress mediators: Central role of the brain. Dialogues Clin Neurosci 8, 367–381 (2006).
McEwen, B. S. Physiology and neurobiology of stress and adaptation: Central role of the brain. Physiol. Rev. 87, 873–904. https://doi.org/10.1152/physrev.00041.2006 (2007).
Ouanes, S. & Popp, J. High cortisol and the risk of dementia and Alzheimer’s disease: A review of the literature. Front. Aging Neurosci. 11, 43. https://doi.org/10.3389/fnagi.2019.00043 (2019).
Tiemensma, J., Andela, C. D., Biermasz, N. R., Romijn, J. A. & Pereira, A. M. Mild cognitive deficits in patients with primary adrenal insufficiency. Psychoneuroendocrinology 63, 170–177. https://doi.org/10.1016/j.psyneuen.2015.09.029 (2016).
Brunello, N. et al. Posttraumatic stress disorder: Diagnosis and epidemiology, comorbidity and social consequences, biology and treatment. Neuropsychobiology 43, 150–162. https://doi.org/10.1159/000054884 (2001).
Ouanes, S. et al. Life events, salivary cortisol, and cognitive performance in nondemented subjects: A population-based study. Neurobiol. Aging 51, 1–8. https://doi.org/10.1016/j.neurobiolaging.2016.11.014 (2017).
Beck, A. T., Steer, R. A., Ball, R. & Ranieri, W. Comparison of beck depression inventories-IA and -II in psychiatric outpatients. J. Pers. Assess. 67, 588–597. https://doi.org/10.1207/s15327752jpa6703_13 (1996).
Wolfe, F. et al. The American College of rheumatology 1990 criteria for the classification of fibromyalgia. Report of the multicenter criteria committee. Arthritis Rheum. 33, 160–172. https://doi.org/10.1002/art.1780330203 (1990).
Folstein, M. F. & Folstein, S. E. McHugh PR Mini-mental state. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 12, 189–198. https://doi.org/10.1016/0022-3956(75)90026-6 (1975).
Chang, C. C. et al. Validating the Chinese version of the verbal learning test for screening Alzheimer’s disease. J. Int. Neuropsychol. Soc. 16, 244–251. https://doi.org/10.1017/S1355617709991184 (2010).
Gong, Y. Manual of Wechsler Memory Scale-Chinese Version. (Human Medical College Press, 1989).
Taylor, L. B. Localisation of cerebral lesions by psychological testing. Clin. Neurosurg. 16, 269–287. https://doi.org/10.1093/neurosurgery/16.cn_suppl_1.269 (1969).
Cheung, R. W., Cheung, M. C. & Chan, A. S. Confrontation naming in Chinese patients with left, right or bilateral brain damage. J. Int. Neuropsychol. Soc. 10, 46–53. https://doi.org/10.1017/s1355617704101069 (2004).
Gomez, R. G. & White, D. A. Using verbal fluency to detect very mild dementia of the Alzheimer type. Arch. Clin. Neuropsychol. 21, 771–775. https://doi.org/10.1016/j.acn.2006.06.012 (2006).
Hester, R. L., Kinsella, G. J. & Ong, B. Effect of age on forward and backward span tasks. J. Int. Neuropsychol. Soc. 10, 475–481. https://doi.org/10.1017/s1355617704104037 (2004).
Kramer, J. H. et al. Distinctive neuropsychological patterns in frontotemporal dementia, semantic dementia, and Alzheimer disease. Cogn. Behav. Neurol. 16, 211–218. https://doi.org/10.1097/00146965-200312000-00002 (2003).
Greve, K. W. The WCST-64: A standardized short-form of the wisconsin card sorting test. Clin. Neuropsychol. 15, 228–234. https://doi.org/10.1076/clin.15.2.228.1901 (2001).
McLean, S. A. et al. Momentary relationship between cortisol secretion and symptoms in patients with fibromyalgia. Arthritis Rheum. 52, 3660–3669. https://doi.org/10.1002/art.21372 (2005).
Doerr, J. M., Fischer, S., Nater, U. M. & Strahler, J. Influence of stress systems and physical activity on different dimensions of fatigue in female fibromyalgia patients. J. Psychosom. Res. 93, 55–61. https://doi.org/10.1016/j.jpsychores.2016.12.005 (2017).
Cohen, J. Statistical power analysis for the behavioral sciences. (L. Erlbaum Associates, 1988).
Cumming, G. Understanding the new statistics: Effect sizes, confidence intervals, and meta-analysis. (2012).
Gelonch, O., Garolera, M., Valls, J., Rossello, L. & Pifarre, J. Cognitive complaints in women with fibromyalgia: Are they due to depression or to objective cognitive dysfunction?. J. Clin. Exp. Neuropsychol. 39, 1013–1025. https://doi.org/10.1080/13803395.2017.1301391 (2017).
Pidal-Miranda, M., Gonzalez-Villar, A. J., Carrillo-de-la-Pena, M. T., Andrade, E. & Rodriguez-Salgado, D. Broad cognitive complaints but subtle objective working memory impairment in fibromyalgia patients. PeerJ 6, e5907. https://doi.org/10.7717/peerj.5907 (2018).
Gelonch, O., Garolera, M., Valls, J., Rossello, L. & Pifarre, J. Executive function in fibromyalgia: Comparing subjective and objective measures. Compr. Psychiatry 66, 113–122. https://doi.org/10.1016/j.comppsych.2016.01.002 (2016).
Williams, D. A., Clauw, D. J. & Glass, J. M. Perceived cognitive dysfunction in fibromyalgia syndrome. J. Musculoskelet. Pain 19, 66–75. https://doi.org/10.3109/10582452.2011.558989 (2011).
Verdejo-Garcia, A., Lopez-Torrecillas, F., Calandre, E. P., Delgado-Rodriguez, A. & Bechara, A. Executive function and decision-making in women with fibromyalgia. Arch. Clin. Neuropsychol. 24, 113–122. https://doi.org/10.1093/arclin/acp014 (2009).
Robinson, M. E., Craggs, J. G., Price, D. D., Perlstein, W. M. & Staud, R. Gray matter volumes of pain-related brain areas are decreased in fibromyalgia syndrome. J. Pain 12, 436–443. https://doi.org/10.1016/j.jpain.2010.10.003 (2011).
McCrae, C. S. et al. Fibromyalgia patients have reduced hippocampal volume compared with healthy controls. J. Pain Res. 8, 47–52. https://doi.org/10.2147/JPR.S71959 (2015).
Harry, A. & Crowe, S. F. Is the boston naming test still fit for purpose?. Clin. Neuropsychol. 28, 486–504. https://doi.org/10.1080/13854046.2014.892155 (2014).
Reilly, J., Peelle, J. E., Antonucci, S. M. & Grossman, M. Anomia as a marker of distinct semantic memory impairments in Alzheimer’s disease and semantic dementia. Neuropsychology 25, 413–426. https://doi.org/10.1037/a0022738 (2011).
Glass, J. M. Review of cognitive dysfunction in fibromyalgia: A convergence on working memory and attentional control impairments. Rheum. Dis. Clin. N. Am. 35, 299–311. https://doi.org/10.1016/j.rdc.2009.06.002 (2009).
Duff, M. C., Covington, N. V., Hilverman, C. & Cohen, N. J. Semantic memory and the hippocampus: Revisiting, reaffirming, and extending the reach of their critical relationship. Front. Hum. Neurosci. 13, 471. https://doi.org/10.3389/fnhum.2019.00471 (2019).
Sawrie, S. M. et al. Visual confrontation naming and hippocampal function: A neural network study using quantitative 1H magnetic resonance spectroscopy. Brain 123, 770–780. https://doi.org/10.1093/brain/123.4.770 (2000).
Hayes, J. P., Vanelzakker, M. B. & Shin, L. M. Emotion and cognition interactions in PTSD: A review of neurocognitive and neuroimaging studies. Front. Integr. Neurosci. 6, 89. https://doi.org/10.3389/fnint.2012.00089 (2012).
Piasecka, M. et al. Psychiatric and neurocognitive consequences of endogenous hypercortisolism. J. Intern. Med. https://doi.org/10.1111/joim.13056 (2020).
Sephton, S. E. et al. Biological and psychological factors associated with memory function in fibromyalgia syndrome. Health Psychol. 22, 592–597. https://doi.org/10.1037/0278-6133.22.6.592 (2003).
Barcelo-Martinez, E. et al. Serum cortisol levels and neuropsychological impairments in patients diagnosed with Fibromyalgia. Actas espanolas de psiquiatria 46, 1–11 (2018).
Vining, R. F., McGinley, R. A., Maksvytis, J. J. & Ho, K. Y. Salivary cortisol: A better measure of adrenal cortical function than serum cortisol. Ann. Clin. Biochem. 20, 329–335. https://doi.org/10.1177/000456328302000601 (1983).
Sapolsky, R. M. Why stress is bad for your brain. Science 273, 749–750. https://doi.org/10.1126/science.273.5276.749 (1996).
Bremner, J. D. et al. MRI-based measurement of hippocampal volume in patients with combat-related posttraumatic stress disorder. Am. J. Psychiatry 152, 973–981. https://doi.org/10.1176/ajp.152.7.973 (1995).
Martins, I. P. et al. Cognitive aging in migraine sufferers is associated with more subjective complaints but similar age-related decline: A 5-year longitudinal study. J. Headache Pain 21, 31. https://doi.org/10.1186/s10194-020-01100-x (2020).
Walitt, B. et al. Characterizing “fibrofog”: Subjective appraisal, objective performance, and task-related brain activity during a working memory task. Neuroimage Clin. 11, 173–180. https://doi.org/10.1016/j.nicl.2016.01.021 (2016).
Glass, J. M. et al. Executive function in chronic pain patients and healthy controls: Different cortical activation during response inhibition in fibromyalgia. J. Pain 12, 1219–1229. https://doi.org/10.1016/j.jpain.2011.06.007 (2011).
Weiss, S., Winkelmann, A. & Duschek, S. Recognition of facially expressed emotions in patients with fibromyalgia syndrome. Behav. Med. 39, 146–154. https://doi.org/10.1080/08964289.2013.818932 (2013).
Naylor, B., Boag, S. & Gustin, S. M. New evidence for a pain personality? A critical review of the last 120 years of pain and personality. Scand. J. Pain 17, 58–67. https://doi.org/10.1016/j.sjpain.2017.07.011 (2017).
Okifuji, A., Turk, D. C., Sinclair, J. D., Starz, T. W. & Marcus, D. A. A standardized manual tender point survey. I. Development and determination of a threshold point for the identification of positive tender points in fibromyalgia syndrome. J. Rheumatol. 24, 377–383 (1997).
Hsiao, F. J. et al. Altered insula-default mode network connectivity in fibromyalgia: A resting-state magnetoencephalographic study. J. Headache Pain 18, 89. https://doi.org/10.1186/s10194-017-0799-x (2017).
Si, T., Xing, G. & Han, Y. Subjective cognitive decline and related cognitive deficits. Front. Neurol. 11, 247. https://doi.org/10.3389/fneur.2020.00247 (2020).
Lockhart, J. & Satya-Murti, S. Symptom exaggeration and symptom validity testing in persons with medically unexplained neurologic presentations. Neurol. Clin. Pract. 5, 17–24. https://doi.org/10.1212/CPJ.0000000000000092 (2015).
Greher, M. R. & Wodushek, T. R. Performance validity testing in neuropsychology: Scientific basis and clinical application-a brief review. J. Psychiatr. Pract. 23, 134–140. https://doi.org/10.1097/PRA.0000000000000218 (2017).
Stalder, T. et al. Assessment of the cortisol awakening response: Expert consensus guidelines. Psychoneuroendocrinology 63, 414–432. https://doi.org/10.1016/j.psyneuen.2015.10.010 (2016).
Acknowledgements
Yi-Ju Lin drew the figure on SigmaPlot version 12.3.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and article publication: Ministry of Science and Technology of Taiwan (MOST 107-2314-B-075-015-MY2-2 to WT Chen, and MOST 104-2314-B-010-016-MY3, and MOST 108-2321-B-010-013-MY2 to PN Wang) and Taipei Veterans General Hospital (V108C-129 and V107C-091 to WT Chen, and V108C-060 to PN Wang). This work was also supported by the Brain Research Center, National Yang-Ming University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education in Taiwan. The funders had no role in the study design, data collection and analysis, decision to publish, or manuscript preparation.
Author information
Authors and Affiliations
Contributions
Y.-J.L. performed the statistical analyses, interpreted the data, and drafted the manuscript. Y.-C.K., L.-H.C., F.-J.H., and H.-Y.L. helped in patient enrolment, questionnaire survey, and data analysis. P.-N.W. and W.-T.C. designed the study, collected and interpreted the data, and revised the manuscript. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lin, YJ., Ko, YC., Chow, LH. et al. Salivary cortisol is associated with cognitive changes in patients with fibromyalgia. Sci Rep 11, 1311 (2021). https://doi.org/10.1038/s41598-020-79349-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-79349-0
This article is cited by
-
Long read sequencing characterises a novel structural variant, revealing underactive AKR1C1 with overactive AKR1C2 as a possible cause of severe chronic fatigue
Journal of Translational Medicine (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.